Abstract
The indirect misuse of animals usually correlated to catastrophic consequences like the one we are facing now, novel coronavirus disease-19. However, the source of infection is not fully confirmed yet. But the finger of blame points to bats and pangolins. Hence, understanding and raising awareness about wild animals and the consequences of their misuse are a must to avoid future pandemics. Herein, we threw light on the most animals involved in the current epidemic as well as possible animals in the shadow. Random serological investigations of samples from animals of different species (including asymptomatic animals) are required to understand the prevalence and severity of the infection, the extent of the transmission, and the monitoring of the situation over time. Moreover, alleviating fear associated with the possibility of human infection from their pets, we highlight that there are no reports that confirm this hypothesis; however, the opposite has been reported. Similarly, there is no proof that pigs can become infected amplify or spread novel coronavirus.
Author Contributions
Academic Editor: Sasho Stoleski, Institute of Occupational Health of R. Macedonia, WHO CC and Ga2len CC, Macedonia.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Shilpi Gupta, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Coronaviruses (CoVs) family consist of four genera, α‐ CoVs, β ‐CoVs, γ ‐CoVs, and δ ‐CoVs. There are seven human coronaviruses (HCoVs) that cause respiratory diseases. Four of them associated with mild cold-like signs including human coronavirus 229E (HCoV‐229E) 1 and human coronavirus NL63 (HCoV‐NL63) in α‐ CoVs genera 2, as well as β ‐CoVs genera, includes two human coronavirus OC43 (HCoV‐OC43) 3 and human coronavirus HKUI (HCoV‐HKU) 4. Three of the seven HCoVs are highly pathogenic, β ‐CoVs that have arisen from the zoonotic origin and infected humans such as middle east respiratory syndrome (MER-CoV), severe acute respiratory syndrome coronavirus -1 and 2 (SARS‐CoV‐1 and SARS‐CoV‐2) with high transmission and mortality rates. Cross-species viral transmission from humans to other animal species and vice-versa requires direct contact between the animal and infected person. Moreover, natural viral spillovers and jumping between species are initiated by high mutation rates to allow the viral escape from the new species‐specific immune defences. Fortunately, SARS‐CoV‐2 sequences available in the database do not show a high level of mutations to date which may be related to the non-structural protein 14 (NSP14) proofreading. Herein, we discuss the veterinary perspective, measurements, and significant role of animals spreading the virus that cause COVID-19.
Coronavirus’s Interspecies Transmission Caused by a High Recombination Rate
Coronaviruses undergo discontinuous transcription leading to a high recombination rate, but the error rate of RNA replication is reduced by proofreading enzyme ExoN (NSP14) 5. Coronaviruses are not only known for spillover from the animal to the human interface but also animal-to-animal transmission in domestic animals with animal ancestors in different species. For instance, canine coronavirus-II (CCoV-II) is the prominent ancestor for swine transmissible gastroenteritis virus 6. In contrast, canine coronavirus primarily originates from the bovine coronavirus, which is the common ancestor of the human HCoV-OC43 coronavirus. As bovine coronavirus and human HCoV-OC43 are closely related. Hence, there is a high possibility that one virus originated in one species and then jumped to another. They further crossed species such as alpaca, wild ruminants, canine, and swine. While feline coronavirus-I (FCoV-I) and canine coronavirus-I (CCoV-I) share one common ancestor. In other words, there may have been recombination between CCoV-I with an unknown coronavirus resulting in CCoV-II. In turn, CCoV-II underwent recombination with FCoV-I in an unknown intermediate host leading to feline coronavirus-II (FCoV-II). The most recent swine acute diarrhoea syndrome coronavirus SAD-CoV HKU2 is considered a spillover from bats to swine 7. Bats and humans (HCoV-NL63) have very similar genetic sequences to those of the porcine epidemic diarrhoea virus, suggesting it as a common evolutionary precursor8. Therefore, the surveillance of coronaviruses in wildlife certainly needed interdisciplinary research studies to better understand and predict potential spillover, identify the intermediate host for novel coronavirus, and limit further human pandemics (Figure 1).
Figure 1.Corona virus's cross-species. SARS-CoV-1 transmitted and adapted to a wild animal, palm civet, which used to be sold for the food in Chinese Wet Markets. MERS-CoV transmitted from bat to camel then from camel to human. HCoV-OC43 and bovine CoV are closely similar ancestry. Bovine CoV has also spread to many different animals, like alpaca, wild ruminants, canine, and wine. While Feline coronavirus I and canine coronavirus I have a similar ancestor, canine coronavirus I underwent recombination with an unknown coronavirus that give rise to canine coronavirus II. Then canine coronavirus II with Feline coronavirus I underwent recombination (in an unknown intermediate host) resulting in feline coronavirus II. Canine COV II transmitted to pigs, give rise transmissible gastroenteritis virus.
The Global Outbreak of COVID-19
A few months ago, the COVID-19 pandemic had escalated to become a tremendous challenge for more than 213 countries and territories and caused the pandemic world-over. It has taken a massive toll on the existing healthcare systems, creating fear, anxiety, and a threat to human life as well as the animal 9. As of August 20, 2020, the world battles with ≥ 22,593,620 COVID-19 confirmed cases and ⁓ 791,200 deaths, with the United States leading to ≥5,701,162 incidents and ⁓ 176,342 fatalities 10. The COVID-19 pandemic has unleashed severe global consequences in all aspects of life and society. Coronaviruses can infect the human respiratory tract system as well as able to cause a variety of human and animal diseases, including intestinal and liver diseases 11, 12. The novel virus has serious ramifications not only on COVID-19 symptoms but also on various other human diseases, including cancer, cardiovascular and kidney diseases, and all continents’ economies.
