Soluble Biobased Substances in soil or Salicylic Acid on leaves affect the foliar pH and soil biovariability of Grapes-as explained by the NIR Spectroscopy of Litterbags and Teabags
Abstract
The purpose of the trial was to check the effects of two grapevine treatments on the plant activity and on the bioactivity and biovariability of the soil. An alkaline complex of Soluble Biobased Substances (SBS) was used in soil at 30 g per plant in a single solution. Salicylic Acid (SA) was used on leaves at 150 mg l-1 every two weeks at 50 ml plant. The plants were examined for their foliar pH and NIR spectra. The soil bioactivity was monitored by means of hay-Litterbag-NIRS (LBN) in combination with the Teabag Index (TBI), using rooibos and green tea that had been buried for 60 days. The evolution of the TBI presented here concerns the TBI-NIRS spectroscopic method used for discriminant analysis. A new algorithm was used to estimate the soil microbiome from the green Teabag spectra. The obtained results showed that the plants and the soil responded to the treatments. In fact, SBS, but not SA, lowered the leaf pH by 5%, an unexpected and original result. Both treatments increased the variability of the leaf composition, with a lower discrimination, based on the NIR spectra, from the Control (75%) to 44% (SA) and 38% (SBS). The TBI method, which is based on weights, was less efficient (67%) than the TBI-NIRS of rooibos (96%) or the LBN of hay (80%), but it was like the TBI-NIRS of green tea (74%). The LBN analyses indicated that the mycorrhizal index had increased by 8% in SA but had reduced by 7% in SBS, while both treatments reduced the activity of the microbes, which did not affect the soil respiration rate. The mineral N in the soil was substantially raised by about 11÷69%. The Taxa profiles showed marked deviations from the Control. Moreover, the SBS treatment reduced the Glomeromycota by 35%, which matched to the reduction in the mycorrhizal index. The most favored Bacteria from the treatments were Proteobateriaand Actinobacteria, with Mortierellomycota being the most penalized. Neither treatment affected the production, but both delayed the technological maturity by 9-11%, while the SBS retarded the phenolic maturity by about 18%. It has been concluded that a simple treatment of vines can affect the bioactivity in the leaves and berries as well as the biovariability of the soil.
Author Contributions
Academic Editor: Mohan Lal, Assistant Professor, Irrigation and Drainage Engineering department, College of Technology, G B Pant University of Agriculture and Technology, Pantnagar, India
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2023 Giorgio Masoero, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Healthy soils form the basis of any sustainable viticulture, and a central task is that of making the living medium that contains micro and macro-organisms, which perform many ecological functions and provide ecosystem services, more efficient 1. The resistance of cultivated plants to climate change can be summarized as the fight against thermal, water and saline stresses. The vine can be considered resilient to irrigation, so much so that it can be considered the norm. Such management practices as green pruning, colored nets, anti-hail nets and grassing are more and more frequently used to counteract thermal and solar excesses. The administration of humic acid and fulvic acids (HA) on the ground or even on the leaves can be included in the virtuous practices that can be evaluated. According to Mohamadineia et al. 2, the use of HA as a soil method is better than as a spray method in vineyards. However, as far as biostimulants are concerned, the foliar route is the preferred one. Popescu and Popescu 3 confirmed that foliar application with HA can improve the growth, yield, and berry quality attributes of grapevines, while according to Irani et al 4, biostimulants have a high potential for promoting fruit yield and the quality of grapevines in drought-prone regions. However, the burial of HA in the soil has shown other positive effects, for example, a nematocidal activity 5,6, while other authors fear negative consequences concerning the microbial biodiversity of the soil 7,8, as well as concerning the stability of the enzymatic system of the soil 9, although positive aspects have been highlighted for a potassium humate application and for soil properties, microbial activity, and Jerusalem artichoke yield components 10, and have been confirmed by Kumar et al. 11. The combination of HA and biostimulants has been experimented by various authors. Eman et al. 12minimized the use of nitrogenous fertilizers on grapevines by using HA and yeasts. Martinez et al. 13 showed that the joint application of compost, mineral fertilizer, and microbial inoculant can improve root development in table grapes (Vitis vinifera L.) and soil quality in integrated nutrient management programs.
Ibrhaim et al. 14, through the foliar application of HA and salicylic acid (SA), showed that SA was more effective than HA and recommended the use of SA as a foliar spray to enhance the productivity and nutritional quality of red sweet peppers. SA is a natural growth regulator of vascular plants that influences several physiological and metabolic processes. The foliar application of SA may increase the yield of vegetable species by reducing stress-induced growth inhibition 15. In general, HA and SA positively affect the growth and development of plants by acting as an organic-mineral fertilizer and a phytohormone, respectively. However, the responses of vegetable species and cultivars can differ significantly. In grapevines according to El-kenawy 16 , the best results were obtained with SA (500 ppm) + fulvic acid (500 ppm) without the addition of chitosan. In salt-stressed grapes the optima growth and yield were realized for a SA 1 mM dose 17. SA treatments of 6 and 9 mM have been found to be more effective in reducing salt stress over the plants of 41 B and 1103 P American grapevine rootstocks 18. In heat stress, an SA pretreatment maintained a higher Rubisco activation state 19 . Moreover, exogenous SA can promote the growth and Se uptake of grapevines under Se stress, with 250 ppm having been shown to be the most effective concentration 20. Using SA at 400 ppm three times was highly effective in palm 21 and in Citrus sinensis 22 . SBS-Humic and/or SA foliar treatments can affect fruit quality. Patel et al. 23 showed that among the considered treatments SBS @ 1.5 % was found to be more effective on Mango fruit than SA @ 1000-1550 and 2000 ppm, and significantly affected the maximum shelf life, TSS, total sugar, reducing sugar, non-reducing sugar and ascorbic acid, whereas the acidity, physiological loss in weight and spoilage were minimum.
Experimental procedure
As highlighted in the introduction, the studies on treatments with HA on the soil are not numerous and there are even fewer that concern soil biodiversity, while none are available about leaf treatments with SA. In this experiment, we have studied an alternative to HA, namely a non-commercial alkaline product derived from municipal biowastes, which is extracted from non-renewable sources. The alternative simple and non-joint effects of a soil treatment with SBS and an SA leaf treatment have been compared in this completely randomized experiment. The phenotypic evaluation on the plants took into consideration the leaf composition, as disclosed by the NIR spectra, and especially the leaf pH, the grape production, and the quality of the grapes pertaining to their technological and phenolic maturity. Indirect methods, based on the detection of the bioactivity of hay or of red and green Tea litters, coupled with NIR spectroscopy, were applied to evaluate the soil microorganisms. An attempt was also made to predict the bacterial and fungal biota from the NIR spectra of green Tea to evaluate the soil biodiversity.
