Primary Leiomyosarcoma of the Thyroid; a Case Report and a Review of Recent Literature
Abstract
A case report of primary thyroid leiomyosarcoma with literature review. It discusses diagnostic pitfalls, histologic criteria, and management options.
Author Contributions
Academic Editor: Esther Diana Rossi, catholic university.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2023 Maysan almegbel, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Leiomyosarcoma (LMS) is an uncommon soft tissue sarcoma (STS) accounting for only 10-20% of all STS 1. Primary LMS of the thyroid gland is an exceptionally rare malignancy comprising only 0.014% of primary thyroid malignancies 2, 3. Up to our knowledge there have been only 34 reported cases in English literature, a case of which was first reported in 1969 by Adachi M et al 2, 3, 4, 5, 6, 7, 7, 8, 9, 10. Here we present a case of primary LMS in a 42-year-old female who presented with progressive thyroid swelling over a few years.
Case Report
42 year old woman who is known to have obesity with a surgical history of sleeve gastrectomy, cholecystectomy, abdominoplasty, and bilateral breast augmentation with implants all occurring in 2016, presented to our hospital in March 2022 with left thyroid nodule that has been growing in size over the last few years. She denied history of dysphonia, dysphagia, any obstructive symptoms or symptoms suggestive of hyper or hypothyroidism. Aside from expected weight loss after her gastric sleeve the patient did not have any night sweats, anorexia or family history of malignancies. Her thyroid laboratory profile was normal and her ultrasound showed multiple left thyroid nodules. The largest one was 3.89 x 2.4 x 2.1 cm, solid, hypoechoic, lobulated nodule with no echogenic foci, Thyroid Imaging Reporting and Data System (TI-RADS) category was 5. Her fine needle aspirate (FNA) showed atypia of undetermined significance, cytologic atypia, bethesda system category III. She underwent left hemithyroidectomy with isthmectomy in June 2022, for which the histopathological diagnosis was reported as leiomyosarcoma, grade 3 with multiple margins are involved by the tumor with extrathyroidal extension. Pathological staging was pT3; therefore, She was called back for total thyroidectomy in early July 2022 and she was referred to the oncology and gynecology services for further workup. Right thyroid lobe pathology result was reported as thyroid papillary carcinoma, stage pT1a NX with negative margins.
Gynecological ultrasound (US) was requested in June 2021, which showed 2 uterine masses, one was submucosal and the other was subserosal fibroid. She also had a pap-smear in July 2022 which was negative for atypical cells. Her fluorodeoxyglucose-positron emission tomography (FDG-PET) scan at the time revealed bilateral subcentimetric cervical lymph nodes with no significant FDG activity and bilateral pulmonary nodules that were too small to be assessed on PET/ computed tomography (CT). Her abdominal solid organs did not demonstrate any metabolic activity. Chest CT was done in early August 2022, which showed multiple bilateral small pulmonary nodules, largest in size was 6mm. Additionally, right breast US showed simple cyst with posterior acoustic enhancement as well as few well-circumscribed avascular lesions with heterogeneous predominantly hypoechoic echotexture, the largest was 1.4x0.6cm. Also, there was heterogeneous predominantly hypoechoic avascular mass seen at the left outer quadrant measuring 2.1x1.2cm with internal calcifications and posterior shadowing. Breast US on the left side showed well-circumscribed avascular lesions seen at 3:00 superficially with heterogeneous predominantly hypoechoic echotexture measuring 1.2 x 0.3 cm. There was no architectural distortion, duct changes nor skin changes seen bilaterally and no enlarged axillary lymphadenopathy. Her evaluation was incomplete for both breasts with Breast Imaging-Reporting and Data System (BI-RADS) score of 0; thus, mammogram was recommended but she refused.
