The Initial Result Of The First Living Donor Lung Transplantation Case In Vietnam
Abstract
Lung transplantation is an established treatment option for patients of end-stage lung diseases. Leading indications include chronic obstructive pulmonary disease, idiopathic pulmonary fibrosis and bronchiectasis, pulmonary arterial hypertension. Living donor lung transplantation (LDLT) was indicated for patients who experience a decline in physical condition and have limited life expectancy and had the better result than from brain dead. The first case of LDLT in Vietnam is 7 year olds boy with difuse congenital bronchiectasis, chronic respiratory failure and cor pulmonale. The right and left lower lobes from father and relative uncle are implanted in a recipient in place of whole right and left lung. The initial result of this recipient showed that lung function recuperated quickly and had no early complications. After 12 months surgery, the recipient had normal physical exercise capacity, subclinical tests in normal limits and no respiratory symptoms, opportunistic infection.
Author Contributions
Academic Editor: Feng Ding, School of Basic Medical Sciences, Wenzhou Medical University, Chashan Campus, Wenzhou 325635, P. R. China
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2018 Do Q, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Lung transplantation is an established treatment option for patients of end-stage lung diseases2, 6, 10. The first lung transplantation case was performed by Hardy in the USA in 1963. Living donor lung transplantation (LDLT) was was introduced by Starnes and colleagues for patients who experience a decline in physical condition and have limited life expectancy14, 15, 18. However, living donor single-lobe transplantation was not good results and developed bilateral LDLT, in which two healthy donors donate their right or lef lower lobes. It was the result for developing LDLT with both two sides3, 4, 19, 20. LDLT was suitable for children and small adults, and LDLT has been applied almost exclusively in patients with cystic fibrosis1, 4. Now this technique is applicable in cases of restrictive, obstructive, infectious, and hypertensive lung disease in both pediatric and adult patients when the size matching is acceptable4, 6, 8, 16. The results of bilateral LDLT have been as good as or better than those of conventional cadaveric lung transplantation10, 13, 16. In Vietnam, the first case of LDLT was performed on the 21st February 2017 in the Military Hospital 103 (University hospital of the Military Medical University). We report the initial result of this living donor lung transplantation case in Vietnam.
Case Presentation
A 7 years old boy was diagnosed of difuse congenital bronchiectasis with chronic respiratory failure and cor pulmonale. At the age of 2 months, he had many respiratory infections. When he was 2 years old, respiratory infection became more frequently, he has been usually hospitalized due to uncontrolled recurent respiratory infection. His main complains were frequent cough with sputum and shortness of breath. On September 2016 patient hospitalized with fever, cough with sputum purulence, severe dyspnea (mMRC of 4 points), cyanosis, difuse crackles in the both lungs, and severe malnutrition (height: 90 cm, weight: 11 kg, BMI: 13.5). The subclinical tests: PaO2: 77 mmHg, PaCO2: 46 mmHg, pH 7.46, SaO2 95% and HCO3-26.7mmol/l. CT-scan: severe difuse bronchiectasis; ECG: sinus tachycardia, right atrial hypertrophy; echocardiography: right ventricle enlargment, pulmonary arterial hypertension (47 mmHg). Blood group: O (+), Rh (D) (+); HLA class I: A02; A24; B13, B52; HLA class II: DRB1 01; DRB1 14. (Image 1)
Image 1.The chest CT-scan image of receipient (difuse bronchiectasis)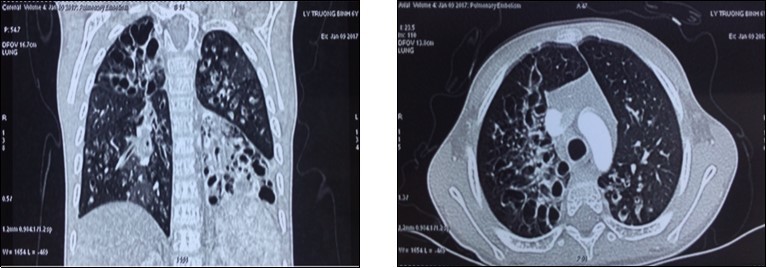
The donors were her biological father, aged 28 years, the weight of 50 kg, height of 165 cm, HLA class I: A02; A24; B46, B52; HLA class II: DRB1 01 and DRB1.14 and real uncle, aged 30 years (Height: 157 cm,weight: 50 kg), HLA class I: A02; A24; B46, B52; HLA class II: DRB1 01; DRB1.14. HLA class II and HLA class I of two donors were suitable with the receipient by the following portions of 100% and 75%.
