Cholesterol-Conjugated siRNA Accumulates in the Different Hematopoietic and Lymphoid Cells.
Abstract
Small interfering RNA (siRNA) based drugs for overcoming multiple drug resistance of hematological malignancies could solve the problem of poor response to the chemotherapy and hight relapse rate. The main factor that significantly limits biomedical application of siRNA is inefficient delivery to target cells and tissues. The attachment of siRNA to molecules, which enter into the cell by natural transport mechanisms, can improve cellular uptake of siRNA. In current study the carrier-free cellular uptake of siRNA containig cholesterol residues conjugated to the 5’-end of the sense strand via oligomethylene linker of various length (here and after Ch-siRNA) was explored. The data demonstrate that cholesterol residue increase the accumulation of siRNA in all tested cell lines and the primary cells. The efficiency of Ch-siRNA accumulation in K562 cells depends greatly on the leangth of the linker connecting cholesterol and siRNA: Ch-siRNAs with linker of 10 - 12 methylene units accumulate the most efficiently in this cells. It was found that Ch-siRNA effectively accumulates in MOLT-3 (acute lymphoblastic leukemia, ALL), HL-60 (acute myelogenous leukemia, AML), K562 (chronic myelogenous leukemia CML) and primary peripheral blood mononuclear cells (PBMC) from patient with non-Hodgkin lymphoma (NHL) or healthy donor resulting in near 100% of transfected cell when used at 1 mM concentration.
Author Contributions
Academic Editor: Xingding Zhang, Department of Lymphoma/Myeloma, UT MD Anderson Cancer Center
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2016 Takuma Hayashi, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Since its discovery in 1998 by Fire and Mellow, siRNA has proved to be an effective tool for gene silencing in various species1, 2. siRNA have great potential as a therapeutic agent for downregulation of the abberant gene expression including expression of viral and mutated genes as welll as hyperexpression of normal genes3. It has been clearly demonstrated, that overexpression of genes, responsible for the drug efflux from cells such as MDR1, could result in clinically significant drug resistance of the tumor. This is why such genes could be used as targets for siRNA-based drugs4. The development of multidrug resistance in haematological malignancies is especially important problem because the surgical removal for this tumor type is not possible and chemotherapy is the only option for the treatment. Inefficient delivery of siRNAs to target cells and tissues is the main obstale that limits their biomedical application. Currently, various approaches have been developed to solve the problem of siRNA delivery5, 6, but they all have limitations for use in therapy. The conjugation of siRNA to the molecules, that can be internalized into the cell via natural transport mechanisms is a promising approach that can be used for overcoming this problem7. Since cholesterol is an important biogenic molecule, and it is a component of cell membranes and there is a natural transport system for cellular internalization of this molecule it has been proposed to attach cholesterol to siRNA for enhancing its carrir-free cellular uptake8. We previously developed conjugates of siRNA with cholesterol attached to the 5’ end of the sense strand, which efficiently penetrate into the cells, silence the expression of MDR1 gene and restore the sensitivity of drug resistant KB-8-5 cancer cells to cytostatics9. It was found, that linker length between siRNA and cholesterol should be optimized for efficient gene silencing in specific cell types. Therefore, to study therapeutic potential of Ch-siRNA we investigated cellular accumulation of Ch-siRNA containig the linkers with different number of carbons in the cells of hematopoetic origin.
Materials and Methods
Synthesis of siRNAs and Conjugates.