Moreover, an abdominal US was carried out which showed 2 hypoechoic hepatic lesions measuring 1cm each with moderate hepatic steatosis. Also the same uterine findings as previously mentioned were seen. Abdomen and pelvis CT were done as well to further evaluate the liver lesions. Her CT showed a non-cirrhotic liver with 1.3cm indeterminate caudate lobe hypodense non-enhancing lesion and 1.2cm hypodense none-enhancing lesion in segment 8. Her common bile duct was slightly dilated with prominent pancreatic duct. There is right breast 2.2 cm in calcified nodule stable since previous imaging. The uterus is bulky with multiple fibroids with one submucosal intracavitary measuring 2x1.7cm which is larger in size than initial US. There was also a left 2.8 cm ovarian cyst.
She also had an abdominal Magnetic Resonance Imaging (MRI) done before presenting to our oncology clinic which showed at least 13 bilobar hepatic lesions, the largest 3 lesions were noted in segments I, VIll and caudate lobe, measuring 2.1 cm, 2cm and 1.9cm respectively which were likely metastatic lesions. There was also 0.6cm distal pancreatic body lesion with MRI characteristics similar to the hepatic lesions concerning for metastasis.
She was seen in our oncology clinic in January of 2023. Her baseline tumor markers including AFP, CEA, CA19.9, CA125, CA15-3 were ordered and these were not elevated. A biopsy for one of her liver masses was arranged. Two weeks later the patient returned to clinic for her biopsy result, which showed non-cirrhotic liver with metastatic spindle cell sarcoma, positive for caldesmon and mitotic index of 60%, consistent with leiomyosarcoma.
She was consented for palliative chemotherapy in the form of Doxorubicin and Trabectedin (as per LMS-04 study 14). She had cycles as an inpatient every 3 weeks. Her disease assessment scans showed a partial response after 3 cycles. She continues on therapy without major toxicity.
Discussion:
LMS arises from smooth muscle cells in the vascular wall; thus, it is more common to present as uterine, gastro-intestinal or retroperitoneal malignancy rather than thyroid gland 3, 7. LMS is more common amongst females and mostly presents at older ages with a mean age of 64 years, the youngest patient reported was 32 years and the oldest was 90 years of age 3, 8. Likewise, our patient matches the more common gender and fits within the age range described. The only exception in reported cases was a pediatric one with a 6 year old child with congenital immunodeficiency whose LMS was attributed to Epstein Barr Virus infection (EBV) 3, 7. Other studies reported LMS in patients who were previously diagnosed with a different malignancy and received radiation therapy or surgery prior to presenting with LMS 3. Therefore, the influence of immunodeficiency, immunosuppression or radiation exposure should be taken into consideration when investigating the etiology of LMS.
Reported patients with LMS involving thyroid gland most commonly present with painless growing mass in the neck. Other symptoms include dysphonia, compression symptoms such as dysphagia/odynophagia or coughing, weight loss, and occasionally arm pain 3, 6. Although the usual presentation is a rapidly growing mass, our patient had noticed an increase in size of the swelling over a period of a few years and has not complained of any other associated symptoms. When looking into the thyroid laboratory profile, most patients had normal TSH, T4 and calcitonin. The exception to this is one case reported by Zou ZY et al. in which the patient had hyperthyroidism and another case reported by Takayama F et al. in which the patient had subclinical hypothyroid profile 10, 11, 12.
When diagnosing thyroid swelling, imaging studies have been helpful in differentiating malignant from benign tumors with US being used as an initial evaluation tool when assessing thyroid nodules. Tumors are categorized based on composition, echogenicity, shape, margin and echogenic foci into added scores determined by the TI-RADS system, which determines the need for FNA 13. Other imaging modalities include MRI and CT. However; radiological assessment does not distinguish between the types of thyroid cancer, and therefore FNA and biopsy are of crucial importance 3, 6. On a pathologic sample, LMS appears as spindled eosinophilic cells with elongated blunt-ended or cigar-like nuclei arranged in fascicles 2, 7 (Figure 1, Figure 2, Figure 3). Despite the unique microscopic morphology, LMS cannot be distinguished from other spindle cell tumors such as spindle cell variant of medullary thyroid cancer, anaplastic thyroid
Figure 1.Left thyroid Low power of the thyroid gland showing complete replacement of the thyroid by the neoplastic cells (H&E, ×4).
