Abstract
Periosteal osteosarcoma is an uncommon primary malignant bone tumor in dogs and humans. This type of tumor is one of the four variants of osteosarcoma. The main feature is a superficial lesion without evidence of bone marrow involvement. The treatment usually performed is limb amputation. There is insufficient data on long-term disease-free time, surgical resection or local recurrence in dogs. The present review aims to gather information on periosteal osteosarcoma in dogs
Author Contributions
Academic Editor: Renita Marshall, Southern University and A&M College
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Gabriela Noronha de Toledo, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors declare that they are not financially or personally related to any individual or organization that could inappropriately influence or bias the content of this article.
Citation:
Introduction
The osteosarcoma in dogs and in humans share several clinical and biological similarities 1, 2. Due to the implantation ability of the distant tumor cells from the original tumor, the patients affected by this tumor die as a result of lung metastases. Regarding all the aspects given, the treatment becomes challenging for veterinary oncologists 2.
Some years ago, among all the varieties of osteosarcoma that have been described, the periosteal osteosarcoma was identified 3, 4.
Periosteal osteosarcoma is an unusual primary malignant bone tumor and is a variant of osteosarcoma. The major feature of periosteal osteosarcoma is surface lesion without evidence of medullary involvement 5, 4. However, there have been some reports describing periosteal osteosarcoma involving the bone marrow 4.
This tumor occurs in a specific anatomical position. Clinical history, radiographic characteristics and different prognosis differ the periosteal osteosarcoma from the parosteal osteosarcoma, central osteosarcoma, and even from conventional high-grade osteosarcoma that occurs in the periosteal region 6, 7.
In veterinary oncology, studies involving periosteal osteosarcoma in dogs are scarce. The aim of this review was to describe the clinical appearance, diagnosis, and treatment of this rare tumor in dogs in order to stimulate an accurate investigation of this tumor.
Predisposing Situations
The pathogenesis of osteosarcoma is not fully understood but multiple factors may promote its development, such as sex, body weight and age 8, 2. Dogs of large and giant breeds with more than 40 kg of body weight have superior tendency than smaller dogs (less than 15 kg) in developing this tumor 9, 2. Several studies stated that this tumor tends to develop more frequently in males than in females 10, 8, 2.
Chronic Lesions
Metal implants that remain in the body for many years after orthopedic procedures have been associated with the development of osteosarcoma in dogs 11, 8. This fact may explain the occurrence of infections, instability, and corrosion at the implant site by the direct effect they cause. However, it should be noticed that not all orthopedic implants are associated with the development of tumors. Considering these facts, the presence of tumors at the implant site may be only a coincidence 8. In addition, other orthopedic diseases are also described as predisposing factors for osteosarcoma in dogs 12, 8.
Genetic Alterations
Multiple studies have been described the expression of several genes in osteosarcoma cell lines in dogs. These studies reported mutations in tumor suppressor genes and proto-oncogenes (p53, PTEN and human epidermal growth factor - HER-2) beyond the expression of hepatocyte growth factor (HGF), matrix metalloproteinases 2 and 9 and constitutive activation of the signal transducer and activator of transcription 3 (STAT3). The expression of all these genes alone or together are related to local disease progression, metastatic spread, decreased survival time, highly aggressive tumor behavior and tumor grade 8.
Clinical Signs
The pain and lameness present in bone tumors are common clinical signs since the skeleton function supports the weight of the dogs during its activities throughout the life. In addition, specifically in bone tumors, the cells that lead to the pain perception are in the periosteum and medullary cavity, and these cells are activated through mechanical and chemical stimulation 13.
The first signs start with sporadic and mild lameness. Considering the progression of the disease, these signs worsen and the pain and lameness become intense. Palpation is usually painful at the tumor site. Commonly, sudden and acute lameness has resulted from a pathological fracture 8.
In humans, the clinical signs of periosteal osteosarcoma are related to sluggish growth and they are present years before the first clinical evidences. The time and severity of the symptoms are regulated by the tumor growth rate 14.
In both species, most of the tumors are located in long bones of appendicular skeleton 15, 16. In humans, as high-grade osteosarcoma, periosteal osteosarcoma affects young patients and the most common sites are tibia (40%) and femur (38%), followed by ulna and humerus (5-10%) 14. In veterinary medicine, especially in dogs, the number of reported cases is lower than in humans. Thus, the place of incidence it is difficult to establish 17.
