Bedside Lung Ultrasound in the Assessment of Volume Status in Chronic Hemodialysis Patients
Abstract
Introduction:
Sonographic B-lines, also known as lung comets, have been shown to correlate with the presence of extravascular lung water. The aim of this study is to assess if chest ultrasound could detect lung water imbibition and its variations induced by dialysis, an experimental model of controlled rapid fluid loss.
Methods:
A cross-sectional study was conducted in a hemodialysis center in the city of Fez. Lung ultrasounds, impedance measurement, and ultrasound of the inferior vena cava were achieved 30-60 minutes before and after the hemodialysis session.
Results:
The values, measured by the different techniques used, decrease significantly after hemodialysis (p <0.001). The values studied before and after the hemodialysis, had shown a significant correlation between the results of the impedance measurement, lung ultrasound, and the maximum and minimum of the VCI index diameter and its collapsibility. The decrease in B-lines was correlated with weight decrease during dialysis (p0.005); none of the parameters concerning the IVC were correlated with fluid removal.
Conclusion:
ultrasound performed at the bedside is now emerging as a reliable, easy-to-apply, and safe method for measuring both lung water and intravascular overload and their decrease after dialysis even on asymptomatic patients. These observations strongly support the use of lung ultrasound in estimating volume overload and monitoring the response to therapy in hemodialysis patients.
Author Contributions
Academic Editor: Ying-Yong Zhao, Northwest University
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2016 Ghita El Bardai, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction:
The reliable and reproducible estimate of dry weight in hemodialysis patients remains a clinical problem to date. Prolonged overestimation of dry weight causes hypertension, left ventricular hypertrophy and heart failure, while the underestimation is responsible for a chronic dehydration leading to the risk of hypotension 1. Both complications have a significant impact on morbidity and mortality in hemodialysis patients 2.
The main issue for the achievement of dry weight by dialysis is that volume subtraction should be tailored to the individual patient’s hemodynamic tolerance taking into full account cardiac performance, which is very often compromised in ESRD patients 3, 4.
It is obvious that using better assessment methods for determining volume changes during hemodialysis are needed to adapt the dry weight of the patient. Such methods have evolved from clinical assessment to sophisticated systems of "biofeedback", which incorporate blood volume, ultrafiltration rate and conductivity, passing by the use of Body Composition Analysis (Body bioimpedance spectroscopy (BIS)), ultrasound of the vena cava, the serum markers (B-type natriuretic peptide: BNP), and detection of lung comets by chest ultrasound (US).
One of the most interesting tools is the use of the lung ultrasound to detect the presence of extravascular lung water (LW). In the presence of excessive LW, the US beam is reflected by subpleural thickened interlobular septa, a low impedance structure surrounded by air with a high acoustic mismatch. This US reflection generates hyperechoic reverberation artifacts between thickened septa and the overlying pleura that are known as “lung comets” or B-Lines 5. Lung comets are defined as vertical artifacts arising from the pleural line and extending to the edge of the screen that move synchronously with respiratory acts 6 (Figure 1).
Figure 1.Hyperechoic microreflections indicating the presence of extravascular lung water known as B-lines in one of our patients.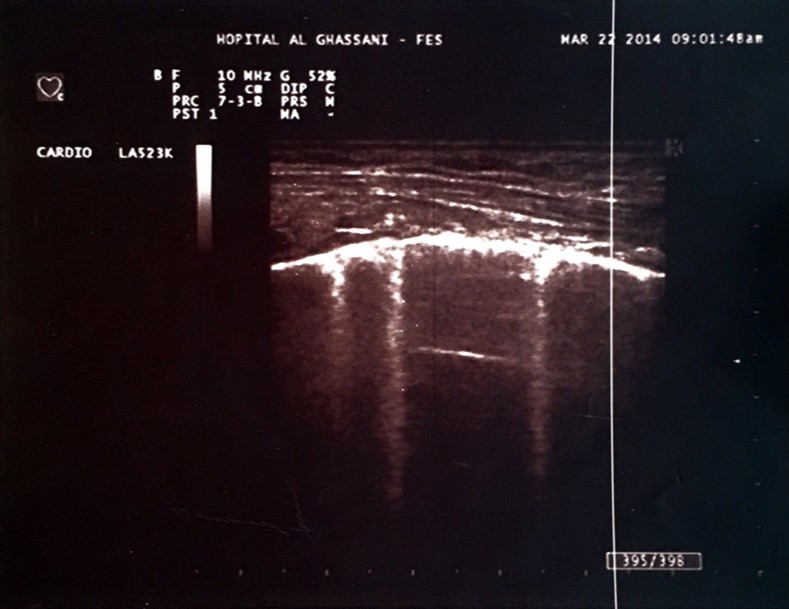
In recent years, the use of chest ultrasound (US) to detect LW has received growing attention in clinical research, in intensive care patients 7, and in patients with heart failure 8. The subclinical phase of pulmonary imbibition and the clearance of B-lines after rapid body fluid removal have been less investigated.