Figure 2.Left thyroid On a Higher power, few thyroid follicles are noted and the lesion is composed of spindle cells with sweeping fascicles (H&E, ×200).
Figure 3.Left thyroid The malignant looking cells are showing diffuse atypia, mitosis and hyper-chromatic nuclei, some remaining follicles could be noted as well (H&E, ×400).
Figure 4.Left thyroid Cells are diffusely positive for Caldesmon immunohistochemistry (IHC, x200)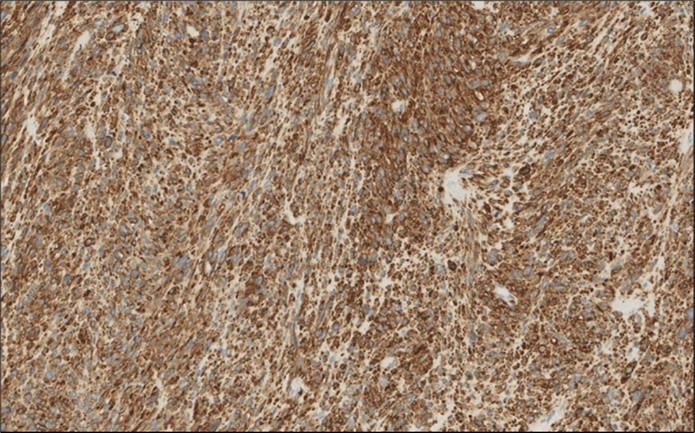
Figure 5.Liver biopsy Lower power of the liver core showing normal liver and part of the metastatic foci (H&E, x4)
Figure 6.Liver Biopsy Higher power showing the metastatic foci is almost identical to what is seen in the thyroid with the highly atypical spindle cells. (H&E, ×200).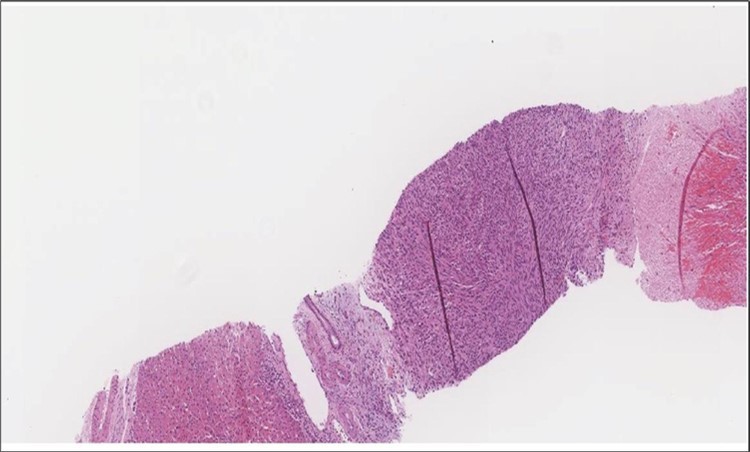
Figure 7.Liver Biopsy IHC of the metastatic foci shows diffuse positivity for Caldesmonstain (IHC,×200).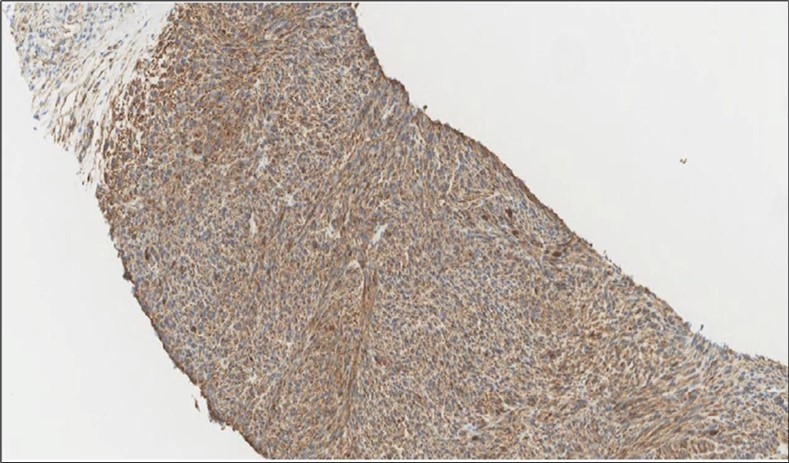
