Abstract
Water availability is one of the fundamental drivers for biological activities and terrestrial carbon cycling. Although the response of soil respiration to precipitation has been well documented in arid and semiarid ecosystems, our understanding of its pattern in forests is rather limited. This study was conducted to examine the difference of precipitation effect on soil respiration under different canopy conditions in a temperate coniferous (Pinus armandii Franch) and broadleaved (Quercus aliena var. acuteserrata) mixed forest in Central China. The results showed that precipitation significantly reduced soil temperature, but increased soil volumetric water content and soil respiration (6.0%-35.3%). Precipitation caused a greater increment in soil respiration beneath the canopy of broadleaved trees (24.2%) than that beneath coniferous ones (13.5%). Precipitation-induced increase in soil respiration was consistently lower beneath the canopy of small size classes (7.1%-32.6%) than large size classes (9.5%-33.3%). Mean soil respiration of forest gaps increased 22.4% following precipitations. Our study highlights the positive response of soil respiration to precipitation pulses in water-unlimited ecosystems. The findings suggest that the spatial heterogeneity of soil respiration to precipitation pulse under different canopy conditions should be emphasized while assessing terrestrial carbon cycling and its feedback to climate change.
Author Contributions
Academic Editor: SYLVESTER IZAH, Department of Biological Science, Niger Delta University, Nigeria
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Yanchun Liu, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Soil respiration, the primary process by which CO2 assimilated by plants returns to the atmosphere 1, is one of the major components of the terrestrial ecosystem carbon (C) cycle 2. Previous researches have shown that this process is influenced by environmental factors 3, 4. Soil temperature and moisture conditions are generally identified as two major factors that are closely related to soil respiration 5, 6. A rapid pulse of CO2 efflux following precipitation has been observed in many ecosystems 1, resulting from rapid microbial growth and mineralization of accessible soil organic content including dead microbial biomass 7. While the presence of CO2 efflux is consistent for the studied ecosystems, the response magnitudes of respiration vary among ecosystems due to different vegetation characteristics, soil structure, and nutrient conditions. In the forest-grassland transition zone of the Loess Plateau, precipitation results in 1.6 times increase in soil respiration for oak forests 5. However, CO2 efflux increased up to 30 times after experimental rewetting in Sonoran desert ecosystem 1. Pulse magnitude of CO2 is also sensitive to the amount, timing, and intensity of precipitation. Munson et al. 8 finds that total C loss and duration of elevated CO2 flux increased with elevated precipitation amounts in a short grass steppe. Heterotrophic respiration is considered to reach an asymptote with increasing precipitation size because of the short lived microbial activity 7, 9, but larger precipitation events are needed to initiate autotrophic respiration due to deeper distribution of root system 1.
Numerous studies conducted in arid and semiarid ecosystems (pulse-driven systems) 4, 7, have addressed the precipitation effects on soil respiration and soil C stocks as well as corresponding microbial activity 1, 5, 7, 10, 11. Although soil water availability is not a limiting factor in humid and semi-humid areas, soil respiration may also be triggered by the discrete rainfall events. The cumulative triggered flush of CO2 of forests is likely contribute more to atmospheric CO2 concentration due to its greater C input and higher occurrence of precipitation events compared to that in arid and semiarid landscapes. For example, Xu et al. 12 finds that the amounts of C lost, in association with summer rain events, are greater at the sites with higher primary productivity and soil C content. In addition, spatial heterogeneity of soil respiration is likely greater than that in grassland and cropland due to its higher heterogeneities in topography and plant distribution. For example, it has been reported that overstory coverage, species and size class can cause variations in plant interception, especially for a small precipitation event 10, 13, 14. Therefore, the responses of soil respiration to precipitation in forest ecosystem may largely depend on the tree species and canopy conditions, which is an important regulator of the pulse size due to its biological and physical effects on soil biotic and abiotic factors. However, how frequent fluctuations in precipitation influence soil respiration in forest ecosystems has not been evaluated to the same extent as in semiarid and arid ecosystems 5.
Extreme weather is happening frequently as a result of climate change 15. Drought does not occur only in arid and semiarid ecosystem any more, it may take place widely in terrestrial ecosystems 16, 17, 18. For the forests, episodic precipitation inputs could impact the photosynthetic and respiratory activity of plants 14 as well as the soil respiration, including respiration by plant roots and from heterotrophy 5, 7. Thus, exploring and quantifying the responses of soil respiration to episodic precipitation in forest ecosystems are critical for assessing terrestrial C cycling under climate change.
This study investigated the spatial heterogeneity of soil respiration response to precipitation pulse in a temperate mixed forest in Central China. The objectives of this study were to (1) determine the effects of tree species with different canopy shape on soil microclimate and respiration beneath canopy condition after rainfall events; (2) compare the responses of soil respiration to precipitation pulse among different size classes; and (3) analyze the diurnal dynamics of soil respiration under canopy and forest gap conditions during precipitation events.
Materials and methods
Site Description
The study site located at Baotianman Forest Ecological Research Stations in the Baotianman National Nature Reserve (111°47′-112°04′ E, 33°20′-33°36′ N), Henan Province, China. Altitude ranges from 600-1800 m above sea level. Mean annual temperature is 15.1 ºC, with monthly air temperature ranging from 1.5 ºC in January to 27.8 ºC in July. Mean annual precipitation is approximately 885.6 mm with 60% distributed in summer (from June to August). Upland soils were dominated by Haplic luvisol (FAO classification). The typical forest in this area is warm-temperate forest, with abundant temperate deciduous broadleaf trees and some plant components from sub-tropical forests. The dominant tree species of the warm temperate forest is Quercus spp., accounting for approximately 70% of the total area 19. The dominant species are Toxicodendronverniciflnum(Stokes) F. A. Barkley, CarpinuscordataBI., Acer spp., PlatycaryastrobilaceaSieb.ET. Zucc, CustaneaseguiniiDode, Lindera obtusilobaBI..