The objectives of our study are:
To estimate the feasibility of LW measurements by chest US in hemodialysis patients and to determine the prevalence of pulmonary congestion in these patients;
To study the relationship-between LW and body fluids volume status- that is assessed by Body bioimpedance spectroscopy (BIS) and ultrasonography of the inferior vena cava (IVC)
To test the regression of lung comets according to the fluids removal on hemodialysis patients
Materials and Methods:
Study Design:
This is a cross-sectional study, conducted during March 2014 in the hemodialysis center of Al Ghassani provincial Hospital of Fez.
Patients:
Using an arteriovenous fistula, the patients included in this study were aged over 18, and have begun chronic hemodialysis since at least 3 months. Were excluded from the study all the patients with the following criteria: An acute event (infectious episode or a hospitalization) within the 3 months preceding the study regardless of the cause. A lung disease with pulmonary fibrosis, or dyspnea stage IV of NYHA showing a heart failure that may affect the results of lung ultrasound regardless of the state of hydration. Inability to wear bioimpedance spectroscopy (BIS) (prostheses, pacemaker...).All our patients received a conventional intermittent dialysis for two to three sessions per week, using low permeability polysulfone membranes with a standard bicarbonate dialysate. The Ultrafiltration rate is prescribed according to the interdialytic weight gains in comparison with the target weight that is clinically determined by the treating nephrologist.
Data Collection:
The survey was carried out by a nephrologist not belonging to the team of hemodialysis center where the study was conducted. A pre-operating sheet was used for the collection of information.
In addition to demographic data and those related to kidney disease, comorbidities and dialysis prescription, we collected all episodes of intradialytic hypotension, cramps or post-dialytic asthenia during the last three sessions of hemodialysis.
The dry weight assessment parameters were measured in two phases: one hour before the hemodialysis (HD) session and between 30 to 60 minutes following the end of the same session.
Therefore, the parameters collected are: Weight, under the usual conditions of the center using an electronic scale. The supine blood pressure measurement carried out after a 10 minutes rest, using a validated electronic device.
Bioimpedance Spectroscopy (BIS):
We used the BCM (Body Composition Monitor, Fresenius Medical Care ®), dedicated to the analysis of body composition and nutritional status of patients. The impedance was performed to the patients, who were lying down flat in bed, after having rested immediately 10 minutes before and 30 to 60 minutes after the hemodialysis session. The electrodes were placed on the wrist of the contralateral arm of the AVF (Arteriovenous Fistula) and on the homolateral ankle.
Lung Ultrasound:
Lung ultrasound was performed by the same trained nephrologist, who did not have access to patients’ clinical data and also to the impedance results. The imaging was made by an ultrasound (KOLTRON Magic Maestro) with a 7 Mhz vascular probe in order to look for an alveolar-interstitial syndrome characterized by the presence of specific artefacts called "B-lines" or "comet tails ". The latter are proof of congestion of the lungs caused by fluid overload in hemodialysis patients 9, 10.Ultrasound examination was performed to the patients in supine position, with a longitudinal scan from the second to the fourth intercostal space of the left hemi-thorax and from the second to the fifth intercostal space of the right hemi-thorax at the medioclavicular and midaxillary lines of each side. B-lines were defined as a hyperechogenic linear artefact that are emerging from the pleural line, up to the bottom of the screen, and are coherent with respiratory movements.
