Management of Absent Capsular Support with a new Intraocular Lens Design
Abstract
Purpose:
To describe a new intraocular lens that can be implanted in case of rupture of the posterior capsule and lack of capsular support.
Methods:
This experimental study was carried out on ten Flanders rabbits. The right eye of each rabbit underwent intra capsular lens extraction and two peripheral iridectomies, and the new lens was implanted with an optic rest in the posterior chamber. Additionally, the haptics rest in the angle of the anterior chamber through peripheral iridectomies. Main outcome measures were lens stability inside the eye and complications regarding this technique.
Results:
Intraocular lenses remained in a stable position with only a mild inflammatory reaction; four eyes developed hyphema that disappeared 10 days after surgery.
Conclusion:
The new lens design has good intraocular stability when posterior capsular support is absent and only produced minimal inflammation inside the eye.
Author Contributions
Academic Editor: Zheng Jiang, Department of Neuroscience, The Johns Hopkins University School of Medicine. Baltimore, MD, 21205 United States.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2015 Ehab M Ghoneim, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
After cataract surgery, the lack of capsular support can cause challenges due to the surgical techniques used for lens implantation and the types of lens implanted.
The current available options are as follows:
1) Implantation of a scleral fixation intraocular lens (IOL) which is relatively a difficult technique. Complications include the possibility of decentration, infection through the suture track and late dislocation after trauma or suture lysis. 1,2
2) Implantation of an anterior chamber IOL although it is an easy technique, however possible endothelial decompensation is a late serious complication. 3
3) Implantation of an iris claw lens which need initially a learning curve. Known complications include problems related to pigment dispersion on the IOL surface, hyphema, chronic uveitis, pupil ovalisation and possible dislocation with eye trauma. 4
The newly designed IOL described here has the advantages of optics located in the posterior chamber away from the corneal endothelium and haptics in the angle of the anterior chamber to give additional stability associated with angle supported IOL.
Methods
The study is an interventional case study of ten eyes from ten Flanders rabbits. The study was performed according to the principles of the Declaration of Helsinki and was approved by the local review boards for scientific research and the bioethics committee of veterinary medicine (study number 20143) at Suez Canal University, Egypt. All institutional and national guidelines for the care and use of laboratory animals were followed.
Lens Description
The lens has an optic diameter of 6.5 mm and an overall diameter of 13 mm.
The level of the haptics is elevated 2 mm above the optic level. Figure 1 shows a diagrammatic cross section of the IOL inside the eye with optics in the posterior chamber and haptics in the angle of the anterior chamber through two peripheral iridectomies.
Figure 1.Diagrammatic cross section of the eye showing the position of the new IOL inside the eye, with the optic in the posterior chamber and the haptics in the anterior chamber angle.
The haptic has two ends:
The first one is a broad end that is 3 mm long, which provides support in the anterior chamber angle.
The other end is a small end that is 1.5 mm long. The function of this end is to prevent haptic slippage from the anterior chamber. The broad end and the small end are back-to-back.
The end of the trailing haptic has a notch for a suture to pass through to facilitate upper haptic insertion. Figure 2 and Figure 3 show different views of the IOL.
Figure 2.Intraocular lens design, with posterior chamber optic and angle-supported haptics. Lateral view (A) and anterior view (B) with different IOL dimensions.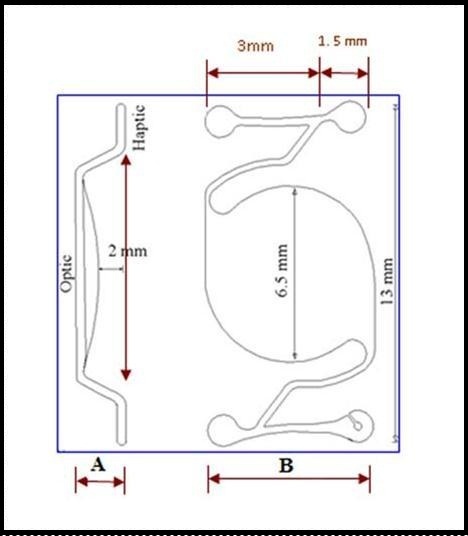
Figure 3.A- Tangential view of the intraocular lens. Note that the trailing haptic has a notch for insertion of a 10/0 nylon suture at the broad end of the haptic. B- Lateral view of the lens. Note that the level of haptics are 2 mm above the level of the optic, the connection between the optic and the haptics pass in a spiral approach to avoid iris chuffing.