Material and Methods
Description and management
The trial was conducted in the western Po valley (45.08180636477463, 7.958740094755527) on an adult vineyard of Malvasia red wine, located on a steep South oriented slope. A conventional management of the vineyard, following an integrated pest management protocol, is adopted, and it had not been fertilized in the three years before the research. The vines are pruned using the mono lateral guyot system with espalier training and at a density of 3.460 plants per hectare. During the vegetative period, the soil management involves three grass mulchings between the rows and three mechanical processings under the rows. The topsoil composition is reported in Table 1.
Table 1. Soil composition.| Component | Content |
| Sand (%) | 29.25 |
| Silt (%) | 50.65 |
| Clay (%) | 19.6 |
| pH soil | 7.9 |
| Ca carbonate total (%) | 6.4 |
| CaCO3 active % | 2.6 |
| Organic matter % | 1.355 |
| N Total % | 0.085 |
| C/N ratio | 9.3 |
| Cation_exchange_capacity (meq 100 g-1) | 17.8 |
| Ca exchangeable (meq 100 g-1) | 19.14 |
| Mg exchangeable (meq 100 g-1) | 1.65 |
| K exchangeable (meq 100 g-1) | 0.21 |
| Mg to K ratio | 7.9 |
| P assimilable (ppm) | 5 |
| Fe assimilable DTPA (ppm) | 29 |
| Fe assimilable ETPA (ppm) | 0 |
| Mn assimilable ETPA (ppm) | 10 |
| Cu assimilable (ppm) | 3 |
Treatments
An experimental alkaline (pH 8.40) complex of Soluble Biobased Substances (SBS) was used for the soil treatment to provide some humic substances, derived from biogas residues (Table 2). A total of 30 ml of the complex, diluted in 750 ml of water, was distributed over each six plants in a single solution on July 6, 2022.
Table 2. Elemental analysis of the Soluble Biobased Substances (SBS).| Constituent | % (res. 60°C)-1 | g 30 g(fluid)-1 |
| Dry matter | 86.58 | 3.90 |
| Solid volatile | 36.82 | 1.66 |
| N | 2.30 | 0.10 |
| C | 27.88 | 1.25 |
| H | 1.14 | 0.05 |
| S | 0.48 | 0.02 |
| C/N ratio | 12.1 | |
| pH 8.40 Dry matter 7.27% | ||
The humic acid extraction process was performed using compost obtained from the anaerobic digestion of municipal green biowastes. The process 24 involves hydrolyzing the compost in an k-alkaline solution and producing value added (SBS). The suspended solids were separated and the liquid solution, containing around 7% of dry matter, was used for the above-mentioned treatments. A typical solution of SBS has 4% of humic extract: humic acids + fulvic acids in a 3:2 ratio (which means 2.4% w/w humic and 1.6% w/w fulvic acids).
The other treatment considered in the experiment was based on Salicylic Acid (SA), a slightly soluble substance (2000 ppm) that was used at a concentration of 150 ppm. The solution reached pH 7.12 (-4% vs. water at pH 7.44) and was sprayed onto the leaves of six plants every two weeks at a dose of 50 ml plant.
Foliar measurements and NIRS analyses.
Two leaves per plant were sampled seven times from July 6 till September 6. The leaves were analyzed for the petiole pH using a Hamilton Peek Double-Pore F, / Knick combined 35 x 6 (LxØ) glass-plastic electrode, at two decimals. The NIR spectroscopy of the leaf blade was conducted in single scan mode using an NIR-SCiOTM smart device, as described in Masoero et al. 25. The foliar pH data were analyzed, by means of the XLSTAT 2019.4.1 26 software, according to a bifactorial ANOVA, with three treatments and seven times replicates. The chemical components of the leaves were predicted, by means of NIRS, through the application of previously published PLS models 27,28, calculated using WinISI-III (FOSS NIRSystem/Tecator, Infrasoft International, LLC) software.
Quality measurements
A total of 30 berries were collected randomly from each plant at two different times near the harvest and subsequently taken to the laboratory. Three subsets of 10 berries were composed and weighed, then the pulp, skins and seeds were divided and each berry was examined for pH using the Hamilton Double-Pore F electrode and by a bench refractometer for the degree brix, while a triplicate NIRS scan was applied to the pooled skins and seeds. The technological index was calculated as pH2 * brix, whereas the phenolic index was expressed as the ratio of the non-extractable to the extractable phenols and was predicted from the NIR spectra according Cugnetto et al. 29.
Set up, properties and spectra elaboration of the Litterbags and Teabags.
The litterbag-NIRS method was developed by Masoero et al. 30 and has been applied in numerous trials with biofertilizers 31,32,33,34. It consists of examining the NIR spectrum of ground alpine hay placed in a package of permeable sachets that have remained buried for 60 days. The chemometric elaboration of the spectra allows a discrimination of the various groups under study to be carried out. The method is that of the Partial Least Squares Discriminant (PLSD), which consists of first preparing the N*G incidence matrix, composed of N observations and G groups, in whose cells a value of 1 is assigned if an observation is present and 0 if an observation is absent. The PLS-Regression method is then used, and G equations fitted to the values 1 and 0 are calculated. The predictions obtained from the equations generate a matrix of values whose G columns are submitted to a linear discriminant analysis to ascertain the percentage of correct classifications (%Correct), which is the diagonal of the G2 confusion matrix. In our case, we used the WinISI-III software (Infrasoft) to obtain the PLS equations from the NIR spectra and the XLSTAT software 26 to perform the discriminant analysis with cross-validation of the predictions.
