Adrenomedullin as a Protein with Multifunctional Behavior and Effects in Various Organs and Tissues
Abstract
In literature, it has been reported that adrenomedullin, which is generally thought to have vasodilator, natriuretic and diuretic effects, is synthesized in almost all body, especially CNS, vascular muscles and endothelium, heart, liver, lung, kidney, gastric mocosa, intestinal endothelium and various blood cells. It has been found that the possible effects of adrenomedullin can be demonstrated directly or indirectly by means of active mediators, neuropeptides, enzymes and hormones. It is also suggested that it regulates the endocrine system by affecting the hypothalamic-pituitary axis. It increases in heart failure, acute coronary syndromes, hypertensive conditions, cerebrovascular accessory, chronic renal failure and periodontitis and decreases in peptic ulcer and intestinal diseases. However, it is still not clear whether increase/decrease in adrenomedullin level is a cause of a disease or is a result of damage due to an illness. This peptide, which could be thought to multifunctional, should be considered as a molecule with genetic coding that may have different effects on different tissues and conditions. For all these reasons, we aimed to review the multifonctional behavior of adrenemedullin in the light of the current literature to pioneer new hypotheses and discuss possible mechanisms.
Author Contributions
Academic Editor: Akmal El-Mazny, Department of Obstetrics and Gynecology Cairo University, Egypt.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Fatih Ozcelik, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Adrenomedullin was first described by Japanese scientists in 1993. These researchers discovered a new peptide while searching for the effects of peptides on cAMP levels in rat thrombocytes. The peptide was named adrenomedullin (ADM) as it was extracted from adrenal medulla 1. Following researches have revealed that this peptide was not only found in adrenal medulla but it was also detected in heart, vessels, kidneys, lungs, gastrointestinal tract, central nervous system (CNS) and endocrine tissues 2. ADM was found to be present in circulation and various biological fluids and synthesized by different tissues. It is reported to function both as a generalized hormone and locally affecting autocrine or paracrine mediator 3, 4.
Altough initially thought a hypotensive and natriuretic peptide related to vascular structures, laterly it was found that adrenomedullin has various effetcs like growth, and differentiation on different tissues like renal, edndocrine, CNS and peripheral tissues 1, 2, 3, 4, 5. It exerts these effects either directly or indirectly via some active mediators, neuropeptides, enzymes and hormones 4, 6, 7, 8. Afterwards, it was found that it was elevated in heart failure, acute coronary syndrome, hipertensive situations, cerebrovascular events, chronical kidney failure, sepsis and periodontitis, while diminished in peptic ulcus and intestinal disease 1, 5, 9, 10, 11, 12, 13, 14, 15. Besides these, adrenomedullin has drawn attention as it stimulates the proinflammatory cytokine IL-6 and supresses cytokines like TNF- α for regulating inflammation, being a potent of inhibitor of apoptosis and stimulating angiogenesis in tumor cells 16, 17, 18, 19. However, there is still a lot of information gap on adrenomedullin. Unfortunately, evidences explaining these multifunctional effects of adrenomedullin have not yet reached. Therefore, there is a need for new hypotheses to discuss possible mechanisms that may explain these effects.
Structure and Synthesis
Adrenomedullin gene is located on chromosome 11 and contains 3 intron and 4 exon domains. Transcriptionally activated TATA, CAAT, GC boxes and, nuclear factor-interleukin 6 (NF-IL-6) and activator protein-2 (AP-2) domains are found at the 5’ end of this gene. The AP-2 region activates the protein kinase C (PKC)-cAMP cascade 20, whereas the NF-IL-6 region is the site where acute phase proteins, which are particularly effective in inflammation, bind. The binding of many cytokines involved in the inflammation to the NF-IL-6 region stimulates human ADM synthesis of 52 amino acids and subsequently accelerates blood flow in the area of inflammation 1, 21.
ADM is included in calcitonin/CGRP/amylin family as it has a slight resemblance in sequence to calcitonin gene-related peptide (CGRP) 1. mRNA responsible for ADM synthesis encodes information for the synthesis of a preprohormone known as preproadrenomedullin with 185-amino acid. Later, 21 amino acid signal peptide is cleaved from preproadrenomedullin (1-185) and the 164 amino acid peptide proadrenomedullin (proADM) (22-185) is formed 22, 23. Proadrenomedullin has three vasoactive peptides: ADM, proadrenomedullin N-terminal 20 peptide (PAMP) and adrenotensin. In addition, it has an inactive domain known as MR-proADM. Afterwards PAMP 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, mid-regional proadrenomedullin (MR-proADM) 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, ADM 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107, 108, 109, 110, 111, 112, 113, 114, 115, 116, 117, 118, 119, 120, 121, 122, 123, 124, 125, 126, 127, 128, 129, 130, 131, 132, 133, 134, 135, 136, 137, 138, 139, 140, 141, 142, 143, 144, 145, 146 and adrenotensin (153-185) are detached from proADM. ADM, detached from proADM precursor, is an immature and inactive molecule of 53 amino acids bound to glycine called “ADM-glycine”. Glycine is then removed by amidation and 52 amino acid active form is obtained. This active form is called “ADM-mature” 24, 25 (Figure 1).
Figure 1.Biosynthesis ADM. Adrenomedullin is inactive form when glycine is bound.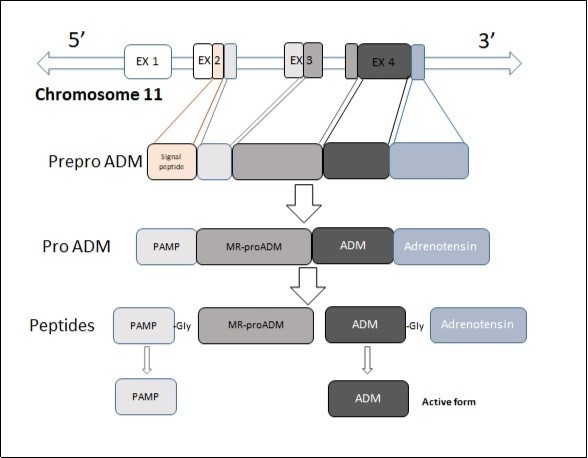
Production and Metabolism
Tissues with highest ADM mRNA levels are reported to be adrenal medulla, heart, lungs and kidneys 22. Besides these, it was found to be synthesized in gastrointestinal, endocrine and central nervous system, reproductive system cells, vascular smooth muscle and endothelial cells 26. It has also been detected in body fluids like blood, urine, saliva, cerebrospinal fluid, sweat, amniotic fluid and breast milk 3, 4, 26. As it is found in almost all tissues and body fluids, it brought out the thought that ADM has multiple biological functions. That is why there are many ongoing studies about ADM currently.
Production and metabolism of ADM is very fast with a half-life of 20 minutes 27. It is carried in circulation with a specific protein called adrenomedullin binding protein-1 (AMBP-1) 28. Another interesting feature of ADM is that it does not have circadian rhythm and plasma levels are not affected by age and gender 29. Plasma ADM levels also were found to be not affected by food or water intake and to be stable all day 30.
Although it is defended that ADM is not stored and secreted continuously 31, 32, there are several studies known to report that ADM is stored in tissues of the pancreas and adrenal medulla 33. The major form of ADM in circulation is immature or inactive ADM-glycine form 25. Metalloproteinases and aminopeptidases are shown to be effective in ADM catabolism 34.
ADM synthesis and release are under the control of many factors. Particularly cytokines (such as IL-1α, IL-1β, TNF-α and TNF-β), liposaccharides 35 and endotoxins 36 strongly stimulate the synthesis of ADM. This, in turn shows that ADM has a strong relationship with inflammation and sepsis. Steroids hormones like glucocorticoids, peptide hormones like thyroxine 37, mediators like AT II 38, endotelin-1 39 and bradykinin 40, atrial natriuretic peptide (ANP) 41 and arginine vasopressin (AVP) 42 stimulate ADM synthesis. Besides these, higher altitudes, hypoxia 43 and pregnancy 44 are known to elevate plasma ADM levels.
ADM Receptors and Signal Pathways
ADM is a member of calcitonin peptide family, which also includes calcitonin, calcitonin-gene related peptide (CGRP) and amylin 1. Its functions in the body are mediated by CGRP and adrenomedullin receptors. These receptors are receptors, which are called calcitonin receptor-like receptor (CLR), which are seven transmembrane folding and a kind of G protein 45. In order for CLR proteins to function, they need proteins (RAMPs) with three types that alter receptor activity. CLR/RAMP1 complex serves as CGRP receptor, while CLR/RAMP2 complex serves as ADM1 receptor and CLR/RAMP3 complex serves as ADM2 receptor 46, 47, 48 (Figure 2).
