Silver Nanoparticles: Cytotoxic and Apoptotic Activity on HT-29 and A549 Cell Lines
Abstract
Silver nanoparticles (Ag-NPs) are versatile materials with a broad range of applications in various fields such as cancer therapy, drug delivery. In this work, cytotoxic and apoptotic activities of silver nanoparticles was evaluation against lung (A549) and colon (HT-29) cell lines. The cytotoxic activity of nanoparticles was performed by MTT assay, while their apoptotic activity was tested through TUNEL method. The results of MTT of A549 have illustrate that fifty percent of cells destruction in concentrations more than 250 µg/ml of Ag-NPs. Apoptotic results of nanoparticles have shown more than fifty percent of apoptosis on A549 cell line. HT-29 display full apoptosis at concentrations more than 500 µg/ml. It seems that synthesized Ag-NPs by using P. farcta extract can be candidate as anti-cancer agent in treatment many cancers through creating or discovering new drug forms
Author Contributions
Academic Editor: Zhe-Sheng Chenz, Professor, Department of Pharmaceutical Sciences, College of Pharmacy and Allied Health Professions, St. John’s University, United States.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2018 Abdolhossien Miri , et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
In recent years, nanoparticles have developed as diagnostic and therapeutic agents for the treatment of many diseases such as diabetes, asthma, allergies and cancer 1. Ag-NPs have special physicochemical properties, which apply such as solar cells, catalyst in chemistry reaction, electric batteries etc. 1. Also, they have contain antimicrobial 2, antiviral 3, antifungal 4, 5, anti-inflammatory 6, and antioxidant effects 7. Studies have showed the usage of nanoparticles in drug delivery and molecular imaging 8, while researches have reported their capability in eliminating cancer cells 9, 10. Some cancer cells have been reported to show resistance toward many drugs, specially platin derivatives such as cis-platin, as well as showing toxic effects. Thus, it is vital to discover new drugs for this particular disease in which Ag-NPs seem to fit as a suitable suggestion 11. Recent studies on Ag-NPs have shown significant anti-cancer activity on breast (MCF-7) 9, mouse fibroblasts (L929) 8, colon (HCT-119) 12, lung (A549) 13, pharynx (Hep-2) 14, rat skeletal muscle (L6) 15, and prostate (MDA-MB-231) 16 cancer cell lines. The aim of this project is to investigate the anticancer activities of synthesized Ag-NPs by using Prosopis farcta extract against lung (A549) and colon (HT-29) cancer cell lines. These nanoparticles, which contain spherical shapes with size about 10.8 nm 17, have been already synthesized by our team members. The cytotoxic and apoptotic activities of the mentioned nanoparticles were investigated through the use of MTT assay and TUNEL test, respectively.
Materials and Methods
Synthesis of Ag-NPs
Ag-NPs were synthesized according to the protocol of Miri et al17. Briefly, 5 ml of P. farcta aqueous extract was added to 95 ml of silver nitrate solution (1 mM) in dark, room temperature and shaken with 150 rpm. After 1 hour, the color of solution changed to brown, which indicates the formation of Ag-NPs. The synthesized nanoparticles were centrifuged at 4000 rpm for 10 min, and sediment was used to prepare stock suspension using water as vehicle.
Cell line and Cell Culture
The lung (A549) and colon (HT-29) cell lines were prepared from Pasteur Institute of Iran. Cells were incubated in RPMI media with 10% FBS, 100 μg/ml of Streptomycin, and 100 U/ml of Penicillin, at a temperature of 37 ºC and the atmosphere of CO2 with 5% moisture.
Evaluation Cytotoxicity Effect by MTT Assay
The cytotoxicity of synthesized nanoparticles was enquired into by 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT). Briefly, 200 µl of cell suspension was added to each well (1x104 Cell/well) and the plates were incubated for 24 h. Afterwards, 50 µl of synthesized nanoparticles (50, 250, 500 and 1000 µg/ml) were separately added to the wells and incubated for certain time periods. 20 µl of MTT solution (5 mg/ml MTT dye in PBS buffer) was added to each well and the plates were incubated for 3 h at 37 ºC. Finally, 100 µl of DMSO was added to the wells. Optical absorbance was measured at 570 nm through the use of a micro-plate reader. Cell viability was expressed as a percent relative to untreated control cells at 24 h treatment time.