Based on previous equations 35, the NIR spectra allowed us to predict 23 Litterbag-NIRS variables. These predictions were the final bromatological composition parameters of the hay residuals in the litter in relation to a feeding value for ruminants. Furthermore, some soil properties previously related to spectra (NO3-N, NH4+N, Soil Induced Respiration) and the mycorrhizal index were estimated. Finally, in agreement with the proposal of Fontaine et al.36 and Blagodatskaya et al.37, it was possible to obtain an indirect evaluation of the microbial activity by comparing the initial composition of the hay and the final one of the litters. This scheme considers two conventional types of microbes: the so-called “r-” (r-labile-strategist) microbe, which refers to a negative allometric variation of substances that decrease during the decay, as opposed to a category of slow “k+” (k+ resistant-strategist) microbes, which refer to a positive allometry of the substances that increase in percentage in the litter. In the works of Masoero et al.33and Hett et al.34, it was highlighted that the components that formed last in the herbs (acid detergent fiber, acid detergent lignin, crude fiber, cellulose) are the first to degrade, while the substances that formed first(ash, hemicellulose, crude protein) are of type "k", whose relative presence in litters increase during the decay process.
The Teabag Index (TBI) method of Keuskamp et al. 38 involves burying two bags of Lipton green tea (EAN 87 22700 05552 5) and rooibos tea (EAN 87 22700 18843 8) for 90 days. At the end of the test, the two sachets are removed and weighed. In the TBI, two parameters are determined by means of two formulas: the stabilization factor TBI_S and the decomposition rate TBI_K. These parameters were elaborated as univariates by means of the ANCOVA model. In this work, for the first time, we have improved the TBI method by applying NIR spectroscopy to green and red tea residues. The spectra were elaborated by means of a PLS-D type procedure, exactly as was done for the hay Litterbags. At present, we cannot consider the allometry of the decomposition of the two types of tea with respect to the original ones, because there is a lack of chemical data to calibrate the procedure. Instead, starting from the NIR spectra of green tea, which were taken from the study of Staffolani et al 39in vineyards, it was possible to obtain a set that was highly correlated with the bacterial and fungal biomass. In that study, which was carried out on nine clusters of Nebbiolo vines, the triplicate samples of soil, kept in a refrigerated environment between 1 and 6°C, reached the Bioma Makers company laboratory within 72 hours of sampling. The analysis performed by the laboratory was based on the sequencing and characterization of the 16SrRNA gene and ITS markers of bacteria, fungi and Archaea using Illumina MiSeq technology. The results reported the presence of taxa expressed as a percentage of the total identified taxa. In the present work, the PLS equations, recalculated after the cited study (unpublished), resulted an R2 coefficient of 0.68 ± 0.13 for both Bacteria and Fungi, and these models were applied to the green tea spectra of the present study.
Results
Foliar pH and NIR spectra.
The SBS lowered the leaf pH by 5% (Table 3).
Table 3. Effects of the treatments on the leaf pH and % Classification based on the NIR spectra of the leaves.| Treatment | No. | pH | NIRS1-VR | % Class | P(%Class) | ||
| Control | 89 | 3.75 | a | 75% | a | 0.0001 | |
| SA-Salicylic | 105 | 3.72 | a | 0.47 | 44% | b | 0.0165 |
| SBS-Humic | 79 | 3.58 | b | 0.66 | 38% | b | 0.3446 |
| Total | 273 | 52% | 0.0001 | ||||
| ln (SA/C)% | -1% | -54% | |||||
| ln (SBS/C)% | -5% | -68% | |||||
This did not happen at the beginning of the test but after 10 days, and it continued until the end (Figure 1). It is worth noting that the pH trend during the summer reached its maximum around August 12, and then decreased .
Figure 1.Trend of the leaf pH at seven moments of the trial in the three treatments. (a>b…>i, P<0.05)
Based on the analyses of the NIR spectra (Table 3), it was observed that the treatments had a strong effect on the leaves, as demonstrated by the 1-VR coefficient, which reached values of 0.47 in the comparison Control vs. Salicylic and of 0.66 in the comparison Control vs. SBS-Humic. The treatments affected the confusion matrix and the correct classifications. In fact, when the Control was set at a 75% level, the classification dropped to 44% for Salicylic and 38% for SBS-Humic, although the latter was not significant (P = 0.34).
%Class = %Classification; 1-VR ≡ R-squares in cross-validation; a>b P<0.05.
This result originated from modifications in the components of the leaf blade (Table 4), with several decreases in hemicellulose (-17% and -15% for SA and SBS, respectively), crude protein (-5% and -1%) and dry matter (-1% and -2%). An increase was instead observed for the cellulose, ADF and not digestile NDF, so that the overall net effect was a rise in the Crop Maturity Index (+3% and +1%).
Table 4. Effects of the treatments on the leaf composition.| Leaf component | Mean Control | ln (SA/C) | ln (SBS/C) | ||
| Hemicellulose (%DM) | 6.29 | -17% | * | -15% | * |
| Crude Protein (%DM) | 7.36 | -5% | * | -1% | |
| Dry Matter ( %) | 32.03 | -1% | -2% | * | |
| NDF (%DM) | 50.41 | 0% | -2% | * | |
| Total Digestibility (%) | 69.56 | -1% | * | 0% | |
| Cellulose (%DM) | 35.74 | 2% | * | -1% | * |
| ADF (%DM) | 44.12 | 2% | * | 0% | |
| NDF not digestible (%DM) | 30.83 | 2% | * | 1% | |
| Crop Maturity Index | 2.68 | 3% | * | 1% |
Litterbags and Teabags.
The average classification rate (Table 5) was 80% from the NIRS of the Litterbags, 96% from the NIRS of the rooibos and 74% from the NIRS of the green tea. The calculations based on the weight loss were explanatory of the experiment, with an average classification of 67% (statistically significant different from the random value of 33%), but inferior to the level obtained from the NIR spectra. Therefore, it could be convenient to use rooibos as a marker of soil bioactivity as it can enhance the resolution power of the experiment.
Table 5. Discrimination analysis: effects of the Treatments on the % Classification (%Class) as obtained from the NIR Spectra of Litterbags and NIR spectra of Teabags.| Treatment | No. of Teabags | Weight of two Teabags (TBI_S,TBI_K) | Litterbags | Rooibos | Green tea | |||
| %Class | NS | %Class | NS | %Class | NS | %Class | ||
| C-Control | 4 | 60% | 36 | 72% | 18 | 89% | 18 | 67% |
| SA-Salicylic | 3 | 80% | 36 | 86% | 21 | 100% | 22 | 82% |
| SBS-Humic | 5 | 60% | 36 | 83% | 18 | 100% | 18 | 72% |
| Total-Average | 12 | 67% c | 108 | 80% b | 57 | 96% a | 58 | 74% bc |
| ln (SA/C) | 28% | 18% | 12% | 20% | ||||
| ln (SBS/C) | 0% | 14% | 12% | 8% | ||||
The two parameters of the TBI showed different features in relation to the acidic treatments. In fact, the regression of TBI_K on TBI_S, which was negative in the Control, became positive in SA and strongly positive in SBS (Figure 2), while at same time, TBI_S increased by 80% in SA, and the TBI_K factor increased by 116% in the SBS treatment (Table 6).