Newly emerged COVID-19 disease is caused by an infection of an aggressive strain of SARS-CoV-2. SARS-CoV-2 is the 7th member of the coronavirus family known to infect humans and contains a positive-sense, single-strand RNA (+ssRNA) genome of ⁓30 kilobases 13. Interestingly, SARS-CoV-2 have ⁓79% sequence homology with SARS-CoV-1 and ⁓50% homology with MER-CoV 14, 15. Different structural and sequence analyses of receptor binding motif (RBM) between SARS-CoV-1 and SARS-CoV-2 demonstrated that novel coronavirus has also utilized angiotensin-converting enzyme-2 (ACE2) receptor. Strong interactions between virus spike protein RBD (receptor-binding domain) and its host ACE2 receptor control both the person-to-person and cross-species spread of the virus. The virus infection and initial attachment are mainly determined by the atomic-interaction between host ACE2 and viral RBD 16, 17.
Expected COVID-19 Intermediate Host
Currently, there is no evidence in which animal species other than bats and pangolins play a critical role in spreading, transmitting, or being infected with SARS-CoV-218. Bats considered the common ancestral reservoir for coronaviruses based on bat’s natural susceptibility to a variety of CoVs. For example, bats and rats have been reported to be the natural host of HCoVs (HCoV-NL63 and HCoV-229E), that cause common cold in humans. Up to date, there are hundreds of novel coronaviruses that have been isolated and identified in bats 19. For these 200 bat viruses to jump and adapt to infect humans, an intermediate host may be needed. The previous two reported human coronaviruses suggest that zoonotic origins such as SARS-CoV-1 and MERS with civet cat and camel were intermediate hosts.
Hence there is a probability that the recombination between pangolin coronavirus and RaTG13 (bats' virus) happened and was the origin of SARS-CoV-2 based on genetic similarities RBD and Furin like cleavage insertion 8. However, the RaTG13 lineage is insufficient to confirm it as an intermediate host 20. There is no evidence to prove the intermediate host, zoonotic source, and factors that drive inter-species transmission. Recent veterinary coronaviruses' studies conducted in 2017 and 2018 in bats living close to other domestic and wild animals highlighted the potential risk of a spillover to humans 21, 22.
Origin and Spread of SARS-CoV-2
Coronaviruses (CoVs) are highly pathogenic zoonotic viruses that can infect humans emerge through cross-host transmission from animals and are first recognized in the mid-1960s 23. Since the start of the twenty-first century, three human coronaviruses (MERS, SARS-CoV-1, and SARS-CoV-2) belonging to the β-coronavirus genus could cross the species barrier to cause human respiratory diseases 15, 24, 25, 26. A bat-borne virus, SARS-CoV-1 first emerged as a new coronavirus in humans in 2002 in Guangdong, China, a causative pathogen of SARS illness and transmitted to more than 30 countries 8,422 confirmed cases and 774 deaths worldwide. The next zoonotic virus outbreak was MERS-CoV initially emerged from bats and transmitted in dromedary camels, and humans act as a transient/terminal host 27, 28. The MERS-CoV outbreak was first originated in 2012 in the Arabian Peninsula, Saudi Arabia, with ⁓2,494 people infected and more than 900 deaths recorded in about 25 countries 29.
The current and third outbreak of pneumonia cases of unexplained novel coronavirus was recorded in the wholesale seafood market of Wuhan, Hubei China, on December 31, 2019 30. This outbreak became an epidemic in China, and later rapidly became pandemic across the world with devastating effects on global public health. Though the first patient with unknown pneumonia was identified on November 17, 2019, in Hubei Province, China, and based on genomic information and subsequent investigations, the virus was possibly started spreading between human-to-human in late November or early December 11. Still, Chinese public health communities have not quickly informed to the World Health Organization (WHO) about the outbreak. Since then, the community spread of the virus has already started in China. A massive number of cases from China have been sporadic imported to several neighbouring countries as well as to other continents of the world.
However, there is still some doubt that novel coronavirus has emerged from animals or during SARS-CoV-like coronavirus laboratory experimental manipulation in Wuhan, China. The novel hypervirulent strain of human coronavirus; SARS-CoV-2 was isolated and sequenced from Seafood Market bronchoalveolar lavage fluid samples, and confirmed to be the causative agent of COVID-19 13, 16, 17. The genome sequences analysis revealed that SARS-CoV-2 has a high nucleotide sequence identity (with 88-96% identity) with two bat-derived SARS-CoV-1 (bat-SL-CoVZC45 and bat-SL-CoVZXC21). However, SARS-CoV-2 showed more top nucleotide sequences of homology with SARS-CoV-1 than MERS-CoV 14, 17. Both SARS-CoV-2 and SARS-CoV-1 have an animal origin, probably related to bat or Malayan pangolin, but the bat most likely serves as a reservoir host for both viruses 8, 17, 31, 32, 33, 34.