Experimental Design
The experiment was carried out in a coniferous and broadleaved mixed forest on a northeastern aspect with a slope of 28°. A watershed of ~ 1.5 ha area with an average slope of 18 ° was selected to conducted the experiment. Dominant species were P. armandii and Q. aliena var. acuteserrata, with average diameters at breast height of 23.6 ± 7.90 cm and 19.1 ± 8.62 cm, accounting for 30.4% and 34.3% of total cover, respectively. All plants within the watershed were surveyed, and dimensions including diameter at breast height (DBH), height and health state were recorded. Totally 16 sampled trees from two diameter size classes (small class (~15 cm) and large class (~30 cm)) for P. armandii (PS and PL) and Q. aliena var. acuteserrata (QS and QL) were selected to investigate soil respiration under the canopy (Table 1). Canopy diameter of each selected tree was estimated from two vertical directions (N-S, E-W) with a meter stick (Table 1). Four forest gaps (FG) with area of about 5 m × 10 m were selected to investigate the soil respiration under open canopy.
Table 1. Canopy characteristics of trees by size class (mean ± SE., n=4)| Species | Size class | Mean DBH (cm) | Mean canopy | Mean crown |
| width (m) | depth (m) | |||
| P. armandii | Small | 16.77±0.55 | 1.83×1.67 | 3.80±1.15 |
| Large | 32.67±0.61 | 5.50×5.17 | 8.77±2.29 | |
| Q. aliena | Small | 14.50±0.89 | 2.63×2.03 | 4.37±2.02 |
| Large | 32.03±3.95 | 7.47×6.50 | 8.97±1.01 |
Soil Respiration Measurement
Soil respiration was measured using an automated soil respiration system (LI-8100, LI-COR, USA) from June to August in 2014. Three polyvinyl chloride (PVC) collars (19.6 cm inside diameter and 8 cm height) were inserted into the forest floor to a depth of 5 cm around each sampled tree at an angle of 120 ° with 1 m distance to the bole. One PVC collar was installed in each forest gap (FG). All collars were left at the site for the whole study period. Living plants and obvious litter were removed 24 hours before efflux measurements.
Soil respiration without precipitation event (baseline) was expressed as the average values investigated in the morning (8:30-11:00) and afternoon (2:30-5:00) on June 19, and before then there had been no effective rainfall events (>1 mm) for nearly 20 days. Flush of soil respiration by precipitation was measured approximately 30 minutes after each rainfall event. Totally 9 rainfall events have been observed during the study period from June 22 to August 14 (June 22 for 5.81 mm, June 24 for 5.01 mm, June 28 for 3.43 mm, July 6 for 14.03 mm, July 16 for 4.13 mm, July 19 for 20.57 mm, July 22 for 4.78 mm, August 1-2 for 24.19 mm, August 16 for 10.28 mm), in which soil respirations of two rainfall events (July 6, July 16) were not included in this study. Besides, two rainfall events (July 22, August 1-2) were selected to measure the diurnal dynamics of soil respiration with 30 minutes interval beneath the canopy and in forest gap during the precipitation.
Measurements of Soil Temperature and Moisture
Soil temperature at 10 cm was measured adjacent to each respiration collar with a portable temperature probe connected with the LI-8100. Soil volumetric water content (SVWC) at 0-5 cm was measured with a portable time domain reflectometer MPKit-B soil moisture gauge (NTZT Inc., Nantong, China) at three points closed to each collar. Precipitation data were collected from a nearby weather station less than 400 m away from the sample plot.
Statistical Analysis
Normality and homogeneity of variance for field data were first tested, and repeated measures ANOVA was then used to test the significance of soil respiration rate, soil volumetric content at depth of 0-5 cm and soil temperature at depth of 10 cm as affected by precipitation, species, size class and their interactions. Then one-way ANOVA and post-hocs (LSD) was used to test the significance of soil respiration rate of baseline, increments of soil respiration rate after each rainfall event among different treatments (species, size class, and forest gap). Data is expressed as mean ±1 standard error (SE). The statistical analyses were performed in SPSS 16.0 for Windows (SPSS Inc., Chicago, IL, USA).
Results
Soil Temperature and Moisture
Soil temperature without precipitation (0 mm) was higher than those following different precipitation events for all canopy conditions (Figure 1a). All precipitation events significantly decreased soil temperature under PS, PL, QS, QL, and FG (Table 2, P < 0.001). Soil temperature of FG was significantly greater than that of PS, PL, QS, and QL in all rainfall events. Soil temperature of the small size classes (PS and QS) was lower than the large ones (PL and QL) following each rainfall event (Table 2, P = 0.035). In addition, coniferous species had a relatively higher soil temperature than broadleaved species (Table 2, P = 0.0214).
