Abstract
Obesity can be defined as a condition of abnormal or excess fat accumulation in adipose tissue, to the extent that health may be impaired. Fennel is one of the oldest spice plants which widely grows in arid and semi-arid and due to its economic importance and pharmaceutical industry usage. This plant has anti-inflammatory and analgesic effect and is effective in gastrointestinal disorder treatment. The aim of the present study was to evaluate the phytochemical characteristics and therapeutic properties of this medicinal plant. The current study demonstrates that the alteration induced by high fat diet causing changes in blood parameters. These changes are exhibited through a decrease in RBCs, Ht, Hb and platelet and these changes could be due to oxidative stress, which lead to lipid peroxidation in RBCs membranes, auto oxidation of hemoglobin. As regards the total WBCs, lymphocytes and monocytes showed marked decrease. While a distinct increase in the percentage of neutrophils and eosinophil. The present, demonstrates that rats treated with (fennel), (ator) and (fennel with ator) exposure provided significant protection to the altered hematological variables. The effect of the treatment with fennel and ator more effect than fennel only and ator only. The finding of this study indicates that the concentration of Malondialdehyde (MDA) and Myeloperoxidase (MPO) in liver homogenates of the fennel and ator group significant decrease than group (3) and group (4) and the obese group. There is a growing awareness that obesity is a prime risk factor for the development of dyslipidemia profile and that oxidative stress may play a role in various adverse effects of obesity.
Author Contributions
Academic Editor: Andrei Alimov, Leading researcher (preclinical studies), Docent (academic teaching) Research Center of Medical Genetics, Moscow, Russia.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 NA El ghazaly, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Triglycerides are esters that play an important role in the metabolism as energy sources and transporters of dietary fat; they are major component of very low density lipoproteins and chylomicrons. Fennel is one of the world’s most dimension medicinal herb 1. It is anti-ulcer and anti-oxidant properties it is used to treat neurological disorders 2, 3. Fennel plants (Foeniculum vulgare) have been widely used as a source of folk medicine 4. The combination of statins and fibrates is a possible treatment 5. Fibrates are a class of lipid-lowering drug, their hypo-triglyceridemic action involves reduced hepatic triglyceride synthesis 6. The aim of the present study was performed to investigate the possible interaction between a conventional drug used for management of cholesterol and traditional herbal remedies on the obesity.
Triglycerides (TGs) are essential fats transported in the blood stream with cholesterol. High blood TG levels can be genetic, or caused by diabetes, thyroid problems, kidney disease, or some medicines. When more calories are consumed than the body requirements, the liver forms triglycerides from the excess energy and these are then stored as fats. High blood triglycerides are associated with an increased risk of developing coronary heart disease. Obese individuals differ not only according to the degree of excess stored fat but also in the regional distribution of the fat within the body 7, 8. Obesity leads to cardiovascular disease, hypertension 9. Obesity is associated with a number of serious medical complications. The most common complications involve alterations in metabolic function that are risk factors for cardiovascular disease, namely hyperglycemia 10. Ectopic fat accumulation; fat accumulation in non-adipose tissues such as in the liver appears to be a key feature distinguishing metabolically healthy from metabolically abnormal subjects 11. Obesity has deleterious effects on metabolic homeostasis 12.
Lipids are essential for membrane synthesis, maintenance of membrane integrity, as an energy source, as hormone precursors 13. Hyperlipidemia is usually characterized by elevated serum total cholesterol and decrease in High density lipoprotein (HDL) cholesterol and increase in Low density lipoprotein (LDL) cholesterols 14. Fennel seeds contain fiber and complex carbohydrates 15. Foeniculum vulgare is a widely distributed plant in most tropical and subtropical countries. The British Herbal Pharmacopoeia reported its action as a carminative is considered to be one of the best additions to purgative medicines. Fennel seed extracts may possess a radical scavenging antioxidant activity. Herbal drugs and essential oil of fennel have antispasmodic, anti-inflammatory, analgesic and antioxidant effects. Fennel reduced the body weight gain. This may be due to several isoflavans constituents of fennel as phytoestrogens, which like estradiol. This enhances satiety and promotes weight gain 16, 17.