The virus can be transmitted to crowed areas from an infected person through inhalation of small respiratory droplets or fomites 25. Recent reports showed that novel coronavirus could be transmitted from animal-to-humans, human-to-human, or human-to-animal 11, 35. However, the method of the virus transmission from human-to-animal or from animal-to-human has not yet been clearly understood. Indirect transmission of SARS-CoV-2 as fomites has also been reported by touching an infected surface or object and then touching the nose or mouth of a healthy person. Typically, like other respiratory viruses, the infection is considered to be more contagious when patients are symptomatic; however, it can also be transmitted with the close contact of asymptomatic patients and before the onset of symptoms 36, 37. The infected droplets of the virus can transfer up to 1-2 meters, and these droplets have been found viable on different surfaces for several hours 38, 39. The virus infection can be asymptomatic, mild-moderately symptomatic (cold, fever, coughing and acute pneumonia resulting in respiratory failure, lung injury) and causing havoc to be elderly and immune-compromised people with a severe type of respiratory problems including pneumonia, breathing difficulty, acute respiratory distress syndrome (ARDS) and multi-organ failure 40. The disease remains a significant threat to those people with existing co-morbidities such as hypertension, obesity, diabetes, cardiovascular diseases, chronic lung/kidney diseases, and malignancy. The COVID-19 patient’s current diagnosis primarily depends on quantitative RT-PCR to detect nucleic acid of the virus. Computed tomography (C.T.) has also significantly been helpful in most symptomatic patients, which showed bilateral involvement in imaging. Recently, ELISA-based techniques and serological diagnosis have also been recommended to detect the virus's immunologic reaction.
Animals as a Source of Novel Coronavirus Infection
For many decades, animals have been speculated to be primary reservoirs of various emerging and re-emerging zoonotic viruses, including SARS-CoV-1 and SARS-CoV-2, which cause severe human diseases. Several animal and human coronaviruses have been identified in both avian and variety of mammal hosts, including a bat, dogs, mouse, camels, pig, and palm civets, and caused mild-moderate animal and human diseases 41. More than 200 new coronaviruses have been recognized in bats, and ⁓35% of the bat-originated viral genome sequenced 42. The first recognized SARS-CoV-2 positive patients were from the Wholesale Seafood Market in Wuhan, China, where the illegal selling of various kinds of live-wild and domesticated animals including bats, pangolins, snakes etc. are occurred 25, 43. This indicates that bats may be considered as a natural reservoir of SARS-CoV-2, and the spread of virus probably transmits in another intermediate animal, possibly pangolins and on its way to humans or from lab animals (mice or guinea pigs) to humans 44 (Figure 2). However, animals as a zoonotic source of infection of SARS-CoV-2 has a high probability; the exact role of animals in the emergence of SARS-CoV-2 is not clearly identified yet. The animal-to-human transmission and cross-species transmission of novel coronavirus led to public-health emergency and significant tolls globally. Thus, an urgent need for a comprehensive understanding of the mechanism of emergence of different pathogenic strains of novel coronaviruses and how the virus crossed species boundaries to infect humans and other animals, which can efficiently help in the prevention and control of future emerging and re-emerging zoonotic virus infections.
Figure 2.The possible mode of transmission of SARS-CoV-2. Bats can act as primary reservoirs for novel coronavirus; SARS-CoV-2. SARS-CoV-2 may cross the species barrier into pangolins, in wild and other live-animals in seafood/wet market. Consumption of infected animals as food items are the major source of infection transmission in humans, and human-to-human transmission through droplets/fomites, which results in community transmission as well as human-to pet animal spread. Dotted arrows showing the possibility of virus spread from bat to animals or humans whereas the solid black arrows indicate the confirmed transmission of virus.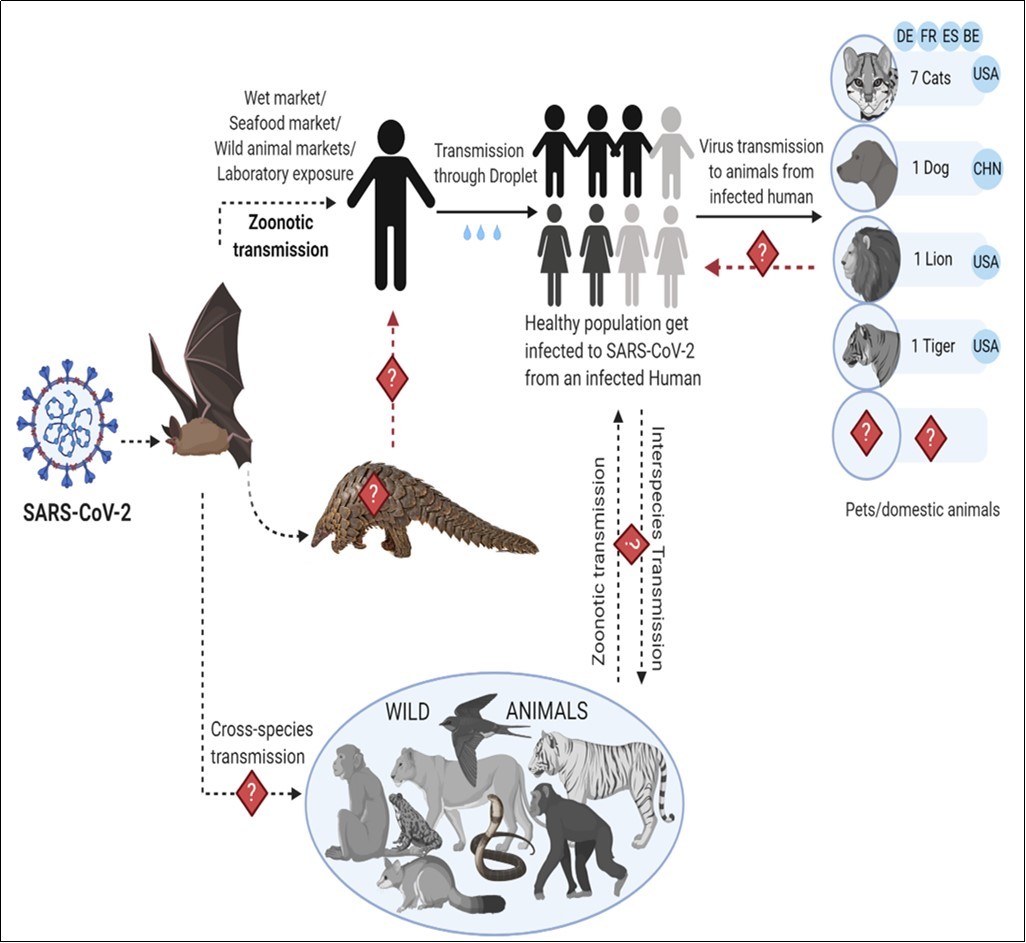