cancer, or other metastatic tumors without further testing with genetic or molecular studies 3, 7. Immunohistochemical testing plays a vital role in differentiating LMS from such similar tumors as it stains positive for vimentin and smooth muscle actin (SMA), and is variably positive for HHF35, desmin (50%-100%), and H-caldesmon 3, 6. Moreover, there were no reported cases where thyroid LMS stained positive for thyroglobulin, cytokeratin, calcitonin, chromogranin, or protein S100 3. Our patient’s left thyroid biopsy showed the characteristic microscopic features and was positive for SMA, desmin, caldesmon, P63 with patchy EMA (Figure 1, Figure 2, Figure 3, Figure 4) as well as her liver biopsy (Figure 5, Figure 6, Figure 7); whereas her right thyroid showed clear papillary thyroid cancer cell features (Figure 8, Figure 9).
Figure 8.Right thyroid Right thyroid lobe shows foci of papillary thyroid carcinoma. (H&E, x4)).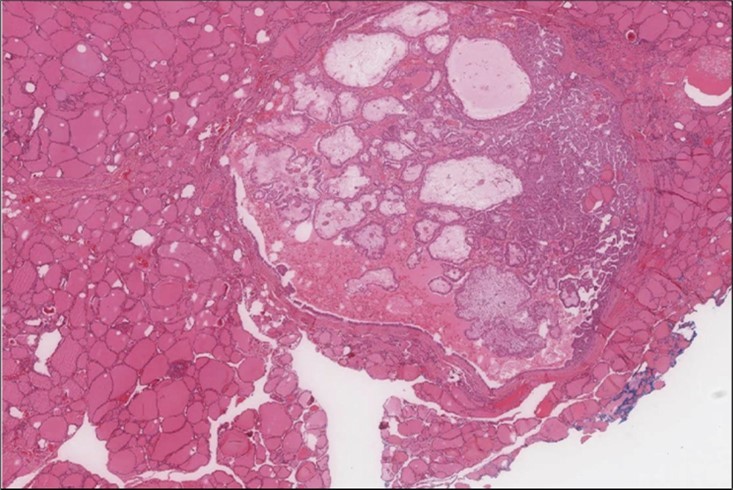
Figure 9.Right thyroid Higher power showing the characteristic nuclear features of papillary thyroid cancer nuclear enlargement, elongation and overlapping, chromatin clearing, margination and glassy nuclei. (H&E, ×200).
