Study of Neuropilin-1/Cd304 Expression in Leukemogenesis
Abstract
Neuropilins are transmembrane glycoproteins that act as receptors for vascular endothelial growth factors (VEGF) and are involved in the process of tumor angiogenesis. Its importance in hematological malignancies such as acute leukemia (AL) remains to be elucidated. The aim of this study was to evaluate the significance of neuropilin-1 expression in acute myeloid leukemia (AML) and acute lymphocytic leukemia (ALL) patients by flowcytometry and the difference between both groups of acute leukemia. Bone marrow aspirates of 52 patients with acute leukemia, 29 patients with de novo AML and 23 ALL patients were examined in this study. 15 subjects with non-hematological malignancy serving as the control group were also included. Neuropilin-1 expression by flow cytometry showed a highly significant increase in de novo AML and ALL patients with a mean of 37.9 ± 20.92% and 32.33±19.8%, respectively, compared to control group’s mean of 11.51 ± 3.04% (p= 0.001, 0.006). There were no statistically significanct difference between ALL and AML patients (p= 0.76). Neuropilin-1 surface expression by flowcytometry showed a significant positive correlation with total leukocyte count, bone marrow blast percentage, CD45 and CD14 and negative correlation with hemoglobin level, RBCs count in AML patients. In ALL patients, positive significant correlations were found with bone marrow blast percentage and negative correlation with hemoglobin level, RBCs count. Neuropilin-1expression was detected significantly in acute leukemias and it is related to the disease severity.
Author Contributions
Academic Editor: Fernando Luiz Affonso Fonseca, Institute of Chemical Sciences, Federal University of São Paulo, UNIFESP, Head of Laboratory of Clinical Analysis of the Faculty of Medicine of ABC, Santo André, SP, Brazil.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2018 Shabaan Radwan, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Neuropilins are single-pass transmembrane glycoproteins that were found to be receptors for the vascular endothelial growth factor (VEGF) family. CD304, or neuropilin-1 (NRP-1), is a transmembrane C-type lectin found on plasmacytoid dendritic cells (PDCs). VEGF is an important cytokine that contributes to disease evolution in various myeloid neoplasms. In particular, VEGF has been described as a mediator of malignant angiogenesis and autocrine growth regulator in leukemic cells 1. It was found that NRP- 1 binds to VEGF that is a critical pro-angiogenic factor that induces proliferation and migration of endothelial cells to tumor vasculature 2.
Also, NRP-1enhances VEGF-mediated mitogenic and chemotactic activity 3. VEGF, as a key factor for angiogenesis and tumor growth, exerts its functions mainly through activation of 2 tyrosine kinase receptors: VEGFR-1 (Flt 1) and VEGFR-2 (KDR).
Experimental and clinical evidence indicates that bone marrow (BM) cells participate in the process of new blood vessel formation. However, the molecular mechanisms underlying their recruitment and their exact role are still elusive. BM cells are recruited to the sites of neoangiogenesis through the NRP-1 receptor and they are essential for the maturation of the activated endothelium and the formation of arteries. The 165-aa isoform of VEGF, which activates the endothelium and recruits NRP-1 myeloid cells, is a powerful arteriogenic agent. In contrast, neither the shortest VEGF121 isoform, which does not bind NRP-1 and thus does not recruit BM cells, nor semaphorin 3A, which attracts cells but inhibits endothelial activation, is capable of sustaining arterial formation 4.
Acute myeloid leukemia (AML) is the most common acute leukemia affecting adults, and its incidence increases with age 5, while acute lymphoblastic leukemia (ALL) is the most common childhood malignancy, representing nearly one-third of all pediatric cancers. With the advent of aggressive multimodality therapy, it has become a curable disease in more than 80% of patients 6. BM angiogenesis is an important requirement for the development and progression of hematological malignancies including leukemia and lymphoma 7. Leukemia is an angiogenesis-dependent malignancy and angiogenesis is strictly dependent on Akt/NF-κB(Protein kinase B (PKB)/ nuclear factor kappa-light-chain-enhancer of activated B cells)activation8.