There are limited research and data available about the risk of the animal's roles. Given it's critical that we are still learning about this virus with a limited report about the probability of cat and dog being infected from the human population. Additional research investigations are highly needed to understand the animals’ superstability and pathogenesis by the virus, and their role in the spread of COVID-19. On the veterinary side, many questions require scientific answers like the original animal reservoir, the intermediate host, the mechanism of transmission, the risk of meat processing, and the dynamic of pet animal infection and transmission. WHO has reported that about 61 percent of pandemic viruses have been transmitted initially from animals through zoonotic transmission 45. Taken into consideration that the combination of environmental pressures, climate changes, and modern human activities could result in a disruption in wildlife and the appearance of pandemic diseases. There are still little known about the determinant at the wild animal-human interface threat. Collaboration between healthcare workers, researchers, scientists, and veterinarians is critical as a one health strategy. Establishment of one approach between the human public health and animal health are needed to identify the host and molecular determinant of pandemic zoonotic viruses' transmission at animal-human interference. This can be achieved by previous animal epidemics control, surveillance of wild animals as a reservoir and source of new emerging pathogens, development of veterinary laboratory support for diagnostic inspection and characterization of new pathogens, including coronaviruses and influenza viruses.
Swine as a Vector for CoVs
Several coronaviruses can infect pigs like the α ‐CoVs porcine transmissible gastroenteritis virus (TEGv) 46, porcine epidemic diarrhoea virus (PEDV) 47, porcine respiratory coronavirus (PRCV) 48, and the newly emerged swine acute diarrhoea syndrome coronavirus (SADS‐CoV) 7, 49, which is closely related to bats viruses exactly like SARS‐CoV-1 and MERS‐CoV. Moreover, swine are also susceptible to porcine hemagglutinating encephalomyelitis virus (PHEV) that related to β ‐CoV 50 and porcine delta coronavirus (PDCoV) in δ ‐CoV 51. Although swine and human have their own range of coronaviruses without overlapping, it is really interesting that most of the coronaviruses in humans cause respiratory infection, while in pigs, it is mainly enteric diseases with one single coronavirus associated with respiratory infections (PRCV).
A few research studies have reported the susceptibility of pigs to SARS‐CoV-1 or MERS‐CoV to determine if swine play a role as a source of infection to humans. The RNA of SARS‐CoV-1 was identified in one pig, and two pigs were SARS‐CoV-1 antibody-positive out of 24252. Another group conducted an experimental study by infecting six-week-old pigs and chickens with SARS‐CoV-1 by different routes of injections, including intranasal, intravenous, oral, and ocular. There were no clinical signs or lesions shown with unsuccessful viral isolation. However, they detected the viral RNA from blood drawn from both pigs and chickens. Two pigs showed neutralizing antibodies specific for the virus. The authors reported that neither chickens nor pigs play a role in virus multiplication53. Recently, a research study investigated the potential susceptibility and transmission of different animal species like pigs, llamas, sheep, and horses to MERS‐CoV. The animals were experimentally infected intranasally, followed by a collection of nasal and rectal swabs collection over time. They detected the MERS‐CoV, RNA, and antigen in the nasal swabs collected from llamas and pigs with a conclusion that MERS‐CoV could be circulated in animals other than camels, like llamas and pigs 54.
COVID-19 and Pigs
Based on the few studies, pigs could be a potential risk for SARS‐CoV-1 spread along with the eliciting of neutralizing antibodies in responses to the infection and the evidence that swine can amplify MERS‐CoV. So far, there is no report about the role of wild pigs as a vector for coronaviruses' transmission and cross-species, and this could be the missing link. To date, there is no proof that pigs can become infected with, can amplify, or spread SARS‐CoV‐2.
