Does a Controlled Diet Improve Cellulite?
Abstract
Abstract:
Several researchers have suggested that a targeted diet for reducing theadipose tissue may interfere with the severity of cellulite. Others emphasize that the diet composition seems to play a more relevant role than the calorie profile for weight loss and cellulite control.
Objective:
The aim of this study was to evaluate the influence of a controlled diet on body composition and cellulite improvement in adult women.
Methods:
Thirty two healthy women, aged from 25 to 40 were included. They receivedan orientation for a controlled diet to be followed for 3 months. Assessments were performed at the baseline and the end of the study which included four parameters: 1) Photonumeric cellulite severity scale; 2) Anthropometric measuremets 3) Skin elasticity (Cutometer®) and 4) Collagen density or echogenicity and length of the dermis-hypodermis interface line (ultrasonography - DermaScan®). The data was compared with the Paired T-test, Wilcoxon and Pearson's correlation for statistical analyses.
Results:
Only 14 completed the study. Although the scores of photonumeric scalereduced, there was no difference in the grade of cellulite severity. There were significant reductions in body measurement. The skin elasticity showed no significant change. On the other hand, the dermal density or echogenicity showed a significant increase in the right and left sides (p = 0.05 and p = 0.005, respectively); however, no difference was observed in the lenght of dermis-hypodermis line.
Conclusion:
the controlled diet was effective for the reduction of weight and body composition, but despite the increase in dermal collagen density, no clinical effect on cellulite could be detected.
Author Contributions
Academic Editor: Guoxun Chen, The University of Tennessee
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2016 Marques N.C , et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction:
Cellulite is a localized skin surface irregularity that affects 85-98% of women after puberty. 1,2 Alquier and Pavot first described it in 1920 as an unaesthetic alteration of skin surface secondary to traumatic, infectious and/or glandular stimuli.3 It is not considered a disease; therefore it does not affect life expectancy or involve risk of secondary diseases. However it may disturbe physical, mental and social wellness, with psychosocial impacts. 1,3,4 It is likely that the etiopathogenesis is multifactorial, and that the anatomical and structural changes in the connective and adipose tissues of women are considered the main factors. 5,6,7,8,9,10,11 Some authors have also cited alterations in microcirculation12,13, hormonal changes, genetic predisposition and nutritionl as factors as well.14, 15
The therapies have common goals such as: induction of lipolysis in subcutaneous tissue; improvement of blood flow, skin elasticity and dermal thickness and reduction of the hypertroph connective tissue septa in the hypodermis. However, the effects are limited and temporary, and therefore the results are variable5, 24, 42, 43.
The role of a specific diet, which reduces the adipose tissue and thus the cellulite severity is controversial8. Some authors believe that the diet composition is more important than the calorie profile for weight loss.11 Changes in the diet composition, like removing simple sugars, since they can increase the level of advanced glycation end products (AGEs) and a consequent collagen breakdown, could be beneficial.16Another change could be a higher consumption of mono and polyunsaturated fatty acids, which is possibly correlated to the reduction of pro-inflammatory factors.17 As well as the increase in nutrients such as zinc and vitamins A, C and D because these may act in the epidermis, derme and collagen fibers.18, 19 And also, ingesting more fruit, vegetables and cereals that are rich in terpenoids and polyphenols, due to its control of adipocyte differentiation.20, 21 These also contain silicon, which contribuite with the connective tissue metabolism.22, 23, 24, 25, 26
There are very few studies evaluating the relationship between dietary patterns and cellulite.However, most of them didn't use the adequate methodology or were inconclusive. Therefore our question was if the possible change in the dietary pattern would reduce the cellulite severity by reducing the subcutaneous fat and/or improving the dermal structure.
Material and Methods:
The participants were recruited in the Cosmetic Dermatology Unit of the Department of Dermatology at the Federal University of Sao Paulo (UNIFESP) after the UNIFESP Ethics Review Board approved. They signed the Consent Form and Photography authorization. The study design was a therapeutic interventionl, open, not randomized and evaluator blinded, including 32 healthy women; aged from 20 to 40. The inclusion criterion was: eutrophic and overweight (according to Body Mass Index - BMI), with visable cellulite on the buttocks, grades II or III, according to Nurnberger-Muller classification5. Exclusion criteria were: pregnancy or desire to become pregnant; currently nursing; history or clinical signs of venous insufficiency, arterial diseases, chronic diseases, mental disorders, and also have undergone any treatment for cellulite in the last 6 months, like gluteoplasty or liposuction in the areas with cellulite.
During their first visit, participants were asked to fill out a 24-hour diet recall for qualitative analysis of their dietary pattern. They were also asked about physical activity, oral contraceptive use and previous pregnancy. They receveid an oral and written orientation for the controlled diet to be followed for 3 months.