Figure 2.Regression of the TBI_K (Decomposition) on the TBI_S (Stabilization) parameters from the Teabag-Index in the three treatments (SBS-Humic, SA-Salicylic, C-Control).
| Treatment | TBI_S Stabilization | TBI_KDecomposition | regression(S/K) | |||
| C-Control | 11.2% | b | 0.59% | b | -0.1163 | c |
| S-Salicylic | 25.0% | a | 1.17% | b | 0.0212 | b |
| SBS-Humic | 12.7% | b | 1.88% | a | 0.0927 | a |
| Average | 16.3% | 1.2% | ||||
| Ln(SA/C) | 80% | 68% | ||||
| Ln(SBS/C) | 12% | 116% |
The Litterbag-NIRS appeared preferable as it allowed the mechanics of the results to be interpreted on the basis of well-established equations. The results of the litterbag decomposition (Table 7) have been very instructive. The microbial k+ resistant-strategists decreased to a great extent (-13 and -28% in SA and SBS, respectively), whereas the r-labile-strategists decreased less (-8 and -4%) and their r-k difference therefore rose, especially in SBS (+19 and +115%). Another sign of a lower microbial activity in the SBS-Humic treatment was the crop maturity index, which decreased by 11%. However, the soil respiration capacity increased (SIR +9 and +5%), albeit not significantly. The most penalized variables of the litter were the type "k+ resistant" ones (protein, ash, hemicellulose), which are linked to the k-strategists, while the "r-labile" variables, which represent the main components of the wall (crude fiber, ADL, NDF not-digestible, ADF, cellulose), were tendentially increased by the treatments. Interestingly, the Mycorrhizal index increased in S (+8%) but clearly decreased in SBS (-7%). A further result was a strong increase in the predicted N content of the soil, that is, of about 17% in S and 46% in SBS, with a prevalence in the NH4 form.
Table 7. Effects of the treatments on the Litterbag-NIRS variables.| Litterbag-NIRS variables | Type | Hay | Control | SA-Salicylic | SBS-Humic | P | Ln(SA/C) | Ln(SBS/C) |
| Microbes k+ "resistant-strategists" (%) | 7.45 a | 6.48 b | 5.38 c | <0.0001 | -13% | -28% | ||
| Protein (%DM) | k+ | 7.88 | 12.72 a | 12.23 a | 10.59 b | <0.0001 | -4% | -17% |
| Ash (%DM) | k+ | 6.77 | 19.54 a | 18.23 b | 17.02 c | <0.0001 | -7% | -13% |
| Hemicellulose (%DM) | k+ | 9.48 | 16.77 a | 15.02 b | 15.75 b | 0.001 | -10% | -6% |
| NDF digestible (%DM) | r- | 28.3 | 23.91 a | 22.96 b | 21.49 c | <0.0001 | -4% | -10% |
| NDF Digestibility (%) | k+ | 46.7 | 53.22 a | 51.22 b | 47.87 c | <0.0001 | -4% | -10% |
| Microbes r- "labile-strategists" (%) | 8.96 a | 8.27 b | 8.62 ab | 0.001 | -8% | -4% | ||
| Crop maturity Index | 1.02 a | 1.05 a | 0.90 b | <0.0001 | 3% | -11% | ||
| Total Digestibility (%) | k+ | 61.8 | 78.99 a | 78.13 b | 76.40 c | <0.0001 | -1% | -3% |
| N-free extracts (%DM) | r- | 46.5 | 49.39 a | 48.95 b | 48.61 c | <0.0001 | -1% | -2% |
| NDF (%DM) | r- | 60 | 44.91 a | 44.82 a | 44.89 a | 0.193 | 0% | 0% |
| Mycorrhizal index | 1.10 ab | 1.20 a | 1.02 b | <0.0001 | 8% | -7% | ||
| Energy (%DM) | r- | 16.95 | 15.69 c | 15.78 b | 15.91 a | <0.0001 | 1% | 1% |
| Lipids (%DM) | r- | 2.87 | 2.85 b | 2.88 a | 2.89 a | 0 | 1% | 2% |
| Cellulose (%DM) | r- | 38.35 | 22.05 b | 22.95 a | 22.20 b | 0.024 | 4% | 1% |
| ADF (%DM) | r- | 21.65 | 28.12 b | 29.80 a | 29.136 b | 0.001 | 6% | 4% |
| Substrate Induced Respiration (µg Cmic g-1 FW) | 119.3 a | 129.85 a | 125.34 a | 0.129 | 9% | 5% | ||
| NDF Non digestible (%DM) | r- | 32.3 | 21.00 c | 21.86 b | 23.39 a | <0.0001 | 4% | 11% |
| ADL-Lignin (%DM) | r- | 12.17 | 6.07 b | 6.85 a | 6.93 a | <0.0001 | 13% | 14% |
| NO3--N in soil (mg kg-1 DM) | 72.52 c | 80.55 b | 88.95 a | <0.0001 | 11% | 23% | ||
| Crude fiber (%DM) | r- | 12.98 | 8.67 b | 11.49 a | 9.48 b | 0 | 33% | 9% |
| NH4+-N in soil (mg kg-1 DM) | 3.15 b | 3.85 b | 5.32 a | <0.0001 | 22% | 69% | ||
| Difference r-k (%) | 1.50 b | 1.79 b | 3.23 a | <0.0001 | 19% | 115% |
The high-degree relationships between the NIR spectra and TBI parameters were assessed through a calibration-validation process (Table 8). In fact, it was possible to predict the two TBI-parameters from all the NIR spectra, but especially from the rooibos.
