Management of Locally Advanced and Metastatic Pediatric Nasopharyngeal Carcinoma. Experience of Children Cancer Hospital – Egypt
Abstract
Background.
Nasopharyngeal carcinoma (NPC) is a rare disease in the pediatric age group; it represents 1% of all pediatrics malignancies, however, it is the predominant malignancy arising in the nasopharynx in this age group. Although NPC is a chemo-radiosensitive disease yet, the optimal dose of radiotherapy and optimal timing of chemotherapy is still not standardized.
Methods.
This is a retrospective study including all the newly diagnosed pediatric NPC who were diagnosed and treated at the Children Cancer Hospital Egypt (CCHE) during the period from July 2007 to December 2012. All imaging studies (e.g., CT or MRI scans) were reviewed by a senior head and neck radiologist for proper staging and assessment of tumor response. Patients were staged according to AJCC staging system. Modified version of the Response Evaluation Criteria in Solid Tumor (RECIST) was used to assess response.
Results.
Twenty-six patients were diagnosed and received treatment as NPC in CCHE. Median age was 12 years (range 7.8-17 years). There was a male predominance. Eleven patients (42.3%) were stratified as stage 4, and 11 (42.3%) as stage 3. All patients received 3 cycles of neo-adjuvant chemotherapy followed by concurrent chemo-radiotherapy. IMRT was used to deliver radiotherapy in all patients. The overall response rate (CR and PR) to induction therapy was 73%, 19.3% had SD, while 7.7% had PD. By the end of the study, 18 patients (69.2%) were alive in CR, 5 patients (19.2%) had PD, and 3 patients (10%) lost for follow up. The Mean duration of follow up was 35 months, range 5-66 months. The 3 year OS and EFS and rates were 84.6% and 69.3 % respectively. OS for M0 was 91.3% and for M1 33.3% with statistical significance (p =0.032).
Conclusion.
Neoadjuvant chemotherapy followed by concurrent chemoradiotherapy using IMRT lead to good clinical end results with limited toxicity. Metastatic disease at presentation was identified as the adverse prognostic factor.
Author Contributions
Academic Editor: Ioannis Chatzistefanou, Aristotele University of Thessaloniki (A.U.Th)
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2015 Hany Abdel Rahman, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction:
Nasopharyngeal carcinoma (NPC) is rare in pediatric age group; however it is the most common malignancy arising in the nasopharynx in this age group1. NPC displays a distinct racial and geographic distribution, a reflection of its multifactorial etiology. It is rare in the United States and Western Europe 2, 3. In contrast, it is endemic in southern China. Intermediate risk regions include south east Asia, north Africa and the middle east. The incidence of nasopharyngeal carcinoma worldwide is two- to three folds higher in males compared with females 3.
Pediatric NPC is distinguishable from the adult form for its strong association with Epstein-Barr virus (EBV) infection, undifferentiated histology, the high incidence of loco-regionally advanced disease and the predominance of WHO grade III undifferentiated histology 2, 4, 5.
It is clinically staged according to the International Union Against Cancer (UICC) and the American Joint Committee on Cancer (AJCC) according to Tumor Node Metastasis (TNM) system which is the most important documented prognostic factor5.
Patient with early disease is recommended to be treated with radiation-therapy only, while patient with loco-regionally advanced or metastatic disease may receive induction chemotherapy followed by chemo-radiotherapy2. Undifferentiated NPC is very sensitive to radiotherapy, which is the mainstay of treatment. With radiotherapy alone, 5-year survival has been reported as 20–60% in most pediatric series 6, 7, 8. Induction chemotherapy has shown a higher response rates in patients with undifferentiated NPC9, 10, 11, 12, 13, 14.
The aim of the present study is to estimate the overall survival (OS) and the event free survival (EFS) of NPC patients treated at Children Cancer Hospital – Egypt (CCHE) during 5.5 years period. Furthermore, common toxicities, the effect of induction chemotherapy regimen followed by concurrent chemo-radiotherapy on survival will be investigated.