The inhibitors of the NF-κB pathway decreasing VEGF secretion in leukemic cells and inhibiting endothelial cell activities may cause the interruption of a reciprocal stimulatory loop between leukemic and endothelial cells. Different reports demonstrating the activation of NF-κB in lymphoid and myeloid malignancies underscore the implication in malignant transformation 9.
While the overexpression of NF-κB may lead to chemo resistance, the appropriate inhibition of this pathway may lead to successful therapy. Moreover, the transcription of VEGF by the classical NF-κB target gene may be repressed, which is one aspect of the participation in tumor genesis of adult T-cell leukemia (ATL) 10. Thus, this work was planned to study of NRP-1 expression in acute leukemia patients by flow cytometry and if there is difference in its expression between AML and ALL.
Patients and Methods
Patients
This study was carried out on 52 newly diagnosed acute leukemia patients attending the Hematology/Oncology Clinic of the Sohag University Hospital. 29 newly diagnosed AML patients (17 males and 12 females) ranged in age from 11 to 72 years with a mean of 33 ±18.05 years. 23 patients with ALL (15 males and 8 females) were also included in this study; their ages ranged from 4 to 65 years with a mean of 33.7±19.19 years. The diagnosis of leukemia subtype was based on the standard morphological and immunophenotyping (IPT) criteria 11, 12. These patients comprised 6 with M1, 8 with M2, 6 with M3, 5 with M4, 2 with M5 and 2 with M6 French-American-British (FAB) subtypes. Fifteen subjects with non-hematological malignancy matching the patients in age and sex were recruited to serve as the control group. The study design was approved by the Research Ethics Committee of the Sohag University Faculty of Medicine. All samples were taken after obtaining written consent from the patients and the controls.
All subjects and controls subjected to the following procedures:
A- Detailed history taking and through clinical examination.
B- Laboratory investigations:
1- Complete blood count (CBC) using Cell-Dyn 3700, automated cell counter, (Abbott diagnostic, Dallas, USA), with examination of leishman-stained peripheral blood (PB) smears for differential leucocytes count and blast cells percentage.
2- BM aspiration with examination of Leishman-stained films for bone marrow blasts.
3- Immunophenotyping (IPT) analysis and assessment of NRP-1 expression of the BM aspirate or peripheral blood using FACS Calibur BD Flowcytometer. Diagnosis of acute leukemia was based on the presence of blast cells > 20% in BM film according to WHO proposal, together with presence of immunophenotyping results consistent with AML and ALL.
Methods
Sampling
Two milliliters of PB was delivered into an EDTA vacutainer tube (1-2 mg/mL blood) for CBC. Two milliliters of BM aspirate was collected from each patient and control under completely aseptic conditions using sterile vacutainers and delivered into 2 EDTA vacutainer tubes. Samples were used for IPT and NRP-1 surface expression by flow cytometry.
Fresh PB or BM samples were kept at ambient temperature and processed for immunophenotyping within 6 hours of collection. The EDTA anticoagulated BM and PB samples were diluted with phosphate buffered saline (PBS), Ph 7.4 (Sigma chemicals, St Louis), the final cell suspension was adjusted at 10 x 103/ml. For each sample, asset of tubes was prepared for a panel of fluroescein isothiocyanate (FITC)/ phycoerythrin (PE)-conjugated MoAbs used for diagnosis of acute leukemia including one for the isotypic matched negative control (supplied by BD Science, San Jose, CA). The monoclonal antibodies used were T-cell lymphoid markers (CD2, CD3, CD5, and CD7), B-cell lymphoid markers (CD10, CD19, and CD20), myeloid markers (CD13, CD33, intracellular MPO). CD61, CD41 and glycophorin in suspected cases. Monocytic marker CD14 and common progenitor markers (CD34 and HLA-DR) and All MoAbs were purchased from (BD Science, San Jose, CA) while PE-conjugated Neuropilin-1 MoAbs were supplied by (Ancell Corporation, USA).
Immunophenotypic Analysis:
Immunophenotypic analysis was performed using whole blood staining method. Fresh samples were obtained from peripheral blood or bone marrow at time of diagnosis. Double and Triple marker labeling was performed, including proper isotype controls. Samples were analyzed on FACSCalibur BD Flowcytometer (Beckman Coulter Corporation, Miami, FL, USA).