Evaluation Apoptotic Effect by TUNEL Stain
Terminal deoxynucleotidyl transferase-mediated dUPT nick end labeling (TUNEL) test was performed according to the manufacturer’s instructions (in situ cell death detection kit, POD; Roche Diagnostics, USA). In brief, samples were treated by Ag-NPs solution (50, 250, 500 and 1000 µg/ml concentrations, separately) for 24 h, while the cells were fixed in 4% paraformaldehyde at 15-25 ˚C for 60 min; then they were washed twice by PBS. Afterwards, 200 µl of blocking solution (3% H2O2 in methanol) was added to each well and the plates were incubated for 10 min at 15-25 ˚C. Cells were washed twice by PBS and then, permeabilization solution (0.1% Triton X-100) was added to them. The results were stored for 2 min at 2-8 ˚C. Cells were washed twice again by PBS, while 50 µl of ‘‘TUNEL reaction mix’’ was added to each sample and incubated in the dark at 37 ˚C for a duration of 1 hour. Later on, they were dried after being washed by PBS and 50 µl of POD-Convertor was added to the samples. The plates were incubated at 37 ˚C for 30 min. After that, 50 µl of DAB substrate was added to the plates and they were stored in 15-25 ˚C for 10 min. At the end, the healthy and apoptotic cells were calculated by direct view through invert microscope.
Statistical Analysis
Statistical analysis was performed by GraphPad Prism 5 and the statistical comparisons of multi-group data were analyzed through the use of two way ANOVA. Values of *p < 0.05 were considered as statistically significant. Each test was performed in triplicates and results were presented as mean ± SD.
Result and Discussion
In our previous studies, Ag-NPs were synthesized by using P. farcta aqueous extract as the reducing agent for the reduction of Ag+ to Ag017. In this study, we survey the cytotoxic and apoptotic activities of synthesized Ag-NPs using P. farcta extract against lung (A549) and colon (HT-29) cancer cell lines. In this regard, the cytotoxic activity of Ag-NPs on A549 and HT-29 cancer cell lines have been tested and performed through the utilization of MTT assay. This method is based on the conversion of tetrazolium salt (solvable, pink) to formazan (unsolvable, purple) by the mitochondria of living cells. The experiment was performed through the application of four concentrations (50, 250, 500 and 1000 µg/ml) of nanoparticles at 24 h treatment time. The achieved results regarding the cytotoxic activity of Ag-NPs against A549 cells has shown a significant decrease in the viability of cells at 24 h in comparison to the of control (*P˂0.05 ), which has gone as far as attaining a fifty percent of cells destruction in concentrations more than 250 µg/ml of Ag-NPs. The results of cytotoxic activity of synthesized Ag-NPs on HT-29 cells show no significant differences between the treated cells and control at 24 h (Figure 1).
Figure 1.MTT cell viability assay of synthesized Ag-NPs on A549 and HT-29 cancer cell lines at 24 h. Data were means ± SD of three independent experiments. *p˂0.05 compared to the control group, by ANOVA.
Moaddab has reported that the cytotoxic activity of Ag-NPs has increased due to chemical and physical interactions of silver ions with functional groups of intracellular proteins such as nitrogen and phosphate groups 18. Lately, cytotoxic effect of biosynthesized Ag-NPs has been reported against many cancer cells such as human cervical (HeLa) 19, prostate (MCF-7) 20, lung (A549) 21 and acute promyelocytic leukemia (HL-60) cells 22.
Apoptotic activity of Ag-NPs was done on lung (A549) and colon (HT-29) cancer cell lines, using TUNEL assay. The basis of this method is the connection of Terminal deoxynucleotidy transferase (TdT) enzyme to broken DNA strands (one of the symptoms of apoptosis). Through the mentioned method, apoptotic cells are characterized by labeling them through attaching the fluorescence color of the kit. In this assay, the apoptotic activity of Ag-NPs (50, 250, 500 and 1000 µg/ml) was put under investigation against A549 and HT-29 cell lines. It is illustrated through the results that as the nanoparticles concentration increases, their apoptotic effect significantly increases as well. The concentration of 1000 µg/ml showed 86% apoptosis effect on A549 cells, and concentrations higher than 500 µg/ml demonstrated 100% apoptotic effect on HT-29 (Figure 2). Previous studies have stated that cellular death, caused by nanoparticles through apoptosis; occur by the changes in cell nuclei such as chromatin agglomerate or nuclear fragmentation 9, while B. Gajendran et al have reported that Ag-NPs cause apoptosis through active oxygen species, increasing ROS level, and creating changes in the mitochondrial membrane potential 23.