The sense and antisense strands of siRNAs were synthesized on an automatic ASM-800 synthesizer using solid phase phosphoramidite synthesis protocols11 optimized for the instrument, with a 10 min coupling step for 2’-O-TBDMS-protected phosphoramidites, 6 min coupling step for 2’-O-methylated phosphoramidites. 3'-PT-amino-modifier C6 CPG (GlenResearch, USA) was used for the synthesis 3'-aminohexyl-contaning antisense strand. A combination of H-phosphonate and phosphoramidite methods was applied to the synthesis of 5’-cholesterol conjugates of siRNAs (Table 1) as described by us earlier10. Conjugates were prepared by analogy with10. Cholesterol derivatives bearing linkers of various lengths were transformed into the corresponding H-phosphonates via interaction with phosphorus triimidazolide followed by hydrolysis. Support-bound 5’-hydroxyl-containing sense strands of siRNA were used for the condensation with H-phosphonates in the presence of pivaloylchloride. All conjugates obtained were fully deprotected according to the standard procedures, using methylamine and TEA·3HF11. Isolation of the oligoribonucleotides conjugated with lipophilic residue from reaction mixtures was accomplished by reverse-phase HPLC. The purified oligoribonucleotides were characterized by electrophoretic mobility in 12% dPAAG, MALDI-TOF-MS and LC-ESI-MS. Alexa-488 was attached the 3’-end of the antisense strand of siRNA equipped with 3'-aminohexyl linker according to the manufacturer’s protocol using Alexa Fluor-488 sulfodichlorophenol ester (Life Technologies, USA) in 0.1 M sodium tetraborate buffer (pH 8.5). siRNAs were obtained by annealing 300 μM sense and antisense strands in the buffer containing 30 mM HEPES-KOH (pH 7.4), 100 mM sodium acetate, and 2 mM magnesium acetate and stored at -20 °C.
Figure 1.Conjugates of siRNA and cholesterol used in the study; n denotes linker length.
Cell Cultures
HL-60, K562 and MOLT-3 leukemia cell lines were obtained from the Institute of Cytology RAS, St. Petersburg, Russia. They were grown in RPMI-1640 medium supplemented with 20 % fetal bovine serum (FBS) for HL-60 cells and 10% for the other cell lines and with 100 U/ml penicillin, 100 µg/ml streptomycin and 0.25 µg/ml amphotericin at 37°C in a humidified atmosphere containing 5% CO2/95% air. Blood samples from patients with lymphoma were provided by Novosibirsk State Medical University, Novosibirsk, Russia, blood of healthy volunteers was provided by Center of New Medicical Technologies SB RAS, Novosibirsk, Russia, all participants provided written informed consent. Blood was collected in heparinized tubes and the experiments were started not later than 12 hours after sampling. The blood was diluted in 10 times with 0.9% sodium chloride (saline solution) and layered onto lymphocyte separation medium (LSM, MP Biomedicals, USA). Tubes were centrifuged at 800 g for 20 minutes, then the rings of PBMC were collected and transferred to a separate tube and washed two times with saline solution. The cells were cultured in Iscove's modified Dulbecco's medium (IMDM) supplemented with 10-20% fetal bovine serum (FBS) 100 U/ml penicillin, 100 µg/ml streptomycin and 0.25 µg/ml amphotericin at 37°C in a humidified atmosphere containing 5% CO2/95% air.
siRNA Cellular Accumulation Assay
The cells were washed with saline solution and plated in 24-well plates at a density 1.5×106 cells/well for PBMC and 3×105 cells/well for other cell lines in 200 µl of serum free medium. Alexa 488-labeled conjugates in 50 µl of Opti-Mem were added to the cells up to 1 µM concentration. Alternatively, the cells were transfected with siRNA (0.2 µM) using Lipofectamine 2000 (Invitrogen, USA) (0.8 µl per well) according to the manufacturer’s protocol. Four hours post-transfection, the cells were washed with saline solution and fixed in 2% formaldehyde in phosphate-buffered saline (PBS). Cells were analyzed using Cytomics FC 500 (Beckman Coulter, USA) flow cytometer (excitation wave length 488 nm, emission 530±30 nm). 15 000 cells from each sample were analyzed. Two parameters reflecting the efficiency of the process were used for the comparison: the transfection efficiency (here and after referred to as ‘TE’) estimated as the percentage of cells with green fluorescence exceeded the maximum level of the auto-fluorescence of untreated cells (here and after referred to as ‘transfected cells’) and the mean values of cell fluorescence intensity measured in relative fluorescent units were also used for data presentation (‘RFU’).