Surgical Technique
The operations were performed on Flanders rabbits because they are available in Egypt. We performed surgery on the right eyes of ten of these rabbits. Average rabbit weight was 2 kg. Their irises were brown in color, which facilitated the operation. The eye of the rabbit is similar to the human eye in many aspects, but the dimensions of the eye are smaller in the rabbit than in the human. The only exception is the anterioposterior diameter of the lens, which is slightly larger in rabbits than in humans, although the equatorial diameter of the human lens is larger than the rabbit.
The postoperative changes that occur in rabbits over two months mimic the changes that occur in humans in six months.
Methods of Anesthesia
Ketamine hydrochloride (Glaxo Smith Kline. Egypt) 50 mg/kg body weight was injected intramuscularly, and then 0.5 ml xylocaine 2% (Astra Zeneca) was injected retrobulbar to provide more anesthesia.
An anaesthiologist monitored the vital signs of the rabbit. Tropicamide 1% eye drops were instilled three times five minutes apart before the operation.
Surgical Procedures
The rabbit’s eye was sterilised with betadine hydrochloride 5% and the eye was covered with sterile draping. An eye speculum was introduced to hold the eyelid open. A small speculum was chosen to accommodate the rabbit eyelid. The anterior chamber was penetrated with a 3.2 mm keratome close to the limbus at the 11 o'clock position. Maintenance of the anterior chamber was obtained with methylcellulose injection. Mechanical zonulysis was performed with a muscle hook to induce 360-degree zonulysis at 2 mm outside the limbus.
Alpha chymotrypsin vial was prepared and diluted 1:1 with saline. Alpha chymotrypsin was injected into the posterior chamber of the eye to induce further zonulysis. The solution was injected with slight force so that the iris ballooned forward, and after three minutes, the anterior chamber was irrigated with 10 ml of ringer’s solution. Extension of the corneal incision was performed with corneal scissors from a 2 o'clock to 10 o'clock positions. The lens was removed within its capsule by applying pressure at the 6 o'clock position with a muscle hook and pressure at the 12 o'clock position, and the eye was left without any capsular support.5
Initially, the peripheral iridectomy was performed at the 6 and 12 o’clock positions, but access to perform the peripheral iridectomy at the 6 o’clock position was difficult, so the position of the iridectomies was modified to the 4 and 10 o’clock positions, which makes it easier to manipulate the haptic at 4 o’clock position. A corneal incision was made at the 4 o’clock position with a 20 gauge MVR Knife, and a vitreous cutter was used to perform the peripheral iridectomy while the peripheral iridectomy at the 10 o’clock position was made with scissors through the main incision. The major problem in the rabbit was extensive bleeding after the peripheral iridectomy, which requires more time for washing out the blood and the use of frequent methylcellulose to clean the anterior and posterior chambers. The human eye does not face this problem, as peripheral iridectomies in humans rarely induce hyphema.
The needle that was tied to the trailing haptic passed through the pupil into the posterior chamber and was retrieved through the peripheral iridectomy at the 10 o’clock position. This step was performed first to facilitate the subsequent upper haptic insertion. The IOL was then introduced through the pupil, and the leading haptic was passed through the peripheral iridectomy at the 4 o’clock position from the posterior aspect of the iris (posterior chamber) to appear in the anterior chamber. Further adjustment of the leading haptic was completed in the anterior chamber using McPherson forceps. The IOL optic now in the posterior chamber and the leading haptic was passed to the anterior chamber through the peripheral iridectomy at the 4 o’clock position. The suture that passed first from the peripheral iridectomy at the 10 o’clock position was now dragged constantly until the trailing haptic was retrieved through the peripheral iridectomy at the 10 o’clock position. Further adjustment of the trailing haptic was completed in the anterior chamber using McPherson forceps. The suture that was tied to the trailing haptic was then cut away. The final position, the IOL optic was placed in the posterior chamber with the two haptics in the anterior chamber adjacent to the angle. Wound closure was carried out using a 10/0 interrupted nylon suture.Figure 4 shows the diagrammatic operative steps for lens insertion.
Figure 4.Operative steps. A- Two peripheral iridectomies at the 4 and 10 o’clock positions. B- Intraocular lens outside the eye with 10 nylon suture attached to the trailing haptic. C- The leading haptic passing through the pupillary area and appearing at the peripheral iridectomy at the 4 o’clock position. D- The broad end and the small end of the leading haptic appeared in the anterior chamber at the 4 o’clock position. E- The needle of the 10 /0 nylon suture that was tied to the trailing haptic passing through the pupillary area and passing through the peripheral iridectomy at the 10 o’clock position. The suture was then dragged away until the trailing haptic appeared in the anterior chamber. F- The broad end and the small end of the trailing haptic appeared in the anterior chamber at the 10 o’clock position.