The diet was based in the maintenance of a nutritional balance with a predominance of fresh food, aimed to increase the ingestion of antioxidants. The distribution of macronutrients comprised: 55-65 %, carbohydrate, 20-35% fat and 10-20%, protein. The original quantitative intake, that was assessed by the 24-hour recall was maintained. The difference was the food choices, with a prioritization of the nutritional quality and, thus, directing the participant to what was permitted and prohibited (Figure 1). The diet was characterized by less industrialized food, low content of saturated and trans fatty acids and low consumption of simple carbohydrates. The participants were advised to consume whole-grain foods, avoid sugar and candies, maintain high levels of animal and vegetable proteins, eat vegetables and legumes freely and completely avoid alcohol. They were also instructed to consume vegetable and red fruit juice once a day. Efficacy assessments were performed at the baseline and at the end of the study, except for the diet recall, which was calculated on the following four days: 01, 30, 60 and 90.
Figure 1.Controlled diet composition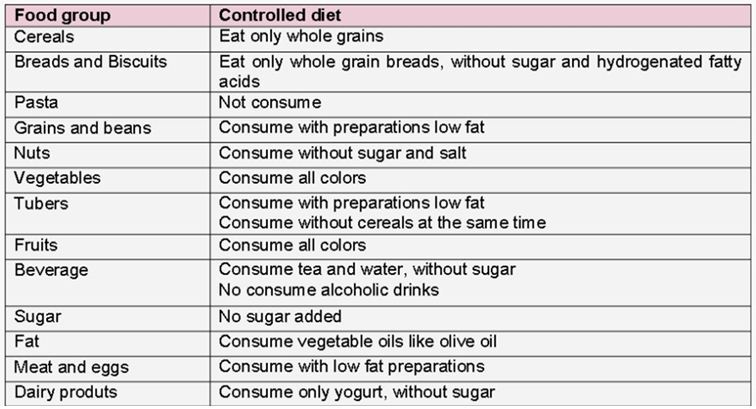
The quantitative analysis was performed by calculating the distribution of macronutrients in the total caloric value,polyunsaturated fatty acids and total fibers, using the Dietwin professional software (version 2979, 2014 / Brazil), which is considered an effective method to assess the amount of nutrients ingested.
The Efficacy Parameters Included:
Photonumeric Cellulite Severity Scale
At the end of the study, three independent observers (all dermatologists) conducted a blind evaluation of before and after through photos that were taken, using the photonumeric cellulite severity scale (PSSC).27
To ensure photograph standardization, participants used similar disposable underwear. Photos were not taken during the first 4 days of menstruation, to avoid possible interference of liquid retention. The photos were taken in a dark room, with a black background and two points of light located at 0,7m from the ground, suspended by a tripod and at 1m from the participant. The camera (Canon EOS - Rebel XTi, USA) was set on a tripod and located at 2 m from the participant. They were asked to remain motionless and relaxed with no contraction of the hip and lower limb muscles. A reference point was marked on the ground to stand by tagging the carpet which ensured standardization of the distances.
Anthropometric Measurement
Circumferences of the waist, abdomen and hips were measured with a non-extensible measuring tape. The thickness of five skinfolds, at the right side of the body, with the participant standing, was taken (biceps, triceps, subscapular, abdominal and suprailiac).28 Sums at baseline and day 90 were generated, which were later used as an indirect method to assess changes. Weight was measured using a simple electronic balance and the participant reported their height.
Body composition was assessed by bioimpedance (BIA 101 Quantum, RJL Systems Detroit, MI, USA), which is a validated non-invasive method.29 It is a fast and accurate form to estimate lean mass and fat mass, and also has relatively low cost.30This was not performed during the menstruation period to avoid the interference of fluid retention.31, 32
Skin Elasticity
Skin elasticity was evaluated using a Cutometer® device (MPA 580, Courage & Khazaka, Cologne, Germany) on standardized points of gluteal region, and the biological elasticity (R7 value) was determined.
Collagen Density and Length of Dermis-Hypodermis Interface Line
A high-frequency (20 MHz) ultrasound (US) device (DermaScan® C USB, Cortex Technology ApS, Denmark) was used, which allows high resolution sectional skin images up to a depth of 3mm, that show both epidermis and dermis. From the images is possible to calculate the echogenicity, which correspond to the dermal extracellular matrix density33, as well as the length of the line between dermis and hypodermis (the longer the line, the greater is the protrusion of the hypodermis toward the deep dermis). The measurements were taken at the same points of gluteal region that was used for skin elasticity.
Statistical Analysis
The data was compared with the Paired T-test, Wilcoxon and Pearson's correlation for statistical analyses, using a statistical program (GraphPad - Prism version 5 - USA). The test used was decided according to the normal curve, two-tailed. The values were considered significant for p value< 0.05.