Patients and Methods
This is a retrospective study including all newly diagnosed pediatric NPC at Children Cancer Hospital Egypt CCHE during the time period from July 2007 to December 2012. The patients' profiles were systematically reviewed for characteristics including pathology, laboratory, radiological work up and documented toxicities. All imaging studies (e.g., CT and MRI scans) were reviewed by a senior head and neck radiologist for proper staging and assessment of tumor response.
Eligibility
Eligibility criteria included age < 18 years, histologically proven undifferentiated NPC (WHO types III). The American Joint Committee on Cancer (AJCC) staging system was used 6.
Patients’ Evaluation
Pretreatment evaluation included a complete history and physical examination, complete blood count, serum biochemistry tests (including evaluation of electrolyte levels, hepatic and renal function tests), CT or MRI scans of the head and neck region, chest and bone scan. During therapy weekly examinations and laboratory evaluations were performed. Tumor response was assessed by clinical examination with appropriate MRI imaging studies after completion of neoadjuvant chemotherapy and at the end of radiotherapy.
Response Criteria
Tumor response was evaluated using the modified version of the Response Evaluation Criteria in Solid Tumor (RECIST) which included volumetric measurement of the primary NPC, and assessment of associated adenopathy. Measurable and non-measurable metastatic foci in the lungs, bones and liver were also evaluated9.
Treatment
Neoadjuvant and concurrent chemotherapy:
Patients received 3 cycles of neo-adjuvant chemotherapy [Cisplatin/5FU: CDDP 100 mg/m2 day 1, and 5-FU 1000 mg/m2 continuous infusion day 1 -5], followed by concurrent chemo-radiotherapy with Cisplatin 100 mg/m2 as consolidation administered every 3 weeks.
Definition of Response Criteria
Complete Response (CR): complete disappearance of the tumor confirmed.
Partial Response (PR): at least 64% decrease in volume compared to the baseline.
Progressive Disease (PD): at least 40% increase in tumor volume compared to the smallest measurement obtained since the beginning of therapy.
Stable Disease (SD): neither sufficient shrinkage to qualify for PR nor sufficient increase to qualify for PD taking as reference the smallest disease measurement since the treatment started.
Radiotherapy
Radiotherapy planning was performed 2-3 weeks after the third course of neoadjuvant chemotherapy. A customized thermoplastic immobilization cast was used for all patients and a CT simulation was applied using iv contrast injection and a 3 mm slice thickness. The gross tumor volumes (GTV) as well as all organs at risk were delineated. Clinical target volume was created by adding a safety margin that encompass all possible microscopic and expected tumor extensions. An isotropic expansion of 5 mm was added to form the planning target volume (PTV) according to the department policy. Intensity modulated radiotherapy (IMRT) technique using KonRad treatment planning system was applied to deliver a dose of 61.2 Gy in 37 fractions to high risk tumor and nodal areas, and 54 Gy to the low risk nodal area applying simultaneous integrated boost technique for CR and PR patients. Those who had SD received 66.6Gy in 37 fraction, while for patients with PD, they received 70.2 Gy in 39 fractions. Toxicity was recorded and graded according the WHO toxicity criteria.
Statistical Methods
Data was analyzed using IBM SPSS advanced statistical version 20 (SPSS Inc., Chicago, IT). Numerical data were expressed as mean and standard deviation or median and range as appropriate. Qualitative data were expressed as frequency and percentage. Survival analysis was done using Kaplan-Meier method. The comparison between two survival curves was performed using log-rank test. A p-value <0.05 was considered significant. The follow-up time for each patient was the time from enrollment to the closing date for analyses or the date of last information. For overall survival, all deaths were counted regardless of the cause, and the survival times for living patients were censored at the closing date. The first progression at any site or death without progression was counted as an event for DFS. One patient excluded from EFS as he was lost for follow up before post induction evaluation.