Figure 2.Signal pathways and effects of adrenomedullin.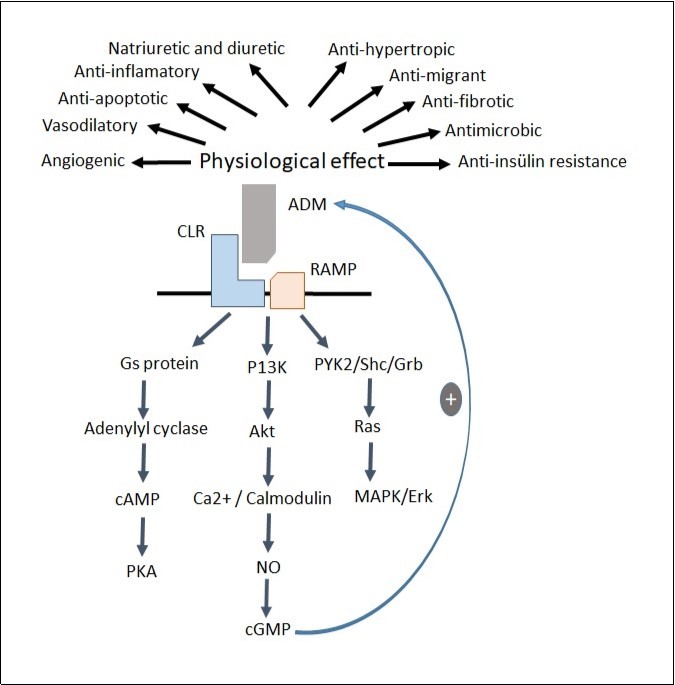
ADM performs its effect mostly by activation of protein kinase A (PKA) by increasing intracellular cAMP levels 49. ADM also increases intracellular Ca++ independent from cAMP and activates endothelial NO synthase (eNOS) which induces NO release and causes vasodilatation 50. It has also been shown to induce NO synthesis by inducible nitric oxide synthase (iNOS) in vascular smooth muscle cells 51. Vasodilatory effect of ADM is known to be caused by NO release 52. ADM also increases NO-activated guanosine 3′, 5′- cyclic monophosphate (cGMP) levels and causes protein kinase G (PKG) activation (NO/cGMP/PKG signal pathway) 53. Besides these pathways, it also uses different signal pathways like mitogen-activated protein kinase (MAPK) 54, K+-ATP channel activation 55, c-fos expression 56 and phosphatidylinositol-3-kinase/akt dependent pathway 57.
ADM Measurement
Reliability of ADM measurement is limited by factors like the molecule being unstable 30, having a short half-life 27 and binding to a specific protein in circulation. MR-proADM generated from prepro ADM is an inactive form, it is more stable and has a longer half-life when compared to ADM. ADM and MR-proADM are produced in equal amounts by posttranslational processes 30. Plasma MR-proADM concentrations are also known to reflect ADM concentrations directly and MR-proADM measurement has some advantages. One of these advantages is that the MR-proADM measurement has a higher diagnostic accuracy 30, 58.
In studies carried out in healthy individuals, plasma ADM reference values were detected with radioimmunoassay (RIA) method ranging from 2.1 ± 0.7 pmol/Lto 3.3 ± 0.39 pmol/L 59, 60. Comparison of different immunoassay methods revealed plasma ADM reference interval to be between 1-10 pmol/L 4. Hence, plasma MR-proADM mean value measured with immunoluminometric method in healthy individuals was reported as 0.33 ± 0.07 nmol/L 30. These values are 1000 times higher than mature ADM levels for which given reference interval is 2.7-10.1 pmol/L 30. That is why it became more possible to detect MR-proADM levels more in a more sensitive and accurate way.
Cardiovascular Effects
Adrenomedullin is synthesized and released in vascular smooth muscle cells 61 and endothelial cells 62. With its strong vasodilator feature, it reduces both systemic and peripheral vascular resistance. Thus, they decreases blood pressure for longer periods and increases blood flow 63. The vasodilatation effect of ADM can be expressed by activating the protein kinase-A cAMP (cAMP/PKA) 64 and NO/cGMP cascade 52 and ATP-sensitive potassium (K-ATP) channels 65. The effect of ADM on vascular smooth muscle cells was found to be bidirectional. It inhibits cell proliferation and migration in medium induced by platelet-derived growth factor (PDGF), but stimulates cell proliferation in medium not induced by PDGF 66.
Plasma ADM concentrations have been observed to increase as arterial stiffness and atherosclerosis increases, which is one of the most important cardiovascular risk factors and can be evaluated by indirect pulse wave velocity 67. Similarly, patients with chronic ischemic stroke, ADM levels were found to be increased in relation to the severity of atherosclerosis in the carotid artery 68. It has been found that ADM has a protective effect against atherosclerosis by anti-apoptotic feature in endothelial cells and anti-proliferative and antimigrate feature in vascular smooth muscle cells 66. In this protection, the anti-inflammatory effect of ADM was considered quite large. It has also been reported to provide the regeneration of the damaged endothelial layer by stimulating angiogenesis 69. In addition, ADM prevents cardiovascular damage by decreasing oxidative stress 70.
ADM was found to be increased in arterial hypertension and even correlated with the degree of hypertension 71. This elevation in ADM is thought to provide a compensatory mechanism by vasodilator, natriuretic and diuretic action 72. ADM was also found to increase cardiac output and heart rate 73, 74.
In the studies, ADM showed a (+) inotropic effect with a Ca++ and CGRP receptor- dependent pathway in the rat heart and (-) inotropic effect with a NO-dependent pathway in the rabbit heart 75, 76, 77. However, different results have been obtained about the effect on the contractility of the human heart. In one study, it was found that ADM in human myocardyocytes had an inotropic effect by inhibiting β-adrenergic stimulation 78. It has been also reported that cAMP/PKA pathway is stimulated in human atrium (compared to ventricles) and has (+) inotropic effect 79. ADM inhibits cardiac hypertrophy by activating cAMP/PKA 80. All these contradictory results show that there is no consensus on the cardiac effects of ADM.
Nishikimi et al. found that ADM levels in heart failure increases as correlating intensity of disease and ADM increases when left ventricular ejection fraction decreases 81. It has been suggested that ADM may be a prognostic indicator in ischemic left ventricular dysfunction 82. Since inflammation is important in the pathophysiology of heart failure 83 and ADM is a strong response to endogenous cytokines in inflammation, it has been suggested that ADM may be useful in determining the prognosis of heart failure 84. According to a recent study, high ADM levels at an early stage of myocardial infarction 85 confirms this prognostic significance. Nakamura et al. showed that ADM increases coronary blood flow by coronary vasodilatation. It is also stated that ADM restricts affected ischemic area by preventing apoptosis of myocytes, oxidative stress and providing cardiac remodeling 86. This information should be considered as evidence that the detection of ADM levels may be critical in the prognostic of coronary heart disease.
Renal Effects
ADM is synthesized by both glomerular and tubular cells in the kidney. The fact that the amount of adrenomedullin detected in urine is higher than that of blood suggests that kidneys may have a role in the clearance of ADM 59. It has been reported that ADM has a diuretic and natriuretic effect by increasing renal blood flow and glomerular filtration by means of vasodilatation resembling ANP and BNP with a NO-dependent mechanism in arterioles 72, 87. In summary, the peptide shows diuretic and natriuretic effect by increasing glomerular filtration and reducing tubular sodium reabsorption.
ADM has an important role in the mesangial cell physiology. ADM induced by proinflammatory cytokines such as IL-1β and TNF-α in mesangial cells shows antioxidant effect by decreasing both anti-inflammatory and free radical formation. ADM activates cAMP/PKA signaling pathway and decreases the generation of reactive oxygen metabolites in mesangial cells dose-dependent, and inhibits cell proliferation. It also suppresses mitogenesis by the MAPK-dependent route in vascular smooth muscle cells and mesangial cells. These effects show that ADM provides both immune and inflammatory protection by suppressing glomerular damage 88. The high level of ADM levels in patients with chronic glomerulonephritis also support this consensus 89.
ADM also plays an important role in the endocrine function of the kidney. However, there are various ideas about that effect on the renin-angiotensin-aldosterone system. ADM reduces arterial pressure while increasing cardiac output by functional antagonist action of angiotensin II in vascular and zona glomerulosa of adrenal gland. The attenuation of angiotensin-inducible aldosterone synthesis while increasing plasma renin activity is thought as evidence that ADM partially interacted with the renin-angiotensin-aldosterone system 90. In rats, there are studies that increase the renin release secondary to the hypotensive effect of ADM 91, but continuously reduces the renin activity of ADM administered externally 92. It was also suggested that plasma ADM levels were elevated in chronic renal failure 93 and correlated with plasma creatinine levels 71. In general, ADM has been widely investigated in the field of nephrology because it has vasodilator and hypotensive effect, antiproliferative effect in mesangial cells and two-way effect on renin release.