Figure 2.Apoptosis analysis of synthesized Ag-NPs using TUNEL assay on A549 and HT-29 cancer cell lines for 24 h.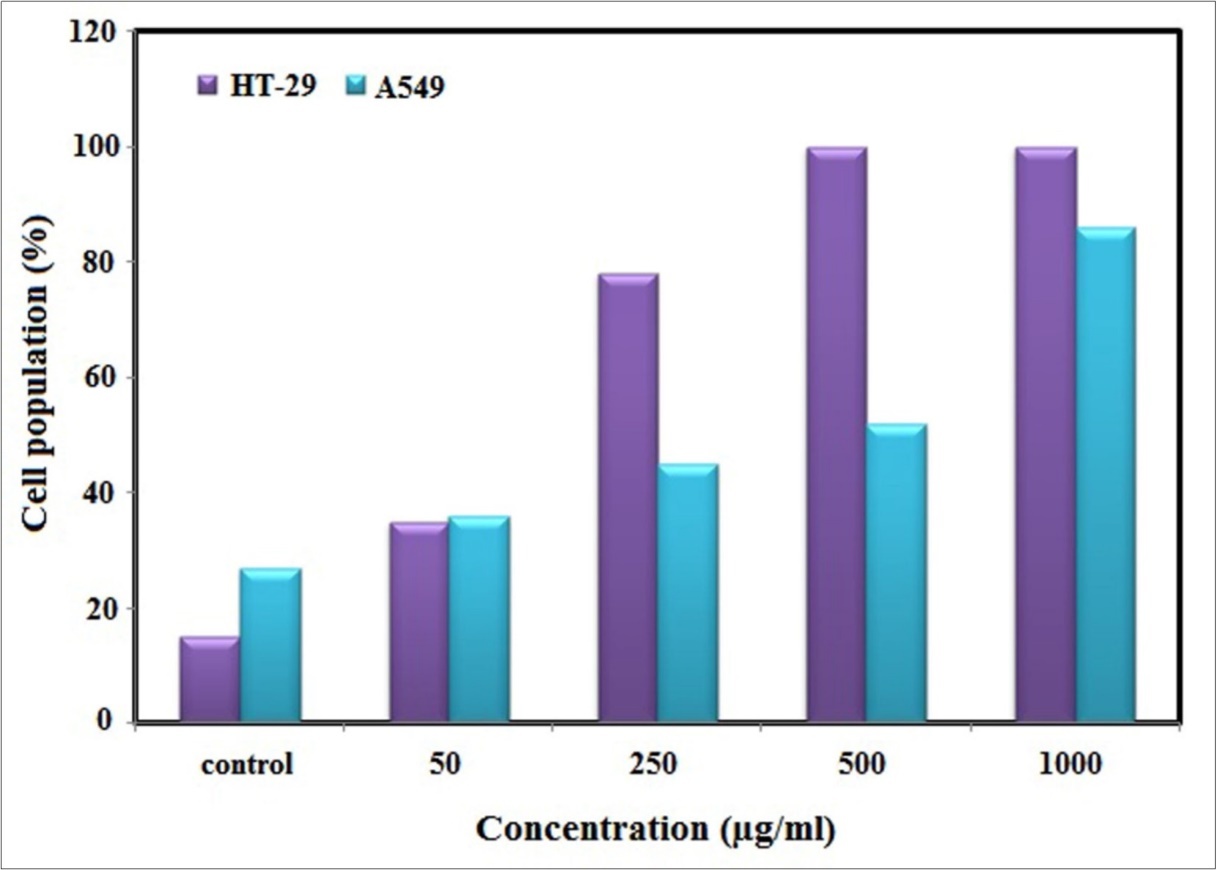
Conclusion
According to various researches and this study, it can be suggested that Ag-NPs is capable of annihilating cancer cell lines. In this research, the cytotoxic effect of synthesized Ag-NPs, using P. farcta extract, has been enquired into through the MTT assay. The results have indicated that the viability of A549 is dependent on the nanoparticles concentration. The apoptotic results of Ag-NPs have shown more than 50% of apoptosis on both cell lines. Also regarding this case, HT-29 has demonstrated full apoptosis at concentrations more than 500 µg/ml. The Size, shape and chemical surface of nanoparticles can be effective on its cytotoxic activity, while on the other hand, the synthesis method can affect these particular factors as well. Therefore, comparisons and supplementary studies on Ag-NPs and in vitro studies may provide a pathway for the treatment of cancer by, creating or discovering new drug forms that can deliver the drug to the site of cancer.