Diagnostic Imaging
Diagnostic imaging is one of the most important complementary exams of several pathologies in dogs and cats. Although, it does not always allow inflammatory or infectious conditions to be differentiated from neoplastic disorders 18.
Periosteal osteosarcoma shows different imaging appearances at the site of the diaphyseal lesion of tibia and femur. Different imaging techniques, including radiographs, computed tomography, and magnetic resonance imaging detect changes present in periosteal osteosarcoma, helping to diagnose this tumor. All these techniques allow observing changes in the tumor site, tumor size, cortical alterations, spinal involvement and other tumor characteristics 14
Radiography
The differential diagnosis of periosteal osteosarcoma includes metastatic lesions, periosteal chondrosarcoma, parosteal osteosarcoma, high-grade surface osteosarcoma and medullary or central osteosarcoma 3.
Radiologically, the high-grade surface osteosarcoma is usually similar to periosteal osteosarcoma. The tumor appears as a dense to moderate mineralization with a light and immature appearance7.
Parosteal osteosarcoma usually surrounds the shaft of the affected bone in its later stages. A radiolucent zone between the tumor and the adjacent bone can be observed 4. In this type, this specific lesion indicates an empty space between tumor mass, outgrowing its pedicle and cortex of the normal bone 14.
In humans, the radiological features of periosteal osteosarcoma are the presence of a dense non-homogeneous cortical lesion, usually situated on the proximal diaphysis of a long bone 3. Radiographic findings include thickening of the diaphyseal cortex with scalloping and perpendicular periosteal reaction extending into the broad soft tissue mass. The periosteal reaction is predominantly perpendicular to the diaphyseal cortex. The most common periosteal reaction is non-aggressive, and solid in nature. The radiographic studies can reveal the magnitude and the extension of mineralization in soft tissue mass 14.
The periosteal osteosarcoma shows as a fusiform elevation of the diaphyseal periosteum. Thin and slightly opaque trabeculae are usually seen perpendicular to the cortex, reminiscent of the “sunburst” appearance. Primarily, the opacities in the tumor occur due to osteoid reactive production. There are no extensive granules, opacities or thickened cortex as observed in chondrosarcoma 6.
In dogs, there are no reports of radiographic studies with periosteal osteosarcoma. The information obtained in veterinary medicine comes from human medicine. Therefore, radiographic changes described in dogs were based on the changes described in humans Figure 1.
Figure 1.Lateral radiograph of an periosteal osteosarcoma involving the canine right tibia. The tumor displays an active periosteal reaction and homogeneous soft tissue mass adjacent on the distal third of tibia (*).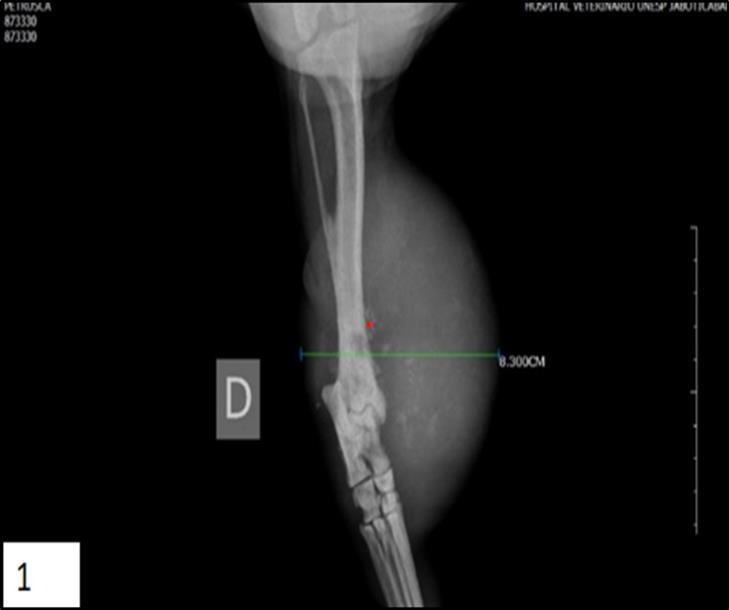
Computed Tomography (CT)
Since its clinical introduction, CT has revolutionized veterinary medicine. This technique is considered to one of the most valuable tools for the imaging work-up of neurological, oncological and orthopedic canine or feline patients 19. The CT is useful for detecting the presence of any soft tissue mass adhered to the cortex or in the medullary canal, cortex thickening, cortical scalloping, and any periosteal reaction perpendicular to diaphysis and extending into the soft tissue mass 20, 14.