Microscopy
In order to prove the intracellular localization of siRNAs, IN Cell Analyzer 2000 (GE Healthcare, USA) with Z-stack imaging feature was utilized. K562 cells in the exponential phase of growth were plated in 96-well plates at a density of 1×103 cells/well in serum-free medium. Alexa Fluor-488 -labeled Ch-siRNAs were added to the cells to final concentration 1 mM. Following 4 h of incubation the cells were fixed in 4% formaldehyde/ PBS at 37°C for 20 minutes, and stained with HCS NuclearMask Blue stain and CellMask Deep Red (Thermo Fisher Scientific) according to the manufacturer protocol. The analysis of intracellular siRNA localization was performed using IN Cell Analyzer 2000 at 60× magnification using DAPI, FITC and Cy5 channels.
Results
The sequence of siRNA (Table 1) targeted to 557–577 nt of the human MDR1 mRNA was selected in our previous study, the nuclease-sensitive sites in siRNA were mapped and protected by introducing 2'-O-Ме-ribonucleotides to protect siRNA from degradation in the presence of serum and in the cytoplasm . 5'-hydroxyl-position of the sense strands of siRNAs was used for the introduction of the lipophilic moieties since the modification of this position is better tolerated by RNAi machinery than the modification of the antisense or both strands. We compared experimentally the accumulation of the Ch-siRNA conjugates connected by aliphatic linker containing 3, 6, 8, 10 or 12 methylene units in K562 cells . The data shows that all siRNA under the study displayed 100% TE when used in 1 mM concentration (Figure 1 A). The mean fluorescence intensity of the cells incubated with Ch-siRNA was much higher than for free siRNA (Figure 1 B). The comparison of the mean fluorescence intensity of the cells incubated with different Ch-siRNAs revealed that the accumulation of Ch-siRNA conjugates in K562 cells increased with the increase of the length of linker connecting cholesterol and siRNA from 3 to 10 carbons. There are two significant gains in the efficiency of accumulation resulted from the augmentation of linker length: the elongation from 3 to 6 carbon atoms (38 and 67 RFU, respectively) and the elongation from 8 to 10 carbon atoms (72 and 101 RFU, respectively), further increase of the linker length to 12 carbon atoms did not improve the accumulation efficiency (104 RFU). siRNA transfected with Lipofectamine 2000 accumulated with similar RFU as conjugate Ch3-siRNA.
Figure 1.Cellular accumulation of Alexa 488-labeled cholesterol derivative of siRNAs containig linker with different length. The percentage of Alexa 488-positive cells in the population (%) (A) and the mean value of the cell fluorescence (RFU) (B) after incubation of K562 cells in the presence of corresponding siRNAs are shown. Data were obtained via flow cytometry. Fifteen thousand events were counted in each sample. Mean values (±SD) from three independent experiments are presented.
To evaluate the efficiency of Ch-siRNA accumulation in different types of hematological malignancy, we used the MOLT-3 (ALL origin), HL-60 (AML origin), K562 (CML origin) cell lines and PBMC obtained from the patient with peripheral T-cell lymphoma (containing NHL tumor cells) or PBMC collected from healthy donors. The flow cytometry data shows (Figure 2) that the efficiency of siRNA transfection by Lipofectamine 2000 reached 100 % for HL-60 and K562 cells, and HL-60 cells were transfected with 75 % efficiency. TE for PBMC both from NHL patient and healthy donor did not reached 40%. Carrier-free accumulation of Ch-siRNA provided 100% of transfected cells in all cell lines under the study including PBMC cells. The comparison of mean fluorescence intensity revealed a significant preference of Ch-siRNA accumulation in PBMC in comparison with free siRNA and siRNA transfected with Lipofectamine 2000. The accumulation of Ch-siRNA increased in the order: PBMC of patient with NHL<PBMC of healthy donor< HL-60< K562<MOLT-3 (26, 34, 45, 67 and 77 RFU, respectively). The cells under the study demonstrated insignificant mean fluorescence intensity after incubation with free siRNA or siRNA transfected by Lipofectamine 2000.
Figure 2.Accumulation Alexa 488-labeled siRNAs in the different types of the cells. The percentage of Alexa 488-positive cells in the population (%) (A, C , E, G and I) and the mean value of the cell fluorescence (RFU) (B, D , F, H and J) after incubation of HL-60 (A and B), K562 (C and D), MOLT-3 cells (E and F), PBMC of patient with NHL (G and H) and PBMC of healthy donor (I, J) incubated in the presence of corresponding siRNAs are shown. Data were obtained via flow cytometry. Fifteen thousand events were counted in each sample. Mean values (±SD) from three independent experiments are presented.