Results:
Out of the 32 women who participated, only 14 completed the study. This represents a dropout rate of 56% (18/32). The main cause of dropout was non-adherence to the proposed diet (10 women). Other causes were: absence in the last assessment (7 women) and unexpected pregnancy (1 woman). Statistically there was no difference in the BMI at the baseline between the participants who finished the study (24, 5±3, 1kg/m2) and those who dropped out (28, 4±6, 8 kg/m2,), therefore the BMI was not a cause for the dropout.
For baseline analysis, the ITT (intention to treat) population (n=32) was considered. Table 1 shows the baseline data, including participants’ mean age, weight and BMI, pregnancy history and oral contraceptive use. Table 2 comprises data for the participants’ level of physical activity, showing that 44% reported a moderate or intense level and 16% reported light physical activity. They were instructed to maintain this practice throughout the study, avoiding the bias of a pattern change. The sedentaries (40%), were instructed to remain so until the end of the study.
Table 1. Mean age, weight, Body Mass Index (BMI); pregnancy history and oral contraceptive use at baseline (ITT population)| n=32 | |
| Age (years) | 32.2±5.0* |
| Weight (kg) | 72.4±3.1* |
| n=32 | |
| Sedentary | (13/32) 40% |
| Light | (5/32) 16% |
| Moderate | (9/32) 28% |
| Intense | (5/32) 16% |
The food consumption data recorded in the 24-hour diet recall at baseline showed that most participants consumed industrial products, foods with high glycemic index and rich in saturated fat, more than twice a day (Table 3).
Table 3. Food groups in the 24-hour diet recall of the participants at baseline (ITT population)| n=32 | <1x /day | 1-2x /day | >2x /day |
| Industrialized food | 10% | 31% | 59% |
| High glycemic food | 10% | 50% | 40% |
| Foods with saturated or trans fat | 6% | 31% | 63% |
| Vegetables | 28% | 38% | 34% |
Photonumeric Cellulite Severity Scale
For data analysis, we considered the mean values attributed to cellulite severity on gluteal regions, by three independent observers, at baseline and day 90 by using the PP (per protocol) population. It was observed that the total scores reduced , but the results were not significant (right side, p=0.3033; left side, p=0.3955). Figure 2 shows the cellulite clinical aspect in the gluteal region of one participant at baseline and day 90, with no noticeble difference.
Figure 2.Cellulite clinical aspect in the gluteal region of one participant at baseline and the end of the intervention
Anthropometric data and bioimpedance
Table 4 presents the mean and standard deviation of the anthropometric measurements and the percentage of lean mass and fat mass obtained by bioimpedance. There were significant reductions in body measurements, such as: weight, BMI, skinfold sum, waist and abdomen circumferences, percentage of lean and fat mass. These data demonstrated the effectiveness of the controlled diet for weight loss and reduction in body measurements.
Table 4. Mean and standard deviation of anthropometric measurements and percentage of bioimpedance data at baseline and end of intervention (PP population)| n=14 | baseline | day 90 | p - value |
| Weight (kg) | 66.8±11.5 | 63.0±11.6* | 0.0001* |
| BMI (kg/m 2 ) | 24.5±3.1 | 23.1±3.1* | 0.0001* |
| ∑ skinfold (mm) | 80.4±24.3 | 64.4±24.3* | 0.0004* |
| Waist (cm) | 75.5±8.5 | 71.5±7.6* | <0.0001* |
| Abdominal (cm) | 85.6±9.3 | 83.3±7.5* | 0.02* |
| FM (%) | 29.3±5.4 | 27.1±4.7 | 0.004* |
| LM (%) | 68.1±7.9 | 72.3±4.9 | 0.015* |
| EW (%) | 46.5±2.6 | 46.1±2.6 | 0.36 |
Skin Elasticity
The skin elasticity showed no significant change (right side, p-=0.4409; left side, p=0.3174) when compared to the mean of the measures at baseline and day 90 (PP population).