Figure 1.Soil temperature at 10 cm (a) and volumetric water content (SVWC) at 5 cm (b) for different canopy conditions following different precipitation events (mean ± SE, n = 4). PS, PL, QS, QL, and FG represent P. armandii with small size class and large size class, Q. aliena with small size class and large size class, and forest gap, respectively.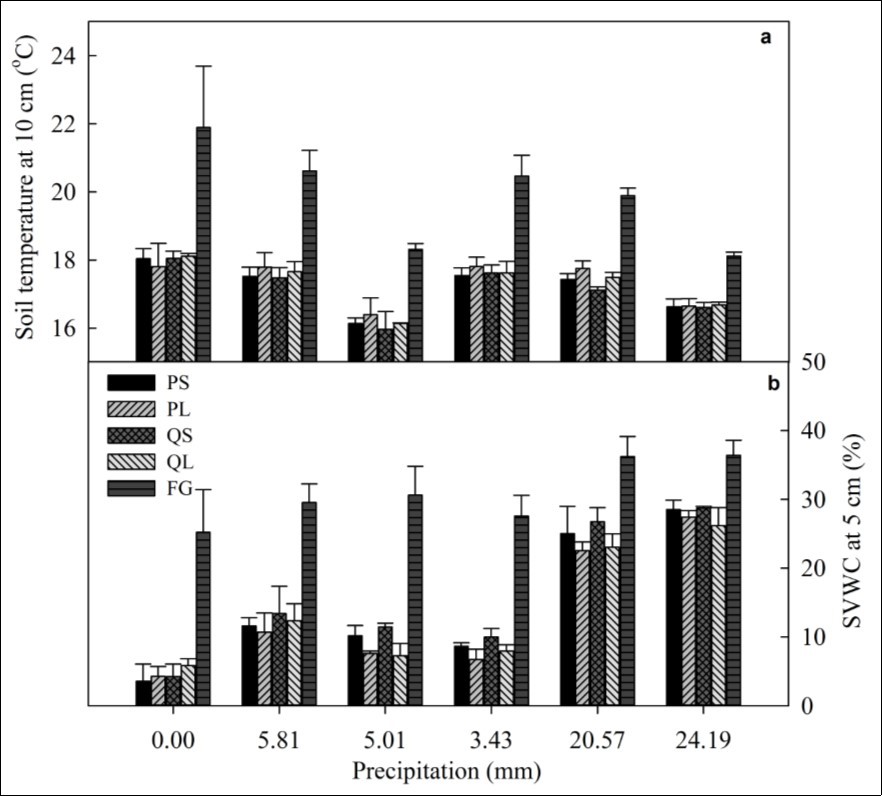
Soil volumetric water content (SVWC) was extremely low in all four canopy conditions without precipitation events (0 mm, all < 6%, Figure 1b). Precipitation led to remarkable increases in SVWC for all canopy conditions (Table 2, P < 0.0001). Similar to soil temperature, the SVWC of FG was consistently greater than those beneath the canopies (Figure 1b). The SVWC for small size classes was significantly higher than larger classes (Table 2, P = 0.0003). In addition, broadleaved species had a higher SVWC than coniferous species (Table 2, P = 0.0461). There were significant effects of precipitation × size class interactions on SVWC (Table 2,P=0.0051), which indicated that the differences of SVWC among different canopy size classes declined with increased precipitation intensity.
Table 2. Results (P value) of repeated-measures ANOVA for soil respiration rate (SR), soil volumetric water content (SVWC) at the depth of 0-5 cm, and soil temperature (ST) at the depth of 10 cm in response to precipitation, species, and size class.| Factors | SR | SVWC | ST |
| Precipitation | < 0.001 | <0.0001 | < 0.001 |
| Species | 0.5891 | 0.0461 | 0.0214 |
| Size class | 0.2191 | 0.0003 | 0.0349 |
| Species × Size class | 0.0071 | 0.4702 | 0.4724 |
| Precipitation × Species | 0.5331 | 0.3841 | 0.5818 |
| Precipitation × Size class | 0.0378 | 0.0051 | 0.1474 |
| Precipitation × Species ×Size class | 0.7141 | 0.5217 | 0.0581 |
Soil Respiration
Under 0 mm precipitation, soil respirations ranged from 2.24 μmol m-2 s-1 for QS to 2.99 μmol m-2 s-1 for QL, with significant differences between QS and the other four types (P < 0.05). Besides, QL had a significantly higher soil respiration than that of PL and FG (P < 0.05, Figure 2).