Intracellular localization of the Ch-siRNAs after accumulation in the cells we analyzed using In Cell Analyzer 2000 with Z-stack imaging feature (Figure 3). Obtained microscopic data indicates that Ch-siRNA accumulated in the cells while free siRNA does not penetrate into them, which is consistent with flow cytometry data. The image clearly shows that Ch-siRNA penetrated into the cell is located within the cytoplasm, and does not remain bound to the membrane.
Figure 3.Accumulation Alexa 488-labeled siRNAs in K562 cells. The cells were incubated for 4 h with cholesterol-containing siRNA Ch6-siRNA. non-modified siRNA or without siRNA (control). Three-channel (BGR) pictures were obtained using staining by HCS NuclearMask Blue stain, staining DNA (B); Alexa 488, attached to cholesterol-modified and non-modified siRNAs (G) and CellMask Deep Red, staining plasma membranes (R).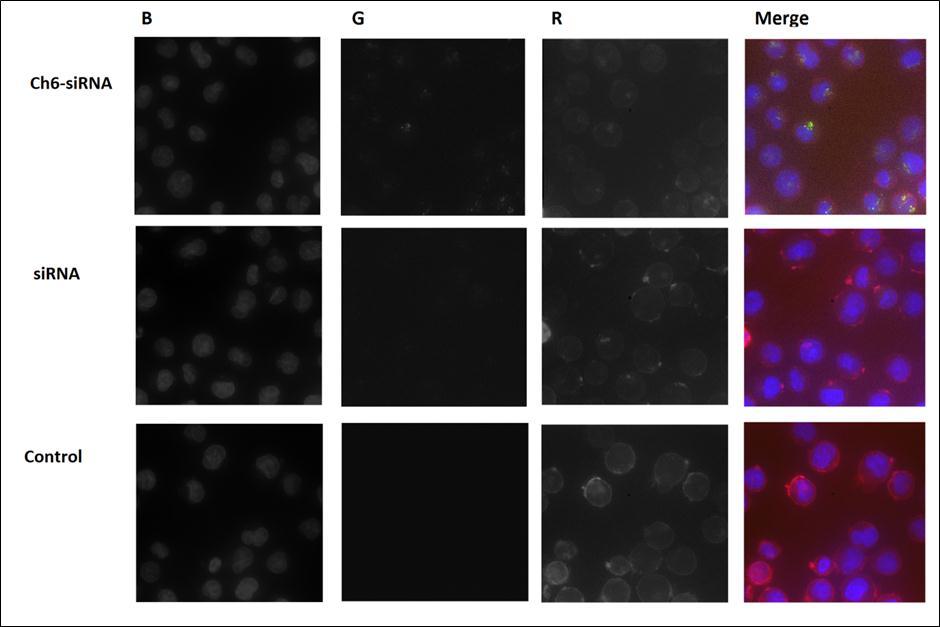
Discussion
In current study we showed that Ch-siRNAs efficiently penetrate both into the normal and malignant cells of hematopoietic origin. The augmentation of the linker length between siRNA and cholesterol from 3 to 10 carbons leads to increased uptake of the conjugates by K562 cells. The mechanism of Ch-siRNA uptake by the cells is not clear enough. However, it could be suggested that the mechanism of internalization of Ch-siRNA into cell in the first stage include "anchoring" of the conjugates in the membrane because of lipophilic nature of the cholesterol. Thus, augmentation of the linker length may facilitate this process by increasing the distance between the negatively charged siRNA and cholesterol residue. Previously we have found that the elongation of the linker length from 10 to 12 methylene units resulted in the decrease of silencing activity of Ch-siRNA in KB-8-5 cells9. Thus, Ch10-siRNA is optimal for the transfection of leukemic cells.