References
- 1.Remya R R, Radhik Rajasree SR, Aranganathan L, Suman T Y. (2015) An investigation on cytotoxic effect of bioactive Ag NPs synthesized using Cassia fistula flower extract on breast cancer cell MCF-7”. , Biotechnol. Rep 8, 110-115.
- 2.Mohammed. (2015) A E “Green synthesis, antimicrobial and cytotoxic effects of silver nanoparticles mediated by Eucalyptus camaldulensis leaf extract”. , Asian. Pac. J. Trop. Biomed 5, 382-386.
- 3.Galdiero S, Falanga A, Vitiello M, Cantisani M, Marra V et al. (2011) Silver nanoparticles as potential antiviral agents”. , Molecules 16(10), 8894-8918.
- 4.Khatami M, S M Mortazavi, Kishani-Farahani Z. (2017) . , Biosynthesis of Silver Nanoparticles Using Pine Pollen and Evaluation of the Antifungal Efficiency”, Iranian. J. Biotech 15, 95-101.
- 5.Azizi Z, Pourseyedi S, Khatami M, Mohammadi H. (2016) Stachys lavandulifolia and Lathyrus sp. Mediated for Green Synthesis of Silver Nanoparticles and Evaluation Its Antifungal Activity Against Dothiorella sarmentorum”. , J. Clust. Sci 27, 1613-1628.
- 6.Hebeish A, M H El-Rafie, M A EL-Sheikh, Seleem A A, M E El-Naggar. (2014) Antimicrobial wound dressing and anti-inflammatory efficacy of silver nanoparticles”. , Int. J. Bio. Macromol 65, 509-515.
- 7.Elemike E E, Fayemi O E, Ekennia A C, Onwudiwe D C. (2017) . Ebenso E E “Silver Nanoparticles Mediated by Costus afer Leaf Extract: Synthesis, Antibacterial, Antioxidant and Electrochemical Properties”, Molecules 22, 701.
- 8.Gogoi N, Jayasekhar Babu P, Mahanta C, Bora U. (2015) Green synthesis and characterization of silver nanoparticles using alcoholic flower extract of Nyctanthes arbortristis and in vitro investigation of their antibacterial and cytotoxic activities”. , Mater. Sci. Eng. C. Mater. Biol. Appl 46, 463-469.
- 9.Venugopal K, H A Rather, Rajagopal K. (2017) Synthesis of silver nanoparticles (Ag NPs) for anticancer activities (MCF 7 breast and A549 lung cell lines) of the crude extract of Syzygium aromaticum”. , J. Photochem. Photobiol. B 167, 282-289.
- 10.Vasanth K, Ilango K, MohanKumar R, Agrawal A, G P Dubey. (2014) Anticancer activity of Moringa oleifera mediated silver nanoparticleson human cervical carcinoma cells by apoptosis induction”. , Colloids. Surf. B. Biointerfaces 117, 354-359.
- 11.W J Youngs, Robishaw N, M J Panzner. (2009) Treatment of Breast Cancer with Silver Antitumor Drugs Encapsulated. In Biodegradable Polymeric Nanoparticles”.NSTI-Nanotech.2 .
- 12.“Antimicrobial Gomaa E Z. (2017) antioxidant and antitumor activities of silver nanoparticles synthesized by Allium cepa extract: A green approach”. , J. Genet. Eng. Biotechnol 15, 49-57.
- 13.Palaniappan P, Sathishkumar G, Sankar R. (2015) Fabrication of nano-silver particles using Cymodocea serrulata and its cytotoxicity effect against human lung cancer A549 cells line”. , Spectrochim. Acta. A. Mol. Biomol. Spectrosc 138, 885-890.