Detection of Surface Markers by Direct Staining:
The whole blood staining method was performed 13. In short, 10 µL labeled MoAb was added to 100 µL whole blood (or BM), incubated in the dark for 20 minutes, hemolyzed by Lysing solution (Becton & Dickinson) and washed by Phosphate Buffer Solution (PBS) (Becton & The collected data were tabulated and analyzed using Statistical Package of Dickinson), then analyzed. The blasts were electronically gated using forward scatter against side scatter.
Detection of Intracellular Markers:
100 µL of whole blood (or BM) was lysed using lysis solution (Becton & Dickinson) for 10 minutes. Cells were washed once and resuspended in 1ml PBS. A mixture of 500 µL 4% paraformaldehyde as fixative, 500 µL PBS and 5 µL tween 20 as detergent was added to the cells and incubated for 10 min. The cells were washed and 10 µL Mo Ab was added and incubated for 30 min. at 4 °C. Cells were washed, resuspended in 500 µL PBS and analyzed 13.
A minimum of 10000 events were acquired. Blast cell population was selected based on its forward and side scatter properties. The percentage of blast cells positive for the relevant studied marker was determined as a percentage from the blast cells population. The negative isotypic control was set at 0.5%. Cells were considered positive for certain marker when 20% or more of the gated cells expressed it (Figure 1).
Statistical Analysis:
Social Science (SPSS-version 17). ALL data was expressed as mean SD. For statistical evaluation Student’s t test and Correlation Coefficient (r) were used. Significance was accepted at p 0.05.
Results
The present study was carried out on 52 newly diagnosed acute leukemia patients, 29 AML and 23 ALL patients. The demographic, clinical and hematological data of the studied AML, ALL patients and control group presented in (Table 1, Table 2).
Table 1. Demographic and clinical data of patients and control.| Control (n =15) | ALL (n =23) | AML (n =29) | Items |
| 2.5 -65 21.67 ± 19.35 | 4-6533.7 ± 19.19 | 11-7233± 18.05 | Age Range Mean ± SD |
| 7(%) 8(%) | 15(65.2%)8 (34.8%) | 17 (58.6%)12 (41.4%) | sex Male Female |
|---|---|---|---|
| 0 | 8(34.8%) | 19 (65.5%) | Hepatomegaly |
| 0 | 19(82.6%) | 22 (75.9%) | Splenomegaly |
| 0 | 20(87%) | 6 (20.7%) | Lymphadenopathy |
| Control(n =15) | ALL(n =23) | AML(n =29) | Items |
| 5 - 18 10.41 ± 4 .4 | 1.3 - 131** 49.87 ± 44.95 | 2-420 *** 113.6 ± 117.02 | WBCs (x 109 /L)RangeMean ± SD |
| 8.6 – 15.6 11.43 ± 1.79 | 5.2 - 12.8***9 ± 2.79 | 3.3 - 10.5 ***7.48 ± 2.24 | Haemoglobin (g/dL)RangeMean ± SD |
| 0 0 | 33 – 88%***65.6 ± 19.48 | 65 – 92%***80.6 ± 9.06 | Blast in PBRangeMean ± SD |
| 0.4 – 2.8% *** 1.55 ± 0.69 | 64 – 93% *** 85.8 ± 10.21 | 70 – 96% *** 84.5 ± 8.11 | Blast in BMRangeMean ± SD |
Results of NRP-1 by Flow Cytometry (Figure 1 and Figure 2)
Figure 1.Dot plot showing negative control (left lower), ALL case with positive Neuropilin -1 expression (41.8%)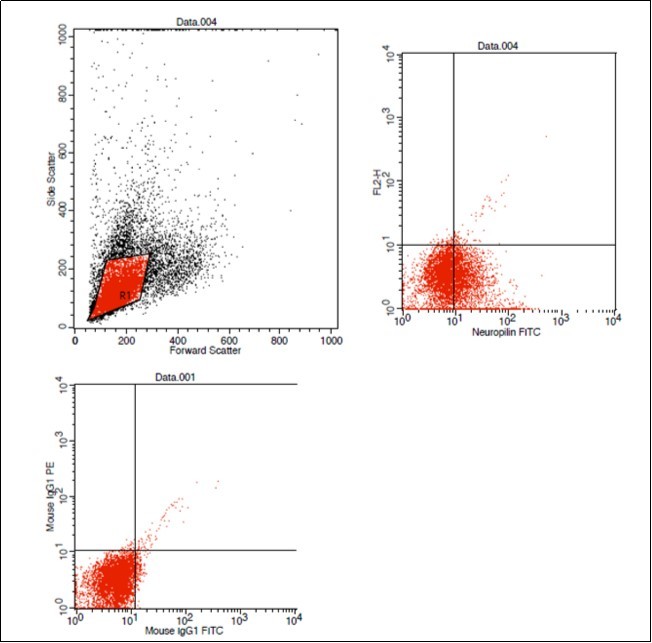