Figure 2.Soil respiration of baseline and corresponding increased values following precipitations for different size classes and canopy conditions (mean ± SE, n=4). Different lowercase letters indicate significant differences (P < 0.05) among different canopy cover conditions (PS, PL, QS, QL, and FG) for the same precipitation event. PS, PL, QS, QL, and FG represent P. armandii with small size class and large size class, Q. aliena with small size class and large size class, and forest gap, respectively.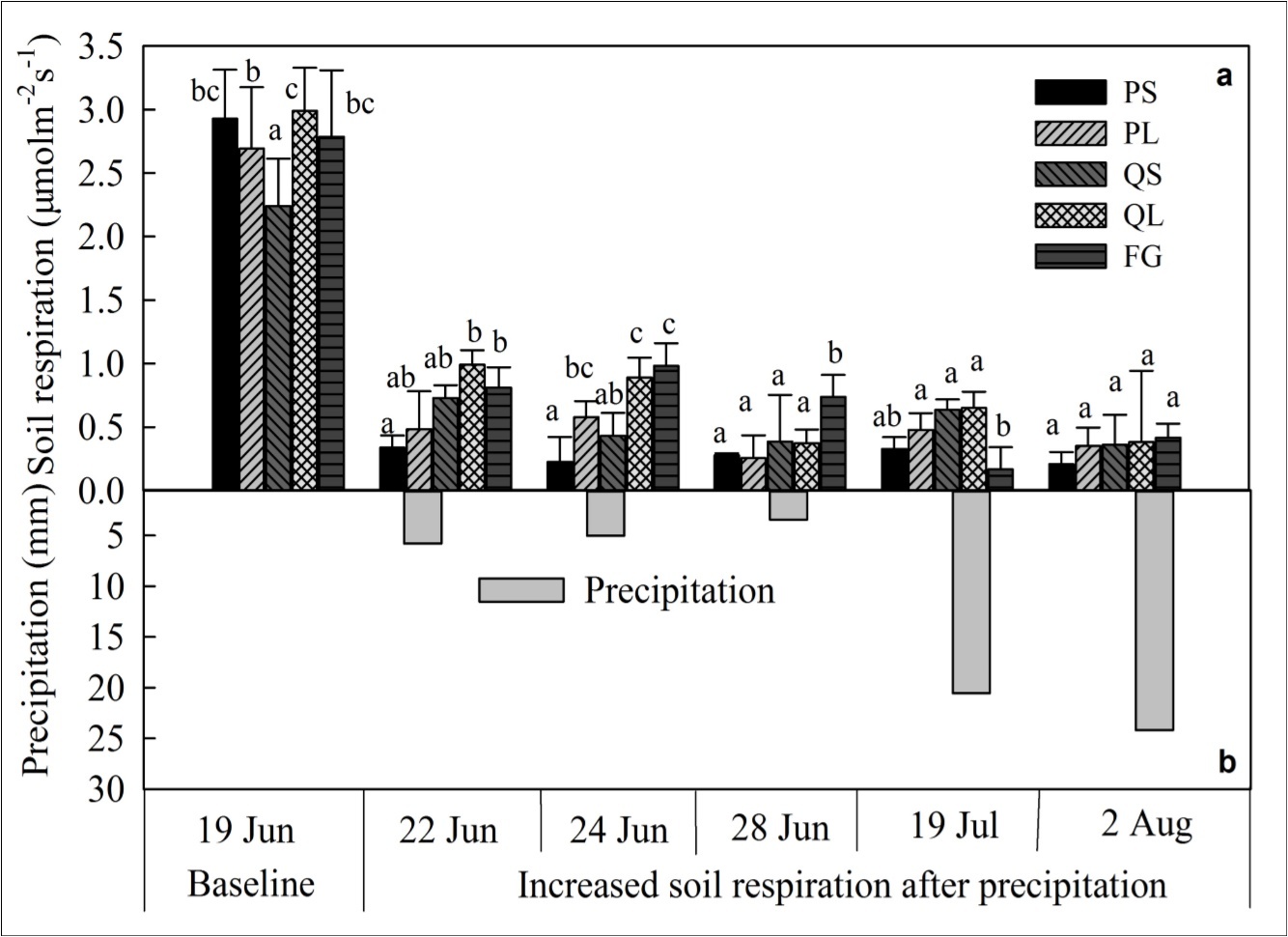
To compare the changed magnitude of soil respiration following different precipitation events, increased soil respiration was calculated based on the soil respiration without precipitation (Figure 2). Precipitation caused significant increases in soil respiration ranging from 6.0% to 35.3% (Table 2, P < 0.001). Enhancements of soil respiration for small size classes (7.1%-11.6% for PS and 16.1%-32.6% for QS) were generally lower than those for large ones (9.5%-21.5% for PL and 12.5%-33.3% for QL) except for the third observed rainfall event (3.43 mm; Table 2, P = 0.2191). In addition, small size classes (0.28 μmol m-2 s-1 for PS and 0.39 μmol m-2 s-1 for QS) showed relative higher soil respiration compared with large classes (0.25 μmol m-2 s-1 for PL and 0.37 μmol m-2 s-1 for QL; Figure 2). Moreover, coniferous species (0.37 μmol m-2 s-1 with 13.5%) had relative lower average increases in soil respiration than broadleaved species (0.63 μmol m-2 s-1 with 24.2%). Soil respiration for FG increased 0.62 μmol m-2 s-1 (0.17-0.98 μmol m-2 s-1) following different precipitations, with a significant higher value (0.74 μmol m-2 s-1) than that beneath the canopies (0.26-0.39 μmol m-2 s-1) at 3.43 mm event. There were significant effects of precipitation × size class and species ×size class interactions on the increases of soil respiration (Table 2, P = 0.0378; P = 0.0071). During the study period, increases of soil respiration following medium rain events (5.81 mm and 5.01 mm) were higher than those following small and large rain events (3.43 mm, 20.57 mm, and 24.19 mm).
Diurnal Response of Soil Respiration to Precipitation
For forest gap, there was a relatively stable emission of soil respiration until the precipitation began. Soil respiration was triggered and generally increased for 1.5 hours after the precipitation (Figure 3). Soil respiration reached its peak value of 4.83 μmol m-2 s-1 3 hours after the rainfall,which was 1.68 times higher than the average soil respiration before rainfall event. Compared to the soil respiration investigated on clear day, up to 20% increases during the same period were observed due to precipitation pulse. Duration of stimulated soil respiration lasted 3 hours and then gradually reduced to pre-rainfall level. No significant decline of soil temperature was observed during the diurnal pattern; however, its peak value was moved up about 3.5 hours later compared with that on a clear day (Figure 3).
Figure 3.Dynamic responses of soil respiration and soil temperature at 10 cm for forest gap to 4.78 mm precipitation (solid circles, from 8:30 on 22 July to 8:30 on 23 July), and corresponding time courses on the following clear day (open circles, from 8:30 on 23 July to 8:30 on 24 July). Gray box indicates time span of precipitation event from 13:50 to 15:45 on 22 July.