CT has been used to investigate primary osteosarcoma lesions, pulmonary metastatic disease, and bone metastases identified on bone scan 21. CT is also used for evaluation of calcification and homogeneity of the non-mineralized component of soft tissue mass 14.
Differential Diagnosis
Differential diagnosis of periosteal osteosarcoma is important to elucidate the nature of osteosarcoma 14. In general, histopathological and morphological findings mingle and may lead to a misleading classification of osteosarcoma. The round cell tumors may be present as a spiculated lesion in the cortical bone. The differential diagnosis should include the other types of osteosarcoma (parosteal osteosarcoma, central osteosarcoma, high-grade surface osteosarcoma and chondroblastic osteosarcoma) 4. Equally important, the guidance for the differential diagnosis of periosteal osteosarcoma can be performed by radiographic characteristics, as well as by clinical significance 14.
Histopathological Findings
The definition of periosteal osteosarcoma is very precise. An accurate diagnosis requires correlation of clinical, radiological and histopathological findings. It is essential to exclude intramedullary osteosarcoma with periosteal extension7.
The basic characteristic of the high-grade surface osteosarcoma is the fast-growing sarcoma arising from the periosteal tissue. The histopathologic description reveals a highly pleomorphic, predominant osteoblastic lesion that have no cartilaginous lobules seen in periosteal osteosarcoma 4, 7. The histopathological finding of the tumor is identical to those of conventional intramedullary osteosarcoma, and even though the medullary involvement is present, most of the tumor is on the surface of the bone 7.
Histopathologically, parosteal osteosarcoma raises from the outer fibrous layer of the periosteum and reveals characteristics of grade I or II osteoblastic osteosarcoma, resembling fibrous dysplasia 4. Parosteal osteosarcoma may be confused with the osteochondroma or osteoma. Other differential diagnoses include periosteal chondroma, periosteal chondrosarcoma and ossifying myositis 17.
The histopathological pattern of the central osteosarcoma include the production of osteoid or immature bone by malignant osteoblasts. However, both the amount and quality of the osteoid matrix can vary markedly between tumors. Osteoid production may be focal, and is often followed by areas of chondroid and/or fibrous differentiation 17.
The periosteal osteosarcoma is an intermediate grade chondroblastic osteosarcoma, corresponding to less than 2% of all osteosarcomas 7. The tumor tends to affect the metaphysis and contains granules or opacities 4, 6. Initially, the periosteal osteosarcoma has the ability to extend out of the soft tissue. Under those circunstances, the lesion may invade the underlying cortex, but rarely extends to the endosteum 17. Based on the histopathological macroscopic and microscopic findings, the bone marrow without secondary involvement defines the diagnosis of periosteal osteosarcoma 4.
In humans, three typical macroscopic lesions of periosteal osteosarcoma were pointed. The first one is a periosteal mass attached to the cortex of the affected diaphysis of the tibia bone. The second is the periosteal tumor showing Codman’s Triangle formations, in the almost intact cortex. Thirdly, the size of the main tumor at the periosteal lesion was larger than the intramedullary lesion 4.
Microscopically, the tumor had mainly a chondroblastic matrix with a malignant osteoid component, and the histological grade of malignancy was grade II or III 4.
Two types of periosteal osteosarcoma have been described in dogs. The first is an aggressive tumor with the same histopathological characteristics and biological behavior of central osteosarcoma. Such tumors may actually be central osteosarcomas that arose in the cancellous bone of the metaphysis of a long bone and for some undetermined reason developed an eccentric growth pattern. The second type of periosteal osteosarcoma is similar to the description found in humans. Only two reports have been described this tumor in the metaphyseal surface of long bones rather than the diaphyseal surface in humans. Both of them, produce dense bone matrix and none of the dogs had metastasis after limb amputation Figure 217.