Figure 2.Dot plot showing AML case with positive Neuropilin -1 expression (63.9%)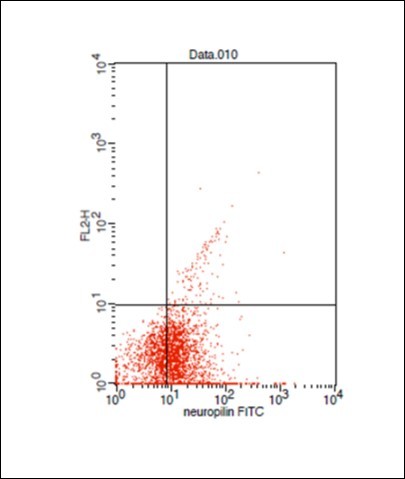
NRP-1 percentage expression for AML cases ranged from 2.7 % to 78% with a mean ± SD value 37.9 ± 20.92 taking all patients as a single group, while its percentage for ALL cases ranged from 8.7% to 82.3 % with a mean ± SD value 32.33 ±18.22.
The mean percentage expression of NRP-1 by flow cytometry in the control group ranged from 7.4 % to 16.8 % with a mean ± SD value 11.51± 3.04, There was a highly statistical significant difference in NRP-1 surface expression, comparing the AML patients or ALL patients to control group (as a whole) (p<0.001& p < 0.006). However, comparing AML and ALL patient groups, no statistical significance was revealed in NRP-1 expression concerning both percentage (p=0.76 and p=0.954, respectively) (Table 1).
In AML patients, Neuropilin-1 surface expression by flowcytometry showed a significant positive correlation with total leukocyte count, blast percentage, CD45 and CD14, but negative correlation was seen between NRP-1 percentage expression and RBCs and hemoglobin (Hb) levels (r= -0.534, p=0.01), with no significant correlation found between NRP-1 percentage expression and any other parameters including age, platelet count, CD33, CD13, CD14, CD117, CD61, myeloperoxidase, CD2, CD5, CD7, CD34, HLA-DR, CD10.
In ALL patients, NRP-1 percentage expression showed a positive highly significant correlation with and age (r= 0.986, p<0.001) and positive significant correlation with BM blast percentage (r= 0.882 and p=0.02). However, for other parameters such as TLC, Hb, platelet count, CD33, CD13, CD117, CD61, myeloperoxidase, CD2, CD5, CD7, CD34, CD117, HLA-DR, and CD10 no significant correlations were found.
Discussion
The aim of this study was to evaluate the significance of NRP-1 expression in AML and ALL patients by flow cytometry. Fifty-two AML and ALL patients were studied for detection of NRP-1 levels by flow cytometry. Patients included in this work were classified into AML and ALL cases. Comparison of patients’ NRP-1 percent positivity to those of the control group showed a highly significant difference between AML and ALL groups when compared with healthy control group. Consistent with these results, Younan, et al., 2012 14 who found NRP-1 was expressed in 80% and 95% of ALL and AML respectively but Kreuter, et al., 200715 reported that the expression of NRP-1 was increased in the bone marrow of AML patients. However, Meyerson et al., 2012 16, found that Neuropilin-1 is frequently expressed on B-ALL blasts, and weakly expressed in normal bone marrow B-cell progenitors, while gradually decreasing during maturation, to be completely lost at later stages of B-cell. The expression of Neuropilin-1 on B-cell progenitors may explain its frequent higher expression in precursor B-ALL than mature ALL.
