Abstract
Purpose of Study:
To study reparative osteogenesis and tissue integration characteristics for implanting three-dimensional mesh structures of titanium nickelide into a bone cavitary defect.
Material and Methods:
The authors modeled cavitary defects of femoral metaphysis experimentally in Wistar rats divided into an experimental group and control one. The study duration was 60 days in total. The methods of radiography, those of light and electron microscopy, X-ray electron probe microanalysis used.
Results:
Under implantation the defect was filled with cancellous bone the volumetric density of which more than 1,5-fold exceeded control values (р < 0.001). The implant had biocompatibility, osteoconductive and osteoinductive properties, it stopped inflammatory processes. The membrane protective barrier which prevented connective tissue sprouting was formed on the implant surface in the defect periosteal zone. The osteointegrative junction was formed being persisted up to the end of the experiment. Reparative osteogenesis was performed by direct intramembranous and apposition type.
Conclusion:
The implant of three-dimensional mesh titanium-nickelide structures has marked osteoplastic properties, and it can be successfully used in orthopedic surgery.
Author Contributions
Academic Editor: Lin Ye, Lecturer,Cardiff University School of Medicine,UK
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2018 Yu.M. Irianov,et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Connective tissue ingrowth from the periosteal surface is the main obstacle for restitution of large-volume bone defects that is caused by a higher rate of migrating fibroblasts comparing with osteogenic cells 1, 2, 3. This can inhibit reparative osteogenesis process completely or partially, as well as be a cause of the defect filling with dense connective tissue of scarry type. In order to create optimal conditions for formation of organotypic regenerated bone the technique of guided reparative osteogenesis was developed using the membrane technology which prevented connective tissue sprouting 4, 5. The membranes of synthetic and natural materials are used for this purpose which, however, are not osteointegrated, they can cause an inflammatory reaction and tissue swelling, in case of their use a repeat surgical intervention is required 6, 7.
The membrane barrier over the bone defect with good contact with the edges of the defect creates a closed space between the "mother" bone and the membrane. The membrane acts as a mechanical protective barrier, preventing the germination of connective tissue into the defect. Only the cells responsible for reparative osteogenesis fall into the defect from the surrounding bone tissue. In this case, there is a process of directed osteogenesis without the influence of other tissues 8. Numerous studies have been performed to elucidate the main features of directed bone regeneration 9, 10, 11, 12, but a number of issues remain unclear to this day. In particular, it is not clear whether there is a difference in the effect of regeneration, from placement in the closed space between the bone and the membrane, osteoconductive or osteoinductive materials. Can individually manufactured membranes be used to achieve the desired shape and volume of bone tissue in the defect?
New possibilities have arisen due to introducing medical technologies related to using the implants based on nickel and titanium which are approached to bone tissue by their mechanic characteristics and are biocompatible 13, 14, 15, 16.
The purpose of this work is to study reparative osteogenesis morphological features when implanting mesh structures of titanium nickel ide into a cavity defect of femoral metaphysis.
Materials and Methods
Description of the Intervention
Cavitary defects of femoral metaphysis modeled in adult male Wistar rats in the experimental group (n=20) and control one (n=20); the defect volume was 0.02 cm3 that amounted for about 40% of total metaphysis volume. The volume of the metaphysis was determined by the volume of water displaced by it. The defect was filled with water using a measuring syringe and its volume. All the manipulations were performed according to the Ministry of Health Order No 708 of 23.08.2010 “Approval of the rules of good laboratory practice”.The implant was introduced into the defect in the animals of the experimental group, no additional manipulations performed in the control group.
Characteristics of the Implant
The implant had a mass of 0.017 ± 0.001 g, a volume of 0.02 ± 0.001 cm3. The implant was a mesh framework made of nickelide-titanium thread (TH-10 brand of 90 µm caliber) formed by the type of knitting with cells – through open pores of 100-300 µm diameter 17. The thread was made of composite material comprising a core of nanostructured monolithic titanium nickelide and a titanium-oxide microporous surface layer of 5-7 µm (Research and Production Enterprise "MITs", Tomsk, Russia; Certificate No РООСRU.АЯ79Н18304). The microrelief of the surface layer of the filament was characterized by sharply pronounced roughness and nanostructuredness, the presence of a multitude of interconnected open capillary micropores (Figure 1a, Figure 1b, Figure 1c). The porosity of the implant was 73.6 ± 3.56%.