Results from the Ultrasonography
Table 5 presents the mean of the measures for dermal echogenicity and the length of the dermis-hypodermis interface line, obtained by the US images. The dermal density showed a significant increase for both sides (right, p=0.02; left, p=0.005). However, the length of dermis-hypodermis interface line showed no significant change.Figure 3 shows the US images of one participant
Table 5. Mean and standard deviation of the measures for dermal echogenicity and the length of the dermis-hypodermis interface line for gluteal regions at baseline and end of intervention (PP population)| n=14 | Dermal echogenicity | Lenght of the dermis-hypodermis interface line | ||||
| Baseline | day 90 | p-value | baseline | day 90 | p-value | |
| Right | 0.23±0.13 | 0.30±0.14 | 0.05 * | 12.61±0.53 | 12.59±0.49 | 0.44 |
| Left | 0.21±0.11 | 0.31±0.09 | 0.005 * | 12.59±0.53 | 12.55±0.40 | 0.73 |
Figure 3.High frequency US images of one participant, showing an increase in dermal echogenicity in gluteal regions by the comparison between baseline and end of intervention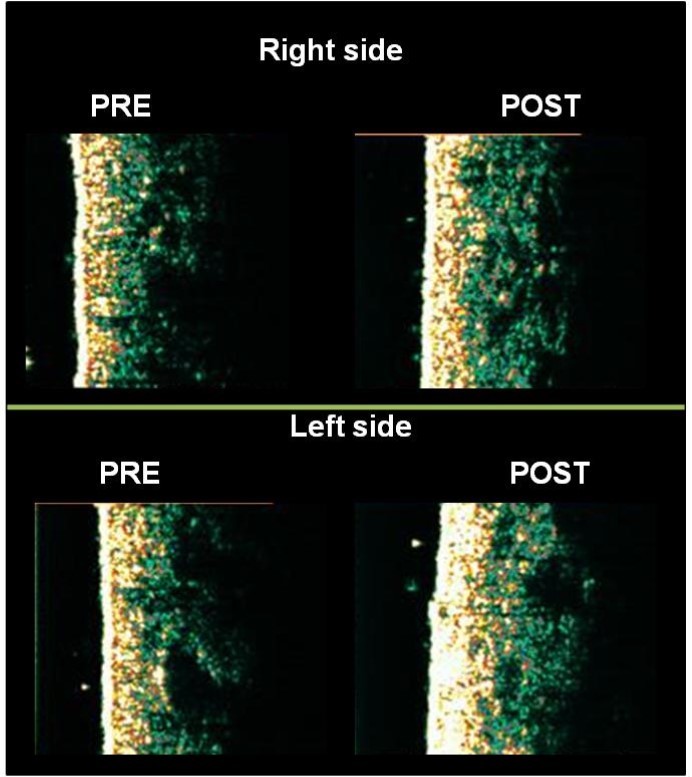
Dietary Consumption
The quantitative calculation demonstrated that the participants followed the controlled diet throughout the intervention period. By observing the macronutrient distribution, it is possible to verify that the participants consumed an average of 35% lipids, 47% carbohydrates and 18% protein from the Total Caloric Values (TCV).
Figure 4 shows a comparison between controlled diet and normal diet. There was a higher consumption of total lipids, polyunsaturaded fatty acids and fiber in the controlled diet, but without statistical significance. Positive correlations were observed between weight loss and lipids consumption (r=0.764; p=0.001) and a negative correlation between weight loss and carbohydrate consumption (r=-0.787; p=0.007).
Figure 4.Comparison of controlled diet and normal diet according to the average of nutritional intake during the intervention.Note: not significant values (Wilcoxon)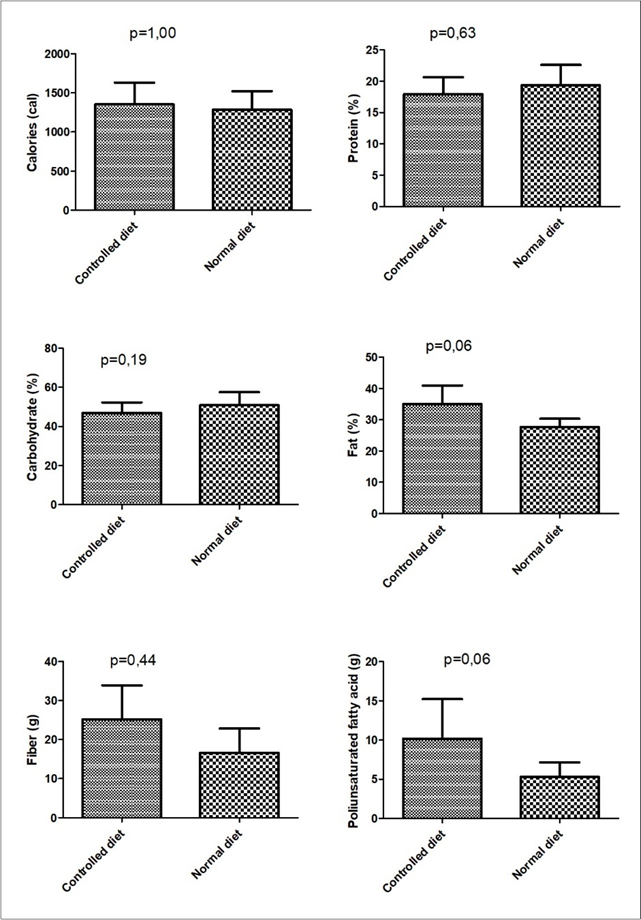
Discussion
Cellulite control depends on the association of various therapeutic interventions, due to its multifactorial etiology, despite the unclear mechanisms. Invasive and non-invasive treatments are based on existing theories about its pathogenesis, as well as trying to mitigate supposed aggravating factors such as unproven changes in the dermis and hypodermis -microcirculation and chronic inflammation. 34, 35In this study, we opted for a non-invasive approach by changing the dietary pattern.