Pulmonary Effects
It has been suggested that lungs are the main tissue where ADM is cleansed from the body 26. It is claimed that ADM acts as a vasodilator and bronchodilator in the lungs and inhibits acetylcholine and histamine-induced bronchoconstriction 94. In particular, it has been reported that blood ADM levels increase in acute asthma exacerbations, and this has a bronchodilator effect 95. Pulmonary vasodilatation due to ADM in hypoxia has been reported to occur via prostaglandins rather than NO 94. In patients with pulmonary hypertension, blood ADM levels were found to be increased as protective mechanism 96. In another study conducted in rats with heart failure with pulmonary hypertension, ADM was found to decrease the right ventricular systolic pressure 10. ADM has also been reported to contribute to the anti-inflammatory process by dose-dependent inhibition of cytokine-induced neutrophil chemoattractant (CIMC) release from lipopolysaccharide-induced alveolar macrophages 97.
Effects of Central Nervous System
ADM receptors have been widely distributed throughout the central nervous system. ADM receptors have been detected in many areas such as cerebral cortex, cerebellum, pons, medulla oblongata, thalamus and hypothalamus 98. The fact that ADM levels in the cerebrospinal fluid are lower than the plasma levels indicate that the secretion of ADM from the cerebrospinal fluid occurs independently of plasma 44. It has been reported that ADM increases the blood flow by vasodilatation in the cerebral circulation and increases the production of cAMP/PKA pathway and NO in the vascular smooth muscle cell, especially in large-scale cerebral vessels, causing vasodilatation 64, 52.
In the case of hypoxic ischemia and hypoglycemia, it has been observed that ADM gene expression is increased in central cortex neurons, endothelium and perivascular glial cells 99. Cortisol, NO and ADM levels were higher in chronic schizophrenia than control group 100. Increased ADM mRNA expression was detected in ischemic cortex in rats with stroke formed by occlusion of the middle cerebral artery 101. ADM infusion after hypertrophic rupture of the middle cerebral artery has been shown to increase regional blood flow and collateral circulation, thus reducing ischemic brain damage 102. As a result, it is understood that ADM has important effects in post-ischemia reperfusion.
Effects on Hypothalamic–Pituitary–Adrenal Axis
ADM has been shown to inhibit ACTH secretion from rat anterior pituitary cells in a dose-dependent manner, reducing CRH-induced ACTH production and performing all of these with a mechanism independent of cAMP 103. ADM levels were found to be quite high and significantly decreased after surgical treatment in patients with Cushing's disease due to pituitary adenoma 104.
ADM has been reported to inhibit the salt appetite in rats by controlling oxytocin (OT) release in the hypothalamus. ADM has been shown to change water intake, salt appetite and food intake when given directly to the brain. It has also been reported that central ADM stimulates OT release by affecting CNS and inhibits more Na consumption 105, 106. The effect of ADM via a neurotransmitter OT suggests that many processes related to OT can be regulated via ADM. It was found that OT stimulates uterine muscle contraction, facilitates delivery and helps protect the brain from hypoxia, regulates the menstrual cycle and ejaculation of men, decreases the repetitive behaviors in diseases such as autism, prevents the proliferation of breast cancer and other tumor cells, stimulates angiogenesis while reducing inflammation, provides cardiac repair in ischemia-reperfusion injury and it also has apoptosis-inhibitory effects in the heart 107, 108, 109, 110, 111, 112. We believe that at least some of these effects of OT may be under the influence of ADM.
CNS-induced ADM is considered as a physiological regulator of thirst. It is suggested that brain-derived ADM increases vasopressin secretion and therefore acts as a physiological regulator of fluid homeostasis 113. ADM is synthesized and released in both adrenal cortex and adrenal medulla. ADM and its receptors have been found to be intense in the adrenal cortex, especially in the area of the zona glomerulose 114. ADM has been shown to inhibit aldosterone production but has no effect on plasma renin activity and plasma corticosterone (or K+) levels 115. However, in another study, an increase in plasma renin activity was observed after ADM infusion to sheeps and cortisol decreased. This study also showed a decrease in ACTH levels. The decrease in cortisol levels was thought to be due to a decrease in ACTH, not a direct effect on the adrenal cortex 116. In addition, it was observed that synthetic ADM application increased the adrenal blood flow in rats and this has led to the use of ADM for therapeutic purposes 117.
Reproductive Effects
ADM is found in the myometrium and endometrium layer of the ovary and uterus of women 118, 119. ADM levels were high in the follicular phase of the menstrual cycle and low in the luteal phase 120. It has been suggested that ADM produced by granulosa cells may play a role in the development of corpus luteum 121. ADM has been found to play an important role in the growth of placenta and fetus and prevention of uterine contraction by providing blood flow to uterus (blood supply to the placenta) and implantation of the embryo 122, 123, 124, 125, 126. ADM is thought to be present in the blood, placenta and amniotic fluid during normal pregnancy due to the important tasks it undertakes in all these stages 44, 127.
ADM was detected in testis, epididymis and prostate in male genital tract 128, 129, 130. The expression of ADM gene is shown in the sertoli and leydig cells in the testis 131, 132. It has been suggested that ADM affects sperm maturation and movement, inhibits contraction in prostate and stimulates prostatic blood flow 128, 133, 134, 135. ADM has also been reported to cause vasodilatation via NO/cGMP in cavernous vascular endothelial cells 57.
Effects on Immune System and Inflammation
ADM has complex effects on inflammation. It has been reported that plasma ADM levels are increased in the local and systemic inflammation and are synthesized in mucosal surfaces and contributes to anti-microbial protection 136. It controls the leukocyte migration and differentiation, while increasing the blood flow in the inflammation zone. It also reduces endothelial permeability and inhibits exudate formation 137. In addition, the fact that it may have local effects suggest that it can be used with topical drugs. Therefore, we consider that it can be used in the treatment of edema.
ADM has been found to play a regulatory and stabilizer role among proinflammatory and anti-inflammatory cytokines. Proinflammatory markers, TNF-α and IL-1β, have been seen as potent stimulators of ADM release 138, 22. ADM also stimulates proinflammatory cytokine release, such as IL-6 and IL-10 while IL-10 also shows anti-inflammatory effect by suppressing TNF-α and IL-1β release 16, 139. ADM is transported in the plasma by binding to the complement factor H, known as adrenomedullin binding protein-1 (AMBP-1). In this way, ADM plays an active role in the regulation of the complement system. It also increases the cleavage of C3b through factor I and it affects the complement regulator function of factor H 28. Since ADM levels were significantly elevated in sepsis and septic shock (especially in endotoxic shock), ADM was thought to be secreted as a preservative against sepsis 140, 141. As a result, ADM regulates the immune system by functioning through both cellular and secretory (cytokines and complement) system. We think that ADM has significant potential as an agent that can be used in the treatment of septic shock due to its anti-inflammatory effects.
Gastro-Intestinal Effects
Immunoreactive ADM has been detected in various tissues including plasma, as well as kidney, pancreas and intestine 142. It is reported that ADM may act as autocrine or paracrine way in these tissues where it is released as a vasoactive hormone in the circulation 71, 143. Subcutaneous ADM was found to have anti-ulcer effect in rats with reserpine-induced gastric mucosal damage. Probable cause of this effect is possibly associated with accelerative effect of ADM on blood flow in the gastric mucosa and partially anti-gastric secretory activity by increasing the release of NO 144. Rossowski et al. previously reported that ADM strongly inhibits gastric acid secretion 145. All these findings suggest that ADM may be an anti-ulcer agent.
ADM and PAMP are able to regulate many physiological and pathological conditions such as intestinal hormones. It has also been reported that it has a regulatory role in small intestine and colon peristaltism. It is also argued that ADM and PAMP regulate the intestinal mucosa and help the mucosal host defense system. In addition, it is also thought that ADM and PAMP deficiency may be effective in the development and progression of intestinal diseases with its effect on microbiota composition 146. However, the use of ADM as an antimicrobial agent requires more investigations that are precise.
Relationship Between Adrenomedullin, Thyroid Hormones and Obesity
In a study investigating the effect of thyroid hormones on ADM, ADM levels were higher in hyperthyroid rats and lower in hypothyroid rats compared to control group 8. Higher ADM and PAMP levels with higher thyroid hormones were detected in cultured vascular smooth muscle cells (VSMC) and thyroid storm due to Graves' disease 147, 37, 148, 149. Therefore, thyroid hormones are thought to regulate the production of ADM in vivo.