It has been known that SARS‐CoV‐2 utilizes the ACE2 receptor isolated from four different animal species to be used in-vitro, including porcine ACE2. Since SARS‐CoV‐2 can enter the cell in-vitro, this can suggest the porcine susceptibility to SARS‐CoV‐2 infection. It is possible that SARS‐CoV‐2 could potentially spillover in other species, such as pigs, which should be closely monitored. Based on the previous detection of SARS‐CoV-1 RNA in pigs, this would suggest that pigs may carry SARS-CoV-2 RNA. However, it is probably unusual that SARS-CoV-2 would be amplified in pigs. Swine farmers, veterinarians, swine caretakers must be free of COVID‐19 disease before being allowed to enter pig farms to minimize the risk of broad range spillover. Close monitoring to wild boar and surveillances is needed to confirm or deny the role of wild boar in the viral spread.
SARS-CoV-2 Transmission to Pet Animals from Infected Personnel
As per the database of animal investigations around the world, animals from different species were reported to get infections from humans and exhibit similar respiratory symptoms 55. To date, there are seven cats from the USA, France, Belgium, Spain, Germany, and one dog from China. In the majority of cases, the pets' owners were infected with SARS-CoV-2 and are assumed to have most probably transmitted infection to their pets. Moreover, SARS-CoV-2 was reported in zoo animals one lion and one tiger from the USA 56, 57 (Figure 2).
Although, there is no data to indicate the route of transmission of the virus in pets. These preliminary reports created fear among the community that pets may get infected from humans and may spread the virus to healthy people or animals. Fortunately, there are no reports about the animal to animal transmission nor human yet. Notably, all SARS-CoV-2 positive pets, the majority of cats are SARS-CoV-2 positive thus, future rapid investigation for SARS-CoV-2 in cats and other pet animals could help to understand the origin and transmission of virus which will facilitate for the development of effective antiviral medications and immunologically safe vaccines for COVID-19 patients.
Possible Experimental Use of Animals to Test the Vaccine for COVID-19
Since animals were able to exhibit similar symptoms to humans and triggered an immune response, they are included in the preclinical trials of novel drugs and vaccines as well as investigating the fundamental mechanism of the disease 58. Animal testing is a critical phase to determine the safety and efficacy of developing a new drug or vaccine. However, no published data regarding preclinical animal models for COVID-19 yet, some trials have been conducted on similar conditions as SARS, and MERS assumed as a good starting point. In 2005, Hamster has been demonstrated to be superior to the mouse as an animal model for SARS-CoV1 59. Also, Ferret was tested and validated as an animal model of SARS-CoV 60.
However, mice are known to be one of the best animal models for testing and validating the cytotoxicity of many drugs. The enormous physiological difference between humans and mice makes it difficult to be included in vaccines or drug experimentation in the past. Due to the genetic engineering technique, the mice can be modified to exhibit similar symptoms to humans through the insertion of ACE2 receptors and the deletion of the native one 44. Similarly, to optimize the pharmacokinetic modelling of antiviral drugs for COVID-19 is one of the important points that should be optimized as well. For instance, carboxylesterase 1c (Ces1c) gene which is responsible for esterase enzyme production, secreted in mice significantly higher than humans and hence affected the drug stability, has been modified genetically for better prediction of the pharmacokinetic response for some drugs as remdesivir (broad-spectrum antiviral medication) 61, 62. Nonhuman primates (NHPs) could be a good model for coronavirus as it develops similar symptoms to human 63.
Recently, a German group in Friedrich-Loeffler-Institute conducted experimental infection studies using pigs, chicken, fruit bats, and ferret to check their susceptibility to SARS CoV2. They inoculated the animals intranasally with SARS-CoV-2 in order to mimic the natural infection route in humans. Their early results showed that fruits bats and ferrets are susceptible to SARS-CoV2 infection, while pigs and chickens are not susceptible (unpublished data). Based on their preliminary study, there are many missing factors like the dose they used, the viral titre, and the time point of sampling before concluding that they do not pose a potential risk to human health. On the other side, Scientists at the Harbin Vet Research Institute reported that SARS-CoV-2 poorly replicated in dogs, pigs, chickens, and ducks but efficiently in ferrets and cats64. Further basic and experimental research and surveillance are needed to confirm whether pigs or wild boar are considered a vector for SARS-CoV2.
Surveillance of New Emerging and Re-Emerging Viruses in Wild Animals
Many research studies on the influenza virus's evolution, transmission, and pathogenesis in domestic and wild animals provided critical knowledge about the aetiology of viral zoonoses and the link between the health of animals, humans, and the ecosystem. Although. Coronaviruses have a wide variety of animal species; little is known about coronaviruses' surveillances. Veterinary epidemiologists have applied a variety of successful surveillance methods for better knowledge about the viral spread and quick diagnostic measures for reducing prevalence by eliminating different infectious diseases in animal populations65. The Centre for Disease Control and Prevention (CDC) has recently published interim guidance for veterinarians and their staff. They may be dealing with animal medical care during the COVID-19 pandemic with recommendations on using infection control considerations. Random serological investigations of samples from known animal numbers and different species, including asymptomatic animals, are required to understand the prevalence of the infection, the extent of the transmission, and to monitor the situation over time. The strategy of prevention and control of zoonotic transmissible diseases is the principal goal of the Veterinary Services of OIE Member Country. In many countries, veterinary institutes and veterinary diagnostic laboratories support the national health care system by performing thousands of tests that have proven their experience in the required technical and scientific abilities to the high request for diagnostic testing of human samples for SARS-CoV-2 during the Covid-19 crisis 30.