Beneath the forest canopy, soil respiration increased dramatically following the precipitation pulse, reaching peak value of 3.96 μmol m-2 s-1 1 hour after rainfall event, which is 1.63 times greater than the average soil respiration before rainfall event. Stimulated soil respiration gradually declined to pre-pulse level after 1 hour duration and then continuously reduced to a lower level partly due to the significant decrease of soil temperature (Figure 4).
Figure 4.Dynamic responses of soil respiration and soil temperature at 10 cm for site beneath the forest canopy to 10.28 mm precipitation (solid circles, from 18:00 on 14 August to 18:00 on 15 August), and corresponding time courses before the rainfall event (open circles, from 18:00 on 13 August to 18:00 on 14 August). Gray box indicates time span of precipitation event from 13:45 to 18:25 on 14 August.
Discussion
Several studies have reported the response of soil respiration to precipitation pulse in a variety of ecosystems 1, 7, 14, 20, 21, most of which show an increase in soil respiration following rainfall events 1, 5, 8. Inconsistent with our study, significant decreases in soil respiration after rainfall has been reported in other studies 22, 23, 24. In this study, elevated soil respiration was observed following precipitation events, but the increment of soil respiration was lower in 24.19 mm precipitation event than moderate rainfall events. Similar to the predictions by Huxman et al. 7, an increase in pulse magnitude from 12 mm to 39 mm did not elicit a larger or longer-lasting soil respiration during precipitation 13. Most studies attribute this to a the rapid up-regulation of soil microbe activity and physical displacement of CO2 from soil pores following shallow soil wetting 1, 13. Because pore space in soil and litter can store CO2 roughly equal to a day’s production, emptying or filling the pore space may enhance or diminish the measured flux relative to biological production 24, 25, 26. Additionally, Gu et al. 27 indicated that short term loss of soil CO2 is enforced by a small, rapidly turning over pool of labile organic matter.
Effects of Soil Temperature on Soil Respiration
It is broadly accepted that soil temperature and moisture play important roles in soil respiration. Linear regression is used in order to compare the differences in sensitivity of soil respiration between species and size classes to temperature (Figure 5a, c). The magnitude of the linear regression slope is interpreted as a quantification of soil respiration sensitivity to temperature 5. For coniferous species, a greater slope in small size class suggests that soil respiration is more sensitive to temperature beneath the canopies of small size trees than large ones (Figure 5a). Conversely, variability in sensitivity is not significant for broadleaved species with an ignorable higher value in large size class (Figure 5c). Broadleaved species show relatively greater temperature sensitivity compared to coniferous species. Similar to the results by Xu and Wan 28 and Shi et al. 5, a negatively linear relationship between soil respiration rate and soil temperature has been observed beneath the canopies (Figure 5a, c) and in forest gap (Figure 6). The finding implies that negative effect of temperature on soil respiration could be expected during dry seasons 28, 29, which is different from other studies 20, 30, 31.