Coustan Smith et al., 201117, demonstrated that NRP-1 is one of the most differentially expressed markers between ALL blast cells and normal B-cells precursors. There were no statistical significant difference in neuropilin-1 surface expression by flowcytometry between ALL and AML patients. These observations may be explained by a common pathway shared by the leukemogenesis of precursor B-cell lymphoblastic leukemia/lymphomas and acute myeloid leukemia. B-cell/myeloid common progenitor that gives rise to both B and myeloid lineages has been identified as a population CD34+CD19+CXCR4- in humans and c-Kit+B220+CD19- in mice (Hou et al., 2005) 18. Additional evidence supporting the existence of a B-cell/myeloid common progenitor comes from a functional study of the transcription factor PU.1 in hematopoiesis (Rosenbauer et al., 2004) 19.
In our study, In AML patients, Neuropilin-1 expression was significantly associated with higher white blood cells Count, BM blasts percentage, CD45 and CD14. In ALL patients, NRP-1 percentage expression showed positive significant correlation with BM blast percentage. Hagag et al., 2015 20 also found that NRP-1/CD304 expression significantly associated with bone marrow blast percentage. In AML, expression was noted in all (100%) acute myeloid leukemias with the M4eo subtype (Meyerson et al 2012) 16. Negative correlation was seen between NRP-1 percentage expression and RBCs and hemoglobin (Hb) levels (r= -0.534, p=0.01), with no significant correlation found between NRP-1 percentage expression and any other parameters These results indicate that higher Neuropilin-1 expression levels correlated with disease severity (Kreuter et al., 2007) 15. Anti-VEGF antibodies have received much attention lately as they are able to block tumor angiogenesis and prolong the life of cancer patients (Pan et al., 2007) 21. In 2004, bevacizumab (Avastin), a humanized monoclonal antibody against VEGF-A, became the first antiangiogenic drug approved by the US Food and Drug Administration. Also another anti-VEGF antibody, ranibizumab (Lucentis), has been successful in the treatment of neo-vascularization associated with wet neovascular age-related macular degeneration, thereby alleviating blindness in patients (Karjalainen et al., 2011) 22. Hu and Jiang 2016 23, Found that NRP-1 activates the signaling pathways of ABL and platelet derived growth factor receptor (PDGFR), which induces resistance to radiotherapy and chemotherapy in cancer cells. Furthermore, NRP-1 activates the tumor microenvironment by binding with fibronectin and activating ABL, thereby promoting tumor growth. Inhibition of NRP-1 may overcome the limitations of individually inhibiting the VEGF-VEGFR2 pathway in cancer therapy and provide new goals for leukemia treatment.
Abbreviations:
*VEGF: vascular endothelial growth factors
* AL: acute leukemia
* AML: acute myeloid leukemia
*ALL: acute lymphocytic leukemia
*NRP-1: neuropilin-1
Declarations
Ethics approval and consent to participate
approval for this work was obtained from Ethics Committee, Faculty of Medicine, Sohag University.
Consent for Publication:
not applicable.
Availability of Data and Materials:
all data generated or analyzed during this study are included in this published article.
Funding:
there is no funding for the research.
Acknowledgment:
not applicable
References
- 1.Dzionek A, Fuchs A, Schmidt P, Cremer S, Zysk M. (2000) BDCA-2, BDCA-3, and BDCA-4: three markers for distinct subsets of dendritic cells in human peripheral blood. , J Immunol 165, 6037-6046.
- 2.Ferrara N, Gerber H P, LeCouter J. (2003) The biology of VEGF and its receptors. , Nat Med 9, 669-76.
- 3.Lu L, Zhang L, Xiao Z, Lu S, Yang R. (2008) Neuropilin-1 in acute myeloid leukemia: expression and role in proliferation and migration of leukemia cells. , Leuk Lymphoma 49, 331-338.
- 4.Zacchigna S, Pattarini L, Zentilin L, Moimas S, Carrer A. (2008) Bone marrow cells recruited through the neuropilin-1 receptor promote arterial formation at the sites of adult neoangiogenesis in mice. , J Clin Invest 118, 2062-2075.