Summary
Several Animals interaction with humans has been noted as a primarily risk-factor for variety of viral diseases. However, the spillover from the animal-to-human leads to the emergence of new viruses, but the link between spillover risk and specific interactions is not fully known. Although the COVID-19 patient zero has not been identified, the initial cases were linked to the Chinese wet market. So, SARS-CoV-2 may originated and spread from the local seafood market of Wuhan, China where bats, raccoon dogs, snakes, palm civets, and other wild animals are traded. The vial zoonotic sources are not known yet, however, genome sequences analysis indicated a strong (88-96% identity) similarity with two bat-derived SARS-CoV-1 (bat-SL-CoVZC45 and bat-SL-CoVZXC21) suggesting that bats could be the reason for cross-species transmission and high infection rate with the aid of unknown intermediate host.
Therefore, continued investigations and the surveillance of coronaviruses in animals are critically needed for better understanding and prediction of the potential spillover. Further, identification of an intermediate host for SARS-CoV-2 will certainly help to elucidate the evolutionary pathway of novel coronaviruses and therefore, support limiting a future pandemics.
To date, there is no specific drug or vaccine has been developed or approved against the novel coronavirus. Thus, the best approaches to fight and limit COVID-19 with respect to better immunity and use of the masks, are to prevent the spread of infection by limiting the direct contact with animals.
Future Perspectives
Throughout the ages, wild animals served as a reservoir of many zoonotic parasites, viruses, and their consumption provides perfect conditions for the emergence of novel viruses. For a broader scope and a little bit away from coronavirus; Armadillos, for example, has served as a reservoir for Mycobacterium leprae, a causative agent of leprosy, "a chronic disease characterized by lesions of the skin and peripheral nerve damage" 66.
Similarly, consuming wild frogs is associated with human sparganosis 25. Although pigs are consumable, Wild boar consider a host of zoonotic pathogens like Toxoplasma gondii, Diplostomidae, Trematoda, and Hepatitis E 67, 68, 69, 70. It is worth mentioning that the hazard index of wild animals' liver was significantly higher than those of farm animals 71. Moreover, it has been demonstrated that Enterococci isolated from wild birds showed high virulence compared to that isolated from human and food animals 72. Also, Toxoplasma infection occurrence has been exhibited widely among wild birds 73. Interestingly, Muslim culture prompt not to eat carnivores either wild (lion, tigers, etc.), domesticated animals (dogs, cats, hamster, etc.), birds have talon or swine flesh 74. Similarly, the Jewish dietary laws (Karshrut) straiten the allowed animals as a food source to those who have cloven hooves and chewing the cud but not predatory animals. Moreover, they argue not to consume rodents, reptiles, and amphibians (http://www.jewfaq.org/kashrut.htm).
The most important lesson we had after the COVID-19 crisis is that the inappropriate use of animals can deteriorate our life. They were created to make our life better, some of them can be an acceptable and safe source of food, some as companion animals and safeguard, and some are an adequate model for testing drugs and vaccines. Thus, humans have to stop wild animal consumption and the distribution of the ecosystem. Further, public education and strict regulations/lows at national and international levels have to be activated to stop hunting and consuming wild animals, which only can reduce and control human’s silent crime and cruelties (Figure 3).
Figure 3.Strict regulations have to be activated to stop hunting and consuming wild animals as a source of food. An ideal approach and comprehensive understanding of wildlife animals ' culture is essential to minimize the risk of the future animal-origin epidemics and pandemics.
Funding Source
None declared.
Ethical Approval
Not applicable.
Acknowledgment
H.E. and S. G. would like to thank BioRender online software for their support.
References
- 1.R A Bucknall. (1972) et al.,Studies with human coronaviruses II. Some properties of strains. 229E and OC43.Proceedings of the Society for Experimental Biology and Medicine 139(3), 722-727.
- 2.Hoek L Van Der. (2004) et al.,Identification of a new human coronavirus.Nature medicine. 10(4), 368-373.
- 3.Brucková M. (1970) The adaptation of two human coronavirus strains (OC38 and OC43) to growth in cell monolayers. Proceedings of the Society for Experimental Biology and Medicine 135(2), 431-435.
- 4.P C Woo.(2005).Characterization and complete genome sequence of a novelcoronavirus,coronavirus HKU1, from patients with pneumonia.Journal of virology. 79(2), 884-895.
- 5.Zhao Y.(2020).Single-cell RNA expression profiling of ACE2, the putative receptor of Wuhan 2019-nCov.bioRxiv.p.2020.01.26.919985.
- 6.Perlman S, Netland J. (2009) post-SARS: update on replication and pathogenesis.Nature reviews microbiology. 7(6), 439-450.
- 7.Zhou P.(2018).Fatal swine acute diarrhoea syndrome caused by an HKU2-related coronavirus of bat origin.Nature. 556(7700), 255-258.
- 9.World Health. (2020) O.,Clinical management of severe acute respiratory infection when novel coronavirus (2019-nCoV) infection is suspected: interim guidance, inClinical management of severe acute respiratory infection when novel coronavirus(2019-nCoV) infection is suspected: Interim guidance. 21-21.
- 11.Decaro N, Lorusso A.(2020).Novel human coronavirus (SARS-CoV-2): A lesson from animal coronaviruses.Vet Microbiol. 244, 108693.