Results
Patients’ Characteristics
Between July 2007 and December 2012, 26 NPC patients were diagnosed and treated at CCHE. The median age at diagnosis was 12 years (range, 7.8–17 years), and a mean 11.9 + 2.7. They were 19 male (73%), and 7 females (27%) with a M:F ratio of 2.7:1.
The most common presentation was dysphagia seen in 23 patients (88.5%), followed by cervical lymphadenopathy in 21 (80.8%), and symptoms of increased intracranial pressure in 19 (73%) patients. The T1, T2, T3 and T4 tumor size was presented in 2 (7.7%), 5 (19.2%), 8 (30.8%) and 11 (42.3%) patients respectively. N0, N1, N2 and N3 was detected in 1 (2.8%), 8 (30.8%), 16 (61.5%) and 1 (3.8%) patients respectively. Three patients (11.5%) were metastatic at presentation.
Four patients (15.4%) were stratified as stage 2b, 11 patients (42.3%) as stage 3, and 11 patients (42.3%) as stage 4 (8 (30.7%) stage 4A, and 3 (11.5%) as stage 4C). Patient characteristics are depicted in Table 1.
The mean follow up period was 35.7 + 17.4 months, median 32.5 months, and ranged between 5-66 months.
Table 1. Patient’s characteristics and response to treatment| Serial number | Age (years) | Sex | TNM | Stage | response post induction | Overall response end of ttt | Final Status |
| 1. | 12 | Male | T4 N2 M1 | 4C | PR | PR | Dead (relapse) |
| 2. | 9 | Male | T3 N0 M0 | 3 | PR | CR | Alive in CR |
| 3. | 13 | Male | T2b N1 M0 | 2b | CR | CR | Alive in CR |
| 4. | 12 | Male | T4 N2 M0 | 4b | PR | PR | Alive in PD (relapse) |
| 5. | 15.3 | Male | T4 N1 M0 | 4a | PR | CR | Alive in CR |
| 6. | 13.3 | Male | T4 N2 M0 | 4a | PR | CR | lost FU |
| 7. | 8.6 | Male | T2b N1 M0 | 2 | CR | CR | Alive in CR |
| 8. | 14 | Male | T4 N2 M1 | 4c | PD | Lost FU | Lost FU |
| 9. | 7.8 | Female | T4 N1 M0 | 4 | PR | CR | Alive in CR |
| 10. | 8.4 | Male | T4 N2 M0 | 4 | PR | CR | Alive in CR |
| 11. | 8.4 | Female | T3 N1 M0 | 3 | SD | CR | Alive in CR |
| 12. | 9.2 | Male | T3 N2 M0 | 3 | PR | CR | Alive in CR |
| 13. | 16.8 | Female | T3 N2 M0 | 3 | PR | CR | Alive in CR |
| 14. | 11.8 | Male | T2b N2 M0 | 3 | PR | CR | Alive in CR |
| 15. | 12.3 | Female | T3 N1 M0 | 3 | PR | CR | Dead (relapse) |
| 16. | 14 yrs | Male | T4 N2 M0 | 4a | PR | CR | Dead (relapse) |
| 17. | 14 yrs | Female | T3 N2 M0 | 3 | PR | CR | Alive in CR |
| 18. | 13 yrs | Male | T3 N1 M0 | 3 | SD | CR | Alive in CR |
| 19. | 12 yrs | Female | T1 N1 M0 | 2b | PR | CR | Alive in CR |
| 20. | 17 yrs | Male | T1 N2 M0 | 3 | SD | PR | Alive in CR |
| 21. | 12 yrs | Male | T2A N2 M0 | 3 | PD | PR | Lost FU |
| 22. | 11 yrs | Male | T2b N2 M0 | 3 | CR | CR | Alive in CR |
| 23. | 13 yrs | Female | T4, N2, M0 | 4a | SD | PR | Alive in CR |
| 24. | 10 yrs | Male | T4 N2 M0 | 4a | SD | CR | Alive in CR |
| 25. | 13 yrs | Male | T3 N2 M0 | 3 | CR | CR | Alive in CR |
| 26. | 10 yrs | Male | T4 N3 M1 | 4c | PR | PD | Dead |
Pathology:
all our patients were undifferentiated NPC (WHO types 3).