Table 8. Predictability of the TBI_S and TBI_K parameters from the NIR spectra of the two types of Tea and the Hay.| NIR spectra | Teabag Index (TBI) Parameters | N | Mean | SD | RSQ | SECV | 1-VR |
| Green Tea | S-Stabilization | 47 | 16.1% | 6.2% | 0.64 | 4.2% | 0.53 |
| Green Tea | K-Decomposition | 47 | 1.1% | 0.5% | 0.53 | 0.3% | 0.47 |
| Rooibos Tea | S-Stabilization | 41 | 18.5% | 6.4% | 0.79 | 3.6% | 0.69 |
| Rooibos Tea | K-Decomposition | 39 | 1.3% | 0.4% | 0.75 | 0.2% | 0.72 |
| Hay | S-Stabilization | 92 | 15.5% | 5.9% | 0.69 | 3.7% | 0.60 |
| Hay | K-Decomposition | 88 | 1.2% | 0.5% | 0.76 | 0.3% | 0.70 |
The abundance of Bacteria and Fungi predicted from the NIR spectra of the green tea (Table 9) was not different, but the Taxa profiles showed marked deviations from the Control. In particular, the SBS treatment altered the Fungi composition and showed a drop of 35% in Glomeromycota, which was correlated with the reduction in the mycorrhizal index. Among the Bacteria, the most favored were Proteobateria and Actinobacteria, while Mortierellomycotaappeared to be the most penalized.
Table 9. Effects of the SA-Salicylic / Control and SBS-Humic / Control treatments on the abundance of the Taxa in Phyla Bacteria and Fungi| Taxa / Phyla | Abundance of the Control | ln (SA/C)% | P | ln (SBS/C)% | P |
| Bacteria | |||||
| Proteobacteria | 96.9 | 3.2 | * | 3.2 | * |
| Actinobacteriota | 87.9 | 4.1 | * | 3.9 | * |
| Firmicutes | 67.8 | 2.8 | 1.3 | ||
| Bacteroidota | 25.7 | 4.2 | * | 3.9 | * |
| Planctomycetota | 8.3 | -6.4 | * | -7.0 | * |
| Chloroflexi | 12.0 | -5.1 | * | -4.3 | * |
| Verrucomicrobiota | 11.0 | 9.8 | * | 5.7 | * |
| Myxococcota | 11.1 | -0.7 | -0.3 | ||
| Acidobacteriota | 6.0 | -5.4 | -0.1 | ||
| Bdellovibrionota | 2.2 | 13.0 | * | 13.1 | * |
| Cyanobacteria | 0.7 | 2.4 | 13.3 | * | |
| Planctomycetes | 1.8 | 17.9 | * | 11.0 | * |
| Spirochaetota | 0.3 | 68.7 | * | 64.2 | * |
| Chlamydiae | 0.1 | 19.2 | -3.5 | ||
| Fungi | |||||
| Ascomycota | 260.6 | 3.9 | -6.3 | * | |
| Basidiomycota | 82.0 | 4.6 | -4.0 | ||
| Mortierellomycota | 14.0 | -8.3 | * | -5.5 | * |
| Glomeromycota | 6.0 | 10.0 | * | -35.0 | * |
| Mucoromycota | 3.9 | 6.0 | 4.0 |
However, the regression line of the SA effect paired with the SBS effect (Figure 3) was positive for the Bacteria, whereas it was negative for the Fungi, due to the deviations of the Glomeromycota
Figure 3.Regression of the Salicylic effect (SA, X axis) on the alkaline SBS-Humic effect (SBS, Y axis) for the Taxa of Phyla Bacteria (blue) and Taxa of Phyla Fungi (red).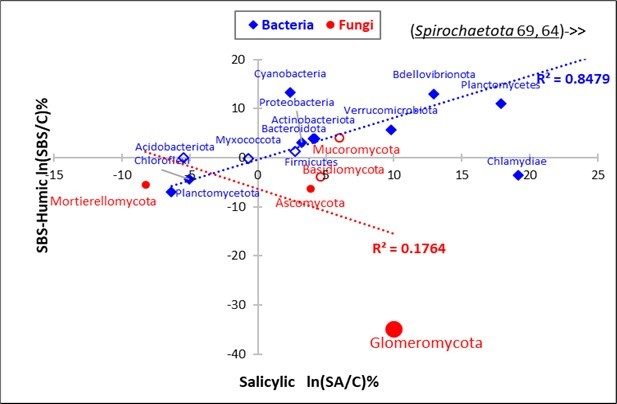
As far as the mineral soil N contents (NO3—N as well as NH4+-N) regressed on the Taxa are concerned, the results (not shown in the table) enhanced the negative coefficients more than the positive ones for Fungi, with a minimum for Ascomycota (-0.045). Similarly, the minima negative taxa from Bacteria were Planctomycetota(-0.06) and Chloroflexi (-0.04), as opposed to the most positive one (Mucoromycota, +0.05).
Production.
The treatments did not modify the productions (Table 10).
Table 10. Effects of the treatments on production| Treatment | No. of bounces per plant | Cluster weight(Kg plant) | Bounce weight(g) |
| C-Control | 27.5 | 5.2 | 186 |
| SA-Salicylic | 36.3 | 5.4 | 150 |
| SBS-Humic | 28.5 | 5.0 | 184 |
| ln(SA/C) | 28% | 4% | -22% |
| ln(SBS/C) | 4% | -4% | -1% |
Quality.
The berries sampled from the treated groups (Table 11) resulted to be smaller than the Control (SA -9% and SBS -4%). In addition to the reduction in the mass of the berries, it has been observed that the treatments delayed the technological maturity by around 10%. This was due to a state of greater acidity, with a -4% reduction in pH for both treatments, albeit for the same amount of sugar. As far as the NIR spectra of the skin is concerned, a higher variability was observed in the SBS group, which was classified at 35%, a non-significant value, and was significantly lower than the other more homogeneous Control (67%) and SA (61%) groups.
Table 11. Effects of the treatments on the technological maturity (TM) and on the Classification from the NIRS of skins.| Treatment | No. | W.10 berries | Brix° | pH | Index TM | % Class. NIRS Skin | |||||
| C-Control | 18 | 21.0 | a | 24.21 | a | 3.20 | a | 248 | a | 67% | a |
| SA-Salicylic | 20 | 19.2 | b | 23.65 | a | 3.09 | b | 226 | b | 61% | a |
| SBS-Humic | 18 | 20.1 | ab | 23.59 | a | 3.07 | b | 223 | b | 35% | b |
| P | 56 | 0.018 | 0.083 | 0.000 | 0.001 | 0.000 | |||||
| ln (SA/C) | -9% | -2% | -4% | -9% | -8% | ||||||
| ln (SBS/C) | -4% | -3% | -4% | -11% | -63% |
The seeds of the three groups were highly and equally characterized by the NIR spectra (about 87%) (Table 12). As for the components of the seeds with similar total phenols, the non-extractables were 6% lower for SBS. This corresponds to a lower phenolic maturity of around 18%, with a prevalence of proanthocyanidines (+9%).