As a rare malignancy with a median survival of 5 months and 10% 1-year survival rate, primary thyroid LMS specific options of management are yet to be studied 3, 4. The current primary option is surgery with complete lobectomy vs total thyroidectomy and neck dissection to gain control of the tumor and symptomatic management; however no survival benefit has been shown with surgery 3, 6. Despite the advancements in oncology to date, there has been no proven chemotherapy agents or radiation therapies that result in reduction of thyroid LMS mortality. There has been some reported data on using imatinib, a tyrosine kinase inhibitor, on patients who had over-expression of the tyrosine kinase receptor, c-kit proto-oncogene, in uterine or thyroid LMS 2, 3, 7. One case by Day AS et al. reported using imatinib as postoperative adjuvant therapy but the patient died 6 months after the surgery 14. Another study evaluated using adriamycin and ifosfamide when the patient had local recurrence after surgery and reported a partial response 7. It is important to mention that these cases get treated as per therapy for metastatic STS with cytotoxic chemotherapy with doxorubicin remaining the first line agent of management 15. LMS02 trial showed encouraging results of using combined doxorubicin and trabectedin showing progression free survival (PFS) of 10.1 months and median overall survival (OS) of 34.4 months 16. A recent randomized multicenter phase III study (LMS-04) compared using doxorubicin vs. doxorubicin and trabectedin combination followed by trabectedin maintenance in managing metastatic or unresectable STS and uterine LMS. The study showed promising results with median PFS of 13.5 months vs. 7.3 when using doxorubicin alone. Moreover, median OS was 30.5 months in doxorubicin and trabectedin compared to 24.1 months for patients who had doxorubicin single agent 15.Our patient was started on doxorubicin and trabectedin and is still following in clinic one year after surgery and diagnosis of primary thyroid LMS.
Worthy of note that certain tumor markers were associated with more aggressive disease such as tumor-associated macrophages which is correlated with the expression of colony-stimulating-factor-1 found in LMS, loss of 10q and gain of 5p DNA copy were also found in aggressive primary, metastatic and recurrent LMS 1, 17. Those markers could aid in the development of appropriate targeted therapy. Another area of interest is tumor receptors that could be expressed on thyroid LMS such as estrogen and progesterone. Although sporadically found in LMS, hormonal therapy may have a role in treating thyroid LMS. All things considered, the behavior of LMS is immensely similar to the heterogeneous behavior of other soft tissue sarcomas as there are some indolent diseases and some aggressive ones 18, 19, 20. Biologically this is known by follow up and treatment. Most cases reported poor prognosis with metastasis or death in 1-51 months after diagnosis 21. We will know how our patient behaves with subsequent imaging after receiving systemic therapy over time.
Conclusions
We report a rare case of thyroid LMS with liver metastases treated with cytotoxic chemotherapy in the form of doxorubicin and trabectedin, with a partial response on therapy. To our knowledge, this is the first case reported from Saudi Arabia. Management of such rare cases requires careful review of pathology, and aggressive chemotherapy as prior reported cases had short median overall survival.
Abbrevations
LMS: Leiomyosarcoma; STS: soft tissue sarcoma; TI-RADS: Thyroid Imaging Reporting and Data System; FNA; fine needle aspirate; US: Ultrasound; PFG-PET: fluorodeoxyglucose-positron emission tomography; PET: positron emission tomography; CT: computed tomography; BI-RADS: Breast Imaging-Reporting and Data System; MRI: Magnetic Resonance Imaging; EBV: Epstein Barr Virus infection; SMA: smooth muscle actin; PFS: progression free survival; OS: overall survival.
References
- 1.Hu J, Rao U N, Jasani S, Khanna V, Yaw K et al. (2005) Loss of DNA copy number of 10q is associated with aggressive behavior of leiomyosarcomas: a comparative genomic hybridization study. Cancer Genetics and Cytogenetics. 161(1), 20-7.
- 2.Gupta A J, Singh M, Rani P, Khurana N, Mishra A. (2017) Primary Sarcomas of Thyroid Gland-Series of Three Cases with Brief Review of Spindle Cell Lesions of Thyroid. J Clin Diagn Res. Feb;11(2):ER01-ER04. doi: 10.7860/JCDR/2017/22907.9164. Epub 1, 28384879-5376816.
- 3.Vujosevic S, Krnjevic D, Bogojevic M, Vuckovic L, Filipovic A et al. (2019) Primary leiomyosarcoma of the thyroid gland with prior malignancy and radiotherapy: a case report and review of literature. World Journal of Clinical Cases. 7(4), 473.
- 4.Adachi M, Wellmann K F, Garcia R. (1969) Metastatic leiomyosarcoma in brain and heart. The Journal of pathology. 98(4), 294-6.