Due to significant effects on oxygen consumption and metabolic rate, thyroid hormones are essential for normal operation of almost all tissues. For this reason, the relationship between thyroid hormones and ADM is also a matter of curiosity. It has been reported that thyroid hormones increase the oxygen capacity of blood by increasing erythropoietin (EPO) production and increase tissue perfusion by vasodilation via accelerating ADM synthesis 150, 37. In addition, it was stated that direct ADM synthesis could be stimulated via thyroid hormones in case of hypoxia 151. Therefore, it is considered that important part of ADM's effect on energy metabolism is carried out by thyroid hormones.
Fat-rich diet increases ADM secretion 152, ADM stimulates lipolysis in brown adipose tissue 153 and adipose tissue adipocyte differentiation 154. Due to these effects, it is important to use it in combating obesity.
There are views that ADM is stored in pancreas secretory granules. ADM has been shown to inhibit insulin secretion by inhibiting β cells in the pancreas 33. It was also emphasized that high ADM levels in DM patients may be caused by hyperglycemia-induced ADM expression 155. On the other hand, it has been suggested that ADM levels are not affected by plasma glucose concentrations 156. Circulating ADM concentrations have been reported to be higher in pregnant women with gestational DM, and have been reported to suppress insulin secretion in pancreatic β-cells in vitro 157.
Obesity, resulting from the expansion of adipose tissues, is characterized by impaired insulin sensitivity of target organs and triggers type 2 diabetes, and also closely related to the proinflammatory (due to its effect on TNF-α) and immune systems 158, 159, 160, 161. We think that the relationship between ADM and proinflammatory and inflammatory regulation is the key mechanism in the relationship between obesity and ADM. However, the role of adaptive immune response in the inflammation of adipose tissue remains unclear.
In a study aiming to indicate the role of adipose tissue inflammation on the pathogenesis of insulin resistance (IR), a short-term high-fat diet was found to increase the number of CD4 (+) T cells in adipose tissues. This increase in the number of CD4 (+) T cells may contribute to the local inflammatory response in the early inflammation phase of adipose tissue and this may have an important role in insulin resistance. Major histocompatibility complex class II (MHCII) dependent antigen presentation for activation of CD4 (+) T cells has been suggested to induce early inflammation and IR in adipose tissue. MHCII inhibition is reported to decrease IR 162, 163.
In a study with transgenic mice, ADM 2 treatment was determined to fix the high fat diet induced early insulin resistance in the fat tissue. It is assumed that this is mainly achieved by inhibiting adipocyte MHCII-dependent antigen presentation and CD4 (+) T-cell activation 163. This finding has been found to be of interest as featured as a peptide with anti-insulin resistance effect.
In a study investigating ADM regulation in obesity and its localization in human adipose tissue, it is considered as a new member of the adipokine family due to being produced from stromal cells of human adipose tissue, including macrophages and synthesized in individuals with metabolic syndrome, especially in the omental region 161.
All of these indicate that ADM may have an important role in energy use and metabolism and obesity-induced insulin resistance as well as its relationship with thyroid hormones. However, since this issue needs clear evidence, there are many things to be investigated.
Conclusion
ADM has Multifunctional Behavior as Outlined Below;
a) have significant potential as an agent for the treatment of septic shock due to its anti-inflammatory effects, b) have a possibility of taking an active role in the breakdown of insulin resistance in obesity, c) may accelerate tissue perfusion by means of thyroid hormones, d) play a regulatory and stabilizing role among proinflammatory and anti-inflammatory cytokines, e) possibly contribute to the implantation of the embryo by providing the blood supply to the uterus and placenta, f) may have bronchodilator activity especially in acute asthma attacks, g) exhibits diuretic and natriuretic effect by increasing renal blood flow and glomerular filtration by vasodilatation with a NO-dependent mechanism in the arterioles, h) effect on plasma renin activity, i) have significant effects in post-ischemia reperfusion at CNS, j) alters thirst, salt appetite and food intake by stimulating OT synthesis k) lowering blood pressure while increasing blood flow, l) protects against atherosclerosis by having anti-apoptotic effect on endothelial cells and anti-proliferative/ anti-migrate effects on vascular smooth muscle cells in ischemic heart disease m) contributes to intestinal mucosal microbiota and provides antimicrobial host defense system, and finally n) stimulates angiogenesis and regenerates the damaged endothelial layer. This multifunctional feature of ADM brings it into the forefront for the purpose of treatment and leads to perform increasing amount of researches in recent years.
References
- 1.Kitamura K, Kangawa K, Kawamoto M, Ichiki Y, Nakamura S. (2012) Adrenomedullin: a novel hypotensive peptide isolated from human pheochromocytoma. , Biochem Biophys Res Commun 425(3), 548-555.
- 2.Kitamura K, Kangawa K, Kojima M, Ichiki Y, Matsuo H. (1994) Complete amino acid sequence of porcine adrenomedullin and cloning of cDNA encoding its precursor. , FEBS Lett 338(3), 306-310.
- 3.Kapas S, Martínez A, Cuttitta F, Hinson J P. (1998) Local production and action of adrenomedullin in the rat adrenal zona glomerulosa. , J Endocrinol 156(3), 477-484.
- 4.Hinson J P, Kapas S, Smith D M. (2000) Adrenomedullin, a Multifunctional Regulatory Peptide,Endocr Rev. 21(2), 138-67.
- 5.Yasue H, Yoshimura M, Sumida H, Kikuta K, Kugiyama K. (1994) Localization and mechanism of secretion of B-type natriuretic peptide in comparison with those of A-type natriuretic peptide in normal subjects and patients with heart failure. , Circulation 90(1), 195-203.
- 6.Daggubati S, Parks J R, Overton R M, Cintron G, Schocken D D. (1997) Adrenomedullin, endothelin, neuropeptide Y, atrial, brain, and C-natriuretic prohormone peptides compared as early heart failure indicators. Cardiovasc Res. 36(2), 246-255.
- 7.Yuksel S, Yurekli M. (2003) The effect of adrenomedullin (ADM) on tyrosine tydroxylase (TH) enzyme activity and blood pressure in cold exposed rats. , Endocr J 50(5), 553-559.
- 8.Murakami Y, Hattori Y, Taniyama M, Kitamura K, Kasai K. (1998) Adrenomedullin regulation by thyroid hormone in the rat. , Life Sci 63, 337-342.
- 9.Nagaya N, Nishikimi T, Horio T, Yoshihara F, Kanazawa A. (1999) Cardiovascular and renal effects of adrenomedullin in rats with heart failure. , Am J Physiol 276(1), 213-218.
- 10.Supeł K, Kacprzak M, Zielińska M. (2017) The prognostic value of MR-proadrenomedullin in patients with acute coronary syndrome complicated by cardiogenic shock. , Biomarkers 22(34), 296-303.
- 11.Kuwasako K, Kida O, Kitamura K, Kato J, Eto T. (1997) Plasma adrenomedullin in cerebrovascular disease: a possible indicator of endothelial injury. , Int Angiol 16(4), 272-279.
- 12.Bernal-Morell E, García-Villalba E, MDC Vera, Medina B, Martinez M. (2018) Usefulness of midregional pro-adrenomedullin as a marker of organ damage and predictor of mortality in patients with sepsis. , J Infect 76(3), 249-257.
- 13.Hussain Q A, McKay I J, Gonzales-Marin C, Allaker R P. (2016) Detection of adrenomedullin and nitric oxide in different forms of periodontal disease. , J Periodontal Res 51(1), 16-25.
- 14.Kaneko H, Mitsuma T, Nagai H, Mori S, Iyo T. (1998) Central action of adrenomedullin to prevent ethanol-induced gastric injury through vagal pathways in rats. , Am J Physiol. 274(6 Pt 2, 1783-1788.
- 15.Martínez-Herrero S, Martínez A. (2016) Adrenomedullin regulates intestinal physiology and pathophysiology. Domest Anim Endocrinol. 56 Suppl 66-83.
- 16.Wong L Y, Cheung B M, Li Y Y, Tang F. (2005) Adrenomedullin is both proinflammatory and antiinflammatory: its effects on gene expression and secretion of cytokines and macrophage migration inhibitory factor in NR8383 macrophage cell line. , Endocrinology 146(3), 1321-1327.
- 17.Yildirim N C, Yurekli M. (2010) The effect of adrenomedullin and cold stress on interleukin-6 levels in some rat tissues. Clin Exp Immunol. 161(1), 171-175.
- 18.Nikitenko L L, Fox S B, Kehoe S, MCP Rees, Bicknell R. (2006) Adrenomedullin and tumour angiogenesis. , Br J Cancer 94(1), 1-7.