Figure 5.Soil respiration in relation to soil temperature at 10 cm and to soil volumetric water content (SVWC) at 5 cm for P. armandii (a and b) and Q. aliena (c and d). PS, PL, QS, and QL represent P. armandii with small and large size class, Q. aliena with small and large size class.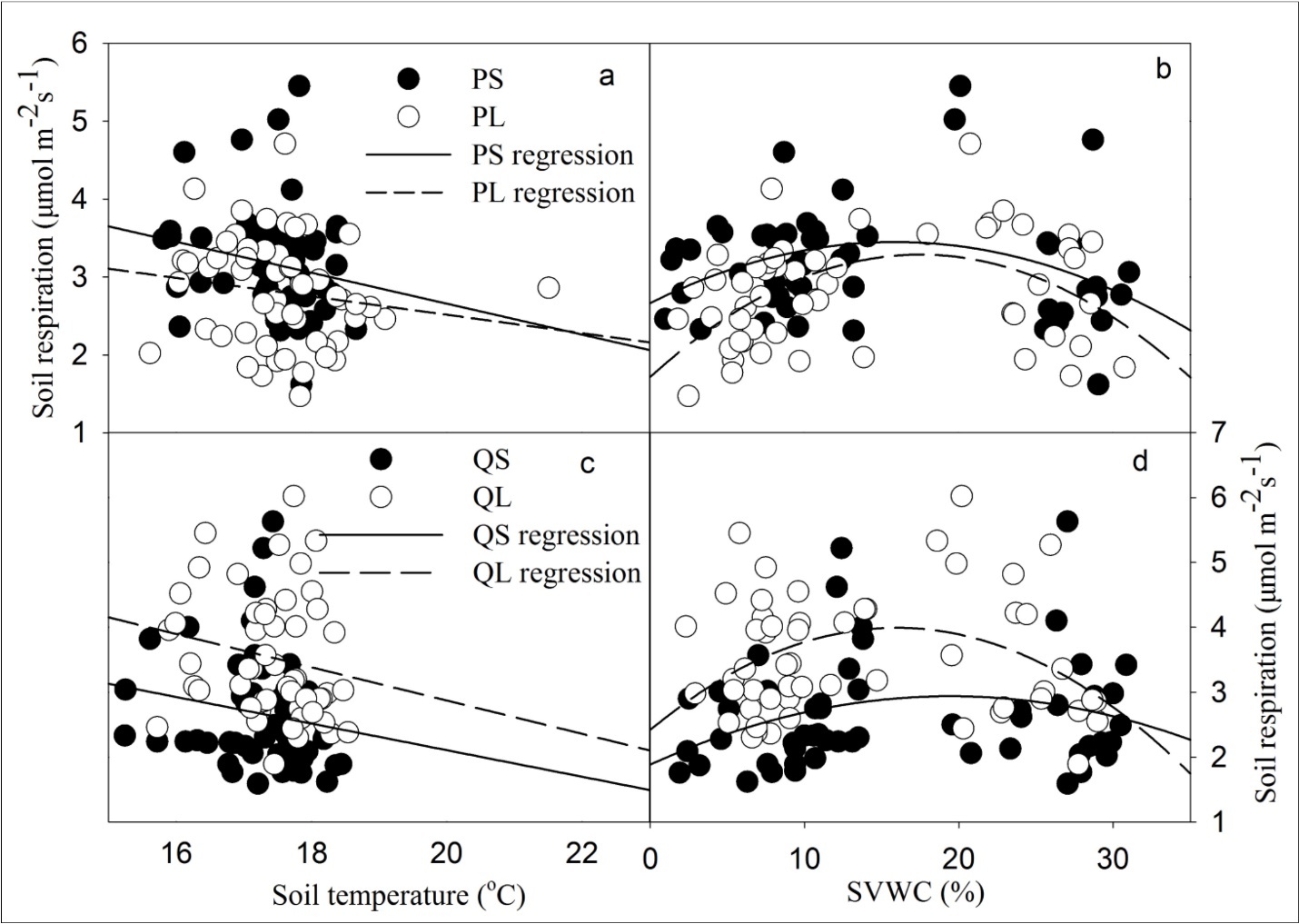
Figure 6.Soil respiration in relation to soil temperature at 10cm (a) and to soil volumetric water content (SVWC) at 5 cm (b) for forest gap.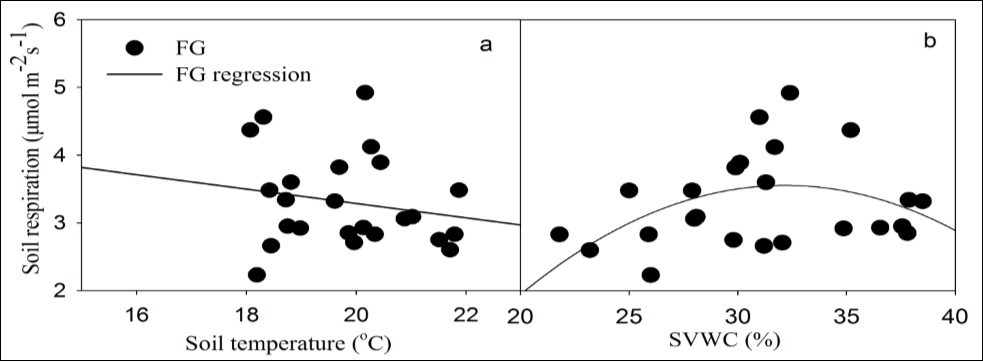
Effects of Soil Moisture on Soil Respiration
The relationships between soil respiration and soil moisture were fitted by polynomial functions, which suggest a significant effect of soil moisture on soil respiration, especially for spots with large trees (PL, P=0.00514; QL, P=0.0368; Figure 5b, d). This may partly explain why the spots beneath the canopy of large size classes have a higher value of soil respiration. Increased soil moisture following rainfall events causes a positive increase of soil respiration at an earlier stage, which is mainly attributable to the increase of microbial activity 25, 32. However, this tends to decline with the continuously rising soil moisture and declining of soil temperature 1. This is in accord with a report indicating that soil water content is negatively correlated with soil respiration at moderate to high water contents 1, 22, 33, probably due to an evolution adaptation of soil microorganism to a long-time mean meteorological situation 22. Moreover, it has long been established that rates of aerobic processes in soils increase with water content up to the point at which activity becomes limited by diffusion of oxygen through water-filled pore spaces 34. Many previous studies reported similar results as this study and found that approximately 60% of water holding capacity to be the threshold at which soil respiration begins to decline with increasing soil water content 5, 35.
Small size class of two species had relatively lower soil temperature and higher SVWC than large classes following each event. We attribute this to its smaller canopy width and crown depth (Table 1), intercepting less water during rainfall. Overall, our results support previous studies in which soil moisture availability is more important than soil temperature in regulating soil respiration in dry seasons 26.
Spatial Heterogeneity of Soil Respiration under Different Canopy Conditions
During the small rainfall event (3.43 mm), soil respiration in FG increases to a greater extent than sites beneath the canopy partly due to the rapid pulse effect and rapid up-regulation of soil microbe activity in FG resulting from no plant interception on rainfalls. But lower pulse effect occurs beneath the canopy due to higher proportions of canopy interception following small rainfall event. Similar results have also been reported recently by Potts et al. 21, who finds open areas in forest tend to have consistently greater soil respiration than sub-canopy (beneath the canopy), particularly after the monsoon period. Among moderate and large precipitations, however, differences in soil respiration beneath the canopy and in forest gap are insignificant, because the water reaching soil surface is sufficient to regulate soil microbe activity 1.
Diurnal patterns of soil respiration show rapid increase and gradual decline after reaching the peak value after 1-1.5 hours following a precipitation pulse. It is because that substrate limitation following rewetting constrains the duration of microbial respiration and thus total CO2 production 1.