The number of B-lines has been determined by the sum of the B-lines found in each examined site. Thus, the selected number reflects the extravascular accumulation of the liquid in the lung.
Ultrasound of the Inferior Vena Cava (IVC):
We used an ultrasound (KOLTRON Magic Maestro) with a 3.5 Mhz cardiac probe. The same trained operator explored the IVC within the sub xiphoid window at 2.5 cm of the IVC-right atrial junction. The measurement of the minimum diameter of IVC (DVCI min) was made while breathing in and the maximum diameter of the IVC (DVCI max) was measured while breathing out.
Studied Variables:
Definitions:
The subjective dry weight: it was the prescribed weight taken from the patient logbook, estimated by the attending nephrologist, and based on clinical criteria such as: weight, blood pressure, presence of edema or vascular congestion, as well as the weight on cardiac index measured by the chest x-ray.
Clinical assessment of fluid status was evaluated according to the subjective dry weight:
Accumulated weight was defined as weight gain from the subjective dry weight.
Weight loss was the difference between weight before and after dialysis.
Residual weight was defined as the difference between the obtained weight after dialysis and the subjective dry weight
Impedance Results:
The reference values determined by the impedance to define euvolemia in the normal population are within the range of -1.1 L to 1.1 L 11. Referring to these values, we have classified our patients into three groups: Dehydrated patients if the fluid volume is less than the reference value - 1.1 LPatients in euvolemia if the volume of fluid is within the reference range ± 1.1 L Patients in overload if the fluid volume is greater than the reference value + 1.1L.
Results of the Inferior Vena Cava Ultrasound:
The index of the IVC diameter (iIVCD) was measured by dividing the IVCD max and IVCD min on body surface (Dubois formula) to obtain respectively the iIVCD max, and the iIVCD min. The collapsibility index of the IVC (CiIVC) was calculated by using the following formula:(IVCD max . Referring to the criteria of ultrasound 12, we classified patients:- Dehydrated if the iIVCD max <8mm / m². - Euvolemic If 8mm / m² ≤ iIVCD max ≤ 11.5mm / m². - In overload if the iIVCD max> 11.5mm / m².
Results of Lung Ultrasound:
Pulmonary congestion (Lung congestion) by fluid overload was retained in patients with the following characteristics: 13
1. Multiple artifacts per scan (at least 3 artifacts)
2. Positivity diffuse in more than one scan per side
3. Bilateral Positivity.
Thus, a positive ultrasound test for AIS was defined as the presence of multiple, diffuse, and bilateral artifacts.
Statistical Analysis:
The data were entered into an Excel sheet and analyzed using SPSS software Version 20. In the descriptive analysis, quantitative variables were expressed as mean ± standard deviation and qualitative variables as percentages.
The comparison of means was made using Student test; whereas, the comparison of percentages was performed using Chi-square test. To measure the correlation between the results of the evaluated methods, we have used the bivariate correlation method and estimated their coefficient for each correlation.
The association was evaluated with linear regression. This was calculated for the following parameters:
Weight loss versus decrease of the B-lines number, minimum diameter of IVC, maximum diameter of IVC, and collapsibility of IVC.
A p value of: 0.05 was considered significant.
Ethical Considerations:
An informed consent for participating in the study was obtained from all patients. No invasive investigation means were used. All additional costs associated with the study were funded by the research budget of the Nephrology Department of the University Hospital Hassan II of Fez. The authors declare no conflict of interest.
Results
We included 77 patients, aged 48.13 ± 16 years with a sex ratio (M/F) of 1.1. The average length of hemodialysis was 10.1 ± 0.5 years. The main causes of end-stage kidney diseases were vascular, glomerular, and diabetic in respectively 35%, 20%, and 8.5% of cases.