Topical tobramycin and dexamethasone (tobradex eye drops, Alcon lab) combination eye drops were administered five times daily, tobramycin and dexamethasone eye ointment (tobradex eye ointment, Alcon lab.), was administered postoperatively at night. The eye drops were gradually tapered over a period of one month.
Topical tropicamide 1% eye drops (Mydriacyl eye drop, Alcon lab.) were administered two times daily for two weeks postoperatively to prevent synechia.
Results
Examination of the anterior segment of the rabbit’s eye included assessment of ciliary injection, corneal clarity, corneal wound healing, anterior chamber reaction, iris pattern and lens position.
The period of follow-up was two months, after which the rabbits were sacrificed and the eyes were enucleated for evaluation. Figure 5 shows a cross section of the back of the anterior segment of the eye with the IOL optic positioned in the posterior chamber with great stability, even when the optic was elevated with forceps.
Figure 5.A- Cross-section of the back of the anterior segment of the right eye of a rabbit two months after the operation. Elevation of the IOL from the back surface of the iris is easy with McPherson forceps, the arrows show the junction between the optic and the haptics.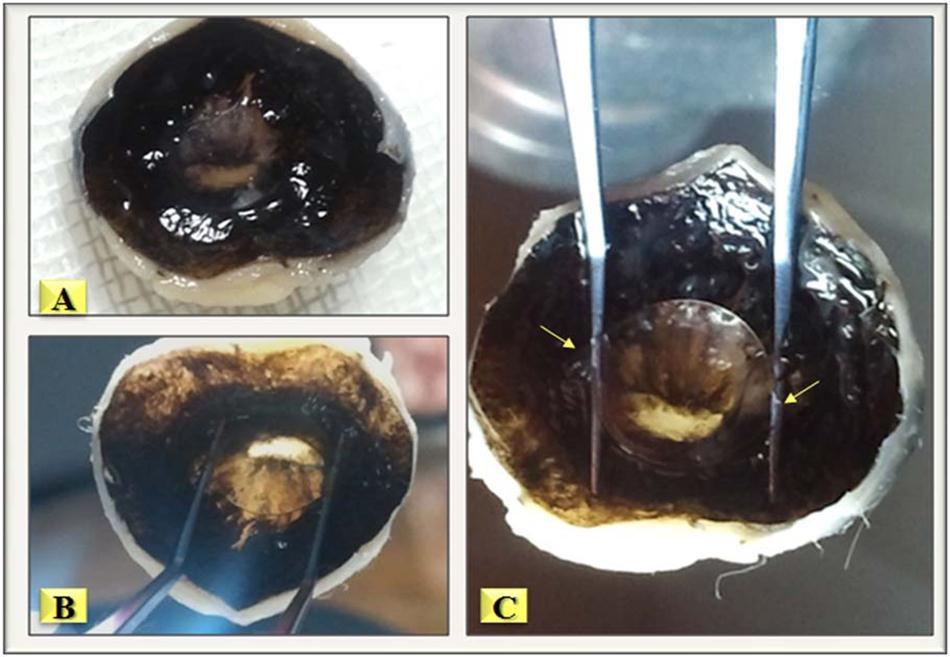
Ciliary injection was marked in the first postoperative week but then resolved. Mild corneal edema was marked for three days after surgery, and then the cornea became clear. Coapted edges of the corneal wound with no epithelial defects were observed one week after surgery.
The problem of hyphema constitutes a major concern in rabbits as peripheral iridectomy induces prolific bleeding. Part of this blood mixed with the vitreous as intracapsular cataract extraction was performed, with the total removal of the posterior capsule to mimic human cases with lack of capsular support. This bleeding prolonged the healing process and induced intraocular fibrotic reactions. Hyphema persisted for one week in four of the operated eyes. Hyphema was mild, had a level of 1.5 mm of the cornea, and resolved after one week. There was a moderate inflammatory reaction in the anterior chamber, which was marked with hyphema and ended with the disappearance of hyphema. Iris patterns were preserved in the postoperative period.
In the four eyes with hyphema, intraocular lenses remained stable in position with mild inflammatory reactions in front of the lens at the level of the pupil. This reaction disappeared 10 days after surgery. No signs of infection were seen during the follow up of the rabbits.
Discussion
Lack of capsular support and zonulysis still poses a challenge to ophthalmic surgeons. The new IOL design presented here has the advantage of the presence of the optic in the posterior chamber, away from the corneal endothelium. The footplate of the lens is 2 mm above the level of the optic, which enables the haptic to rest in the angle of the anterior chamber.
The connection between the optic and the footplate occurs in a spiral pattern to prevent friction between the back of the iris and the haptic and to avoid pigment dispersion during pupillary contraction on lens surface (Figure 3).