The occurrence of a 56% dropout rate illustrates the poor adherence of participants to a restricted diet. Dropouts in clinical studies about cellulite are frequent, due to the absence of visible short-term results and the participant’s lack of interest in maintaining the therapeutic proposals.27, 36 Following a controlled diet during medium and long term, even though providing many benefits, is difficult to mantain considering individual habits and food preferences.37, 38, 39
The weight and BMI at baseline, as well as the level of physical activity corroborate with the assumption that women who are bothered by cellulite and seek treatment, generally with high expectations, are those who possess healthier life habits and higher aesthetic concern37, 38, 39, but, whose food habits at baseline were characterized by large amounts of high glycemic food.
Although there is no consistent evidence on which food groups affect cellulite, the analysis of the diet recall before the intervention showed that most participants were used to eating industrialized food, with high glycemic index and rich in trans fatty acids. It is possible that these food groups can be associated with cellulite. And the lack of vitamins and minerals, which act as antioxidants, may also have some impact in the dermis structure, as was pointed out by several authors.18, 40, 41, 42, 43, 44, 45, 46
The photography analysis demonstrated no significant reduction in the scores. But it has been suggested that reducing adipose tissue, independent of body obesity, can improve cellulite severity7, 8 in healthy persons. It depends greatly on the quality and quantity of their daily diet.
Some studies have discussed that the consumption of foods with high glycemic index can accelerate the collagen tissue glycation.47, 48 The presence of foods rich in saturated and trans fatty acids seems to increase fat deposition, with weight gain and development of inflammatory processes.49 A study conducted in 2013 compared three diets with the same caloric intake and different profiles of saturated fat (6%, 12%, 24%) for 16 weeks in an animal model (rat). It was observed that the diet with 12% saturated fat increased adiposity, inflammatory markers and promoted metabolic dysfunction; the diet with 24% saturated fat displayed an increase in insulin resistance and a higher ratio of triglycerides and LDL-cholesterol which are known as cardiovascular risk factors.50
The controlled diet resulted in weight loss, reduction in the skinfolds sum and percentage of fat. It was observed, a visible change in body composition and an increase in dermal echogenicity, possibly by a positive effect in collagen density and organization, in the areas with cellulite51, 52, 53, 54, 55. However, as weight loss is distributed throughout the body, there is no guarantee that the fat in areas affected by cellulite was also reduced. Additionally, it is not possible to assert that this result can always cause improvement in the dimpled appearance of the skin surface, as previously described in the literature.56However, this study, despite its limitations, suggests that weight loss by a controlled diet may provide structural dermal changes..
The presence of macronutrients in the controlled diet, seems to have had a large contribution to weight loss, as the caloric food intake was maintained.57 The controlled diet included a higher ingestion of fat when compared to the normal diet. It is important to note that this corresponded to a greater consumption of polyunsaturated fatty acids, which may have contributed to the weight loss.
A negative correlation was found between the consumption of carbohydrates and weight loss. It is probable that the participants experienced weight loss due to the limitation of high glycemic index of processed foods, such as pasta, sweets and desserts with added sugar or other sweeteners.. This result is consistent with the findings of another study that demonstrated the greater effectiveness of a low glycemic index diet for weight loss as compared with a low fat and higher glycemic index58In addition, to maintain weight after the initial loss, a low glycemic food load appears to be more effective, due to the positive correlation between low glycemic index and slower digestion, enhancing the intake of proteins and fats as they are related to satiety.59
In the present study, protein intake was maintained within normal limits according to DRI,57 that is, the controlled diet was normoproteic. Clifton et al in a similar intervention did not demonstrate significant difference in weight loss among participants who followed a high proteic diet compared to a standard diet, which is in accordance to our findings.60However, the influence of protein intake on weight loss remains controversial, since other investigators have reported that high proteic diets contribute to satiety and consequent reduction in food consumption.61
Considering only diet and its influence on cellulite, a study showed that a high proteic diet combined with reduced carbohydrate consumption, showed better response in reducing cellulite severity. The participants followed a proteic diet also experienced weight loss. But this diet was also rich in fat, which was not discussed.62
Our study demonstrated that weight loss was obtained with a diet rich in fat consumption, normal range of reduced carbohydrates and changes oriented only in relation to type of foods. There was a significant increase in dermal echogenicity in gluteal regions that could be related to an improvement in collagen tissue. We do not have an explanation about the mechanism involved. It is possible that a reduction in collagen glycation and inflammation in the dermis and adipose tissue may have occurred, despite the short time of the intervention. The study duration was chosen so that the results could be compared to the scarce literature available63, 64, 65, 66, 67, 68, 69, 70.