Implication for C Cycling
Although pulses of soil respiration following rainfall are small in relative to the annual loss, they play an important role in ecosystem C balance due to high occurrence frequency in the forest ecosystems. It is estimated that almost 60% of precipitation events fall below 5 mm based on the precipitation data from 2009 to 2010 in this region (Table 3), which is suggested as a lower threshold for plant response to rainfall 36. According to this threshold, therefore, about 60% of the events are too small to induce plant responses, but are sufficient to elicit microbe-based CO2 losses. In contrast to water-limited ecosystems, the number of precipitation events is higher than in water-unlimited region with higher pre-rainfall soil respiration. It means that more precipitation pulse occur and a greater soil CO2 emission is induced, even though relative changes of increased CO2 emission (6.0-35.3%) is lower than that in water-limited ecosystems (4-8 times 37; 4-5 times 21; 1.6-2.3 times 5). As a result, water-unlimited ecosystem under rainfall circumstances might act as net sources of CO2 to the atmosphere 1. In addition to the measurements of diurnal patterns of two collars, we did not keep continuous measurement for all soil respiration, which may lead to the overestimation of our result.
Table 3. Number of precipitation events in 2009 and 2010 in Baotianman. Data of 2011 was not included, because od missed data due to thunder damage and storage battery problems.| Year | Size class (mm) | No. of precipitation event | Precipitation (mm) |
| 2009 | <1 | 37 | 8.45 |
| 01-May | 21 | 46.1 | |
| 05-Oct | 11 | 76.91 | |
| Oct-20 | 11 | 143.83 | |
| >20 | 19 | 655.96 | |
| Total | 99 | 931.25 | |
| 2010 | <1 | 25 | 9.81 |
| 01-May | 19 | 85.8 | |
| 05-Oct | 5 | 41.68 | |
| Oct-20 | 8 | 97.28 | |
| >20 | 15 | 752.32 | |
| Total | 72 | 986.89 |
Conclusions
In a temperate mixed forest in Central China, we found that episode precipitation events have significant influences on soil respiration, although water availability is not a limiting factor in this ecosystem. Soil respiration beneath broadleaved species have a relatively greater response to precipitation pulse compared with coniferous ones. In addition, the increment of soil respiration beneath the trees with small canopy size was lower than that with large canopy. Our study suggests that the differences in canopy morphology between different species play an important role in regulating magnitude of soil respiration following precipitation events. The findings indicate that the spatial heterogeneity soil respiration response to precipitation pulse under different canopy conditions should be highlighted while assessing terrestrial carbon cycling and its feedback to climate change.
Acknowledgements
This study was funded by the China National Science Foundation (No. 31600379, 31640011), Key Research Project of Colleges and Universities in Henan Province (No.17A180001), and Scientific Research Fund Project of Henan University (2015YBZR035). We thank Ye Tian, Yin Wu, and Xiaojing Liu for field assistance during this experiment and Jingxin Wang from West Virginia University for helpful comments on earlier versions of this manuscript.
References
- 1.R A Sponseller. (2007) Precipitation pulses and soil CO2 flux in a Sonoran Desert ecosystem. , Glob. Change Biol 13, 426-436.
- 2.Zhang X, Tan Y, Zhang B, Li A, Daryanto S. (2017) The impacts of precipitation increase and nitrogen addition on soil respiration in a semiarid temperate steppe. , Ecosphere 8, 01655.
- 3.M, Egea G, Martin-Gorriz B, P A Nortes, Baille A. (2017) Spatial variability of soil CO2 efflux in drip-irrigated old and young citrus orchards and its dependence on biotic and abiotic factors. , Geoderma 294, 29-37.
- 4.Rey A, Oyonarte C, Morán-López T, Raimundo J, Pegoraro E. (2017) Changes in soil moisture predict soil carbon losses upon rewetting in a perennial semiarid steppe in SE Spain. , Geoderma 287, 135-146.
- 5.W Y Shi, Tateno R, J G Zhang, Y L Wang, Yamanaka N. (2011) Response of soil respiration to precipitation during the dry season in two typical forest stands in the forest–grassland transition zone of the Loess Plateau. , Agric. For. Meteorol 151, 854-863.
- 6.Liu Y, Liu S, Wan S, Wang J, Luan J. (2016) Differential responses of soil respiration to soil warming and experimental throughfall reduction in a transitional oak forest in central China. , Agric. For. Meteorol 226, 186-198.
- 7.T E Huxman, K A Snyder, Tissue D, A J Leffler, Ogle K. (2004) Precipitation pulses and carbon fluxes in semiarid and arid ecosystems. , Oecologia 141, 254-268.
- 8.S M Munson, T J Benton, W K Lauenroth, I C Burke. (2009) Soil carbon flux following pulse precipitation events in the shortgrass steppe. , Ecol. Res 25, 205-211.
- 9.J R Welter, S G Fisher, N B Grimm. (2005) Nitrogen transport and retention in an arid land water shed: influence of storm characteristics on terrestrial-aquatic linkages. , Biogeochemistry 76, 421-440.
- 10.J M Cable, T E Huxman. (2004) Precipitation pulse size effects on Sonoran Desert soil microbial crusts. , Oecologia 141, 317-324.
- 11.Muhr J, Franke J, Borken W. (2010) Drying–rewetting events reduce C and N losses from a Norway spruce forest floor. , Soil. Biol. Biochem 42, 1303-1312.
- 12.Xu L, D, Tang J. (2004) How soil moisture, rain pulses, and growth alter the response of ecosystem respiration to temperature. , Glob. Biogeochem. Cycle 18, 4002.
- 13.D L Potts, T E Huxman, J M Cable, N B English, D. (2006) Antecedent moisture and seasonal precipitation influence the response of canopy‐scale carbon and water exchange to rainfall pulses in a semi‐arid grassland. , New Phytol 170, 849-860.