Response to Treatment:
Following induction chemotherapy, the overall response rate (CR and PR) was seen in 19 patients (73%); (4 patients (15.3%) achieved CR, 15 (57.6%) PR. Five patients (19.3%) had SD and 2 patients (7.7%) had PD. Whereas, at time of final analysis, 18 patients (69.2%) achieved CR, 5 patients (19.2%) had PD (4 deaths (15.3%), and 1 patient (3.8%) alive under palliative treatment), and 3 patients (11.5%) lost follow up. Table 2 describes patient's response to therapy.
Table 2. Assessment of response to chemotherapy.| Response | Post induction (percentage) | End of treatment(percentage) | Final status(Percentage) |
|---|---|---|---|
| CR | 4 (15.4%) | 19 (73%) | 18 (69.2%) |
| PR | 15 (57.6%) | 5 (19.3%) | --- |
| SD | 5 (19.3%) | - | --- |
| PD | 2 (7.6%) | 1 (3.8%) | 1 (3.8%) |
| Lost FU | -- | 1 (3.8%) | 3 (11.5%) |
| Dead | -- | -- | 4 (15.4%) |
| Total | 26 | 26 | 26 |
Survival:
The 3 year estimated OS and EFS rates were 83.3% and 80.7% respectively (Figure 1 and Figure 2).
Figure 1.3 years OS of the whole group of patients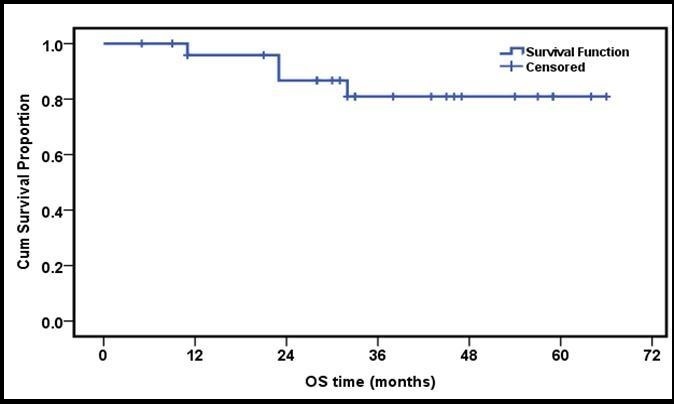
Figure 2.3 years EFS of the whole group of patients
Survival analysis was done in correlation with age, sex, clinical presentation, presence or absence of signs of increased intracranial tension, TNM staging, chemotherapy response and showed no statistical significance.
The OS for early stages (T1 and T2) was 100%, T3 (87%) while T4 cases were 72.7% with no statistical significance (p value =0.2). Similarly, OS for N0,1 was 88.8%, and for N2,3 82.3%, again with no statistical significance (p=0.39). On the other hand, OS for M0 was 91.3% and for M1 33.3% with statistical significance (p =0.032)( Figure 3 and Figure 4).
Figure 3.3 years OS of metastatic versus non metastatic group of patients
Figure 4.3 years EFS of metastatic versus non metastatic group of patients
Events and Causes of Death
Four patients (15.3%) developed disease recurrence, while one patient (3.8%) had PD. Three patients (11.5%) hand distant metastasis (lung, bone or both), while one patient (3.8%) had locoregional recurrence. Disease recurrence occurred at a median of 17.5 months (range, 9–24 months), with a survival range of (3 - 26 months after relapse).