- 5.Bashir M T, Bradish T, Rasul U, Shakeel M. (2021) Primary thyroid leiomyosarcoma: a diagnostic and therapeutic challenge. BMJ Case Reports CP.
- 6.Cherry T J, Tasevski R, Li R. (2019) Primary thyroid leiomyosarcoma - case report and literature review. Surg Rehabil 3: DOI:. 10-15761.
- 7.Canu G L, Bulla J S, Lai M L, Medas F, Baghino G et al. (2018) Primary thyroid leiomyosarcoma: a case report and review of the literature. G Chir. Jan-Feb;39(1): 51-56. doi: 10.11138/gchir/2018.39.1.051. PMID: 29549682; PMCID: PMC5902146 .
- 8.Wei J, Yang J, Liang W, Xu C, Wen Y. (2019) Clinicopathological features of primary thyroid leiomyosarcoma without Epstein-Barr virus infection: A case report. Oncology Letters. 17(1), 281-7.
- 9.Mİ Şahin, Vural A, Yüce İ, Çağlı S, Deniz K et al.Thyroid leiomyosarcoma: presentation of two cases and review of the literature. , Braz J Otorhinolaryngol 2016, 715-21.
- 10.Mevio E, Mevio N, Cazzaniga M, Sbrocca M, Migliorini L et al. (2015) Leiomyosarcoma of the thyroid gland: A Review of the Literature and our experience. Journal of thyroid Cancer. 1.
- 11.Zou Z Y, Ning N, Li S Y, Li J, Du X H et al. (2016) Primary thyroid leiomyosarcoma: A case report and literature review. Oncology Letters. 11(6), 3982-6.
- 12.Takayama F, Takashima S, Matsuba H, Kobayashi S, Ito N et al. (2001) MR imaging of primary leiomyosarcoma of the thyroid gland. European journal of radiology. 37(1), 36-41.
- 13.Ross D S, Cooper D S. (2023) Diagnostic approach to and treatment of thyroid nodules. , UpToDate. Wellesley, MA: UpToDate. Last accessed
- 14.Day A S, Lou P J, Lin W C, Chou C C. (2007) Over-expression of c-kit in a primary leiomyosarcoma of the thyroid gland. European archives of oto-rhino-laryngology. Jun;264: 705-8.
- 15.Pautier P, Italiano A, Piperno-Neumann S, Chevreau C M, Penel N et al. (2021) LBA59 LMS-04 study: A randomised, multicenter, phase III study comparing doxorubicin alone versus doxorubicin with trabectedin followed by trabectedin in non-progressive patients as first-line therapy, in patients with metastatic or unresectable leiomyosarcoma-A French Sarcoma Group study. Annals of Oncology.
- 16.Pautier P, Floquet A, Chevreau C, Penel N, Guillemet C et al. (2015) Trabectedin in combination with doxorubicin for first-line treatment of advanced uterine or soft-tissue leiomyosarcoma (LMS-02): a non-randomised, multicentre, phase 2 trial. The Lancet Oncology. 16(4), 457-64.
- 17.Edris B, Weiskopf K, Volkmer A K, Volkmer J P, Willingham S B et al. (2012) Antibody therapy targeting the CD47 protein is effective in a model of aggressive metastatic leiomyosarcoma. Proceedings of the National Academy of Sciences 109(17), 6656-61.
- 18.Svarvar C, Böhling T, Berlin Ö, Gustafson P, Follerås G et al. (2007) Scandinavian Sarcoma Group Leiomyosarcoma Working Group. Clinical course of nonvisceral soft tissue leiomyosarcoma in 225 patients from the Scandinavian Sarcoma Group. Cancer. 109(2), 282-91.
- 19.Scurr M. (2011) Histology-driven chemotherapy in soft tissue sarcomas. Current treatment options in oncology. Mar;12: 32-45.