Forty-four percent of patients are hypertensive. Dyspnea was found in 35.1% of cases, mostly stage I (24.7%) and no patient had dyspnea stage III or IV. Anemia was found in 52.6% with a mean hemoglobin rate of 9.62 ± 2.17 g / dl. Nutritional status is satisfactory in 61.6% of cases (albumin> 40 g / l) with a mean albumin of 39.73 ± 7.5 g / l. Weight status estimates were taken both clinically (with clinical accumulated weight and clinical residual weight). Biological and demographic characteristics are presented in Table 1.
Table 1. Baseline characteristics (mean ± SD)| Parameters | N=77 |
| Age (years; mean±SD) | 48.13 ± 16 |
| Ejection fraction (%; mean±SD) | 66,5± 13,6 |
| Ejection fraction<55% (%) | 13,9% |
| NYHA % | 35,1 |
| stage I | 24,7 |
| Stage II | 10,4 |
| Stage III, IV | 0 |
| Intradialytic hypotension % | 7,8% |
| Dry weight (kg; mean±SD) | 58,73±10,28 |
| Clinical accumulated weight (kg; mean ±SD) | 2,96±1,39 |
| Clinical residual weight (kg; mean ±SD) | 0,56±1,18 |
| Weekly Kt/v | 3,71±0,89 |
| Residual diuresis (ml/day) | 146±394 |
| Rate UF / session (ml) | 2,6 ± 0,76 |
| Hemoglobin level g/dl | 9,6 ± 2,1 |
| Albumine g/l | 39,73 ± 7,5 |
The values of fluid status assessment, which are measured by the impedance, the number of B-lines in lung ultrasound, and the IVC index, decreased significantly (p <0.001) after hemodialysis compared to predialytic values (Table 2).
Table 2. Fluid status before and after hemodialysis session (mean ± SD)| Before HD | After HD | P | |
| Bioimpedance (liter) | +1,2 ± 1,2 | -0,36 ± 1,3 | < 0,001 |
| Lung ultrasound | |||
| Number of B-lines | 4,4 ± 3,4 | 1,3 ± 1,6 | < 0,001 |
| Ultrasound of VCI | |||
| iDVCI max (mm) | 10,2 ± 2,6 | 7,6 ± 2,6 | < 0,001 |
| iDVCI min (mm) | 5,1 ± 3,1 | 2,4 ± 3 | < 0,001 |
| CVCIi % | 53,2 ± 24,1 | 75,1 ± 29,2 | < 0,001 |
The correlation analysis between the results of different techniques before and after hemodialysis is summarized in (Table 3). There was a very significant correlation, between the impedance, lung ultrasound, the index of the maximum and minimum IVC, and its collapsibility both before and after HD.
Table 3. Correlation between different techniques before and after hemodialysis session| B-Lines | iDVCI min | iDVCI max | CVCIi | |
| Before hemodialysis session | ||||
| Over hydratation by bioimpedance | 0,691** | 0,372** | 0,461** | -0,244* |
| B-Lines | _ | 0,610** | 0,607** | -0,485** |
| iDVCI min | _ | _ | 0,788** | -0,558** |
| iDVCI max | _ | _ | _ | -0,916** |
| CVCIi | _ | _ | _ | _ |
| After hemodialysis session | ||||
| Over hydratation by bioimpedance | 0,756** | 0,559** | 0,581** | -0,507** |
| B-Lines | _ | 0,679** | 0,749** | -0,592** |
| iDVCI min | _ | _ | 0,837** | -0,959** |
| iDVCI max | _ | _ | -0,737** | |
| CVCIi | _ | _ | _ | _ |
| * 0,001<p<0,05 | ||||
| **p<0,001 |
After hemodialysis sessions, according to the number of B-lines, 20% of patients were able to have lung congestion, and according to fluid status determined by bioimpedance spectroscopy also 20% of patients were able to be overhydrated. However there were more patients who were classified hypovolemic by ultrasound of the IVC (57%) than by impedance (38%) (Figure 2).