Atorvastatin calcium is a synthetic lipid-lowering agent. Inhibition of HMG-CoA reductase leads to up regulation of LDL cholesterol receptors in the liver mediated. Atorvastatin reduces LDL-C in patients with homozygous and heterozygous familial hypercholesterolemia (FH) of hypercholesterolemia, and mixed dyslipidemia. Atorvastatin also reduces VLDL-C and TG and produces variable increases in HDL-C. Obesity is one of the most pressing public health disorders in the westernized societies. Its prevalence is increasing worldwide and it is associated with concerning medical comorbidities. Obesity results from multiple environmental factors 18.
Material and Methods
This research contains material and methods that were used in the treatment of adult albino rats after receiving a high fat diet.
Experimental animals and treatment: Adult male Albino rats weighing 150–160gm were obtained from the animal house of the Faculty agriculture, Alexandria University, Egypt. They were housed in a clean and well ventilated animal house with a constant 12 hr light and 12 hr dark schedule. They live in five different cages (seven animals per cage) at 25-27oC.The animals were provided with standard diet and tap water was supplied. They were acclimatized under laboratory conditions for one week before the experiment. Diets: Composition of the experimental diet (g/kg diet) was according to the formula of 18. It is including the normal diet for the control rats (fat 5%, carbohydrates 65%, proteins 20.3%, fiber 5%, salt mixture 3.7% and vitamin mixture 1%). The high fat diet contained fat 46%, carbohydrates 24%, proteins 20.3%, fiber5%, salt mixture 3.7% and vitamin mixture 1%. Normal and HFD high fat diet constituents were purchased from El-Gomhoria Company, Cairo, Egypt. HFD was preserved at 4oC until being used.
Atorvastatin
Atorvastatin was obtained from local pharmacies, Cairo, Egypt and ground using a mortar. The drugs were administered orally once a day.
Fennel (Foeniculum Vulgare)
Fennel seeds were obtained from Haraz market for herbs and medicinal plants market, Cairo, Egypt. Dried fennel seeds (Shamar) were washed with tap water to remove possible potential dust. Afterwards, it was dried by cotton cloth to remove the excess liquid prior to drying. Drying was achieved at room temperature for 48 hr. Then a grinder mill and sieves were used to obtain a powder particle size of less than 0.2mm 18.
Expermintal Design
Thirty five Male Albino rats were randomly divided into five groups (7 rats/group as follows: Group 1: (control group): Animals of this group were fed normal diet. Group 2: (obese group): rats of this group were fed high fat diet for two weeks. Composition of the experimental diet (g/kg diet) was according to the formula of 18. HFD was preserved at 4oC until used. Group 3: (obese rats treated with fennel): Animals of this group were fed high fat diet H.F.D. for two weeks then treated with the fennel herb fennel 300 mg/kg body weight 18. Group 4: (obese rats treated with atorvastatin drug): rats of this group were received high fat diet two weeks then treated with the atorvastatin drug 10 mg/kg 19. Group 5: (obese rats treated with atorvastatin drug and fennel herb): rats of this group were received high fat diet for two weeks then treated with atorvastatin drug and fennel herb. Treatment with fennel and atorvastatin drug started after two weeks.
Studies of Blood Samples
For the determination of the blood parameters, some of the blood was collected into Ethylene Diamine Tetra Acetic acid (EDTA) EDTA–treated tubes for determination the hematological parameters as: Determination of the RBCs count was done according to Wintrobe 20. Since erythrocytes concentration in the blood is very high, blood samples were diluted with an isotonic solution and the erythrocytes in apportion of the diluted blood were counted using hemocytometer under the microscope. Determination of hemoglobin (Hb) content was done according to Dacie and Lewis 21. Hemoglobin reacts with potassium ferricyanide and potassium cyanide, and converted into cyanmethemo globin which can be measured colometrically, where the intensity of the color is proportional to the hemoglobin concentration. Determination of haematocrit (HCT) value (Packed Cell volume) was measured according to Oser 22. The Platelet (PLT) count was measured according to Seivered 23. The total leucocytic count using the haemocytometer method was measured according to Miale 24. The differential leucocytic count was done according to Miale 24.