The results for skin elasticity, contrary to our expectation, were not significant. This aspect may be related to weight loss, as it leads to metabolic abnormalities that can compromise skin elasticity.2Other authors have suggested that weight loss may be associated with changes in the dermis, the reduction of skin elasticity and negative interference in cellulite too.71, 72 Non-invasive interventions for cellulite, in limited period, are targeted to reduce subcutaneous fat and/or edema and not to improve skin elasticity.
The majority of the results in this study was non-significant. It is important to highlight the complexity in assessing cellulite severity, the disparity between researchers’ and participants’ opinion and, above all, the challenge to control this common and disconfortable disorder.
Conclusion:
This study demonstrated the efficacy of a controlled diet in reducing weight and body composition, accompained by a possible increase in collagen density, despite no effect in cellulite clinical appearance.
We believe that further studies on genes related to obesity, as well as studies on cellulite pathogenesis, epidemiology, genetic predisposition, triggers, aggravating conditions, cellular and molecular mechanisms would be useful to direct the therapy. It is important to combine a healthy lifestyle, including diet and exercises, with long-term non-invasive or invasive therapeutic interventions. The successful treatment remains distant. Unfortunately as cellulite continues to bother women, many of them become hostages of costly treatment proposals, most of which, without a solid scientific basis.
Acknowledgements
for the financial support from Foundation for the Support of Research of the State of Sao Paulo (FAPESP) #2011/51633-8
References
- 1.Avram M M. (2004) Cellulite: a review of its physiology and treatment. , J. Cosmet. Laser. Ther 6(4), 181-5.
- 2.L K Smalls, C Y Lee, Whitestone J. (2005) Quantitative model of cellulite: three-dimensional skin surface topography, biophysical characterization, and relationship to human perception. , J. Cosmet. Sci 56(2), 105-20.
- 3.Rossi A B R, A L Vergnanini. (2000) Cellulite: a review. , J. Eur. Acad. Dermatol. Venerol 14, 251-62.
- 5.Nurnberger F, Müller G. (1978) So-called cellulite: an invented disease. , J. Dermatol. Surg. Oncol 4(3), 221-9.
- 6.E H Rose, L M Vistnes, G A Ksander. (1978) A microarchitectural model of regional variations in hypodermal mobility in porcine and human skin. , Ann. Plast. Surg 1(3), 252-66.
- 7.Z D Draelos, K D Marenus. (1997) Cellulite: Etiology and purported treatment. , Dermatol. Surg 23(12), 1177-81.
- 8.Rosenbaum M, Prieto V, Hellmer J. (1998) An exploratory investigation of the morphology and biochemistry of cellulite. , Plast. Reconstr. Surg 101(7), 1934-9.
- 9.G E Piérard, J L Nizet, Piérard-Franchimont C. (2000) Cellulite: from standing fat herniation to hypodermal stretch marks. , Am. J. Dermatopathol 22(1), 34-7.
- 10.Mirrashed F, J C Sharp, Krause V. (2004) Pilot study of dermal and subcutaneous fat structures by MRI in individuals who differ in gender, BMI, and cellulite grading. , Skin. Res. Technol; 10(3), 161-8.
- 11.Hexsel D, Siega C, Schilling-Souza J. (2013) Noninvasive treatment of cellulite utilizing an expedited treatment protocol with a dual wavelength laser-suction and massage device. , J. Cosmet. Laser. Ther 15(2), 65-9.
- 13.Terranova F, Berardesca E, Maibach H. (2006) Cellulite: nature and aetiopathogenesis. , Int. J. Cosmet. Sci; 28(3), 157-67.
- 14.Mole B, Blanchemaison P, Elia D. (2004) High frequency ultrasonography and celluscore: an improvement in the objective evaluation of cellulite phenomenon. , Ann. Chir. Plast. Esthet 49(4), 387-95.
- 15.Emanuele E, Bertona M, Geroldi D. (2010) A multilocus candidate approach identifies ACE and HIF1A as susceptibility genes for cellulite. , J. Eur. Acad. Dermatol. Venereol 24(8), 930-5.
- 16.Kamalov M, P W Harris, C G Hartinger, G M Miskelly, G J Cooper. (2015) Physicochemical studies on the copper (II) binding by glycated collagen telopeptides. , Org. Biomol. Chem 13(10), 3058-63.
- 18.E C Naylor, R E Watson, M J Sherratt. (2011) Molecular aspects of skin aging. , Maturitas 69(3), 249-56.