Figure 2.Distribution of groups according to fluid status determined by lung ultrasound comets (a), inferior vena cava diameter (b), bioimpedance spectroscopy (c), before and after hemodialysis session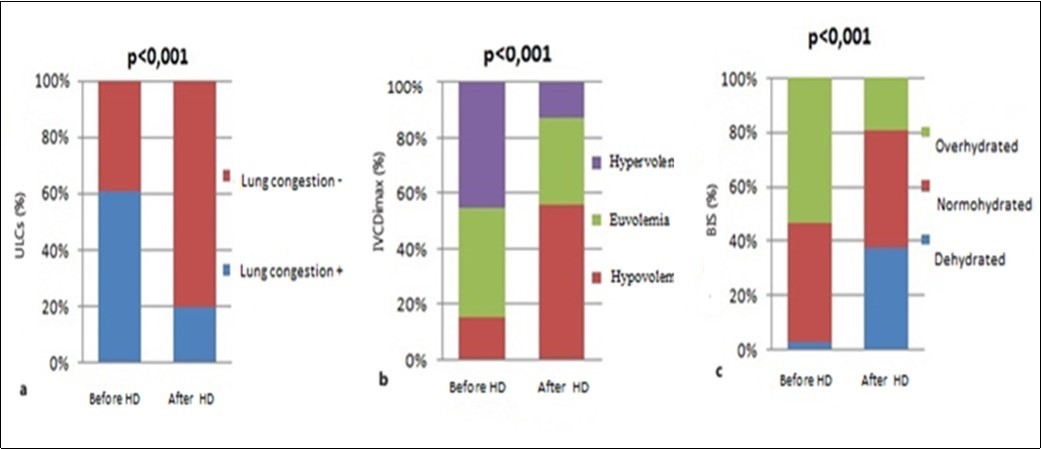
We also studied the correlation between the change of the ultrasound techniques’ results and the movement of water during hemodialysis expressed by weight loss. Only the decrease of the number of the B-lines was significantly correlated with weight loss (β coefficient = 0.36, p<0.005) (Table 4).
Table 4. Linear regression with weight loss| between weight loss and: : | Coefficient β | P |
| B-lines reduction (%) | 0,36 | 0,005 |
| DVCI max reduction (%) | 0,12 | NS |
| DVCI min reduction (%) | 0,04 | NS |
| CVCIi reduction (%) | -0,11 | NS |
Discussion:
Our study aimed to evaluate the efficacy of ultrasound lung for quantifying rapid body fluid changes in asymptomatic patients undergoing HD. We decided to compare ultrasound measurements to bioimpedance estimates, in order to better clarify the relationship between different technological methods and to compare the novel approach with a widely studied technique.
All the techniques investigated in this study show that there is a decrease in overload after hemodialysis session, and there is a good correlation between these different methods. This result fits in with the one found by F Basso et al in 2013 14. Although the new techniques are promising, they have important practical and theoretical limits. For example, skin lesions, wrongly placed electrode, electrical interference, and obesity are limitations of the impedance 15. In addition, the data of the impedance change 120 minutes after the end of hemodialysis 16. Similarly, Agarwal et al. has shown that the measurement of IVCD is a method that well reflects the intravascular volume but has allowed sensitivity for detecting a change in the fluid in post-dialysis time 17. In the same vein, the results show that in post-dialysis, there are more patients who are classified hypovolemic by ultrasound measurements of the IVC than by conductivity measurement. This difference is due to the time required for transferring the fluid from the interstitial sector to the intravascular one "refilling".
Lung ultrasound is a simple method, that is easy, inexpensive, without irradiation, and which can be used at the bedside of the patients 18. In accordance with other recent studies 10, 18, 19, we confirmed a significant decrease in B-lines numbers during the HD session. Importantly, the difference was still a significant event if the mean number of B-lines in our population was particularly small: despite the small predialysis number, we observed a significant decrease in the number of B lines confirming how lung ultrasound can also detect modest variations in the pulmonary imbibition status. By contrast, other studies had a higher number of B-lines, probably to testify a poorer hemodynamic status. There was a higher number of decompensated heart failure patients in the other studies (14.7–25 %) 10, 18. Noticeably, our population had a smaller percentage of patients with NYHA class II (10%), and patients with NYHA class III–IV were explicitly excluded.