Studies of Serum Samples
Non-heparinized blood was allowed to clot in a centrifuge tube and the serum was separated from blood cells by centrifugation at 8000rpm for 5 min. The serum were separated and stored at -2oC until biochemical parameters assay. Determination of serum total protein was measured according to Gornall et al. 25. Determination of serum albumin was measured according to Doumas et al. 26. Determination of serum total cholesterol was done according to Allain et al. 27. Determination of serum triglycerides was measured according to Fassati and Prencipe 28. Determination of serum high density lipoproteins (HDL) Cholesterol levels was according to Fruchart 29.
Preparation of Liver Homogenates
Whole tissue of liver were obtained by dissection, cleaned from adhering matters, washed with physiological saline, then apportion of liver tissues from each rat was stored at -20oC until analyzed. The reminder portion were minced and homogenized in 5-10 ml cold buffer (50mMpotassium phosphate, pH 7.4, 1m Methylene diamine tetracetic acid (EDTA). Homogenates were centrifuged at 10,000xg for -20 minutes at 4oC according to Goldberg and Spooner 30 and the clear supernatants were used for: Determination of Oxidative stress biomarkers. Determination of lipid peroxide (Malondialdehyde) (MDA) was measured according to Ohkawa et al. 31. Determination of Myeloperoxidase (MPO) was measured according to Zhang et al. 32. Determination of Catalase (CAT) was done according to Aebi 33. Determination of Superoxide dismutase (SOD) was done according to Nishikimi et al. 34. Determination of Glutathione peroxide (GPx) was measured according to Paglia and Valentine 35.
Histopathological Examination of the Heart
Conventional techniques of Paraffin - wax sectioning and haematoxylin eosin staining were used for histological studies according to Drury and Wallington 36. Immediately the heart was fixed in formalin 10% solution for 12 hours, dehydrated through ascending grades of ethyl alcohol until it reached the absolute alcohol (1 hour). They were then transferred to xylol. The heart was placed in a mixture of melted wax and xylol (1:1) for about 10 minutes and transferred to paraffin wax 56C. Sections were dehydrated by passing them down 96%, 90%, 80%, 70% and 50% alcohol for a minute in each. The double or counter-staining routine method was used, because the nucleus is usually acidic in reaction and has the affinity for basic dyes. Haematoxylin counterstained by eosin. Slides were dipped in haematoxylin for ten minutes and then washed with distilled water to remove the excess stain then, slides were transferred to 70% alcohol till the proper density of color is attained. The slides were then rinsed in alkaline water to neutralize any acid present and then counterstained with eosin. For five minutes, the excess stain was removed with distilled water till the correct staining effect was attained .Sections were again dehydrated by passing in a series of 70, 80, 90 and 96% alcohol for two minutes in each, then twice in 100% alcohol. Sections were cleared by passing twice in xylol for two minutes each to ensure getting rid of the alcohol. Finally, sections were embedded in Canada balsam, covered with a thin cover glass, and then dried in an oven (40C) to harden the balsam. Sections were examined for histopathological changes.
Statistical Analysis
All data are presented as means. Means with different letters are significant. F, p: F and p values for ANOVA test, Significance between groups was done using Post Hoc Test (LSD), Statistically significant at p ≤ 0.05.