- 12.S R Weiss, Navas-Martin S. (2005) pathogenesis and the emerging pathogen severe acute respiratory syndrome coronavirus.Microbiol. , Mol. Biol. Rev 69(4), 635-664.
- 13.Chen N.(2020).Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study.Lancet. 395(10223), 507-513.
- 14.Lu R.(2020).Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding.Lancet. 395(10224), 565-574.
- 15.Zhu N.. (2020).A Novel Coronavirus from Patients with Pneumonia in China, 2019.N Engl J Med 382(8), 727-733.
- 16.Wu F. (2020) A new coronavirus associated with human respiratory disease in China. , Nature 579(7798), 265-269.
- 17.Zhou P. (2020) A pneumonia outbreak associated with a new coronavirus of probable bat origin. , Nature 579(7798), 270-273.
- 18.Zhang T, Wu Q, Zhang Z. (2020) Probable pangolin origin of 2019-nCoV associated with outbreak of COVID-19.
- 20.L van Dorp.(2020).Emergence of genomic diversity and recurrent mutations in SARS-CoV-2.Infection, genetics and evolution : journal of molecular epidemiology and evolutionary genetics in infectious diseases. 83, 104351-104351.
- 21.Rizzo F.(2017).Coronavirus and paramyxovirus in bats from Northwest Italy.BMC Vet Res. 13(1), 396.
- 22.Lecis R.(2019).Molecular identification ofBetacoronavirusin bats from Sardinia (Italy): first detection and phylogeny.Virus genes. 55(1), 60-67.
- 23.Yang P, Wang X.(2020).COVID-19: a new challenge for human beings.Cellular. , Molecular Immunology 17(5), 555-557.
- 24.Drosten C. (2003) et al.,Identification of a novel coronavirus in patients with severe acute respiratory syndrome.N Engl. 348(20), 1967-76.
- 25.Huang C.(2020).Clinical features of patients infected with 2019 novel coronavirus in. , Wuhan, China.Lancet 395(10223), 497-506.
- 26.A M Zaki.(2012).Isolation of a novel coronavirus from a man with pneumonia in. , Saudi Arabia.N Engl J Med 367(19), 1814-20.
- 27.B L Haagmans, D A. (2014) Osterhaus,Neutralizing the MERS coronavirus threat.Sci Transl Med. 6(235), 235-19.
- 28.Z A Memish, A.(2013).Public health management of mass gatherings: the Saudi Arabian experience with MERS-CoV.Bull World Health Organ. 91(12), 899-899.
- 29.S van Boheemen.(2012).Genomic characterization of a newly discovered coronavirus associated with acute respiratory distress syndrome in humans.mBio. 3-6.
- 30.Lorusso A.(2020).Novel coronavirus (SARS-CoV-2) epidemic: a veterinary perspective.Veterinaria italiana. 56(1), 5-10.
- 31.X Y Ge.(2013).Isolation and characterization of a bat SARS-like coronavirus that uses the ACE2 receptor.Nature. 503(7477), 535-8.
- 32.Hu B.(2017).Discovery of a rich gene pool of bat SARS-related coronaviruses provides new insights into the origin of SARS coronavirus.PLoS. , Pathog 13(11), 1006698.
- 34.X L Yang.(2016).Isolation and Characterization of a Novel Bat Coronavirus Closely Related to the Direct Progenitor of Severe Acute Respiratory Syndrome Coronavirus.J Virol. 90(6), 3253-6.
- 35.A J Rodriguez-Morales.(2020).Clinical, laboratory and imaging features of COVID-19: A systematic review and meta-analysis.Travel Med Infect Dis. 34, 101623.
- 36.Nishiura H.(2020).Estimation of the asymptomatic ratio of novel coronavirus infections (COVID-19).Int. , J Infect Dis 94, 154-155.
- 37.Rothe C.(2020).Transmission of 2019-nCoV Infection from an Asymptomatic Contact in. , Germany.N Engl J Med 382(10), 970-971.
- 38.N van Doremalen.(2020).Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1.N. , Engl J Med 382(16), 1564-1567.
- 39.Kampf G.(2020).COVID-19-associated shortage of alcohol-based hand rubs, face masks, medical gloves and gowns - proposal for a risk-adapted approach to ensure patient and healthcare worker safety.J Hosp Infect.
- 40.Wan Y.(2019).Molecular Mechanism for Antibody-Dependent. , Enhancement of Coronavirus Entry.J Virol 94(5).
- 41.Guan Y.(2003).Isolation and characterization of viruses related to the SARS coronavirus from animals in southern China.Science. 302(5643), 276-278.
- 42.Han Z.(2015).Altered pathogenicity of atl/CH/LDT3/03 genotype infectious bronchitis coronavirus due to natural recombination in the 5'- 17kb region of the genome.Virus Res. 213, 140-148.
- 43.D S Hui.(2020).The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health - The latest 2019 novel coronavirus outbreak in Wuhan. , China.Int J Infect Dis 91, 264-266.
- 44.Cyranoski D.(2020).This scientist hopes totest coronavirusdrugs on animals. in locked-down Wuhan.Nature 577(7792), 607.
- 45.L H Taylor, S M Latham. (2001) M.E.J.P.T.o.t.R.S.o.L.S.B.B.S. Woolhouse,Risk factors for human disease emergence. 356(1411), 983-989.