On the one hand, we find a significant decrease of pulmonary B-lines following dialysis, demonstrating a rapid clearance of lung imbibition after the removal of volume overload. This decrease is significantly related to weight loss occurring during dialysis, confirming the direct relationship between pulmonary B-lines and water balance. Noble et al. 20 also report on B-lines’ decrease at mid- and end-dialysis session. Our data confirm that lung echography may recognize rapid changes in pulmonary imbibition. On the other hand, the correlation between weight loss and ultrasound data showed that only the decrease of B-lines was significantly correlated; whereas, no correlation existed with the decrease of IVCD. This confirms that the two preceding techniques evaluate two different fluid compartments: the IVCD reflects the volume of intra-vascular water, and the number of B-lines reflects the volume of extravascular water. Thus, the IVCD is not sensitive in assessing rapid changes of fluids during hemodialysis 19, whereas, lung ultrasound can be performed immediately after hemodialysis 20.
Moreover we confirmed a significant association between residual weight determined by bioimpedance spectroscopy and B-lines artifacts measured after HD. These results seem to indicate that lung ultrasound might be an important aid in the estimation of the dry weight, a gold standard examination for its determination being not yet established.
In addition, our data add more evidence to the potential use of lung US in the evaluation and management of the fluid overload of patients on long-term hemodialysis, a crucial issue being that fluid retention is strongly associated with cardiovascular mortality in patients on dialysis 21. Recently, Mallamaci et al. 10 report a significant correlation between B-lines and various indices of cardiac performance in hemodialysis patients, but they found that B-lines’ number is not significantly different with various hydration statuses (evaluated with BIS). We believe that such result might just have been biased by the large proportion of patients with cardiac dysfunction. In our study, 79% of patients were not affected by cardiac disease, thus confirming that, in our population, pulmonary interstitial imbibition is frequently the result of the hypervolemic status, and this also explains the significant correlation between overhydration assessed by BIA and B-lines.
Finally many studies have shown that the interobserver concordance coefficient between an expert sonographer and a nephrology trainee after a 2-h training session on chest US was as high as 0.96; and the inter-probes (standard 3.0-mHZ echocardiography probe and standard 3.5-mHz abdominal probe) coefficient of concordance were as high as 0.98, denoting the considerable simplicity and reliability of the technique 10, 14, 18, 19.
Conclusion:
In conclusion, ultrasound performed at the bedside is now emerging as a reliable, easy-to-apply and safe method for measuring lung water and intravascular overload and their decrease after dialysis even on asymptomatic patients. These observations strongly support the use of lung ultrasound in estimating volume overload and monitoring the response to therapy in hemodialysis patients.
References
- 1.Seibert E, Levin N W, Kuhlmann M K, Zhu F. (2005) Bioimpedance, dry weight and blood pressure control: new methods and consequences. Curr Opin Nephrol Hypertens. 14(6), 543-549.
- 2.Ozkahya M, Ok E, Toz H, Asci G, Duman S et al. (2006) Long-term survival rates in haemodialysis patients treated with strict volume control.Nephrol Dial Transplant. 21(12), 3506-3513.
- 3.Parfrey P S, Foley R N, Harnett J D, Kent G M, Murray D C et al. (1996) Outcome and risk factors for left ventricular disorders in chronic uraemia. Nephrol Dial Transplant. 11, 1277-1285.
- 4.Zoccali C, Benedetto F A, Mallamaci F. (2004) Prognostic value of echocardiographic indicators of left ventricular systolic function in asymptomatic dialysis patients. , J Am Soc Nephrol 15, 1029-37.
- 5.Picano E, Frassi F, Agricola E, Gligorova S, Gargani L et al. (2006) Ultrasound lung comets: a clinically useful sign of extravascular lung water. , J Am Soc Echocardiogr 19, 356-363.
- 6.Lichtenstein D, Mézière G, Biderman P. (1997) The comet-tail artifact. An ultrasound sign of alveolar-interstitial syndrome. , Am J Respir Crit Care Med 156(5), 1640-1646.