(BMI: Body mass index; PaO2: Partial pressure of arterial oxygen; PaCO2: Ppartial pressure of arterial carbon dioxide; SaO2: arterial oxygen saturation:, CT: Computed tomography; HLA: Human Leucocyte Antigen.)
The technique of lung transplatation: The technologies used in this recipient accepted by ethical council of Vietnam Health Ministry and followed ethical disciplines of International Society for Heart and Lung Transplantation (ISHLT). The recipient and two donors have concerns and accepted surgery. Before the operation, the receipient and donors were examined and undergone subclinical tests to evaluate the function of their organs7, 12. The receipient was treated respitatory infection and no infection before surgery. Three surgical teams perform this operation. The recipient and the right-side donor are brought to the operating room at the same time. The lef-side donor is brought to the operating room 30 minutes later. The right lower lobectomy from uncle and a lef lower lobectomy from father. The evaluation of anatomic and functional size matchings between the receipient and three donors based on the standards of Japanese authors (Date H. et al.)4. The recipient is anesthetized and intubated with a single-lumen endotracheal tube. Afer the bilateral pneumonectomy, the right lower lobe is implanted frist, followed by implantation of the lef lower lobe. After the bilateral implantations are completed, both lungs have been reperfused and ventilated, the patient is gradually weaned from cardiopulmonary bypass. The total surgical time in the recipient was 10 hours.
Early outcome: After the operation, the patient was ventilated via endotrachial tube, controlled the status of respiration, hemodynamics and draignage care. Recipient was weaned off ventilator in 36 hours. Postoperative immunosuppression consists of triple drug therapy with neoral, cellcep, and prednisolone. We judge acute rejection on the clinical, radiographic fndings, other subclinical tests and control opportunistic infection. At 1, 3, 6, 9 and 12 months after LDLT, the recipient has recovered lung function very fast and there were not any early complications and adverse events after the operation. Now the recipient had normal physical exercise capacity, subclinical tests in normal limits and not respiratory symptoms, opportunistic infection and no evidence of rejection. (Image 2)
Image 2.The chest X-ray images at the first (a), 7th day (b) and 12th month (c). CT-scan images at the 3th day (d) and 8th day (e) after lung transplantation.
Discussion
In recent years, the number of lung transplantation patients has increased remarkbly with the trend to bilateral lung transplantation more than single lung transplantation. The sources of donors are extended, with non-heart beating donors, ex-vivo lung perfusion and living donors. LDLT is a last option chosen to save critically ill patient with end-stage pulmonary disease who cannot wait for organs from cadaveric donor4. In our receipient, the indication of lung transplantation was resonable because he always had recurent respiratory infection, decreased PaO2, increased oxygen requirements, hypercapnia, pulmonary arterial hypertension. Two donors were relative and young. The anatomic and functional size matchings between the receipient and two donors played a very important role in the success of this lung transplantation case. We use a formula to estimate graft FVC (Forced vital capacity) on the basis of the donor’s measured FVC and the number of pulmonary segments being implanted4. The right lower lobe FVC of uncle estimated 0.71 (L), the left lower lobe FVC of father estimated 0.8 (L), the receipient FVC estimated 0.98 (L). Total FVC of the two grafs/ FVC of the recipient was 1.5 (according to the Japanese authors, it is acceptable when this ratio > 0.45). Therefore, after vetilation these 2 lung lobes expanded nearly completly with the size of the paient’s chest cavity and by the 7th day, both 2 lung lobes had expanded completly. At the 1st, 3th, 6th , 9th and 12th month, the chest X-ray image are normal. The clinical change of the recipient: fever immediately after operation and recovered in 2 days, sputum in 5 days, oxygen therapy in 5 days; weaned off ventilator in 36 hours. After 4 days, he was withdrawn from thoracic drainage and could walk himself in the room at the 6th day after the operation. In the 1st, 3th, 6th, 9th and 12th month, he had normal physical activities, not any clinical symptoms (now he returns to school and takes part in all activities as his classmates), subclinical tests in normal limits and no evidence of opportunistic infection or rejection.