Table 12. Effects of the treatments on the quality and phenolic maturity (PM) of the grape seeds.| Treatment | No. | NIRS%Class | Phenolic | |||||||||
| Total | Extractable | Non extractable | Phenolic Maturity | Proanthocyanidines | ||||||||
| C-Control | 60 | 88% | 1.51 | a | 0.35 | ab | 1.04 | ab | 2.29 | a | 0.47 | b |
| SA-Salicylic | 60 | 85% | 1.60 | a | 0.39 | a | 1.12 | a | 2.32 | a | 0.48 | b |
| SBS-Humic | 60 | 87% | 1.49 | a | 0.32 | b | 0.98 | b | 1.91 | b | 0.51 | a |
| P | 180 | 0.084 | 0.002 | 0.027 | 0.000 | 0.000 | ||||||
| ln (SA/C) | -4% | 6% | 11% | 7% | 1% | 3% | ||||||
| ln (SBS/C) | -2% | -1% | -7% | -6% | -18% | 9% | ||||||
Therefore, the treatment with humic acids generated a maturation delay of 18%.
Discussion
Soil traits.
How is it possible to imagine that a minimal treatment with acids could affect the bioactivity of the soil so much? All soil investigation systems show differences between treatments. In our case, the couple of TBI parameters derived from weighing the residues indicated that the TBI_S (Stabilization) factor, which depended on the green tea degradation, increased by 80% in SA, while the TBI_K (Decomposition) factor, linked to the rooibos degradation, increased by 116% in SBS. According to Mori 40,the two parameters are related in the positive mode, and greater emphasis should be placed on the TBI_S parameter, linked to green tea. Therefore, in the framework of the regression features exposed in Figure 2, the Control group with the strong negative trend should be considered an outlier, thus making the comparison results invalid. As far as the chemical composition and quality degradation of the two teas are concerned, the study by Duddigan et al.41 was based on Nuclear Magnetic Resonance Spectroscopy, where the variability depended on the relative abundance of the components that are traditionally considered labile (O-alkyl C and carbohydrate C) against more recalcitrant components (alkyl C and methoxyl C of lignin), and the tea was disregarded as a feed decomposed in a rumen, which is the approach adopted in the LBN method. The mass decay of the green tea was biphasic, with a rapid decline to 14 d, and the loss of soluble tannins, followed by a slower decline rate to 91 d, whereas the mass of rooibos tea declined at a slow rate over the whole incubation period. The percentages of decomposition at 91 d were 80% and 52% for green and rooibos tea, respectively.
Since the NIR spectra and residual weight were well correlated (Table 8), the conclusions obtained from the weight tend to converge, but the spectra, as they also reflected finer quality variations, showed higher power of discrimination. TBI only has agroecological purposes, and is only rarely used for agronomic purposes
In a framework of a multifaceted experiment, Tóth et al. 42 compared set-aside areas and cultivated areas in Hungary, which were cultivated using only rooibos buried for one month. The decomposition rate in the cultivated fields was 18.7%, whereas it was 25.83 (+32%) in the set-aside fields. However, in that study, the type of habitat did not affect the bacterial alpha diversity or community composition to any great extent (P = 0.122).
The Litterbag-NIRS variables predicted from the hay NIR spectra clearly indicate that SBS decreased the components of the k+ type (protein, ash, crop maturity index, NDF digestibility, hemicellulose) by about 28% and the r-type traits by about 4%. The SA treatment showed a similar but lower reduction of the k-variables (-13%), which was stronger in the r-variables (-8%). In short, the bioactivity detected from the quality of the hay litters was limited by both the SBS and SA. Our results are in agreement with those of Allison9 about the strong HA inhibition of enzyme activity, but are not in agreement with the results of a study on HA in pots of chicory 8, in which the increase in bacterial biomass was related to the availability of carbonaceous substrates for heterotrophic growth: in fact their dose threshold resulted to be 2 g kg-1 of soil, a value that is not comparable with the amendment used for our vines, which was instead around 0.125 g per kg of vineyard soil, that is, 16 times lower.
Contrary to expectations, no significant differences were found between the cultivated and set-aside soils in the Hungarian experiment Tóth et al 42, regarding bacterial alpha diversity and community composition. In the present trial, we have been able to profile the reduction in bioactivity in the litters through a biodiversity pattern in the bacterial and fungal taxa, a result that was beyond any expectation.
Our data confirm the rise in mineral N levels observed by Jin et al.7in Panax ginseng, which amounted to 90-125% after dosing humic and fulvic acid at 0.8 g⋅Kg− 1 soil, an 8 times higher level than that applied in the present work. Moreover, their finding indicated that the microbial diversity of the rhizosphere soil and bulk soil was reduced after the humic acidic treatments. The results of the present work confirm the reduction in Chloroflexi, Actinobacteria and Acidobacteria.
Leaf traits.
The treatments affected the foliar composition, regardless of whether they were administered per leaf or in the soil. The most surprising result was the lowering of the leaf pH following the treatment with SBS. This physiological measure is not usual in agronomy. A first comparative analysis on forty-nine Mediterranean species in different environmental conditions 43 was followed by observations on tree species located along 3300 km of a water gradient 44 and on desert species in China 45. From a physiological point of view, a lowering of pH can be determined by a stress condition, be it water or heat 43, such as an excess of UV-A, B 25 or by a reduction of the solar cycle over the years 46. A negative relationship between artificial mycorrhization and leaf pH has been observed on corn 47, sorghum 27, and on seven crops 43, in the sense that a biofertilization with mycorrhizae corresponded to a lowering of pH.
Product quality.
The injection of SA into grapevine berries is an elicitor of phenolics and hydrolytic enzymes that delay, or even inhibit the ripening of the berries 48. Ranjbaran et al.49, who sprayed SA on harvested berries,enhanced the increase of the total phenolic content of the berry skins after the shelf-life period. Similarly, in our trial, SA delayed the commercial maturity of the grape by 9%, but preserved the phenolic maturity of the seeds, and both these results could be interesting for winemakers to counteract climatic warming.