Figure 2.Photomicrography of a sample of canine bone (tibia) showing neoplastic mesenchymal proliferation, originated in bone cells, HE. Periosteal array of tumor cells, bar 200 μm.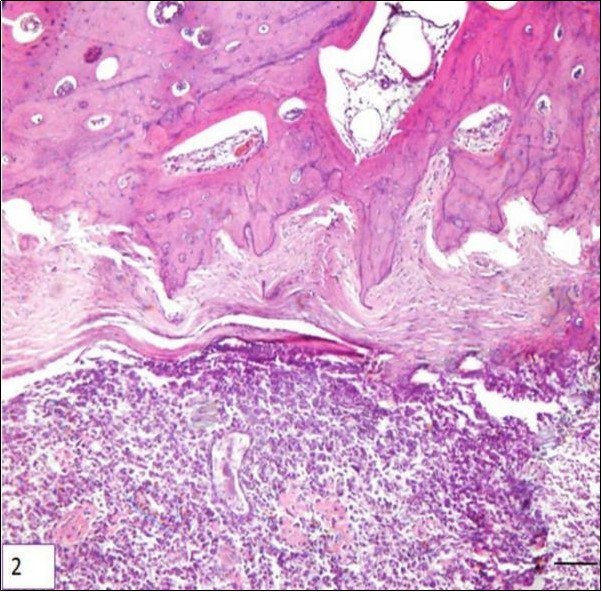
Surgery
In humans, before the surgery, it is essential to establish whether the tumor involves the medullary canal and CT provides this information. If the medullary canal is involved the tumor should be treated like a classical central osteosarcoma or chondrosarcoma 15.
As well as in humans, adequate surgical treatment seems to be essential for tumor control. Limb amputation or limb-sparing surgery is the first method of treatment 2. Limb-sparing surgery is an alternative method to limb amputation 15, 2 and this technique is recommended for dogs with compromising neurologic or orthopedic problems 22. This method can be beneficial for owners who refuse to perform limb amputation 22. Moreover, tumor recurrence appears in 15–25 % of cases. If surgical treatment is timely and adequate, the survival rate is increased and metastasis is significantly reduced 14.
In humans, the prognosis of periosteal osteosarcoma is based on several factors. These include the anatomical position of the tumor, the degree of invasion into the cortex or medulla, and the histopathological grade. Some studies have shown that the anatomical position could influence survival time. This trend was extrapolated to patients with periosteal osteosarcoma. However, according to the results of this study, the anatomical position did not influence the survival rate 15, 14. The number of periosteal osteosarcoma cases reported in dogs is insufficient to determine the prognosis 17.
Chemotherapy
In all types of osteosarcoma, adjuvant chemotherapy may be performed to provide pharmacological support following the surgical procedure. Manifold studies have been performed to assess if the survival time of dogs with osteosarcoma can be prolonged by adjuvant therapy 23. Latest reports showed the efficiency of few chemotherapeutic drugs. The adjuvant chemotherapeutic protocols include the use of cisplatin, carboplatin, and doxorubicin. However, the outcome remains inconclusive 2. Chemotherapy is usually indicated for patients with osteosarcoma, but the advantages are still uncertain 14.
Carboplatin
In a veterinarian study, a comparison of the median survival time of 48 dogs with appendicular osteosarcoma after receiving single-agent carboplatin (300 mg/ m (2) IV q 21 days for 4 cycles) were performed after the following amputation to amputation alone. Dogs treated with adjuvant therapy had a prolonged median survival time (307 days) in comparison to those after surgery alone (138 days) 24, 25, 2.
Cisplatin
Some authors indicated significantly longer median survival time for dogs with appendicular osteosarcoma treated with adjuvant chemotherapy with cisplatin followed by amputation or limb-sparing surgery (322 days) than with surgery alone (138 days) 26, 27. The results indicated the effectiveness of the treatment with cisplatin, but it did not inhibit metastases 2.
Doxorubicin
The use of doxorubicin in osteosarcoma’s treatment is considered as effective as cisplatin or carboplatin. Doxorubicin demonstrated efficacy in slowing down metastasis in dogs with appendicular osteosarcoma with one, two, and three-year median survival time of 35%, 17%, and 9% respectively 28. These results were similar to those with carboplatin as the adjuvant method, which indicates that both drugs might be used to prolong patients’ lives; however, neither of them inhibits metastasis 2.