Figure 1.A mesh design made of titanium nickelide implanted into the defect of the metaphysis of the femur: a - general view of the implant, b - microrelief of the surface layer of the implant filament, c - adhesion of malodifferentiated cells and newly formed capillary terminals (arrow) on the surface of the filament 7 days after the operation. Scanning electron microscopy, magnification: a - × 13, b - × 190, c - × 475.
Methods of Research and Registration of Outcomes
The animals were withdrawn from the experiment after 7, 14, 30 and 60 days (five animals were used for each time point). The bone meta-epiphyseal zone was sewn out, fixed in 2% paraformaldehyde and glutaraldehyde solution and embedded in araldite (without decalcification). The araldite blocks studied using INCA-200 Energy X-ray electron probe micro-analyzer (“Oxford Instruments”, England) and JSM-840 scanning electron microscope (“Jeol”, Japan). With the help of this device, images (maps) were obtained in the characteristic X-ray radiation (emission) of calcium atoms. The structures of bone tissue were visualized and the necessary measurements were made. The bulk density of bone tissue was determined (in %). As the ratio of the area occupied by bone structures in the image to the total area of the map. The bone tissue compactness index (ratio of bone structure and non-mineralized components) and concentration of osteotropic macronutrients (calcium, phosphorus, magnesium, sodium and sulfur) in the bone regenerate were calculated. 14, 15, 16.
Methods of a Statistical Analysis
We used the program "Microsoft Excel - 2010" and StatSoftStatistica v6.0. The mean (M) and standard error of the mean (SEM) were calculated. The Mann-Whitney (U Test) and Student t-test was used to compare the differences between the two independent groups. A probability value of P<0.05 was considered statistically significant.
Results
The process of bone formation which occurred from periosteum, endosteum, bone marrow and the damaged bone structures of the defect edges was observed in the both groups of animals 7, 14 days after surgery. In the control group of animals,a non-matured regenerated bone (of connective-tissue type) was formed in the periosteal zone, its collagen fibers grew from periosteum into the defect central zone as strip-like bundles. The defect was filled with loose connective and granulation tissue where the foci of lymphocytic and plasmacytic infiltration and fibrin clots were located. The islets of reticulofibrous bone tissue represented as a fine-cellular network of interwoven bone-osteoid trabeculae were formed near the defect outer edges in the periosteum, as well as in the endosteum. Randomly arranged fragments of the damaged bone structures, as well as separate unrelated foci of newly formed bone tissue as short bone-osteoid trabeculae lining the defect inner surface located near the defect inner edges. The bone tissue volumetric density in the defect and the compactness index of the regenerated bone amounted to about 30% (P < 0.001) of the intact metaphysis values 14 days after surgery in the control group of animals (Table 1).
Table 1. Bone tissue volumetric density in the metaphysis defect in the animals from the control group (Cont) and the experimental one (Exp) and in the intact metaphysis of the contralateral limb (М±SEM)| Parameters | Period of the experiment, days | Metaphysis of contralateral limb | |||||
| 14 | 30 | 60 | |||||
| Bone tissue, % | Cont | Exp | Cont | Exp | Cont | Exp | |
| 8.443±0.381 | 12.174±0.6131 | 13.543±0.681 | 19.764±0.9621 | 15.022±0.744 | 22.984±1.0941 | 25,072±1,212 | |
| Compactness index | 0.092±0.005 | 0.143±0.0123 | 0.164±0.013 | 0.252±0.0212 | 0.181±0.013 | 0.310±0.0212 | 0,333±0,024 |
A thin membrane-like cover of connective tissue was formed round the implant threads and cells in the defect periosteal zone in the experimental group animals 7, 14 days after surgery (Figure 2a). The cover formation began on the surface of the thread in the places of its weavings and spread from the periphery of the cells to their center. The cover had a layered structure. The inner layer consisted of dense formalized connective tissue. Collagen fibers were collected into dense circularly oriented lace-like bundles, and they braided the implant threads in the form of a sleeve, they were firmly fixed to their microporous surface, grew into the gaps between them and provided the fixation of the implant threads both between each other and in the bone defect (Figure 2b).