Result
In the present experimental groups which were studied, the rats received a high fat diet (HFD). Normal and HFD constituents were purchased from El-Gomhoria Company, Cairo, Egypt. HFD was preserved at 4oC until used for two weeks. Treatment with fennel and atorvastatin drug started after two weeks. Haematological investigations were carried among different experimental groups. These studies included red blood cell (RBCs) count, haemoglobin (Hb) content, haematocrit value (Ht) and Platelet count. In addition to, total white blood cells (WBC) count and differential counts. The haematological parameters of rats of different groups are represented in (Table 1 and Table 2). Effect of the Fennel and Ator on erythrocytes count of obese male albino rats; the RBCs count was greatly affected by high fat diet (HFD) administration, since the number was significantly decreased from (4.79±0.33) to (3.64±0.18) at p<0.05 as compared to the control group. Rats treated with (fennel), (ator) and (fennel with ator) showed a significant increase in RBCs count reaching (4.12±0.13), (4.12±0.10) and (4.72±0.25); respectively, compared to (3.64±0.18) in the obesity group. The hemoglobin content (Hb) in the control rats was found to be (12.25±0.24), male rats with HFD significantly decreased this value to (9.98±0.32). Administration of the fennel with ator restored the Hb to (12.10±0.16). The hematocrit value (Ht) was reported in Table 1 as; 42.95±1.62, 34.20±1.40, 39.08±0.80, 39.25±0.78 and 41.25±0.88; respectively whereas, (gp1) is significantly decreased than (gp2), (gp3) and (gp4). The HFD caused significant decrease in platelet counts from (387.50±26.30) to (241.0±25.11) as compared to the control group. Treatment with (fennel), (ator) caused a partial recovery in platelet count as compared to the obese group (Table 1), on the other direction the treatment with both together caused significant increase in the platelet count (361.75±24.84) as compared to the obesity group.
Table 1. Effect of Fennel and Ator on the erythrocytes count of obese male albino rats| Parameters | Control (gp1) | Obesity(gp2) | Fennel(gp3) | Ator (gp4) | Fennel +Ator (gp5) |
| Red blood cells (RBC)(10^6/mm3) | 4.79a+0.33 | 3.64c+ 0.18 | 4.12b+0.13 | 4.12b+0.10 | 4.72a+0.25 |
| Hemoglobin (Hb) (g/dl) | 12.25a+0.24 | 9.98c+0.32 | 11.48b+0.48 | 11.30b+0.54 | 12.10a+0.16 |
| Hematocrit (Ht) (%) | 42.95a+1.62 | 34.20c+1.40 | 39.08b+0.80 | 39.25b+0.78 | 41.25a+0.88 |
| Platelet count (mm3) | 387.50a+26.3 | 241.0c+25.11 | 297.50b+15.0 | 310.0b+18.26 | 361.75a+24.84 |
Effect of the Fennel and Ator on the total leucocytes count and the differential count of the obese male albino rats: At the end of the experimental period, clear WBCs were observed after HFD eating. The count was (4.99±0.17%10^3) as compared to the basal value of (6.59±0.34 %10^3) as shown in Table 2. The (HFD)+ (fennel), (ator) and (fennel with ator) treated group showed significantly increased (5.84±0.22%10^3), (5.93±0.23%10^3) and (6.41±0.23 %10^3); respectively as compared to obesity group (4.99± 0.17%10^3). A distinct increase in the percentage of neutrophils was observed (68.58±1.80) after HFD eating compared with the control value (60.20±0.22). Obese rats treated with (fennel), (ator) and (fennel with ator) showed an improvement in neutrophil percentage (66.47±0.96), (66.53±0.51) and (62.38±2.65) respectively. The Eosinophil percentage was reported in Table 2 as follow control group (1.50±0.14), obesity group (2.45±0.29), fennel group (2.0±0.18), ator group (2.0±0.36) and fennel with ator group (1.70±0.23), whereas, control group is significant decrease than obesity group, fennel group and ator group. Concerning the percentage of lymphocytes, an evident drop was observed at the end of the experimental period from a basal value of (37.28±1.6) in the control group to (26.78±1.96) in the obesity rats. The fennel group and ator treated group showed an amelioration of lymphocytes suffered rats (29.73±0.68) and (31.30±1.28); respectively. In addition to the fennel and ator group showed more increase in the percentage of lymphocytes (34.65±0.44) than the obesity group. Table 2 showed a significant (P<0.05) increase in the monocytes in fennel group, ator group and fennel with ator group (4.13±0.17), (4.3±0.23) and (4.70±0.18); respectively compared to the obesity group (3.98±0.22). While the monocytes of group (5) were near to the normal value of the control (4.98±0.21).