- 14.W E Emmerich, C L Verdugo. (2008) Precipitation thresholds for CO2 uptake in grass and shrub plant communities on Walnut Gulch Experimental Watershed. , Water Resources Research 44, n/a-n/a..
- 15.IPCC. (2014) Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. , Team, C
- 16.Martinho F, Leitão R, Viegas I, Dolbeth M, J M Neto. (2007) The influence of an extreme drought event in the fish community of a southern Europe temperate estuary. , Estuar. Coast. Shelf Sci 75, 537-546.
- 17.Shen C, Wang W-C, Hao Z, Gong W. (2007) Exceptional drought events over eastern China during the last five centuries. , Climatic Change 85, 453-471.
- 18.J L Chen, C R Wilson, B D Tapley, Z L Yang, G Y Niu. (2009) drought event in the Amazon River basin as measured by GRACE and estimated by climate models. , J. Geophys. Res 114, 05404.
- 19.C S Song. (1994) Scientific survey of the Baotianman Mountial national nature reserve, China Forestry publishing House. , Beijing
- 20.Chen S, Lin G, Huang J, He M. (2008) Responses of soil respiration to simulated precipitation pulses in semiarid steppe under different grazing regimes. , J. Plant Ecol 1, 237-246.
- 21.D L Potts, R L Scott, J M Cable, T E Huxman, D G Williams. (2008) Sensitivity of mesquite shrubland CO2 exchange to precipitation in contrasting landscape settings. , Ecology 89, 2900-2910.
- 22.DÖRR H, K O Münnich. (1987) Annual variation in soil respiration in selected areas of the temperate zone. , Tellus B 39, 114-121.
- 23.B C, Scott A, J P Parker. (1999) Field N2O, CO2 and CH4 fluxes in relation to tillage, compaction and soil quality in Scotland. , Soil Till. Res 53, 29-39.
- 24.M L Thomey, S L Collins, Vargas R, J E, R F Brown. (2011) Effect of precipitation variability on net primary production and soil respiration in a Chihuahuan Desert grassland. , Glob. Change Biol 17, 1505-1515.
- 25.M G Ryan, B E Law. (2005) Interpreting, measuring, and modeling soil respiration. , Biogeochemistry 73, 3-27.
- 26.Liu Y, Liu S, Wang J, Zhu X, Zhang Y. (2014) Variation in soil respiration under the tree canopy in a temperate mixed forest, central China, under different soil water conditions. , Ecol. Res 29, 133-142.
- 27.Gu L, W M Post, A W King. (2004) Fast labile carbon turnover obscures sensitivity of heterotrophic respiration from soil to temperature: a model analysis. , Glob. Biogeochem. Cycle 18, 1022.
- 28.Xu W, Wan S. (2008) Water-and plant-mediated responses of soil respiration to topography, fire, and nitrogen fertilization in a semiarid grassland in northern China. , Soil. Biol. Biochem 40, 679-687.
- 29.Keith H, Jacobsen K, Raison R. (1997) Effects of soil phosphorus availability, temperature and moisture on soil respiration in Eucalyptus pauciflora forest. , Plant Soil 190, 127-141.
- 30.Hashimoto T, Miura S, Ishizuka S. (2009) Temperature controls temporal variation in soil CO2 efflux in a secondary beech forest in Appi Highlands. , Japan, J. For. Res 14, 44-50.
- 31.G M Malcolm, J C López-Gutiérrez, R T Koide. (2009) Temperature sensitivity of respiration differs among forest floor layers in a Pinus resinosa plantation. , Soil. Biol. Biochem 41, 1075-1079.
- 32.V A Orchard, Cook F. (1983) Relationship between soil respiration and soil moisture. , Soil. Biol. Biochem 15, 447-453.
- 33.Davidson E, Belk E, R D Boone. (1998) Soil water content and temperature as independent or confounded factors controlling soil respiration in a temperate mixed hardwood forest. , Glob. Change Biol 4, 217-227.
- 34.R D Miller, Johnson D. (1964) The effect of soil moisture tension on carbon dioxide evolution, nitrification, and nitrogen mineralization. 644-647.
- 35.Linn D, Doran J. (1984) Effect of water-filled pore space on carbon dioxide and nitrous oxide production in tilled and nontilled soils. , Soil Sci Soc Amer J 48, 1267-1272.
Cited by (3)
- 1.Yao Hu, Peng Haijun, Hong Bing, Ding Hanwei, Hong Yetang, et al, 2022, Seasonal and diurnal variation in ecosystem respiration and environmental controls from an alpine wetland in arid northwest China, Journal of Plant Ecology, 15(5), 933, 10.1093/jpe/rtac050
- 2.Kivalov Sergey, Lopes de Gerenyu Valentin, Khoroshaev Dmitry, Myakshina Tatiana, Sapronov Dmitry, et al, 2023, Soil Temperature, Organic-Carbon Storage, and Water-Holding Ability Should Be Accounted for the Empirical Soil Respiration Model Selection in Two Forest Ecosystems, Forests, 14(8), 1568, 10.3390/f14081568
- 3.Kurganova I. N., Lopes de Gerenyu V. O., Myakshina T. N., Sapronov D. V., Romashkin I. V., et al, 2020, Experimental and Model Estimates of Respiration of the Forest Sod-Podzolic Soil in the Prioksko-Terrasny Nature Reserve, Contemporary Problems of Ecology, 13(7), 813, 10.1134/S1995425520070057