However, we cannot comment on the benefic effects obtained from massive SBS spraying on mango pointed out by Patel et al 23 (SBS @ 1.5% or SA @ 1500 ppm) or on the advantages in grape quality pointed out by Popescu and Popescu 3.
Conclusion
The systematic lowering of the leaf pH (-5%) by the SBS product containing Humic Acid is the first milestone that has been achieved in the present trial, and this result is in conflict with the expectations of a worse (-7%) predicted mycorrhization index of the vines. However, the better mycorrhization index obtained from the use of Salicylic Acid (+8%) left the pH unchanged (-1%). Both results need confirmation, possibly by resorting to artificial mycorrhization and verifying the effectiveness of the desired symbiosis in a batch experiment. However, the deviation of the leaf pH after a mild SBS soil treatment could be interesting to progress against the phytoplasmosis flavescence dorée.
The second milestone has been the confirmation of an always latent antagonism between chemical treatments and soil bioactivity. In the ongoing challenge of progress toward soil knowledge, the third milestone seems to us to be the demonstration of the efficacy of Litterbags-NIRS and TBI-NIRS in untangling the relationship between microbial bioactivity and biovariability quickly and indirectly.
Acknowledgments
The research was supported by the “Fondazione Cassa di Risparmio di Asti” Asti, Italy.
The AA are grateful to Luisella Celi and Daniela Vindrola from DISAFA for the elemental analyses
References
- 1.Giffard B, Winter S, Guidoni S, Nicolai A, Castaldini M et al. (2022) . Vineyard Management and Its Impacts on Soil Biodiversity, Functions, and Ecosystem Services.Frontiers in Ecology andEvolutionJul;22,10 .
- 2.Mohamadineia G H, Farahi M H, Dastyaran M E. (2015) Foliar and soil drench application of humic acid on yield and berry properties of ‘Askari’grapevine.Agricultural. 3(2), 21-7.
- 3.Popescu G C, Popescu M. (2018) Yield, berry quality and physiological response of grapevine to foliar humic acid application.BragantiaMar;22;77:. 273-82.
- 4.Irani H, ValizadehKaji B, Naeini M R. (2021) Biostimulant-induced drought tolerance in grapevine is associated with physiological and biochemical changes.Chemical and Biological Technologies in Agriculture8(1):. 1-3.
- 5.Nagachandrabose S. (2022) Nematode Management by Humic Acids. InSustainable Management of Nematodes in Agriculture, Vol. 1:Organic ManagementSep 13 , Cham: 135-155.
- 6.Nagachandrabose S, Baidoo R. (2021) Humic acid–a potential bioresource for nematode control.Nematology7;24(1):. 1-0.
- 7.Jin Q, Zhang Y, Wang Q, Li M, Sun H et al. (2022) Effects of potassium fulvic acid and potassium humate on microbial biodiversity in bulk soil and rhizosphere soil ofPanax ginseng.Microbiological ResearchJan 1;254:. 126914.
- 8.Valdrighi M M, Pera A, Agnolucci M, Frassinetti S, Lunardi D et al. (1996) Effects of compost-derived humic acids on vegetable biomass production and microbial growth within a plant (Cichorium intybus)-soil system: a comparative study.Agriculture. , Ecosystems &EnvironmentJul 58(2), 133-44.
- 9.Allison S D. (2006) Soil minerals and humic acids alter enzyme stability: implications for ecosystem processes.BiogeochemistryDec. 81, 361-73.
- 10.Moustafa Y, Hammam A, Haddad S. (2018) Potassium Humate Application and Cutting Immature Flowers affect Soil Properties. , Microbial Activity and Jerusalem Artichoke Yield Components.Journal of Soil Sciences and Agricultural EngineeringJan 9(1), 33-41.
- 11.Kumar D, Singh A P, Raha P, Rakshit A, Singh C M et al. (2013) Potassium humate: A potential soil conditioner and plant growth promoter.International. , Journal of Agriculture, Environment and 6(3), 441-6.
- 12.Eman A, El-Monem A, Saleh M, Mostafa E. (2008) Minimizing the quantity of mineral nitrogen fertilizers on grapevine by using humic acid, organic and biofertilizers.Research. , Journal of Agriculture and Biological 4(1), 46-50.
- 13.Martínez M M, Ortega R, Janssens M, Fincheira P. (2018) Use of organic amendments in table grape: effect on plant root system and soil quality indicators.Journal of soil science and plant. 18(1), 100-12.
- 14.Ibrahim A, Abdel-Razzak H, Wahb-Allah M, Alenazi M, Alsadon A et al. (2019) Improvement in growth, yield, and fruit quality of three red sweet pepper cultivars by foliar application of humic and salicylic acids.HortTechnologyApr 1;29(2):. 170-8.
- 15.M I Khan, Fatma M, T S Per, N A Anjum, N A Khan. (2015) Salicylic acid-induced abiotic stress tolerance and underlying mechanisms in plantsFront. , Plant Sci. Jun 30, 462.
- 16.El-kenawy M A. (2017) Effect of chitosan, salicylic acid and fulvic acid on vegetative growth, yield and fruit quality of Thompson seedless grapevines.Egyptian. , Journal of HorticultureJun 44(1), 45-59.
- 17.Roustakhiz J, Saboki E. (2017) Effect of salicylic acid on yield and yield component of grapevine (Vitis vinifera) under salinity stress condition.Intl. , J. Farm & Alli Sci6: 39-42.
- 18.Ekbic H B, Ozcan N, Erdem H. (2020) Impacts of salicylic acid treatments on salt resistance of some American grapevine rootstocks.Fresenius. , Environmental BulletinJan 29(2), 685-92.
- 19.Wang L J, Fan L, Loescher W, Duan W, Liu G J et al. (2010) Salicylic acid alleviates decreases in photosynthesis under heat stress and accelerates recovery in grapevine leaves.BMC plant biologyDec;10:. 1-0.
- 20.Li Z, Fan R, Peng X, Shu J, Liu L et al. (2022) Salicylic acid alleviates selenium stress and promotes selenium uptake of grapevine.Physiology and. , Molecular Biology of 28(3), 625-35.
- 21.Ahmed E F S. (2011) Response of Sakkoti date palms to foliar application of salicylic acid.MiniaJ. , Res. Dev 21(2), 305-316.