- 19.Costa A, S Pegas Pereira E, E C Assumpção, B Calixto dos Santos F, F S Ota. (2015) Assessment of clinical effects and safety of an oral supplement based on marine protein, vitamin C, grape seed extract, zinc, and tomato extract in the improvement of visible signs of skin aging in men. , Clin. Cosmet. Investig. Dermatol 8, 319-28.
- 20.Kuenzli S, J H Saurat. (2003) Peroxisome proliferator-activated receptors in cutaneous biology. , Br. J. Dermatol 149(2), 229-36.
- 21.Gupta M, V K Mahajan, K S Mehta, P S Chauhan, Rawat R. (2015) Peroxisome proliferator-activated receptors (PPARs) and PPAR agonists: the 'future' in dermatology therapeutics?. , Arch. Dermatol. Res 307(9), 767-80.
- 22.Chen F, Cole P, When L. (1994) Estimates of trace element intakes in Chinese farmers. , J. Nutr; 124(2), 196-20.
- 23.Anasuya A, Bapurao S, Paranjape P K. (1996) Fluoride and silicon intake in normaland endemic fluorotic areas. , J. Trace. Elem. Med. Biol 10(3), 149-55.
- 24.Jugdaohsingh R, S H Anderson, K L Tucker. (2002) Dietary silicon intake and absorption. , Am. J. Clin. Nutr 75(5), 887-93.
- 25.K R Marin. (2007) The chemistry of silica and its potential health benefits. , J. Nutr. Health. Aging 11(2), 94-7.
- 26.R, Kossmann E, Barel A. (2007) Effect of oral intake of choline-stabilized orthosilicic acid on hair tensile strength and morphology in women with fine hair. , Arch. Dermatol. Res 299(10), 499-505.
- 27.D M Hexsel, Dal`Forno T, C L Hexsel. (2009) A validated photonumeric cellulite severity scale. , J. Eur. Acad. Dermatol. Venereol; 23(5), 523-8.
- 29.E O Diaz, Villar J, Immink M. (1989) Bioimpedance or antrhopometry. , Eur. J. Clin. Nutr 43(2), 129-37.
- 30.M Y Jaffrin, Morel H. (2008) Body fluid volumes measuremenys by impedance: A review of bioimpedance spectroscopy (BIS) andbioimpedance analysis (BIA) methods. , Med. Eng. Phys 30(10), 1257-69.
- 31.C N Gleichauf, D A Roe. (1989) The menstrual cycle's effect on the reliability of bioimpedance measurements for assessing body composition. , Am. J. Clin. Nutr 50(5), 903-7.
- 32.Seoane F, Abtahi S, Abtahi F. (2015) Mean expected error in prediction of total body water: a true accuracy comparison between bioimpedance spectroscopy and single frequency regression equations. , Biomed. Res. Int 656323.
- 33.Caetano L V N, Soares J L M, Bagatin E. (2015) Reliable assessment of forearm photoageing by high-frequency ultrasound: a cross-sectional study. , Int J Cosmet Sci 1-8.
- 34.Savoia A, Landi S, Vannini F. (2013) Low level laser therapy and vibration therapy for the treatment of localized adiposity fibrous cellulite. , Dermatol. Ther 3(1), 41-52.
- 35.Wanner M, Avram M. (2008) An evidence-based assessment of treatments for cellulite. , J. Drugs. Dermatol 7, 341-45.
- 36.Schonvvetter B, Soares J L M, Bagatin E. (2014) Longitudinal evaluation of manual lymphatic drainage for the treatment of gynoid lipodystrophy. , An. Bras. Dermatol 89(5), 712-8.
- 37.F M Sacks, G A Bray, V J Carey. (2009) Comparison of weight-loss diet with different compositions of fat, protein, and carbohydrates. , N. Engl. J. Med 360(9), 859-73.
- 38.B M Chimenti, Bruno M L M, Nakasato M. (2006) Estudo sobre adesão: fatores intervenientes na dieta hipocalórica de coronariopatas internados em um hospital público de São Paulo. Rev Bras Nutr Clin. 21(3), 204-10.
- 39.G P Kurita, Pimenta C A M. (2003) Adesão ao tratamento da dor crônica: estudo de variáveis demográficas, terapêuticas e psicossociais. , Arq. Neur. Psiquiatr 61(2), 416-25.
- 40.Scheinfeld N, M J Dahdah. (2007) Vitamins and minerals: their role in nails health and disease. , J. Drugs. Dermatol 6(8), 782-7.
- 41.M V Catani, Savini I, Rossi A. (2005) Biological role of vitamin C in keratinocytes. , Nutr. Rev 63(3), 81-90.
- 42.M C Cosgrove, O H Franco, S P Granger. (2007) Dietary nutrient intakes and skin-aging appearance among middleaged American. , Am. J. Clin. Nutr; 86, 1225-31.