- 19.Si H, Zhang Y, Song Y, Li L. (2018) Overexpression of adrenomedullin protects mesenchymal stem cells against hypoxia and serum deprivation-induced apoptosis via the Akt/GSK3β and Bcl-2 signaling pathways. , Int J Mol Med 41(6), 3342-3352.
- 20.Ishimitsu T, Kojima M, Kangawa K, Hino J, Matsuoka H. (1994) Genomic structure of human adrenomedullin gene. , Biochem Biophys Res Commun 203(1), 631-639.
- 21.Shoji H, Minamino N, Kangawa K, Matsuo H. (1995) Endotoxin markedly elevates plasma concentration and gene transcription of adrenomedullin in rat. , Biochem Biophys Res Commun 215(2), 531-537.
- 22.Kitamura K, Sakata J, Kangawa K, Kojima M, Matsuo M. (1993) Cloning and characterization of cDNA encoding a precursor for human adrenomedullin. , Biochem Biophys Res Commun,194(2): 720-725.
- 23.Sakata J, Shimokubo T, Kitamura K, Nakamura S, Kangawa K. (1993) Molecular cloning and biological activities of rat adrenomedullin, a hypotensive peptide. , Biochem Biophys Res Commun.195(2) 921-927.
- 24.Sugo S, Minamino N, Kangawa K, Miyamoto K, Kitamura K. (1994) Endothelial cells actively synthetize and secrete adrenomedullin. , Biochem Biophys Res Commun 201, 1160-6.
- 25.Kitamura K, Kato J, Kawamoto M. (1998) The intermediate form of glycine-extended adrenomedullin is the major circulating molecular form in human plasma. , Biochem Biophys Res Commun 244(2), 551-555.
- 26.Nishikimi T, Kitamura K, Saito Y, Shimada K, Ishimitsu T. (1994) Clinical studies on the sites of production and clearance of circulating adrenomedullin in human subjects. , Hypertension 24(5), 600-604.
- 27.Hirayama N, Kitamura K, Imamura T, Kato J, Koiwaya Y. (1999) Secretion and clearance of the mature form of adrenomedullin in humans. , Life Sci 64(26), 2505-2509.
- 28.Pio R, Martinez A, Unsworth E J, Kowalak J A, Bengoechea J A. (2001) Complement factor H is a serum-binding protein for adrenomedullin, and the resulting complex modulates the bioactivities of both partners. , J Biol Chem 276(15), 12292-12300.
- 29.Nishikimi T, Horio T, Kohmoto Y, Yoshihara F, Nagaya N. (2001) Molecular forms of plasma and urinary adrenomedullin in normal, essential hypertension and chronic renal failure. , J Hypertens 19(4), 765-773.
- 30.Morgenthaler N G, Struck J, Alonso C, Bergmann A. (2005) Measurement of midregional proadrenomedullin in plasma with an immunoluminometric assay.Clin Chem. 51(10), 1823-1829.
- 31.Takahashi K, Satoh F, Sone M, Totsune K, Arihara Z. (1998) Expression of adrenomedullin mRNA in adrenocortical tumors and secretion of adrenomedullin by cultured adrenocortical carcinoma cells,Peptides,19(10):. 1719-1724.
- 32.Isumi Y, Shoji H, Sugo S, Tochimoto T, Yoshioka M. (1998) Regulation of adrenomedullin production in rat endothelial cells. , Endocrinology 139(3), 838-846.
- 33.Martínez A, Weaver C, López J, Bhathena S J, Elsasser T H. (1996) Regulation of insulin secretion and blood glucose metabolism by adrenomedullin. , Endocrinology 137(6), 2626-2632.
- 34.Lewis L K, Smith M W, Brennan S O, Yandle T G, Richards A M. (1997) Degradation of human adrenomedullin (1–52) by plasma membrane enzymes and identification of metabolites. , Peptides 18(5), 733-739.
- 35.Sugo S, Minamino N, Shoji H, Kangawa K, Kitamura K. (1995) Interleukin-1, tumor necrosis factor and lipopolysaccharide additively stimulate production of adrenomedullin in vascular smooth muscle cells. , Biochem Biophys Res Commun,207(1): 25-32.
- 36.So S, Hattori Y, Kasai K, Shimoda S, Gross S S. (1996) Up-regulation of rat adrenomedullin gene expression by endotoxin: relation to nitric oxide synthesis. Life Sci. 58(19), 309-315.
- 37.Imai T, Hirata Y, Iwashina M, Marumo F. (1995) Hormonal regulation of rat adrenomedullin gene in vasculature. , Endocrinology 136(4), 1544-1548.
- 38.Tsuruda T, Kato J, Kitamura K, Kuwasako K, Imamura T. (1998) Adrenomedullin: a possible autocrine or paracrine inhibitor of hypertrophy of cardiomyocytes. , Hypertension 31(12), 505-510.
- 39.Mishima K, Kato J, Kuwasako K, Ito K, Imamura T. (2001) Effects of endothelin on adrenomedullin secretion and expression of adrenomedullin receptors in rat cardiomyocytes. , Biochem Biophys Res Commun 287(1), 264-269.
- 40.Marutsuka K, Hatakeyama K, Yamashita A, Sato Y, Sumiyoshi A. (2003) Adrenomedullin augments the release and production of tissue factor pathway inhibitor in human aortic endothelial cells. Cardiovasc Res. 57(1), 232-237.
- 41.Vesely D L, Blankenship M, Douglass M A, McCormick M T, Rodriguez-Paz G. (1996) Atrial natriuretic peptide increases adrenomedullin in the circulation of healthy humans. , Life Sci 59(3), 243-254.
- 42.Romppanen H, Marttila M, Magga J, Vuolteenaho O, Kinnunen P. (1997) Adrenomedullin Gene Expression in the Rat Heart is Stimulated by Acute Pressure Overload: Blunted Effect in Experimental Hypertension. , Endocrinology 138(6), 2636-2639.
- 43.Toepfer M, Hartmann G, Schlosshauer M, Hautmann H, Tschöp M. (1998) Adrenomedullin: a player at high altitude?. , Chest 113(5), 1428.
- 44.Nagata N, Kato J, Kitamura K, Kawamoto M, Tanaka N. (1998) Dissociation of adrenomedullin concentrations in plasma and cerebrospinal fluid in pregnant and non-pregnant women. , Eur J Endocrinol 139(6), 611-614.
- 45.Kuwasako K, Kitamura K, Nagata S, Hikosaka T, Takei Y. (2011) Shared and separate functions of the RAMP-based adrenomedullin receptors.Peptides. 32(7), 1540-1550.
- 46.Morfis M, Christopoulos A, Sexton P M. (2003) RAMPs: 5 years on, where to now? Trends Pharmacol Sci. 24(11), 596-601.
- 47.Poyner D R, Sexton P M, Marshall I, Smith D M, Quirion R. (2002) International Union of Pharmacology. XXXII. The mammalian calcitonin gene-related peptides, adrenomedullin, amylin, and calcitonin receptors. Pharmacological reviews. 54(2), 233-246.
- 48.Febres A, Vanegas O, Giammarresi M, Gomes C, Díaz E. (2018) Is the activity of CGRP and Adrenomedullin regulated by RAMP (-2) and (-3) in Trypanosomatidae? An in-silico approach. , Infect Genet Evol 61, 197-206.
- 49.Coppock H A, Owji A A, Bloom S R, Smith D M. (1996) A rat skeletal muscle cell line (L6) expresses specific adrenomedullin binding sites but activates adenylate cyclase via calcitonin gene-related peptide receptors. , Biochem J. 318 (Pt 1, 241-245.
- 50.Shimekake Y, Nagata K, Ohta S, Kambayashi Y, Teraoka H. (1995) Adrenomedullin stimulates two signal transduction pathways, cAMP accumulation and Ca2+ mobilization, in bovine aortic endothelial cells. , J Biol Chem 270(9), 4412-4417.
- 51.Ikeda U, Kanbe T, Shimada K. (1996) Adrenomedullin increases inducible nitric oxide synthase in rat vascular smooth muscle cells stimulated with interleukin-1. , Hypertension 27(6), 1240-1244.
- 52.Hirata Y, Hayakawa H, Suzuki Y, Suzuki E, Ikenouchi H. (1995) Mechanisms of adrenomedullin-induced vasodilation in the rat kidney. , Hypertension. 25(4 Pt 2, 790-795.
- 53.R De Matteo, May C N. (2003) Direct coronary vasodilator action of adrenomedullin is mediated by nitric oxide.Br. , J Pharmacol 140(8), 1414-1420.