The clinical and subclinical datas have shown that the receipient has recovered lung function very fast and no complications after transplantation. Previous studies have reported some complications in the recipient after lung transplantation, such as pulmonary edema, acute rejection, anatomous airways, chronic rejection and opportunistic infection, especially lung transplantation from brain dead donor4, 5, 11. The best initial result in our receipient may relate to some factors: young and relative donors (≤ 30 years old); the anatomic and functional size matchings, as well as the HLA matchings between the receipient and two donors. Two donors had no complication in and after the operation. They recovered after 1 week and had normal lung function and return their normal life after 3 months. In previous studies, there was no mortality in live lobar donors and 15–20% donors suffered some kind of morbidity which is acceptable and similar to the standard lung resection8, 9, 17.
Conclusions
In the first case of LDLT in children in Vietnam, the initial results have shown that lung function recuperated quickly and had no early complications. After 3 months’ surgery, the recipient had normal physical exercise capacity, subclinical tests in normal limits and no respiratory symptoms, opportunistic infection.
References
- 1.Antonoff M B, Alexander Patterson G. (2016) Bilateral sequential lung transplantation: Technical aspects, Lung transplantation: Principles and Practice, Edited Vigneswaran. , NewYork 151-158.
- 2.Christie J D, Edwards L B, Kucheryavaya A Y. (2010) Registry of the International Society for Heart and Lung Transplantation: Twenty-Seventh Official Adult Lung and Heart-Lung Transplantation Report, J Heart Lung Transplant. 29-1104.
- 3.Date H, Aoe M, Sano Y. (2004) Improved survival after living-donor lobar lung transplantation. J Thorac Cardiovasc Surg. 128-933.
- 4.Date H. (2016) Living Related Lung Transplantation, Lung transplantation: Principles and Practice, Edited Vigneswaran. , NewYork 165-174.
- 5.Keller C A, Díaz-Gómez J L. (2016) Posttransplantation critical care management, Lung transplantation: Principles and Practice, Edited Vigneswaran. , NewYork 229-250.
- 6.Kotsimbos T, Williams T J, Anderson G P. (2012) Update on lung transplantation: programmes, patients and prospects, Eur Respir Rev. 21-126.
- 7.Kreider M, Kotloff R M. (2009) Selection of Candidates for Lung Transplantation. , Proc Am Thorac Soc 6, pp.
- 8.Mohite P N, Popov A F, Yacoub M H, Simon A R.Live related donor lobar lung transplantation recipients surviving well over a decade: still an option in times of advanced donor management. , Journal of Cardiothoracic Surgery 2013, 37.
- 9.Hayes D, Meyer K C. (2010) Lung Transplantation for Advanced Bronchiectasis, Seminars in respiratory and critical care medicine. 31, 123-138.
- 10.Orens J B, Garrity E R. (2009) General Overview of Lung Transplantation and Review of Organ Allocation. , Proc Am Thorac Soc 6, 13-19.
- 11.Paraskeva M A, Westall G P, Snell G I. (2016) Immunosuppression strategies in lung transplantation, Lung transplantation: Principles and Practice, Edited Vigneswaran. , NewYork 279-291.
- 12.Romána A, Ussettic P, Soléd A, Zurbanoe F. (2011) Guidelines for the Selection of Lung Transplantation Candidates, Arch Bronconeumol. 47(6), 303-309.
- 13.Silhan L L, Orens J B. (2014) Update to the guidelines for selection of lung-transplant candidates, Curr Respir Care Rep. 3, 103-111.
- 14.Starnes V A, Barr M L, Cohen R G. (1996) Living-donor lobar lung transplantation experience: Intermediate results. J Thorac Cardiovasc Surg. 112-1284.
- 15.Starnes V A, Barr M L, Schenkel F A. (1997) Experience with living-donor lobar lung transplantation for indications other than cystic fbrosis. J Thorac Cardiovasc Surg. 114-917.
- 16.Starnes V A, Lewiston N J, Luikart H. (1992) Current trends in lung transplantation: Lobar transplantation and expanded use of single lungs. , J Thorac Cardiovasc Surg 104-1060.
- 17.S M, R D Levy, McNeil K, J B Orensz. (2004) Lung transplant outcomes: a review of survival, graft function, physiology, health-related quality of life and cost-effectivenessLung transplant outcomes: a review of survival, graft function, physiology, health-related quality of life and cost-effectiveness, Eur Respir J. 24, 674-685.
- 18.Todd J L, Christie J D, Palmer S M. (2013) Update in Lung Transplantation. , Am J Respir Crit Care Med 190, 19-24.