- 6.He Z, Tessier-Lavigne M. (1997) Neuropilin is a receptor for the axonal chemorepellent semaphorin III. , Cell 90, 739-751.
- 7.Pui C H, Evans W E. (2006) Treatment of acute lymphoblastic leukemia. , N Engl J Med.12; 354(2), 166-78.
- 8.Bellamy W T, Richter L, Sirjani D. (2001) Vascular endothelial cell growth factor is an autocrine promoter of abnormal localized immature myeloid precursors and leukemia progenitor formation in myelodysplastic syndromes. , Blood 97, 1427-1434.
- 9.Braun T, Carvalho G, Fabre C, Grosjean J, Fenaux P. (2006) Targeting NF-kappaB in hematologic malignancies. , Cell Death Differ 13, 748-758.
- 10.Sujobert P, Bardet V, Cornillet-Lefebvre P. (2005) Essential role for the p110delta isoform in phosphoinositide 3-kinase activation and cell proliferation in acute myeloid leukemia. , Blood 106, 1063-1066.
- 11.Pui C H, Campana D, Pei D, Bowman W P, Sandlund J T. (2009) Treating childhood acute lymphoblastic leukemia without cranial irradiation. , N Engl J Med 360(26), 2730-41.
- 12.Veerman A J, Kamps W A, Berg H van den, Berg E van den, Bökkerink J P. (2009) Dexamethasone-based therapy for childhood acute lymphoblastic leukaemia: Results of the prospective Dutch Childhood Oncology Group (DCOG) protocol ALL-9 (1997–2004) Lancet Oncol. 10(10), 957-66.
- 13.Jung R T, Schauer U, Heusser C, Neumann C, Rieger C. (1993) Detection of intracellular cytokines by flow cytometry. Immunol Method.159(1-2): 159-197.
- 14.Younan S, Elhoseiny S, Hammam A, Gawdat R, El-Wakil M. (2011) Role of neuropilin-1 and its expression in Egyptian acute myeloid and acute lymphoid leukemia patients. Leuk Res. 36(2), 169-73.
- 15.Kreuter M, Steins M, Woelke K, Buechner T, Berdel W E. (2007) Downregulation of neuropilin-1 in patients with acute myeloid leukemia treated with thalidomide. , Eur J Haematol 79, 392-397.
- 16.Meyerson H J, Blidaru G, Edinger A, Osei E, Schweitzer K. (2012) A potential marker for minimal residual disease detection in precursor B-cell acute lymphoblastic leukemia. , Am J Clin Pathol 137, 39-50.
- 17.Coustan-Smith E, Song G, Clark C, Key L, Liu P. (2011) New markers for minimal residual disease detection in acute lymphoblastic leukemia. , Blood 117(23), 6267-6276.
- 18.Hou Y H, Srour E F, Ramsey H, Dahl R, Broxmeyer H E. (2005) Identification of a human B-cell/myeloid common progenitor by the absence of CXCR4. , Blood 105, 3488-3492.
- 19.Rosenbauer F, Wagner K, Kutok J L, Iwasaki H, Le Beau MM. (2004) Acute myeloid leukemia induced by graded reduction of a lineage-specific transcription factor, PU.1. , Nat Genet 36, 624-630.
- 20.Hagag A A, Nosair N A. (2015) Prognostic impact of neuropilin-1 expression in Egyptian children with B-lineage acute lymphoblastic leukemia. , Mediterr J Hematol Infect Dis 7(1), 2015009.
- 21.Pan Q, Chanthery Y, Liang W C, Stawicki S, Mak J et al. (2007) Blocking neuropilin-1 function has an additive effect with anti-VEGF to inhibit tumor growth. , Cancer Cell 11(1), 53-67.
Cited by (3)
This article has been cited by 3 scholarly works according to:
Citing Articles:
The Egyptian Journal of Hospital Medicine (2019) OpenAlex
Nawal M. Abdel Qawy, Alshaymaa M. Alhabibi, S. Khalil, Marwa M. Abdelfattah - The Egyptian Journal of Hospital Medicine (2019) Semantic Scholar
Journal of Hematology and Oncology Research (2018) OpenAlex