Toxicity
Toxicity data during chemotherapy and FU period were reported for all 26 patients. As expected, most severe toxicities were myelosuppression and oral mucositis. Twenty patients (76.9%) had grade 3-4 neutropenia, with no major infectious complications, 18 patients (69.2%) had Grade 2 - 4 oral mucositis, 5 (19.2%) had grade 2-4 ototoxicity, 4 (15,3%) had grade 2-3 nephrotoxicity and 3 (11.5%) had neuro-psychiatric complications.
Discussion
Nasopharyngeal carcinoma (NPC) remains one of the greatest oncological challenges because of its highly aggressive natural behavior and the anatomical proximity to critical structures1. In the young population, it is a rare malignancy, although it is one of the most common epithelial tumors in children. The incidence varies extensively according to racial and geographical factors2.
In the present study, the age of patients ranged from 7.8 to 17 years with a median of 12 years. This is relatively in agreement with other studies 15, 16, 17, 18, 19, 20, 21. Our demographic data showed a male predominance, similar to most of the published pediatric NPC series 18, 19, 21.
The majority (84.6 %) of the patients had an advanced stage 3 or 4 disease . This is in agreement with an Indian study reported by Laskar et al. 2008, where stage IV represented 61% of their patients, stage III 27.7% and stage IIb 11% 22. In three other studies, stage IV presentation ranged from 50-56% 23, 24, 25. Late presentation may be explained by the fact that NPC is highly malignant with early lymphatic spread and predilection for distant metastasis. Furthermore, early detection is difficult due to its silent deep- seated location26.
Nineteen of our patients (73%) achieved good response to induction therapy (CR-PR), 5 (19.3%) had SD, while 2 (7.7%) had PD during therapy. The same findings were seen in most of studies 20, 21, 22, 23, 24, 26, 27.
All our patients were histologically WHO type 3, which is in accordance with other studies 20, 21, 23. On the other hand, Daoud et al, reported 56.3% of patients as undifferentiated carcinoma (WHO type 3), and the remainder had a non-keratinising carcinoma (WHO type 2)26.
At the end of our study, 69.2% of the patients were alive in CR, 4 (15.4%) died, 3.8% had PD, while three patients (11.2%) was lost for follow up. Similar results were reported by Venkitaraman et al., and Shen et al.21, 22, 23. Achieving CR at end of treatment was highest in the German Pediatric study (NPC-91-GPOH). With a response rate of 91% where patients were treated by neoadjuvant chemotherapy with (cisplatin, 5 FU, and methotrexate), followed by radiation-therapy and adjuvant interferon β for 6 months27. Similarly, Galindo et al reported good responders to neoadjuvant chemotherapy were 93.75%, and 6.3% of patients had PD24.These results were better than that reported (33.3%) in the Turkish study conducted on 84 Patients. 25.
In a study conducted by Laskar et al., good responders were 47.2%, while 52.8% of cases had SD or PD 22. Another study showed 82.1% of patients had good response, 10.9% had PD and 6% had SD28. Mertens et al reported in their study good responders to neo-adjuvant chemotherapy as 94.9% 29. Similarly, in a multicenter study performed by Ozyar et al., 2006, 96.3% of patients either responded completely or partially to neoadjuvant chemotherapy27.
The 3 year estimated OS and EFS rates were 83.3% and 80.7% respectively, that was similar to what has been reported by other groups 28, 29, 30
In the present study, T stage did not show significant prognostic influence. Similar results were reported by other groups21, 25. However, other reported that T- stage carried a statistically significant influence on event free survival only, with no effect on overall survival20, 22. Zaghloul et al. reported, that the survival worsened with the advancement in T stage 31.
The OS for N0-N1 vs. N2-N3 was 88.8% vs.82.3% respectively with no statistical difference. Same results were reported by Shen et al and Zaghloul et al. 23, 31. On the contrary, other studies, reported a better EFS for N0-N1 20, 25, 27 .
Twenty-three patients (88.4%) in the present series were stratified as M0, while 3 patients (11.6%) were M1. Their OS survival was 91.3% versus 33.3%. This was the only prognostic factor identified in our study.