- 14.Venugopal K, Ahmad H, Manikandan E. (2017) et al.,”The impact of anticancer activity upon Beta vulgaris extract mediated biosynthesized silver nanoparticles (Ag-NPs) against human breast (MCF-7), lung (A549) and pharynx (Hep-2) cancer cell lines”. , J. Photochem. Photobiol. B 173, 99-107.
- 15.M K Swamy, M S Akhtar, S K Mohanty, Sinniah.U.R.,”Synthesis and characterization of silver nanoparticles using fruit extract of Momordica cymbalaria and assessment of their in vitro antimicrobial, antioxidant and cytotoxicity activities”. , Spectrochim. Acta. A. Mol. Biomol. Spectrosc 151(2015), 939-944.
- 16.Krishnaraj C, Muthukumaran P, Ramachandran R, Balakumaran Kalaichelvan, T P. (2014) Acalypha indica Linn: Biogenic synthesis of silver and gold nanoparticles and their cytotoxic effects against MDA-MB-231, human breast cancer cells”. , Biotechnol. Rep 4, 42-49.
- 17.Miri A, Sarani M, Rezazade Bazaz M, Darroudi M. (2015) Plant-mediated biosynthesis of silver nanoparticles using Prosopis farcta extract and its antibacterial properties”. , Spectrochim. Acta. A. Mol. Biomol. Spectrosc 141, 287-291.
- 18.Moaddab S, Ahari H, Shahbazzadeh D. (2011) Toxicity study of nanosilver (Nanocid®) on osteoblast cancer cell line”. , Int. Nano. Lett 1, 11-16.
- 19.Jeyaraj M, Rajesh M, Arun R. (2013) An investigation on the cytotoxicity and caspase-mediated apoptotic effect of biologically synthesized silver nanoparticles using Podophyllum hexandrum on human cervical carcinoma cells. , Colloids. Surf. B. Biointerfaces 102, 708-717.
- 20.Sathishkumar G, Gobinath C, Wilson A. (2014) Dendrophthoe falcata (Lf) Ettingsh (Neem mistletoe): a potent bioresource to fabricate silver nanoparticles for anticancer effect against human breast cancer cells (MCF-7). , Spectrochim. Acta. A. Mol. Biomol. Spectrosc 128, 285-290.
- 21.Sankar R, Karthik A, Prabu A. (2013) Anticancer activity of Ficus religiosa engineered copper oxide nanoparticles. , Colloids. Surf. B. Biointerfaces 108, 80-84.
Cited by (14)
This article has been cited by 14 scholarly works according to:
Citing Articles:
Applied Biochemistry and Biotechnology (2024) Crossref
International Journal of Applied Pharmaceutics (2024) OpenAlex
S. K., A. Shettar, R. S. - International Journal of Applied Pharmaceutics (2024) Semantic Scholar
International Journal of Applied Pharmaceutics (2024) Crossref
Applied Biochemistry and Biotechnology (2023) OpenAlex
Fareeha Arshad, G. Naikoo, Israr U. Hassan, S. Chava, Mohamed El-Tanani et al. - Applied Biochemistry and Biotechnology (2023) Semantic Scholar
K. Hamidian, M. Sarani, Mahmood Barani, Faeze Khakbaz - Arabian Journal of Chemistry (2022) Semantic Scholar
Research Square (Research Square) (2021) OpenAlex
G. D. Reddy, M. Haseena, M. Noorjahan, A. Ratnamala, Chandra Babu Naidu - Journal of Inorganic and Organometallic Polymers and Materials (2021) Semantic Scholar
Applied Physics A (2020) OpenAlex
G. D. Reddy, M. Noorjahan, A. Ratnamala, M. Haseena, H. Manjunatha et al. - Applied Physics A (2020) Semantic Scholar
Materials Today Proceedings (2020) OpenAlex
Materials Today: Proceedings (2020) Crossref
Shalini Jain, N. Saxena, M. Sharma, Sreemoyee Chatterjee - (2020) Semantic Scholar
M. Barbinta-Patrascu, N. Badea, M. Bacalum, Camelia Ungureanu, Ioana-Raluca Șuică-Bunghez et al. - Materials Science and Engineering C: Materials for Biological Applications (2019) Semantic Scholar
Materials Science and Engineering C (2019) OpenAlex
Materials Science and Engineering: C (2019) Crossref
Journal of new developments in Chemistry (2018) OpenAlex