- 54.Kim W, Moon S O, Sung M J, Kim S H, Lee S. (2003) Angiogenic role of adrenomedullin through activation of Akt, mitogen-activated protein kinase, and focal adhesion kinase in endothelial cells. , FASEB J 17(13), 1937-1939.
- 55.Terata K, Miura H, Liu Y, Loberiza F, Gutterman D D. (2000) Human coronary arteriolar dilation to adrenomedullin: role of nitric oxide and K+ channels. , J Physiol Heart Circ Physiol 279(6), 2620-2626.
- 56.Sato A, Autelitano D J. (1995) Adrenomedullin induces expression of c-fos and AP-1 activity in rat vascular smooth muscle cells and cardiomyocytes. , Biochem Biophys Res Commun 217(1), 211-216.
- 57.Nishimatsu H, Hirata Y, Hayakawa H, Nagata D, Satonaka H. (2001) Effects of intracavernous administration of adrenomedullin on erectile function in rats. , Peptides 22(11), 1913-1928.
- 58.Funke-Kaiser A, Havulinna A S, Zeller T, Appelbaum S, Jousilahti P. (2014) Predictive value of midregional pro-adrenomedullin compared to natriuretic peptides for incident cardiovascular disease and heart failure in the population-based FINRISK. , cohort.Ann Med 46(3), 155-162.
- 59.Sato K, Hirata Y, Imai T, Iwashina M, Marumo F. (1995) Characterization of immunoreactive adrenomedullin in human plasma and urine. , Life Sci 57(2), 189-194.
- 60.Kitamura K, Ichiki Y, Tanaka M, Kawamoto M, Emura J et al. (1994) Immunoreactive adrenomedullin in human plasma. , FEBS Lett 341(23), 288-290.
- 61.Uemura T, Kato J, Kuwasako K, Kitamura K, Kangawa K. (2002) Aldosterone augments adrenomedullin production without stimulating pro-adrenomedullin N-terminal 20 peptide secretion in vascular smooth muscle cells. , J Hypertens 20(6), 1209-1214.
- 62.Ishihara T, Kato J, Kitamura K, Katoh F, Fujimoto S. (1997) Production of adrenomedullin in human vascular endothelial cells. , Life Sci 60(20), 1763-1769.
- 63.Nakamura M, Yoshida H, Makita S, Arakawa N, Niinuma H. (1997) Potent and long-lasting vasodilatory effects of adrenomedullin in humans. Comparisons between normal subjects and patients with chronic heart failure. , Circulation 95(5), 1214-1221.
- 64.Ishizaka Y, Ishizaka Y, Tanaka M, Kitamura K, Kangawa K. (1994) Adrenomedullin stimulates cyclic AMP formation in rat vascular smooth muscle cells. , Biochem Biophys Res Commun,200(1): 642-646.
- 65.Sakai K, Saito K, Ishizuka N. (1998) Adrenomedullin synergistically interacts with endogenous vasodilators in rats: a possible role of K-ATP channels. , Eur J Pharmacol 359(2), 151-159.
- 66.Kano H, Kohno M, Yasunari K, Yokokawa K, Horio T. (1996) Adrenomedullin as a novel antiproliferative factor of vascular smooth muscle cells. , J Hypertens 14(2), 209-213.
- 67.Kita T, Kitamura K, Hashida S, Morishita K, Eto T. (2003) Plasma adrenomedullin is closely correlated with pulse wave velocity in middle-aged and elderly patients. Hypertens Res. 26(11), 887-893.
- 68.Shinomiya K, Ohmori K, Ohyama H, Hosomi N, Takahashi T. (2001) Association of plasma adrenomedullin with carotid atherosclerosis in chronic ischemic stroke. , Peptides 22, 1873-1880.
- 69.Iimuro S, Shindo T, Moriyama N, Amaki T, Niu P. (2004) Angiogenic effects of adrenomedullin in ischemia and tumor growth. Circ Res. 95(4), 415-423.
- 70.Tsuruda T, Kato J, Kuwasako K, Kitamura K. (2019) Adrenomedullin: Continuing to explore cardioprotection. , Peptides 111, 47-54.
- 71.Ishimitsu T, Nishikimi T, Saito Y, Kitamura K, Eto T. (1994) Plasma levels of adrenomedullin, a newly identified hypotensive peptide, in patients with hypertension and renal failure. , J Clin Invest 94(5), 2158-2161.
- 72.Kato J, Kitamura K, Matsui E, Tanaka M, Ishizaka Y. (1999) Plasma adrenomedullin and natriuretic peptides in patients with essential or malignant hypertension. Hypertens Res. 22(1), 61-65.
- 73.Del Bene R, Lazzeri C, Barletta G, Vecchiarino S, Guerra C T. (2000) Effects of low-dose adrenomedullin on cardiac function and systemic haemodynamics in man. , Clin Physiol 20(6), 457-465.
- 74.Nagaya N, Satoh T, Nishikimi T, Uematsu M, Furuichi S. (2000) Hemodynamic, renal, and hormonal effects of adrenomedullin infusion in patients with congestive heart failure. , Circulation 101(5), 498-503.
- 75.Szokodi I, Kinnunen P, Tavi P, Weckström M, Tóth M. (1998) Evidence for cAMP-independent mechanisms mediating the effects of adrenomedullin, a new inotropic peptide. , Circulation 97(11), 1062-1070.
- 76.Szokodi I, Kinnunen P, Ruskoaho H. (1996) Inotropic effect of adrenomedullin in the isolated perfused rat heart.Acta Physiol Scand. 156(2), 151-152.
- 77.Ikenouchi H, Kangawa K, Matsuo H, Hirata Y. (1997) Negative inotropic effect of adrenomedullin in isolated adult rabbit cardiac ventricular myocytes. , Circulation 95(9), 2318-2324.
- 78.Mukherjee R, Multani M M, Sample J A, Dowdy K B, Zellner J L. (2002) Effects of adrenomedullin on human myocyte contractile function and beta-adrenergic response. , J Cardiovasc Pharmacol Ther 7(4), 235-240.
- 79.Bisping E, Tenderich G, Barckhausen P, Stumme B, Bruns S. (2007) Atrial myocardium is the predominant inotropic target of adrenomedullin in the human heart.Am J Physiol Heart Circ Physiol. 293(5), 3001-3007.
- 80.Nishikimi T, Horio T, Yoshihara F, Nagaya N, Matsuo H. (1998) Effect of adrenomedullin on cAMP and cGMP levels in rat cardiac myocytes and nonmyocytes. , Eur J Pharmacol 353(2), 337-344.
- 81.Nishikimi T, Saito Y, Kitamura K, Ishimitsu T, Eto T. (1995) Increased plasma levels of adrenomedullin in patients with heart failure. , J Am Coll Cardiol 26(6), 1424-1431.
- 82.Richards A M, Doughty R, Nicholls M G, MacMahon S, Sharpe N. (2001) Australia-New Zealand Heart Failure Group. Plasma N-terminal pro-brain natriuretic peptide and adrenomedullin: prognostic utility and prediction of benefit from carvedilol in chronic ischemic left ventricular dysfunction. Australia-New Zealand Heart Failure Group. , J Am Coll Cardiol 37(7), 1781-1787.
- 83.Mann D L. (2015) Innate immunity and the failing heart: cytokine hypothesis revisited. , Circ Res 116, 1254-1268.
- 84.S von Haehling, Filippatos G S, Papassotiriou J, Cicoira M, Jankowska E A. (2010) Mid-regional pro-adrenomedullin as a novel predictor of mortality in patients with chronic heart failure. , Eur J Heart Fail 12(5), 484-491.
- 85.Nagaya N, Nishikimi T, Uematsu M, Yoshitomi Y, Miyao Y. (1999) Plasma adrenomedullin as an indicator of prognosis after acute myocardial infarction. , Heart 81(5), 483-487.
- 86.Nakamura R, Kato J, Kitamura K, Onitsuka H, Imamura T. (2002) Beneficial effects of adrenomedullin on left ventricular remodeling after myocardial infarction in rats. Cardiovasc Res. 56(3), 373-380.
- 87.McGregor D O, Troughton R W, Frampton C, Lynn K L, Yandle T. (2001) Hypotensive and natriuretic actions of adrenomedullin in subjects with chronic renal impairment. , Hypertension 37(5), 1279-1284.
- 88.Chini E N, Chini C C, Bolliger C, Jougasaki M, Grande J P. (1997) Cytoprotective effects of adrenomedullin in glomerular cell injury: central role of cAMP signaling pathway.Kidney Int. 52(4), 917-925.
- 89.Kubo A, Kurioka H, Minamino N, Nishitani Y, Sato H. (1998) Plasma and urinary levels of adrenomedullin in chronic glomerulonephritis patients with proteinuria. , Nephron 80(2), 227-230.