The role of chemotherapy in addition to radiotherapy is becoming the standard of care, and has been proved by many studies 32, 33, 34. Concurrent chemo-radiotherapy27, and adjuvant chemotherapy, both improve OS when compared with RT alone 20, 22, 24, 32.Neoadjuvant chemotherapy failed to improve long term survival as reported by few Chinese studies23, 35, 36. In our study, there was no OS statistical difference between patients according to their response to neoadjuvant chemotherapy. On the other hand, some studies showed that neoadjuvant chemotherapy has better OS, and is better tolerated than adjuvant chemotherapy. Furthermore, early use of a potent combination of cytotoxic drugs at full dose would theoretically be more effective for eradicating micrometastases, shrink the primary tumor, and give a wider margin for irradiation sparing critical neural structures accompanied with less toxicity 20, 29, 30, 31 .
On the other hand, controversy exists about the best chemotherapeutic agent to be used. Huang et al. in 2009, showed that concurrent chemo-radiotherapy with carboplatin didn’t improve the survival when compared with induction chemotherapy with carboplatin and 5- fleurouracil 35. No solid conclusion could be drawn pointing to the need for further assessment in larger trials 36. The optimal treatment strategy for NPC patients has not been established through randomized clinical trials yet 25, 26.
The development of RT technique to 3-dimensional (3D) conformal (CRT), and then Intensity-Modulated Radiotherapy (IMRT) not only improved tumor coverage and ensured precision of delineation, but also had better sparing of normal structures and lowered toxicity 26. In our study, all our patients were treated with IMRT with minimal, generally acceptable immediate and late toxicities.
The optimal dose of radiotherapy is still controversial, particularly when combined with chemotherapy. Recently, several studies showed that dose reduction to 60 Gy or less was feasible in multimodality treatment without compromising the outcome in chemo responsive patients 21, 22, 25, 27, 30, 37. On the contrary, several authors reported a better OS with radiation dose > 66 Gy in comparison with lower doses 12, 14, 20, 24.
Prognostic Factors:
In our study, the presence or absence of distant metastasis at presentation was found to be the only factor of statistical significance. Similar finding was reported by many other authors who reported age and sex having no prognostic significance20, 21, 24. Good responders to neo-adjuvant chemotherapy were found to have a better OS and EFS in some studies 20, 22, 25, 30, while others -including the present study showed no statistical difference according to initial response 21, 24, 26, 27.
Survival:
By the end of our study, with a median follow up period of 35 months. Four patients (15.3%) in the present study relapsed and 1 had PD at a median of 17.5 months. Four of them died due to disease progression. Similar results were reported where the relapse rate was 36%20. Other studies showed 31% 19, 33% 24 relapse rate. Lower rate of recurrence was similarly reported by other study groups ranging from 17-21% of cases 20, 23, 29.
In all studies including the present one, distant failure predominates and remained a challenge to overcome. This could be explained by good local control achieved by concurrent chemo radiotherapy. Because distant failure is the major cause of death in nasopharyngeal carcinoma, development of more potent and less toxic chemotherapy regimens and targeted therapy bring new opportunities26.
Conclusion:
Neoadjuvant chemotherapy followed by concurrent chemoradiotherapy using IMRT lead to good clinical end results with limited toxicity. Metastatic disease at presentation was identified as the adverse prognostic factor.
Supplementary Figures
References
- 2.Parkin D M, Bray F, Ferlay J, Pisani P. (2005) . Global cancer statistics,2002.CA Cancer J Clin. 55 : 74-108.
- 3.Chang E T, Adami H O. (2006) The enigmatic epidemiology of nasopharyngeal carcinoma. Cancer Epidemiol Biomarkers Prev. 15, 1765.
- 4.Teo P M, Chan A T, Lee W Y, Leung T W, Johnson P J. (1999) Enhancement of local control in locally advanced node-positive nasopharyngeal carcinoma by adjunctive chemotherapy. , Int J Radiat Oncol Biol Phys 43, 261-71.