Figure 2.Connective-tissue cover on the implant surface in the defect periosteal zone 7 (a) and 14 (b, c) days after surgery. Scanning electron microscopy, magnification × 160.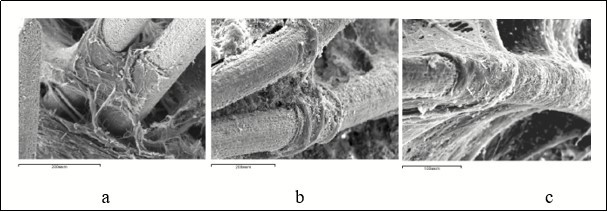
The areas of active appositional bone formation were observed below the connective-tissue cover in the endosteal and central zone of the defect and at its edges round the implant structures, as well as on their surface. A layer of reticulofibrous bone tissue of 300-400 µm thickness emerged on the implant thread surface directly forming osteointegration connection (Figure 3a).
The implant threads in the osteointegration areas were coated with mineralizing bone matrix. Newly formed trabeculae grew into the implant fine-cellular structure (Figure 3b,Figure 3c). The results of quantitative studies (Table 1) evidenced of reparative osteogenesis significant activation, as well as of increasing the maturity degree of newly formed bone tissue in the regenerated bones of the animals of the experimental group comparing with those in the control group. The bulk density of bone tissue in the defect in the animals of the experimental group was greater by 44.19%, and the compactness index by 55.56% compared to the values in the control group of animals (P < 0.001).
Figure 3.Reparative osteogenesis in the metaphyseal defect 7 (a) and 14 (b, c) days after surgery, arrows indicate the areas of osteointegration; a, b – maps of x-ray electron probe microanalysis, image in characteristic X-ray emission of calcium atoms, c – scanning electron microscopy (organic components removed with 6% sodium hypochlorite solution), magnification: а - × 100, b - × 25, c - × 70.
The defect was filled with regenerated bone 30-60 days after surgery in the control group of animals, where little-mineralized dense unformed connective tissue growing into from the periosteal surface prevailed. The initial stages revealed for periosteal-intermediary uniting and forming cortex resembling cancellous bone by structure (Figure 4a, Figure 4d).
The operated metaphyseal zone acquired a marked conical shape. Little-calcified dense connective tissue prevailed in the periosteal zone of the regenerated bone. The fine-cellular bone structures of the periosteal regenerated bone fused with endostea newly formed trabeculae, arcuately grew into the defect central zone and formed a thin crescent layer of newly formed cortical bone The initial stages revealed for periosteal-intermediary uniting and forming cortex resembling cancellous bone by structure (Figure 4a). Osteogenesis foci were observed in the defect central and marginal zones where osteoid areas revealed, as well as fragments of newly formed little-mineralized reticulofibrous bone trabeculae isolated from each other by wide interlayers of loose connective tissue with the cavities filled with lymphocytic and macrophage elements. Bone tissue volumetric density in the defect, index of compactness of the regenerated bone, the content of calcium and phosphorus amounted to 50-60% of the intact metaphysis values (p < 0.001) (Table 1,Table 2).