- 90.Charles C J, Lainchbury J G, Nicholls M H, Rademaker M T, Richards A M. (2003) Adrenomedullin and the renin-angiotensin-aldosterone system. Regul Pept. 112(1), 41-49.
- 91.Jensen B L, Kramer B K, Kurtz A. (1997) Adrenomedullin stimulates renin release and renin mRNA in mouse juxtaglomerular granular cells. , Hypertension 29(5), 1148-1155.
- 92.Jougasaki M, Burnett Jr JC. (2000) Adrenomedullin as a regulator peptide. , Nephrol Dial Transplant 15(3), 293-295.
- 93.Toepfer M, Lang S M, Hartmann G, Sitter T, Schiffl H. (1998) Plasma adrenomedullin levels body fluid status, and end-stage renal failure. , Nephron 80(2), 232-233.
- 94.Yang B C, Lippton H, Gumusel B, Hyman A, Mehta J L. (1996) Adrenomedullin dilates rat pulmonary artery rings during hypoxia: role of nitric oxide and vasodilator prostaglandins. , J Cardiovasc Pharmacol 28, 458-462.
- 95.Kohno M, Hanehira T, Hirata K, Kawaguchi T, Okishio K. (1996) An accelerated increase of plasma adrenomedullin in acute asthma. , Metabolism 45, 1323-1325.
- 96.Kakishita M, Nishikimi T, Okano Y, Satoh T, Kyotani S. (1999) Increased plasma levels of adrenomedullin in patients with pulmonary hypertension.Clin Sci(Lond). 96(1), 33-39.
- 97.Kamoi H, Kanazawa H, Hirata K, Kurihara N, Yano Y. (1995) Adrenomedullin inhibits the secretion of cytokine-induced neutrophil chemoattractant, a member of the interleukin-8 family, from rat alveolar macrophages. , Biochem Biophys Res Commun 211(3), 1031-1035.
- 98.Sone M, Takahashi K, Satoh F, Murakami O, Totsune K. (1997) Specific adrenomedullinbinding sites in the human brain.Peptides. 18(8), 1125-1129.
- 99.Serrano J, Alonso D, Fernández A P, Encinas J M, López J C. (2002) Adrenomedullin in the central nervous system.Microsc Res Tech. 57(2), 76-90.
- 100.Yilmaz N, Herken H, Cicek H K, Celik A, Yürekli M. (2007) Increased levels of nitric oxide, cortisol and adrenomedullin in patients with chronic schizophrenia. Med Princ Pract. 16(2), 137-141.
- 101.Wang X, Yue T L, Barone F C, White R F, Clark R K. (1995) Discovery of adrenomedullin in rat ischemic cortex and evidence for its role in exacerbating focal brain ischemic damage. Proc Natl Acad Sci USA 92, 11480-11484.
- 102.Doğan A, Suzuki Y, Koketsu N, Osuka K, Saıto K. (1997) Intravenous infusion of adrenomedullin and increase in regional cerebral blood flow and prevention of ischemic brain injury after middle cerebral artery occlusion in rats. , J Cereb Blood Flow Metab 17(1), 19-25.
- 103.Samson W K, Murphy T, Schell D A. (1995) A novel vasoactive peptide, adrenomedullin, inhibits pituitary adrenocorticotropin release.Endocrinology. 136(5), 2349-2352.
- 104.Letizia C, R Di Iorio, G De Toma, Marinoni E, Cerci S. (2000) Circulating adrenomedullin is increased in patients with corticotropin-dependent Cushing's syndrome due to pituitary adenoma. , Metabolism 49(6), 760-763.
- 105.White M M, Samson W K. (2009) A possible relationship between brain-derived adrenomedullin and oxytocin in the regulation of sodium balance. , J Endocrinol 203(2), 253-262.
- 106.Murphy T C, Samson W K. (1995) The novel vasoactive hormone, adrenomedullin, inhibits water drinking in the rat. , Endocrinology 136(6), 2459-2463.
- 107.Salonia A, Nappi R E, Pontillo M, Daverio R, Smeraldi A. (2005) Menstrual cycle-related changes in plasma oxytocin are relevant to normal sexual function in healthy women. , Horm Behav 47(2), 164-9.
- 108.Khazipov R, Tyzio R, Ben-Ari Y. (2008) Effects of oxytocin on GABA signalling in the foetal brain during delivery. Prog Brain Res. 170, 243-257.
- 109.Burbach J P, Young L J, Russell J. (2006) Oxytocin: synthesis, secretion, and reproductive functions. Knobil and Neill’s Physiology of Reproduction In: Neill JD, editor , New York: 3055-3127.
- 110.Modahl C, Green L, Fein D, Morris M, Waterhouse L. (1998) Plasma oxytocin levels in autistic children. , Biol Psychiatry 43(4), 270-277.
- 111.Oyama T, Nagai T, Wada H, Naito A T, Matsuura K. (2007) Cardiac side population cells have a potential to migrate and differentiate into cardiomyocytes in vitro and in vivo. , J Cell Biol 176(3), 329-341.
- 112.Gutkowska J, Jankowski M, Antunes-Rodrigues J. (2014) The role of oxytocin in cardiovascular regulation. , Braz J Med Biol Res 47(3), 206-214.
- 113.Taylor M M, Baker J R, Samson W K. (2005) Brain-derived adrenomedullin controls blood volume through the regulation of arginine vasopressin production and release.Am. , J Physiol Regul Integr Comp Physiol 288(5), 1203-1210.
- 115.Yamaguchi T, Baba K, Doi Y, Yano K, Kitamura K. (1996) Inhibition of aldosterone production by adrenomedullin, a hypotensive peptide, in the rat.Hypertension. 28(2), 308-314.
- 116.Parkes D G, May C N. (1995) ACTH-suppressive and vasodilator actions of adrenomedullin in conscious sheep. , J Neuroendocrinol 7(12), 923-929.
- 117.He H, Bessho H, Fujisawa Y, Horiuchi K. (1995) Tomohiro A et al. , Eur J Pharmacol 273(3), 209-214.
- 118.Moriyama T, Otani T, Maruo T. (2000) Expression of adrenomedullin by human granulosa lutein cells and its effect on progesterone production.Eur. , J Endocrinol 142(6), 671-676.
- 119.Laoag-Fernandez J B, Otani T, Maruo T. (2000) Adrenomedullin expression in the human endometrium. , Endocrine 12(1), 15-19.
- 120.Marinoni E, R Di Iorio, Letizia C, Lucchini C, Alò P. (2000) Changes in plasma adrenomedullin levels during the menstrual cycle. Regul Pept. 87(1), 15-18.
- 121.Abe K, Minegishi T, Ibuki Y, Kojima M, Kangawa K. (2000) Expression of adrenomedullin in the human corpus luteum. , Fertil Steril 74(1), 141-145.
- 122.Makino I, Shibata K, Makino Y, Kangawa K, Kawarabayashi T. (1999) Adrenomedullin attenuates the hypertension in hypertensive pregnant rats induced by N(G)-nitro-L-arginine methyl ester. , Eur J Pharmacol 371(2), 159-167.
- 123.Zhang X, Green K E, Yallampalli C, Dong Y L. (2005) Adrenomedullin enhances invasion by trophoblast cell lines. , Biol Reprod 73(4), 619-626.
- 124.Wilson C, LLl Nikitenko, Sargent I L, Rees M C. (2004) Adrenomedullin: Multiple functions in human pregnancy. , Angiogenesis 7(3), 203-212.
- 125.Witlin A G, Li Z Y, Wimalawansa S J, Grady J J, Grafe M R. (2002) Placental and fetal growth and development in late rat gestation is dependent on adrenomedullin. , Biol Reprod 67(3), 1025-1031.
- 126.Yanagita T, Yamamoto R, Sugano T, Kobayashi H, Uezono Y. (2000) Adrenomedullin inhibits spontaneous and bradykinininduced but not oxytocin- or prostaglandin F2a-incuded periodic contraction of rat uterus. , Brit J Pharmacol 130(8), 1727-1730.
- 127.Kanenishi K, Kuwabara H, Ueno M, Sato C, Sakamoto H. (2002) Change of adrenomedullin concentrations in plasma and amniotic fluid, and human placental adrenomedullin expression with advancing gestation. , Placenta 22, 244-50.
- 128.Marinoni E, R Di Iorio, Villaccio B, Vellucci O, T Di Netta. (2005) Adrenomedullin in human male reproductive system. , Eur J Obstet Gynecol Reprod Biol 122(2), 195-198.
- 129.Asada Y, Hara S, Marutsuka K, Kitamura K, Tsuji T. (1999) Novel distribution of adrenomedullin-immunoreactive cells in human tissues. , Histochem Cell Biol 112(3), 185-191.