All types of tumor cells of hematopoetic origin under the study efficiently accumulate Ch-siRNA. The localization of Ch-siRNA in the cytoplasm as revealed by microscopy of K562 cells allows to expect the internalized siRNA to participate in RNA interference and to silence the target gene expression. However, the limitation of this study is that the cells under the study are not characterized by overxpression of our MDR1 gene - the target gene for the designed Ch-siRNA and the biological effect was not studied. Nevertheless, the data obtained previously on KB-8-5 carcinoma cells demonstrate that Ch-siRNA conjugates can effectively silence the target gene when accumulated in the cells and exhibit therapeutically significant effects.
Conclusion
Conjugate siRNA and cholesterol with linker length of 10 carbons is optimal for the transfection of cells of hematopoietic origin, including leukemic cells. The use of siRNA conjugates and cholesterol may be a promising method for the delivery of siRNA in blood cells, which is essential for creating drugs based on siRNA for the treatment of hematological malignancies.
Acknowledgements
The authors acknowledge Marat Kasakin for MS analysis, Ms A. Iglina for her assistance in the synthesis of the conjugates, Kolesnikova M.A. and Prof. Pospelova T.I. for providing blood samples. This work was supported by the Russian Foundation for Basic Research (no. 14-04-00869-a).
References
- 1.Song E, Zhu P, S K Lee, Chowdhury D, Kussman S. (2005) Antibody mediated in vivo delivery of small interfering RNAs via cell-surface receptors. , Nat. Biotechnol 23(6), 709-717.
- 2.B J Li, Tang Q, Cheng D, Qin C, F Y Xie. (2005) Using siRNA in prophylactic and therapeutic regimens against SARS coronavirus in Rhesus macaque. , Nat. Med 11(9), 944-951.
- 3.Jankovic R, Radulovic S, Brankovic-Magic M. (2009) siRNA and miRNA for the treatment of cancer. , J BUON 14(1), 43-9.
- 4.M, C A Hrycyna, P V Schoenlein, U A Germann, Pastan I. (1995) Genetic analysis of the multidrug transporter. , Annu Rev Genet 29, 607-49.
- 5.Landry B, H M Aliabadi, Samuel A, Gül-Uludağ H, Jiang X. (2012) Effective non-viral delivery of siRNA to acute myeloid leukemia cells with lipid-substituted polyethylenimines. 10-1371.
- 6.He W, M J Bennett, Luistro L, Carvajal D, Nevins T.Discovery of siRNA lipid nanoparticles to transfect suspension leukemia cells and provide in vivo delivery capability. (Sep 3, 2014) Mol Ther. 10-1038.
- 7.J H, Mok H, Y K Oh, T G Park. (2009) siRNA conjugate delivery systems. , Bioconjug Chem 20(1), 5-14.
- 8.Soutschek J, Akinc A, Bramlage B, Charisse K, Constien R. (2004) Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAs. , Nature 7014, 173-178.
- 9.N S Petrova, I V Chernikov, M I, I S Dovydenko, A G Venyaminova. (2012) Carrier-free cellular uptake and the gene-silencing activity of the lipophilic siRNAs is strongly affected by the length of the linker between siRNA and lipophilic group. , NAR 40(5), 2330-2344.
Cited by (6)
This article has been cited by 6 scholarly works according to:
Citing Articles:
Expert Opinion on Drug Discovery (2023) Crossref
Ahmed Abosalha, Waqar Ahmad, J. Boyajian, P. Islam, Merry Ghebretatios et al. - Expert Opinion on Drug Discovery (2022) Semantic Scholar
Expert Opinion on Drug Discovery (2022) OpenAlex
I. V. Chernikov, M. Meschaninova, D. Gladkikh, A. G. Ven'yaminova, M. Zenkova et al. - Russian journal of bioorganic chemistry (2021) Semantic Scholar
Russian Journal of Bioorganic Chemistry (2021) OpenAlex
Russian Journal of Bioorganic Chemistry (2021) Crossref
I. V. Chernikov, Ulyana A Karelina, M. Meschaninova, Aliya G Ven'yaminova, M. Zenkova et al. - Russian journal of bioorganic chemistry (2019) Semantic Scholar
Russian Journal of Bioorganic Chemistry (2019) OpenAlex
Russian Journal of Bioorganic Chemistry (2019) Crossref