Table 2. Content of osteotropic chemical elements in the regenerated bone of the control and experimental groups of animals 60 days after surgery and in the intact metaphysis of the contralateral limb (M±SEM, %)| Elements | Control(n=5) | Experiment(n=5) | Metaphysis ofcontralateral limb(n=5) |
| Sodium | 0.41±0.02 | 0.44±0.02 | 0.40±0.02 |
| Magnesium | 0.22±0.01 | 0.25±0.01 | 0.22±0.01 |
| Phosphorus | 1.93±0.04 | 2.94±0.131 | 3.22±0.15 |
| Sulfur | 0.23±0.01 | 0.30±0.022 | 0.22±0.01 |
| Calcium | 3.85±0.16 | 5.89±0.261 | 6.44±0.31 |
Figure 4.Reparative bone formation in the defect of femoral metaphysis in the control (a, d) and experimental (b, c, e, f) group of animals; a, b, c - 30 days after operation; d, t, f - 60 days after operation; a, b, d, e – maps of x-ray electron probe microanalysis, image in characteristic X-ray emission of calcium atoms, magnification × 20; c, f – scanning electron microscopy, magnification: c - × 670, f – 800.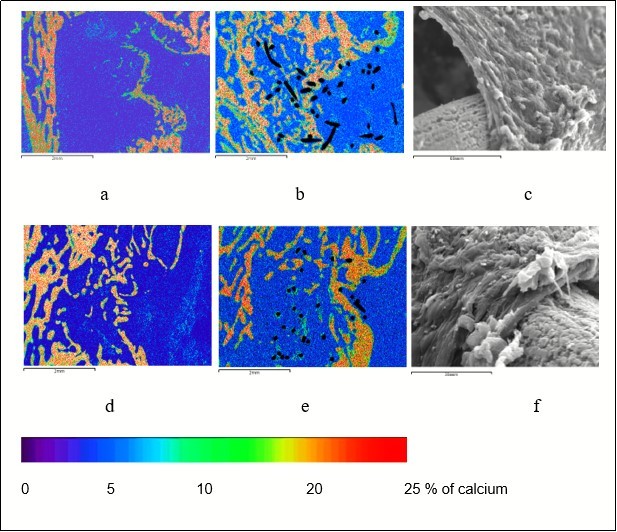
In the experimental group of animals, the area of bone defect was filled with regenerated bone where cancellous bone prevailed 30-60 days after surgery (Figure 4b, Figure 4e). A new area of cortical layer was formed represented as compact bone of lamellar structure. The bundles of collagen fibers of the protective cover dense connective tissue located on the defect periosteal surface round the implant which formed the interweaving of village fence type (Figure 4c, Figure 4f). The implant threads were surrounded by osteoid or they were completely overgrown by newly formed bone tissue thereby forming a composite – compact bone reinforced by titanium nickelide. Bone tissue volumetric density, index of compactness and mineralization degree of the regenerated bone were somewhat less comparing with the intact metaphysis values, but these differences were not statistically significant by the end of the experiment, and, at the same time, they more than 1.5-fold exceeded (p < 0.001) the values in the control group of animals (Table 1,Table 2).
Discussion.
Substitution or reconstruction of extensive bone cavity defects caused by congenital or acquired pathology is an actual medical and social problem The main methods of surgical treatment are variants of bone plastic with the use of various synthetic, biological and composite materials. However, when studying the long-term results, it was found that most of these materials are not osseointegrated, but surrounded by a fibrous capsule. The use of own bone (autotransplantation) is associated with an additional traumatic effect and is limited by the inability to take the necessary amount of autologous bone material, especially in children. In this case, there is a risk of transmission of various diseases and the development of a number of serious complications of the immune nature, which is often accompanied by graft rejection and suppuration in the postoperative period 18. The results of this study showed that the implant studied well performed the defect form, had good biocompatibility, expressed osteoconductive properties. The microporous structure of the surface layer of the implant filaments ensured the adhesion of the cells of the regenerate and the formation of the osseointegrative compound, which was maintained until the end of the experiment. The development of osteogenic differentiation of cells on the surface of the implant was proved by the development of a specific calcified matrix. Regenerate tissues and blood vessels easily germinated into the implant without disrupting the integrity of the implant. In the periosteal region of the defect, a layer of dense connective tissue was formed on the surface of the implant, which served as a biological protective barrier preventing the germination of paraosal connective tissue. The defect was compensated by a spongy bone, the bulk density of which at all stages of the experiment was more than one and a half times higher than the control indices, and its mineral composition approximated the parameters of the spongy bone of the intact metaphysis. Reparative bone formation was carried out by the type of direct intramembranous osteogenesis. In none of the cases there were signs of an inflammatory process, which confirmed the data we received earlier 15. For the first time during the implantation, an artificial composite biological tissue reinforced with titanium nickelide filaments was obtained: dense fibrous connective tissue, spongy and compact bone. The fine-celled structure and microporosity of the surface of the implanted structures created capillary properties, due to which adsorption (mechanical impregnation) of endogenous bone morphogenetic proteins (BMP) and growth factors (cytokines) occurred. The functional activity of the latter, providing proliferation and accelerated differentiation of osteogenic cells, stimulating the synthesis of collagen, osteocalcin, alkaline phosphatase, activating the mineralization of the organic matrix of bone 18, 19, ensured the osteoinductiveness of the implant.
Conclusion.