- 5.Barnes L, Eveson J W, Reichart P, Sidransky D. (2005) Pathology and Genetics of Head and Neck Tumours. In: World Health Organization Classification of Tumors,IARC Press. , Lyon
- 6.BeahrsOH HensonDE, RVP Hutter, Kennedy B J. (1992) editors Manual for staging of cancer. 4th edition. , Philadelphia: J.B.Lippincott: 33-38.
- 7.M Cvitkovic E Bachouchi, Armand J P. (1991) Nasopharyngeal carcinoma: biology, natural history and therapeutic implications. Hematol Oncol Clin North Am. 5, 821-23.
- 8.Vokes E E, Liebowitz D N, Weichselbaum R R. (1997) . , Nasopharyngeal carcinoma. Lancet: 350, 1087-91.
- 9.Eisenhauer E A, Therasse P, Bogaerts J, Schwartz L H, Sargent D et al. (2009) New response evaluation criteria in solid tumours: Revised RECIST guideline .European Journal of Cancer,45. 228-47.
- 10.Halperin E C, Constine L S, Tarbell N J, Kun L E. (1999) Endocrine, aerodigestive tract, and breast tumors. In: Pediatric Radiation Oncology, 3rd edn. , Philadelphia: Lippincott Williams,Wilkins 395-421.
- 11.Jenkin R D, Anderson J R, Jereb B, Thompson J C, Pyesmany A et al. (1981) Nasopharyngeal carcinoma—a retrospective review of patients less than thirty years of age: a report of Children’s Cancer Study Group. Cancer. 47, 360-66.
- 12.JST Sham, Poon Y F, Wei W I, Choy D. (1990) Nasopharyngeal carcinoma in young patients. Cancer. 65, 2606-10.
- 13.Ayan I, Altun M. (1996) Nasopharyngeal carcinoma in children: retrospective review of 50 patients. , Int J Radiat Oncol Biol Phys 35, 485-92.
- 14.Serin M, Erkal H, Elhan A H, Cakmak A. (1998) Nasopharyngeal carcinoma in childhood and adolescence. Med Pediatr Oncol. 31, 498-505.
- 15.Sahraoui S, Acharki A, Benider A, Bouras N, Kahlain A. (1999) Nasopharyngeal carcinoma in children under 15 years of age: a retrospective review of 65 patients. Annal Oncol. 10, 1499-502.
- 16.Wolden S L, Steinherz P G, Kraus D H, Zelefsky M J, Pfister D G et al. (2000) Improved long term survival with combined modality therapy for pediatric nasopharynx cancer. , Int J Radiat Oncol Biol Phys 46, 859-64.
- 17.Ingersoll L, Woo S Y, Donaldson S, Giesler J, Maor M H et al. (1990) Nasopharyngeal carcinoma in the young: combined MD Anderson and Stanford experience. , Int J Radiat Oncol Biol Phys 4, 881-87.
- 18.Berry M P, Smith C R, Brown T C, Jenkin R D, Rider W D. (1980) Nasopharyngeal carcinoma in the young. , Int J Radiat Oncol Biol Phys 6, 415-21.
- 19.Lombardi F, Gasparini M, Gianni C, M De, Molinari R et al. (1982) Nasopharyngeal carcinoma in childhood. Med Pediatr Oncol. 10, 243-50.
- 20.Kupeli S, Varan A, Ozyar E, Atahan I L, Yalcin B et al. (2006) Treatment results of 84 patients with nasopharyngeal carcinoma in chicldhood. Pediat Blood canc. 46, 454-58.
- 21.Venkitaraman R, Ramanan S G, Sagar T G. (2007) Nasopharyngeal cancer of childhood and adolescence:a single institution experience. Ped. Hemat and Oncol. 24, 493-502.