- 130.Jiménez N, Calvo A, Martínez A, Rosell D, Cuttitta F. (1999) Expression of adrenomedullin and proadrenomedullin N-terminal 20 peptide in human and rat prostate. , J Histochem Cytochem 47(9), 1167-1178.
- 131.Chan Y F, Tang F, O W S. (2008) Adrenomedullin in the rat testis. II: Its production, actions on inhibin secretion, regulation by follicle-stimulating hormone, and its interaction with endothelin 1 in the Sertoli cell. , Biol Reprod 78(4), 780-785.
- 132.Chan Y F, O W S, Tang F. (2008) Adrenomedullin in the rat testis. I: Its production, actions on testosterone secretion, regulation by human chorionic gonadotropin, and its interaction with endothelin 1 in the Leydig cell. , Biol Reprod 78, 773-779.
- 133.Hwang I S, Autelitano D J, Wong P Y, Leung G P, Tang F. (2003) Coexpression of adrenomedullin and adrenomedullin receptors in rat epididymis: Distinct physiological actions on anion transport. , Biol Reprod 68, 2005-2012.
- 134.Ventura S, Lau W A, Buljubasich S, Pennefather J N. (2000) Calcitonin gene-related peptide (CGRP) inhibits contractions of the prostatic stroma of the rat but not the guinea-pig. Regul Pept. 91, 63-73.
- 135.Shibata Y, Kashiwagi B, Arai S, Magari T, Suzuki K. (2006) Participation of adrenomedullin and its relation with vascular endothelial growth factor in androgen regulation of prostatic blood flow in vivo. , Urology 68, 1127-1131.
- 136.Allaker R P, Zihni C, Kapas S. (1999) An investigation into the antimicrobial effects of adrenomedullin on members of the skin, oral, respiratory tract and gut microflora. , FEMS Immunol Med Microbiol 23(4), 289-293.
- 137. (2002) Hippenstiel S,Witzenrath M,Schmeck B,Hocke A,Krisp Met al. Adrenomedullin reduces endothelial hyperpermeability.Circ Res.91(7) 618-625.
- 138.LYF Wong, BMY Cheung, CYY Li, Lan H Y, Tang F. (2005) Adrenomedullin modulates MIF secretion and cytokine responses in rat macrophages stimulated with lipopolysaccharide. , Endocrinology 146(3), 1321-1327.
- 139.Yang S, Zhou M, Chaudry I H, Wang P. (2002) Novel approach to prevent the transition from the hyperdynamic phase to the hypodynamic phase of sepsis: Role of adrenomedullin and adrenomedullin binding protein-1. Ann Surg;. 236(5), 625-633.
- 140.Hirata Y, Mitaka C, Sato K, Nagura T, Tsunoda Y. (1996) Increased circulating adrenomedullin, a novel vasodilatory peptide, in sepsis. , J Clin Endocrinol Metab 81(4), 1449-1453.
- 141.Shindo T, Kurihara H, Kurihara Y, Morita H, Yazaki Y. (1998) Upregulation of endothelin-1 and adrenomedullin gene expression in the mouse endotoxin shock model. , J Cardiovasc Pharmacol 31, 541-544.
- 142.Ichiki Y, Kitamura K, Kangawa K, Kawamoto M, Matsuo H. (1994) Distribution and characterization of immunoreactive adrenomedullin in human tissue and plasma. , FEBS Lett 338(1), 6-10.
- 143.Eguchi S, Hirata Y, Kano H, Sato K, Watanabe Y. (1994) Specific receptors for adrenomedullin in cultured rat vascular smooth muscle cells. , FEBS Lett 340(3), 226-230.
- 144.Salomone S, Caruso A, Cutuli V M, Mangano N G, Prato A. (2003) Effects of adrenomedullin on the contraction of gastric arteries during reserpine-induced gastric ulcer. , Peptides 24(1), 117-122.
- 145.Rossowski W J, Jiang N Y, Coy D H. (1997) Adrenomedullin, amylin, calcitonin gene-related peptide and their fragments are potent inhibitors of gastric acid secretion in rats. , Eur J Pharmacol 336(1), 51-63.
- 146.Martínez-Herrero S, Martínez A. (2016) Adrenomedullin regulates intestinal physiology and pathophysiology. Domest Anim Endocrinol. 56 Suppl 66-83.
- 147.Minamino N, Shoji H, Sugo S, Kangawa K, Matsuo H. (1995) Adrenocortical steroids, thyroid hormones and retinoic acid augment the production of adrenomedullin in vascular smooth muscle cells. , Biochem Biophys Res Commun 211(2), 686-693.
- 148.Taniyama M, Kitamura K, Ban Y, Sugita E, Ito K. (1997) Elevation of circulating proadrenomedullin-N terminal 20-peptide in thyrotoxicosis. , Clin Endocrinol (Oxf) 46(3), 271-274.
- 149.Taniyama M, Kitamura K, Ban Y, Eto T, Katagiri T. (1996) Elevated plasma adrenomedullin level in hyperthyroidism. , Eur J Clin Invest 26(6), 454-456.
- 150.Ma Y, Freitag P, Zhou J, Brüne B, Frede S. (2004) Thyroid hormone induces erythropoietin gene expression through augmented accumulation of hypoxia-inducible factor-1. , Am J Physiol Regul Integr Comp Physiol 287(3), 600-607.
- 151.Cormier-Regard S, Nguyen S V, Claycomb W C. (1998) Adrenomedullin gene expression is developmentally regulated and induced by hypoxia in rat ventricular cardiac myocytes. , J Biol Chem 273, 17787-17792.
- 152.Nambu T, Arai H, Komatsu Y, Yasoda A, Moriyama K. (2005) Expression of the adrenomedullin gene in adipose tissue. , Regul Pept 132, 17-22.
- 153.Go A D, Chow K H, Hwang I S, Tang F. (2007) Adrenomedullin and its receptor components in adipose tissues: Differences between White and brown fats and the effects of adrenergic stimulation. , Peptides 28, 920-927.
- 154.Li Y, Jiang C, Wang X, Zhang Y, Shibahara S. (2007) Adrenomedullin is a novel adipokine: Adrenomedullin in adipocytes and adipose tissues. , Peptides 28, 1128-1143.
- 155.Hayashi M, Shimosawa T, Fujita T. (1999) Hyperglycemia increases vascular adrenomedullinexpression. , Biochem Biophys Res 258(2), 453-456.
- 156.Nakamura T, Honda K, Ishikawa S, Kitamura K, Eto T. (1998) Plasma adrenomedullin levels in patients with noninsulin dependent diabetes mellitus: close relationships with diabetic complications. , Endocr J 45(2), 241-246.
- 157.Dong Y, Banadakoppa M, Chauhan M, Balakrishnan M, Belfort M. (2019) Circulating Adrenomedullin Is Elevated. in Gestational Diabetes and Its Role in Impaired Insulin Production by β-Cells.J Clin Endocrinol Metab 104(3), 697-706.
- 158.Tewari N, Awad S, Macdonald I A, Lobo D N. (2015) Obesity-related insulin resistance: implications for the surgical patient. , Int J Obes 39, 1575-1588.
- 159.Grimble R F. (2002) Inflammatory status and insulin resistance. , Curr Opin Clin Nutr Metab Care 5, 551-559.
- 160.De Taeye BM, Novitskaya T, McGuinness O P, Gleaves L, Medda M. (2007) Macrophage TNF-alpha contributes to insulin resistance and hepatic steatosis in diet-induced obesity. , Am J Physiol Endocrinol Metab 293, 713-725.
- 161.Paulmyer-Lacroix O, Desbriere R, Poggi M, Achard V, Alessi M C. (2006) Expression of adrenomedullin in adipose tissue of lean and obese women. , Eur J Endocrinol 155(1), 177-185.
Cited by (8)
This article has been cited by 8 scholarly works according to:
Citing Articles:
(2025) OpenAlex
(2025) OpenAlex
Stem Cell Research & Therapy (2025) OpenAlex
Stem Cell Research & Therapy (2025) Crossref
A. Bayer, Natalia Pinzón, Axel You, Cinthia Bergman, Nadine Dragin et al. - Stem cell research & therapeutics (2025) Semantic Scholar
International Journal of Molecular Sciences (2023) OpenAlex
International Journal of Molecular Sciences (2023) Crossref
Benjamin Wende, Anna-Sophia Lieselott Beyer, Niklas Ruhnke, Daniel Kaemmerer, J. Sänger et al. - International Journal of Molecular Sciences (2023) Semantic Scholar
Journal of Biochemical and Molecular Toxicology (2021) OpenAlex
Journal of Biochemical and Molecular Toxicology (2021) Crossref