- 46.Lee K. (1956) Propagation of transmissible gastroenteritis virus in tissue culture. , NYASA 66(1), 191-195.
- 47.Pensaert M, P De Bouck. (1978) A new coronavirus-like particle associated with diarrhea in swine. Archives of virology. 58(3), 243-247.
- 48.Pensaert M, Callebaut P, Vergote J. (1986) Isolation of a porcine respiratory, non‐enteric coronavirus related to transmissible gastroenteritis. Veterinary Quarterly. 8(3), 257-261.
- 49.Pan Y. (2017) Discovery of a novel swine enteric alphacoronavirus (SeACoV) in southern China. Veterinary microbiology. 211, 15-21.
- 50.Greig A. (1962) A hemagglutinating virus producing encephalomyelitis in baby pigs. Canadian journal of comparative medicine and veterinary science. 26(3), 49.
- 51.P C Woo. (2012) Discovery of seven novel Mammalian and avian coronaviruses in the genus deltacoronavirus supports bat coronaviruses as the gene source of alphacoronavirus and betacoronavirus and avian coronaviruses as the gene source of gammacoronavirus and deltacoronavirus. , J Virol 86(7), 3995-4008.
- 52.Chen W. (2005) SARS-associated coronavirus transmitted from human to pig. Emerging infectious diseases. 11(3), 446-448.
- 53.H M Weingartl. (2004) Susceptibility of pigs and chickens to SARS coronavirus. Emerging infectious diseases. 10(2), 179.
- 54.Vergara-Alert J.(2017).Livestock susceptibility to infection with Middle East respiratory syndrome coronavirus.Emerging infectious diseases. 23(2), 232.
- 55.Health W O f A.. (2020).Questions and Answers on the 2019 Coronavirus Disease (COVID-19). [cited 17 May ]; Available from:https://rr-africa.oie.int/en/news/questions-and-answers-on-the-2019-coronavirus-disease-covid-19/ .
- 56.Bryner J. (2020) Cat infected with COVID-19 from owner in Belgium. Live Science. [acesso. Abr 3]. DisponÃvel em: https://www. livescience. com/cat-infectedcovid-19-from-owner. html
- 57.Shi J.(2020).Susceptibility of ferrets, cats, dogs, and other domesticated animals toSARS–coronavirus 2.Science.
- 58.Cohen J.(2020).From mice to monkeys, animals studied for coronavirus answers.Science. 368(6488), 221-222.
- 59.Roberts A.(2005).Severe acute respiratory syndrome coronavirus infection of golden. , Syrian hamsters.J Virol 79(1), 503-11.
- 60.Y K Chu.(2008).The SARS-CoVferret model in an infection-challenge study.Virology. 374(1), 151-63.
- 61.T P Sheahan.(2020).Comparative therapeutic efficacy ofremdesivirand combination lopinavir, ritonavir, and interferon beta against MERS-CoV.Nature Communications. 11(1), 222.
- 62.T P Sheahan.(2017).Broad-spectrum antiviral GS-5734 inhibits both epidemic and zoonotic coronaviruses.Science translational medicine. 9(396)
- 63.Roberts A.(2005).Severe acute respiratory syndrome coronavirus infection of golden Syrian hamsters.Journal of virology. 79(1), 503-511.
- 64.Shi J.(2020).Susceptibility of ferrets, cats, dogs, and other domesticated animals to SARS–coronavirus 2.Science. 368(6494):. 1016-1020.
- 65.Hoinville L.(2013).Proposed terms and concepts for describing and evaluating animal-health surveillance systems.Preventive veterinary medicine.112(1-2):. 1-12.
- 66.Silva da, B M.(2018).Evidence of zoonotic leprosy in Para, Brazilian Amazon, and risks associated with human contact or consumption of armadillos.PLoS Negl Trop Dis. 12(6), 0006532.
- 67.C K Laforet.(2019).Toxoplasma gondii seroprevalence in extensively farmed wild boars (Sus scrofa). in Denmark.Acta Vet Scand 61(1), 4.
- 68.Gavrilovic P, Pavlovic I, Todorovic I. (2019) domestic pigs and wild boars. in South Banat, northern Serbia.Comp Immunol Microbiol Infect Dis 63, 142-144.
- 69.Santoro M.(2019).Real-time PCR detection of Toxoplasma gondii in tissue samples of wild boars (Sus scrofa) from southern Italy reveals high prevalence and parasite load.Parasit Vectors. 12(1), 335.
- 70.Montone A M I.. (2019).Occurrence of HEV-RNA in Italian Regional Pork and Wild Boar Food Products.Food Environ Virol 11(4), 420-426.
- 71.Kicinska A, Glichowska P, Mamak M.(2019).Micro- andmacroelementcontents in the liver of farm and wild animals and the health risks involved in liver consumption.Environ Monit Assess.191(3):. 132.
- 72.Stepien-Pysniak D.(2019).Biofilm formation capacity and presence of virulence factors among commensal Enterococcus spp. from wild birds.Sci Rep. 9(1), 11204.
Cited by (1)
- 1.Elkhenany Hoda, Gupta Shilpi, Abdelhai Mostafa F., Turabian Jose Luis, 2021, Cell Therapy as an Alternative approach for COVID-19 Infection Consequences: A Non-Systematic Review, International Journal of Coronaviruses, 2(3), 23, 10.14302/issn.2692-1537.ijcv-20-3685