This novel design takes advantage of the stability of anterior–chamber-angle supported IOLs and the optic in the posterior chamber, away from the corneal endothelium.
This sutureless IOL avoids the complications related to sclera fixation IOLs, which include suture erosion, suture knot exposure and recurrent dislocation caused by suture lysis. In addition, complications related to anterior chamber IOL designs, such as endothelial decompensation, will be avoided.6, 7, 8, 9
The two peripheral iridectomies were initially carried out at the 6 and 12 o’clock position, but it was very difficult to insert the haptic at the 6 o’clock position, so the site of the peripheral iridectomy was modulated to the 4 and 10 o’clock positions, which made it easier to manipulate the haptic. Vitreous cutter was used to make iridectomy at 4 o’clock position to minimise iridectomy size, If the peripheral iridectomy was too wide, it was easy to make a stitch in the iris with 10/0 nylon suture to narrow the iridectomy gap after insertion of the haptic to prevent light from passing through the iridectomy. The peripheral iridectomy at the 10 o’clock position will be covered by the upper eyelid, with little possibility for light to pass through.
The new IOL design could also be applicable to small incision cataract surgery through the use of acrylic optics and polypropylene haptics to pass through a 3 mm incision.
The current options available for the correction of aphakia with lack of capsular support include sclera fixation IOL, anterior chamber IOL and iris claw lenses, with unique complications related to each type.
These complications include endothelial decompensation, lens subluxation and infection through the suture track. This novel design opens a new era in the management of cases with aphakia with lack of capsular support, lens subluxation, lens dislocation, ectopia lentis, after complicated cataract surgery with rupture of the posterior capsule and extensive zonulysis. The new lens was stable without suturing with the optic in the posterior chamber, away from the corneal endothelium, and the haptic in the anterior chamber to provide good stability for the intraocular lens. The number of eyes used in the study was limited, but the study was conclusive. Further studies on a larger scale in experimental animals may be needed to support the use of this new intraocular lens before it is used in human subjects.
Acknowledgments / Disclosure
A- Funding/Support:
No financial support or financial conflict of interest
b- Financial Disclosures:
The author has no proprietary or commercial interest in any materials discussed in this article.
c-Contributions of the Authors in each of these Areas:
The author was involved in design; conduct of study; collection of data; analysis, management, interpretation of data; review and preparation of the manuscript
d- Other Acknowledgments:
I would like to thank Dr. Alaa Ghomaa for facilitating the operative procedures at the El Salam Eye Center, and give special thanks to Dr. Ashraf El Bayomi for his help with Anesthesia and Miss Hoda Eesa for her help in the preparation of the theatre for surgery. I am also very grateful to Engineer Hasan Abdo for preparing the graphics presented in this work and to Dr. Khalid Saif for his assistance with the graphics.
The trial is registered with the Iranian Registry of Clinical Trials (IRCT)
Registration ID in IRCT: IRCT2013031412817N1
References
- 1.Asadi R, Kheirkhah A. (2008) Long-term results of scleral fixation of posterior chamber intraocular lenses in children. , Ophthalmology2008;(1): 115, 67-72.
- 2.Krause L, Bechrakis N E, Heimann H, Salditt S, Foerster M H. (2009) Implantation of scleral fixated sutured posterior chamber lenses: a retrospective analysis of 119 cases. Int Ophthalmol. 29(4), 207-12.
- 3.Kwong Y Y, Yuen H K, Lam R F, Lee V Y, Rao S K et al. (2007) Comparison of outcomes of primary scleral-fixated versus primary anterior chamber intraocular lens implantation in complicated cataract surgeries.Ophthalmology2007;114(1):. 80-5.
- 4.Gonnermann J, Klamann M K, Maier A K, Rjasanow J, Joussen A M et al. (2012) Visual outcome and complications after posterior iris-claw aphakic intraocular lens implantation. J Cataract Refract Surg. 38(12), 2139-43.
- 5.Harris G S. (1961) Alpha-Chymotrypsin in Cataract Surgery. , Can Med Assoc J1961;22; 85(4), 186-8.
- 6.Donaldson K E, Gorscak J J, Budenz D L, Feuer W J, Benz M S et al. (2005) Anterior chamber and sutured posterior chamber intraocular lenses in eyes with poor capsular support. J Cataract Refract Surg. 31(5), 903-9.
- 7.Dong Z, Wang N L, Hao L, Wang H Z, Zhang H. (2012) Clinical analysis of long term safety after implantation of iris-fixed phakic intraocular lens Verisyse. Zhonghua Yan Ke Za Zhi. 48(8), 707-12.
Cited by (1)
This article has been cited by 1 scholarly work according to:
Citing Articles:
Journal of Ophthalmic Science (2015) OpenAlex