- 22.Laskar S, Bahl G, Muckaden M, Pai S K, Gupta T et al. (2008) Nasopharyngeal carcinoma in children: comparison of conventional and intensity-modulated radiotherapy. Int.J.Radiat Oncol Biol Phys. 72, 728-36.
- 23.Shen C, Gao Y, Xu T, Wang X, Ying H et al. (2009) Carcinoma of the nasopharynx in young patients: a single institution experience. , Clinic. Oncol 21, 617-22.
- 24.Galindo C R, Wofford M, Castleberry R P, Gp Swanson, London W B et al.Preradiation chemotherapy with methotrexate, cisplatin, 5-fluorouracil, and leucovorin for pediatric nasopharyngeal carcinoma.Cancer2005 ;103:. 850-7.
- 25.Ayan I, Kaytan E, Ayan N. (2003) Childhood nasopharyngeal carcinoma: from biology to treatment. Lancet Oncol . 4, 13-21.
- 26.Daoud J, Toum N, Bouaziz M, Ghorbel A, Jlidi R et al. (2003) Nasopharyngeal carcinoma in childhood and adolescence: Analysis of a series of 32 patients treated with combined chemotherapy and radiotherapy. , Euro J Cancer 39, 2349-54.
- 27.Ozyar E, Selek U, Laskar S, Uzel O, Anacak Y et al. (2006) Treatmentv results of 165 pediatric patients with non-metastatic nasopharyneal carcinoma: a rare cancer network study. , Radiotherpy Oncol; 81, 39-46.
- 28.Lee A W, Lin J C, Ng W T. (2012) Current management of nasopharyngeal cancer. Semin Radiat Oncol. 22, 233-44.
- 29.Mertens R, Granzen B, Lassay L, Bucsky M, Hundgen M et al. (2005) . , Treatment of Nasopharyngeal Carcinoma in Children and Adolescents. Cancer; 104, 1083-9.
- 30.Al Sarraf M, M Le Blanc, Giri P G, Fu K K, Cooper J et al. (1998) Chemoradiotherapy versus radiotherapy in patients with advanced nasopharyngeal cancer: phase III randomized intergroup study 0099. J Clin Oncol;. 16, 1310-17.
- 31.Zaghloul M S, Dahba N M, Wahab A A, Hussien M H, El-Koutbey M. (1993) Nasopharyngeal carcinoma in children and adolescents successful role of retrieval therapy. Tumori. 79, 123-127.
- 32.Rischin D, Corry J, Smith J, Stewart J, Hughes P et al. (2002) Excellent disease control and survival in patients with advanced nasopharyngeal cancer treated with chemoradiation. , J Clin Oncol 20, 1845-1852.
- 33.Lee A W, Ng W T, Chan Y H, Sze H, Chan C et al. (2012) The battle against nasopharyngeal cancer. Radiotherapy and Oncol;. 104, 272-278.
- 34.Baujat B, Audry H, Bourhis J, Chan A T, Onat H et al.The phamongkhol K, Pignon JP; MAC-NPC Collaborative Group .(2006) Chemotherapy in locally advanced nasopharyngeal carcinoma: an individual patient data meta-analysis of 8 randomized trails and 1753 patients. , Int J Radiat Oncol Biol Phys,56-2006,46: 47-56.
- 35.Huang P Y, Mai H Q, Luo D H, Qiu F, Li N W et al. (2009) Induction-concurrent chemoradiotherapy versus induction chemotherapy and radiotherapy for locoregionally advanced nasopharyngeal carcinoma. Ai Zheng. 28, 1033-42.
Cited by (2)
This article has been cited by 2 scholarly works according to:
Citing Articles:
Journal of Pediatric Hematology/Oncology (2020) Crossref
Wael Zekri, M. Wahed, Eman I. Attia, E. Khalil - Journal of pediatric hematology/oncology (2019) Semantic Scholar
Journal of Pediatric Hematology/Oncology (2019) OpenAlex