Table 2. Effect of Fennel and Ator on total leucocytes count and the differential count of obese male albino rats| Parameters | Control (gp1) | Obesity(gp2) | Fennel(gp3) | Ator (gp4) | Fennel+Ator (gp5) |
| TotalWBC 10^3/mm | 6.59a±0.34 | 4.99c±0.17 | 5.84b±0.22 | 5.93b±0.23 | 6.41a±0.23 |
| Neutrophils (%) | 60.20b±0.22 | 68.58a±1.80 | 66.47a±0.96 | 66.53a±0.51 | 62.38b±2.65 |
| Eosinophil (%) | 1.50c±0.14 | 2.45a±0.29 | 2.0b±0.18 | 2.0b±0.36 | 1.70bc±0.23 |
| Lymphocytes (%) | 37.28a±1.60 | 26.78d±1.96 | 29.73c±0.68 | 31.30c±1.28 | 34.65b±0.44 |
| Monocytes (%) | 4.98a±0.21 | 3.98c±0.22 | 4.13bc±0.17 | 4.30b±0.23 | 4.70a±0.18 |
The serum levels of the total protein and albumin in the obesity group (7.1± 0.22) and (4.78 ± 0.19); respectively were significantly (p ≤ 0.05) more than those of the control group (6.50±0.13) and (3.95± 0.13); (Table 3). While, the administration of (fennel group) revealed significant (P<0.05) decrease in the serum levels of the albumin and total protein (4.38± 0.10) and (6.65± 0.24); respectively as compared to the obesity group (4.78 ± 0.19) and (7.10± 0.22). Table 4
Table 3. Effect of Fennel and Ator on serum total protein level and albumin level of obese male albino rats| Parameters | Control (gp1) | Obesity (gp2) | Fennel(gp3) | Ator(gp4) | Fennel +Ator(gp5) |
| Total protein (g/dl) | 6.50b+0.13 | 7.10c+0.22 | 6.65b+0.24 | 6.63b+0.22 | 6.53a+0.10 |
| Albumin (g/dl) | 3.95d± 0.13 | 4.78a±0.19 | 4.38b±0.10 | 4.28bc±0.17 | 4.15cd ± 0.13 |
| Parameters Cholesterol (mg/dl) | Control (gp1) | Obesity (gp2) | Fennel(gp3) | Ator (gp4) | Fennel +Ator (gp5) | LSD (5%) |
| After two week form high fat diet | 78.0b ± 52.23 | 144.14a ± 8.49 | 141.71a ± 5.96 | 145.0a ± 6.78 | 142.86a ± 5.90 | 26.365 |
| One week form treatment | 78.71c ± 53.48 | 145.57a ± 6.78 | 118.14ab ± 5.76 | 118.29ab± 12.85 | 100.43bc ± 8.68 | 27.531 |
| Two week from treatment | 84.43c ± 42.11 | 149.14a ± 5.58 | 111.71b ± 10.06 | 97.29bc ± 20.11 | 93.29bc ± 5.99 | 23.645 |
| three week from treatment | 89.86b ± 11.54 | 154.43a ± 21.88 | 101.0b ± 12.15 | 91.57b ± 24.42 | 87.14b ± 11.28 | 18.799 |
| Four week from treatment | 97.14b ± 36.59 | 166.14a ± 17.14 | 92.57 b± 36.66 | 85.14b ± 9.96 | 80.43b ± 21.55 | 29.045 |
| Fiveweek from treatment | 105.0b± 8.04 | 174.43a ± 31.95 | 88.86bc ± 11.16 | 79.43c ± 7.44 | 78.14c ± 6.72 | 17.671 |
| Sixweek from treatment | 106.86b ± 7.65 | 178.0a ± 31.33 | 85.43c ± 8.81 | 78.14c ± 7.78 | 77.14c ± 6.87 | 17.087 |