- 22.El-Rahman Abd, M, El-Masry S M A. (2012) Response of Valencia trees to foliar application of some vitamins, salicylic acid and turmeric extract.MiniaJ. , Agric. Res. Dev 32(5), 851-867.
- 23.Patel S J, Parekh D D, Rathwa J C, Sindha D J. (2020) Effect of foliar application of humic acid, salicylic acid and novel liquid on shelflife and quality of mango (Mangifera indicaL.) cv. , Amrapali.Journal of Pharmacognosy and 9(6), 1515-7.
- 24.Montoneri E, Koutinas M, Padoan E, Negro V, Licignano C et al. (2022) Integrated chemical and biochemical technology to produce biogas with a reduced ammonia content from municipal biowaste. Validating lab-scale research in a real operational environment.Environ. , Sci.: Adv 1(5), 746-68.
- 25.Masoero G, Cugnetto A, D’Amore F, Giovannetti G, Nuti M. (2020) . UV rays Decrease Foliar pH in Cress (Lepidium Sativum) and Modify NIR Spectrum.Journal of Agronomy Research3(2): 17-27.
- 26.Addinsoft. (2021) XLSTAT statistical and data analysis solution. , New York, USA. https://www.xlstat.com
- 27.Masoero G, Peiretti P G, Cugnetto A, Giovannetti G. (2018) Raw pH fall-out as a sign of a mycorrhizal modifier ofSorghumsudanensis.Journal of Agronomy Research1:(2):. 1-11.
- 28.Peiretti P G, Masoero G, Tassone S. (2020) Near infrared reflectance spectroscopy (NIRS) evaluation of the nutritive value of leaf and green pruning residues of grapevine (Vitis viniferaL.). In:Grapevines at a Glance. , NY, Chapter 3, 67-89.
- 29.Cugnetto A, Lajolo L, Vitaloni G, Sarasso G, Borgogno Mondino EC et al. (2021) Vineyard clusters monitored by means of Litterbag-NIRS and Foliar-NIRS spectroscopic methods.Journal of Agronomy Research3(2):. 39-56.
- 30.Masoero G, G Delmastro M Cugnetto A, Nuti M. (2018) NIRS footprint of bio-fertilizers from hay litter-bags.Journal of Agronomy Research1:. 22-33.
- 31.Volpato S, Masoero G, Giovannetti G, Nuti M. (2020) Arbuscular Mycorrhizal Biofertilizers sources in the potato (Solanum tuberosum) plant show interactions with cultivars on yield and litter-bags spectral features.Journal of Agronomy Research2(4):. 10-17.
- 32.Baldi E, Toselli M, Masoero G, Nuti M. (2020) Organic and symbiotic fertilization of tomato plants monitored by Litterbag-NIRS and Foliar-NIRS rapid spectroscopic methods.Journal of Agronomy Research3(1):. 9-26.
- 33.Masoero G, Oggiano P, Migliorini P, Taran A, Nuti M et al. (2021) Litterbag-NIRS to forecast yield: a horticultural case with biofertilizer effectors.Journal. of Soil Science and Plant Nutrition22: 186-200.
- 34.Hett J, Neuhoff D, Döring T F, Masoero G, Ercole E et al. (2022) Effect of multi-species microbial inoculants on early wheat growth and soil microbial activity.Agronomy MDPIApr. 12(4), 899.
- 35.Baldi E, Gioacchini P, Montecchio D, Mocali S, Antonielli L et al. (2021) . Effect of Biofertilizers Application on Soil Biodiversity and Litter Degradation in a Commercial Apricot Orchard.Agronomy11: 1116.
- 36.Fontaine S, Mariotti A, Abbadie L. (2003) The priming effect of organic matter: a question of microbial competition?Soil. Biol.Biochem.35: 837-843.
- 37.Blagodatskaya E V, Blagodatsky S A, Anderson T H, Kuzyakov Y. (2009) Contrasting effects of glucose, living roots and maize straw on microbial growth kinetics and substrate availability in soil.EJSS60:. 186-197.
- 38.Keuskamp J A, Dingemans B J, Lehtinen T, Sarneel J M, Hefting M M. (2013) Tea Bag Index: a novel approach to collect uniform decomposition data across ecosystems.Methodsin Ecology and EvolutionNov;4(11):. 1070-5.
- 39.Staffolani N, Stuardi M, Masoero G, Guidoni S. (2023) Esplorazione del microbiota del suolo di vigneti e sue relazioni con la pianta. S.O.I. , Torino 233.
- 40.Mori T. (2022) Is the Tea Bag Index (TBI) Useful for Comparing Decomposition Rates among Soils?Ecologies. , Nov 3(4), 521-9.
- 41.Duddigan S, Shaw L J, Alexander P D, Collins C D. (2020) Chemical Underpinning of the Tea Bag Index: An Examination of the Decomposition of Tea Leaves.Applied and Environmental Soil Science6085180:. 1-8.
- 42.Tóth Z, Hornung E, Báldi A. () Effects of set-aside management on certain elements of soil biota and early stage organic matter decomposition in a High Nature Value Area. , Hungary.Nature ConservationAug 29, 1-26.
- 43.Masoero G, Cugnetto A. (2018) The raw pH in plants: a multifaceted parameter.Journal of Agronomy Research1(2):. 18-34.
- 44.Lu H, Sun M, Ma Y, Si M, Xie J et al. (2022) Contrasting patterns of variation in foliar pH between a woody species and an herbaceous species along a 3300 km water availability gradient in China.Catena. 1-216.
- 45.Luo Y, Yan Z, Liu S, Chen J, Li K et al. (2021) Variation in desert shrub foliar pH in relation to drought and salinity in Xinjiang, China.Journal of Vegetation ScienceMay;32(3):e13031.
- 46.Masoero G, Cugnetto A, Sarasso G, Giovannetti G, Nuti M. (2019) Sunspots are correlated with foliar pH in grapevine.Journal of Agronomy Research2(3):. 31-41.
- 47.Masoero G, Giovannetti G. (2015) In vivo Stem pH can testify the acidification of the maize treated by mycorrhizal and microbial consortium.Journal of Environmental. , Agricultural Sciences 3, 23-30.
Cited by (2)
This article has been cited by 2 scholarly works according to:
Citing Articles:
Silicon (2024) Crossref
Silicon (2023) OpenAlex