Alternative chemotherapy protocols for osteosarcomas include using lobaplatin or ifosfamide. The protocol with ifosfamide was evaluated in dogs with osteosarcoma and previously managed with standard chemotherapy. The findings indicated that ifosfamide was well tolerated but had lower anti-tumor activity 29. Promising results showed the efficiency of lobaplatin on the group of 28 dogs with osteosarcoma 30. Compared to dogs used as controls treated only with surgery alone, the results suggest that lobaplatin prolonged the disease-free interval and survival time in dogs with osteosarcoma. More than 20 % of dogs reached one-year disease-free interval and more than 30 % of dogs reached one-year survival time 31, 2.
Another way to improve the efficacy of chemotherapy was comparing the effects of two different antineoplastic drugs administered on the alternative way. The alternating efficiency of administration of cisplatin and doxorubicin after amputation was evaluated in a study. Thirty-eight dogs treated with combination therapy after amputation had significantly longer survival free time than dogs treated with isolated amputation, but this result was similar to that obtained during monotherapy involving carboplatin or doxorubicin 31, 32, 33. These results demonstrated that the combination of the two drugs did not increase the free survival time compared to monotherapy. However, lower adverse effects were observed in these patients 2. On the other hand, dogs with appendicular osteosarcoma receiving carboplatin alone had significantly longer disease-free intervals than dogs receiving carboplatin and doxorubicin in an alternate scheme 23. In summary, there is no clear evidence that combined chemotherapy is more effective than single-agent therapy; however, it may reduce adverse effects, which may indicate that the use of multidrug in a long-term therapy increases the quality of life 2.
Conclusions
The clinical evidence based on the biological behavior of osteosarcomas suggested that the first choice of treatment for periosteal osteosarcoma should be surgery. The recommendation of chemotherapy protocols after surgery remains unclear since complete remission and the occurrence of metastases cannot be warranted. In veterinary medicine, the information is scarce for this type of osteosarcoma. Most of them come from human case reports. Therefore, a study involving periosteal osteosarcoma in dogs should be stimulated to obtain more information about the incidence, biological behavior, tumor site, prognosis and free survival time.
References
- 1.Wycislo K L, Fan T M. (2015) The immunotherapy of canine osteosarcoma: a historical and systematic review.J Vet Int. Med.29 759-769.
- 2.Szewczyk M, Lechowski R, Zabielska K. (2015) What do we know about canine osteosarcoma treatment? – review. , Vet Res Commun 39, 61-67.
- 3.Santos L A, Murray J A, Finklestein J B, Spjut H J, Ayala A G. (1978) The Radiographic Spectrum of Periosteal Osteosarcoma. , Radiol 127, 123-129.
- 4.Suehara Y, Yazawa Y, Hitachi K, Yazawa M. (2004) Periosteal osteosarcoma with secondary bone marrow involvement: a case report. , J Orthop Sci 9, 646-649.
- 6.Bertoni S, Boriani F, Laus M, Campanacci M. (1982) Periosteal chondrosarcoma and periosteal osteosarcoma two distinct entities. , J Bone Joint Surg Br 64, 370-376.
- 7.Wang G D, Zhao Y F, Liu Y, Jiang L, Z J Jiang. (2011) Periosteal Osteosarcoma of the Mandible: Case Report and Review of the Literature. , J Oral Maxillofac Surg 69, 1831-1835.
- 8.Morello E, Martano M, Buracco P. (2011) Biology, diagnosis and treatment of canine appendicular osteosarcoma: similarities and differences with human osteosarcoma.Vet. J.189 268-277.
- 9.Bergman P J, MacEwen E G, Kurzman I D. (1996) Amputation and carboplatin for treatment of dogs with osteosarcoma:. 48 cases(1991to1993).J Vet Intern Med 10, 76-81.
- 10.Kirpensteijn J, Kik M, G R Rutteman, Teske F. (2002) Prognostic significance of a new histologic grading system for canine osteosarcoma. Vet. Pathol.39 240-246.