The implant made of mesh structures of titanium nickelide is an effective osteoconductor and osteoinductor, provides prolonged activation of reparative bone formation and spatial development of bone tissue in the defect. Atraumatism of surgical intervention, absence of biological rejection reaction place the implant in the range of the most optimal osteoplastic materials, and its application seems theoretically grounded and promising, especially in patients with reduced osteogenetic and reparative potential, including in mature and elderly patients, as well as in children.
References
- 1.Liu J, Kerns D G. (2014) Mechanisms of guided bone regeneration: a review. doi: 10.2174/1874210601408010056.Open DentJ,eCollection2014,8;56.
- 2.Jwa-Young Kim, Byoung-Eun Yang, Jin-Hee Ahn, Park Sang O, Hye-Won Shim.Comparable efficacy of silk fibroin with the collagen membranes for guided bone regeneration. in rat calvarial defects.J AdvProsthodont.6 (2014) 539. doi: 10.4047/jap.2014.6.6.539 PMCID: PMC4279054 .
- 3.Jung R E, Fenner N, Hämmerle C H, Zitzmann N U.Long-term outcome of implants placed with guided bone regeneration (GBR) using resorbable and non-resorbable membranes after 12-14 years.Clin Oral Implants Res.24(2013)1065. 10-1111.
- 4.Hämmerle C H, Jung R E. (2003) Bone augmentation by means of barrier membranes.Periodontol2000. 33(1), 36-53.
- 5.Karring T, Nyman S, Gottlow J, Laurell L. (1993) Development of the biological concept of guided tissue regeneration--animal and human studies.Periodontol2000;1(1993):. 26-35.
- 6.SchmidmaierG BaehrK, Mohr S, Kretschmar M, Beck S, WildemannB. (2006) Biodegradable polylactide membranes for bone defect coverage: biocompatibility testing, radiological and histological evaluation in a sheep model.ClinOralImplantsRes.;. 17(4), 439-444.
- 7.van Leeuwen AC, Huddleston Slater JJ, GielkensPF de Jong JR, Bos RR GrijpmaDW. (2012) Guided bone regeneration in rat mandibular defects using resorbable poly(trimethylene carbonate) barrier membranes. doi: 10.1016/j.actbio.2011.12.004.Acta Biomater.8:. 1422. Epub2011Dec13
- 8.Dahlin C, Linde A, Gottlow J, Nyman S. (2008) Healing of bone defects by guided tissue regeneration. Plast Reconstr Surg. 81, 672-6.
- 9.Urist M R, McLean F C. (2003) Recent advances in physiology of bone. , I. J Bone Joint Surg Am 45, 1305-13.
- 10.Dahlin C, Sennerby L, Lekholm U, Linde A, Nyman S. (2009) Generation of new bone around titanium implants using a membrane technique: an experimental study in rabbits. , Int J Oral Maxillofac Implants;4: 19, 25.
- 11.Schropp L, Wenzel A, Kostopoulos L, Karring T. (2003) Bone healing and soft tissue contour changes following single-tooth extraction: a clinical and radiographic 12-month prospective study. Int J Periodontics Restorative Dent. 23, 313-23.
- 12.Iasella J M, Greenwell H, Miller R L. (2003) Ridge preservation with freeze-dried bone allograft and a collagen membrane compared toextraction alone for implant site development: a clinical and histologic study in humans. , J Periodontol 74, 990-9.
- 13.Chernov V F, Bevzyuk A N, Chernov A V, Iryanov Y M. (2008) The study of the implantation of mesh structures of NiTi.Med Science Education Urals.2;98. [Article. in Russian
- 14.YuM Iryanov.Iryanova TYu Bone defect replacement under condition of transosseous osteosynthesis and titanium nicelide implant application.Morphology.4 (2012) 83. [Article. in Russian
- 15.Iriyanov Y M, Chernov V F, Radchenko S A, Chernov A V. (2013) Plastic efficiency of different implants used for repair of soft and bone tissue defects. doi: 10.1007/s10517-013-2191-4.Bull Exp Biol Med.155. 518.
- 16.Irianov Y M, Diuriagina O V, Karaseva T Y.Karasev ЕА The osteoplastic effectiveness of the implants made of mesh titanium nickelide constructs.BosnJ Basic Med Sci.1(2014)4.
- 17.YuM Iryanov.Iryanova TYu. (2011) The implant to replace bone defect]. RF Patent 111759A61F2/28;12. [Article. in Russian