- 44.S B Wu, Y T Wu, T P Wu. (2014) Role of AMPK-mediated adaptive responses in human cells with mitochondrial dysfunction to oxidative stress. , Biochim. Biophys. Acta; 1840(4), 1331-44.
- 45.Quan C, M K Chon, Perry D. (2015) Age-associated reduction of cell spreading induces mitochondrial DNA common deletion by oxidative stress in human skin dermal fibroblasts: implication for human skin connective tissue aging. , J. Biomed. Sci 22, 62.
- 46.Bagdon Allemann I, Baumann L. (2008) Antioxidants used in skin care formulations. , Skin. Therapy. Lett 13(7), 5-9.
- 47.Varani J, M K Dame, Rittie L. (2006) Decreased collagen production in chronologically aged skin: roles of age-dependent alteration in fibroblast function and defective mechanical stimulation. , Am. J. Pathol 168(6), 1861-8.
- 48.L B, A E Mitchell, Rucker R.B.etal.(2000) Accumulation of advanced glycation endproducts in aging male Fischer 344 rats duringlong-term feeding of various dietarycarbohydrates. , J. Nutr 130(5), 1247-55.
- 49.K A Harford, C M Reynolds, F C Mcgilicuddy.et al.(2011) Fats, inflammation and insulin resistance: insights to the role of macrophage and T-cell accumulation in adipose tissue. Proc. Nutr. Soc 70(4), 408-17.
- 50.R T Enos, J M Davis, K T Velazquez. (2013) Influence of dietary saturated fat contentonadiposity, macrophage behavior, inflammation, and metabolism: composition matters. , J. Lipid. Res; 54(1), 152-63.
- 51.R M Trueb. (2015) Effect of Ultraviolet Radiation. , Smoking and Nutrition on Hair. Curr. Probl. Dermatol; 47, 107-20.
- 52.Tsuda K, Nakatani T, Sugama J. (2010) Influence of the timing of switching a protein-free to a protein-containing diet on the wound healing process in a rat all-layer skin defect. , Int. Wound. J; 7(3), 135-46.
- 53.Czerwonka M, Szterk A. (2015) The effect of meat cuts and thermal processing on selected mineral concentration in beef from Holstein-Friesian bulls. , Meat. Sci; 105, 75-80.
- 54.SoongY Y, Lim J, Sun L. (2015) Effect of co-ingestion of amino acids with rice on glycaemic and insulinaemic response. , Br. J. Nutr 114(11), 1845-51.
- 55.Ros E, L C Tapsell, Sabaté J. (2010) Nuts and berries for heart health. , Curr. Atheroscler. Rep 12(6), 397-406.
- 56.L K Smalls, Hicks M, Passeretti D. (2006) Effect of weight loss on cellulite: gynoid lypodystrophy. Plast Reconstr Surg. 118, 510-6.
- 57.Institute of Medicineof the National Academies (2005) Dietary reference intakes (DRI): Energy, carbohydrates, fiber, fat, fatty acids, cholesterol, protein and amino acids , National Academies Press. , Washington, DC
- 58.Juanola-Falgarona M, Salas-Salvadó J, Ibarrola-Jurado N. (2014) Effect of the glycemic index of the diet on weight loss, modulation of satiety, inflammation, and other metabolic risk factors: a randomized controlled trial. , Am. J. Clin. Nutr 100(1), 27-35.
- 59.Bosy-Westphal A, Muller M J. (2015) Impact of carbohydrates on weight regain. , Curr. Opin. Clin. Nutr. Metab. Care 18(4), 389-94.
- 60.O M Clifton, Bastiaans K, J B Keogh. (2009) High protein diets decrease total and abdominal fat and improve CVD risk profile in overweight and obese men and women with elevated triacylglycerol. Nutr Metab Cardiovasc Dis. , Nutr. Metab. Cardiovasc. Dis; 19(8), 548-54.
- 61.T L Halton, F B Hu. (2004) High protein diets decrease total and abdominal fat and improve CVD risk profile in overweight and obese men and women with elevated triacylglycerol. , J. Am. Coll. Nutr 23(5), 373-85.
Cited by (6)
This article has been cited by 6 scholarly works according to:
Citing Articles:
RCMOS - Revista Científica Multidisciplinar O Saber (2025) Crossref
RCMOS - Revista Científica Multidisciplinar O Saber (2025) OpenAlex
B. Silva, Márcia Gorny, Igor Dantas de Medeiros - RCMOS - Revista Científica Multidisciplinar O Saber (2025) Semantic Scholar
The Egyptian Journal of Hospital Medicine (2022) OpenAlex
H. T. Mohamed, A. Nashed, Amir A. Gabr, Wafaa M. Kamal - The Egyptian Journal of Hospital Medicine (2022) Semantic Scholar
Saúde (Santa Maria) (2021) OpenAlex