- 11.Harasen GLG, Simko E. (2008) Histiocytic sarcoma of the stifle in a dog with cranial cruciate ligament failure and TPLO treatment.Comp Orthop Traumatol.21. 375-377.
- 12.Holmberg B J, Farese J P, Taylor D, Uhl E W. (2004) Osteosarcoma of the humeral head associated with osteochondritis dissecans in a dog. , J Am Anim Hosp Assoc 40, 246-249.
- 14.Liu X W, Zi Y, Xiang L B, Han T Y. (2015) Periosteal osteosarcoma: a review of clinical evidence.Int. , J Clin Exp Med 8, 37-44.
- 15.Rose P S, Dickey I D, Wenger D E, Unni K K, Sim F H. (2006) Periosteal osteosarcoma long-term outcome and risk of late recurrence. Clin Orthop Relat Res. 453, 314-317.
- 16.Fenger J M, London C A, Kisseberth W C. (2014) Canine osteosarcoma: a naturally occurring disease to inform pediatric oncology.ILAR. J.55 69-85.
- 17.Thompson K G, Dittmer K E. (2017) Tumor of Bones: Tumors. in Domestic Animals, 5th Ed., Meuten DJ, Ames 356-424.
- 18.Vignoli M, Saunders J H.(2011)Image-guided interventional procedures in the dog and cat.Vet. , J 187, 297-303.
- 19.Ohlerth S, Scharf G. (2007) Computed tomography in small animals – Basic principles and state of the art applications.Vet J.173,254–27.
- 20.Murphey M D, Temple H T, Flemming D J, Gannon F H, Jelinek J S. (2004) Imaging of periosteal osteosarcoma: radiologic-pathologic comparison. , Radiol 233, 129-138.
- 21. (2017) Retrospective Evaluation of Whole Body Computed Tomography for Tumor Staging in Dogs with Primary Appendicular Osteosarcoma. Vet Surg. 46, 75-80.
- 22.MacDonald T L, Schiller D. (2010) Limb-sparing surgery using tantalum metal endoprosthesis in a dog with osteosarcoma of the distal radius. , Can Vet J 51, 497-500.
- 23. (2013) Carboplatin versus alternating carboplatin and doxorubicin for the adjuvant treatment of canine appendicular osteosarcoma: a random.Vet Comp Oncol. 14, 81-87.
- 24.Phillips B, Powers B E, Dernell W S, Straw R C, Khanna C. (2009) Use of single-agent carboplatin as adjuvant or neoadjuvant therapy in conjunction with amputation for appendicular osteosarcoma in dogs.J Am Anim Hosp Assoc. 45, 33-38.
- 25.Saam D E, Liptak J M, Stalker M J, Chun R. (2011) Predictors of outcome in dogs treated with adjuvant carboplatin for appendicular osteosarcoma: 65 cases (1996–2006). , J Am Vet Med Assoc 238, 195-206.
- 26. (1991) Amputation and cisplatin for treatment of canine osteosarcoma. , J Vet Int Med 5, 205-210.
- 27.Berg J, Weinstein M J, Schelling S H, Rand W N. (1992) Treatment of dogs with osteosarcoma by administration of cisplatin after amputation or limb-sparing surgery: 22cases(1987–1990). , J Am Vet Med Assoc 200, 2005-2008.
- 28.Ogilvie G K, Kristal O, Moore A S, Dernell W S, Elmslie R. (2007) Doxorubicin and BAY 12–9566 for the treatment of osteosarcoma in dogs: a randomized, double blind, placebo-controlled study. , J Vet Intern Med 21, 783-779.
- 29.Batschinski K, Dervisi N G, Kitchell B E. (2012) Evaluation of ifosfamide salvage therapy for metastatic canine osteosarcoma. , Vet Comp Oncol 12, 249-57.
- 30.Kirpensteijn J, Teske E, Kik M, Klenner T, Rutteman G R. (2002) b Lobaplatin as an adjuvant chemotherapy to surgery in canine appendicular osteosarcoma: a phase II evaluation. , Anticancer Res 22, 2765-2777.
- 31.Chun R, Kurzman I D, Couto C G, Klausner J, Henry C et al. (2000) Cisplatin and doxorubicin combination chemotherapy for the treatment of canine osteosarcoma: a pilot study. , J Vet Intern Med 14, 495-498.