- 7.Dexheimer L, Dalcin P, Teixeira C, Beltrami F. (2012) Lung ultrasound in critically ill patients: a new diagnostic tool. , J Bras Pneumol 38, 246-56.
- 8.Picano E, Gargani L, Gheorghiade M Why. (2010) when, and how to assess pulmonary congestion in heart failure: pathophysiological, clinical, and methodological implications. Heart Fail Rev. 15, 63-72.
- 9.Frassi F, Gargani L, Tesorio P, Raciti M, Mottola G et al. (2007) Prognostic value of extravascular lung water assessed with ultrasound lung comets by chest sonography in patients with dyspnea and/or chest pain.JCardFail. 13(10), 830-835.
- 10.Mallamaci F, Benedetto F A, Tripepi R, Rastelli S, Castellino P et al. (2010) Detection of pulmonary congestion by chest ultrasound in dialysis patients. JACC Cardiovasc Imaging. 3(6), 586-594.
- 11.Machek P, Jirka T, Moissl U, Chamney P, Wabel P. (2010) Guided optimization of fluid status in haemodialysis patients. , Nephrol Dial Transplant 25(2), 538-544.
- 12.Brennan J M, Ronan A, Goonewardena S, Blair J E, Hammes M et al. (2006) Handcarried ultrasound measurement of the inferior vena cava for assessment of intravascular volume status in the outpatient hemodialysis clinic. Clin J Am SocNephrol. 1(4), 749-753.
- 13.Volpicelli G, Mussa A, Garofalo G, Cardinale L, Casoli G et al. (2006) Bedside lung ultrasound in the assessment of alveolar-interstitial syndrome. , Am J Emerg Med 24(6), 689-696.
- 14.Basso F, Milan Manani S, Cruz D N, Teixeira C, Brendolan A et al. (2013) Comparison and Reproducibility of Techniques for Fluid Status Assessment in Chronic Hemodialysis Patients. Cardiorenal Med. 3(2), 104-112.
- 15.Peacock W F, Soto K M. (2010) Current techniques of fluid status assessment. , ContribNephrol 164, 128-142.
- 16.Di Iorio BR, Scalfi L, Terracciano V, Bellizzi V. (2004) A systematic evaluation of bioelectrical impedance measurement after hemodialysis session. Kidney Int. 65(6), 2435-2440.
- 17.Agarwal R, Bouldin J M, Light R P, Garg A. (2011) Inferior vena cavadiameter and left atrial diameter measure volume but not dry weight. , Clin J Am SocNephrol 6(5), 1066-1072.
- 18.Trezzi M, Torzillo D, Ceriani E, Costantino G, Caruso S et al. (2013) Lung ultrasonography for the assessment of rapid extravascular water variation: evidence from hemodialysis patients. , Intern Emerg Med 8(5), 409-415.
- 19.N Dugo M Vitturi, M Simoni F Soattin, L Zagatti R Maresca, Maresca M C. (2014) Lung ultrasound during hemodialysis: the role in the assessment of volume status. Int Urol Nephrol. 46, 169-174.
Cited by (8)
This article has been cited by 8 scholarly works according to:
Citing Articles:
Medicine theory and practice (2023) OpenAlex
J. Jeswani, A. Bhardwaj, S. Bhatt - EMJ Radiology (2023) Semantic Scholar
EMJ Radiology (2023) Crossref
EMJ Radiology (2023) OpenAlex
Craig D. Nowadly, K. Kelley, Desiree H Crane, J. S. Rose - Wilderness & environmental medicine (Print) (2021) Semantic Scholar
Yuan-Yuan Chen, Dan-Qian Chen, Lin Chen, Jing-Ru Liu, N. Vaziri et al. - Journal of Translational Medicine (2019) Semantic Scholar
Journal of Translational Medicine (2019) OpenAlex
Shi-Xing Ma, You-Quan Shang, Huan-Qiao Zhang, Wei Su - Journal Of Nephrology Advances (2018) Semantic Scholar
Journal of Nephrology Advances (2018) Crossref
Journal Of Nephrology Advances (2018) OpenAlex