Effect of Fennel and Ator on triglyceride of obese rats: Table 5 showed triglyceride of the five experimental groups of animals after two weeks form high fat diet then treatment with fennel and Ator through six weeks. Triglyceride of the group (5) (fennel and ator) after two weeks form high fat diet then treatment with fennel and Ator through six weeks equal 228.14 ± 33.82, 186.43 ± 37.29, 173.57 ± 24.09, 153.29± 34.99, 130.43 ± 32.47, 121.43 ± 19.28 and 120.57 ± 19.18; respectively, it indicates a decrease in the triglyceride and reaches to the normal control which record in Table 5 as 100.43 ± 9.95, 102.29 ± 7.20, 110.29 ± 10.81, 124.14 ± 37.02, 127.29 ± 31.14, 131.14± 25.67 and 135.14± 27.39; respectively. While the triglyceride of group(3) (fennel) indicated in Table 6 as 225.71 ± 32.98, 196.86 ± 40.77, 189.57 ± 38.30, 165.71 ± 34.51, 136.29 ± 37.32, 126.57 ± 35.68 and 125.71 ± 35.26 as compared to the group (4) (ator group) 229.43± 26.15, 195.43 ± 40.85, 182.0 ± 26.63, 165.0 ± 11.28, 135.86 ± 39.26, 125.43 ± 13.33 and 123.29 ± 13.74 showed no significant form each other but significant decrease than group(2) (obese group) which the triglyceride 239.71 ± 40.67, 300.86 ± 13.80, 308.43 ± 37.64, 315.29 ± 45.64, 327.0 ± 49.04, 341.29 ± 28.74 and 345.0± 27.02 increase more than the control group.
Table 5. Effect of Fennel and Ator on triglyceride of obese rats| Parameters: Triglyceride (mg/dl) | Control (gp1) | Obesity (gp2) | Fennel (gp3) | Ator (gp4) | Fennel+Ator(gp5) |
| After two week form high fat diet | 100.43b±9.95 | 239.71a±40.67 | 225.71a±32.98 | 229.43a±26.15 | 228.14a±33.82 |
| One week form treatment | 102.29c±7.20 | 300.86a±13.80 | 196.86b±40.77 | 195.43b±40.85 | 186.43b±37.29 |
| Two week from treatment | 110.29c±10.81 | 308.43a±37.64 | 189.57b±38.30 | 182.0b± 26.63 | 173.57b±24.09 |
| three week from treatment | 124.14c±37.02 | 315.29a±45.64 | 165.71b±34.51 | 165.0b± 11.28 | 153.29bc±34.99 |
| Four week from treatment | 127.29b±31.14 | 327.0a± 49.04 | 136.29b±37.32 | 135.86b±39.26 | 130.43b± 32.47 |
| Fiveweek from treatment | 131.14b±25.67 | 341.29a±28.74 | 126.57b±35.68 | 125.43b±13.33 | 121.43b± 19.28 |
| Sixweek from treatment | 135.14b±27.39 | 345.0a± 27.02 | 125.71b±35.26 | 123.29b±13.74 | 120.57b± 19.18 |
(Table 6) showed high density lipoprotein (HDL) of the five experimental groups of animals after two week form high fat diet then treatment with fennel and Ator through six weeks. High density lipoprotein (HDL) of the group(5) (fennel and ator) after two week form high fat diet then treatment with fennel and Ator through six weeks equal 34.29 ± 5.99, 35.86 ± 4.45, 36.86 ± 3.44, 37.71 ± 6.13, 39.14± 7.40,40.29 ± 7.72and 42.0± 6.53; respectively, its indicate increase the high density lipoprotein (HDL) and reaches to the normal control which record in Table 6 as 46.86 ± 8.28, 46.29 ± 9.01, 44.57 ± 10.91, 43.29 ± 7.67, 42.43± 8.81, 42.0 ± 11.96 and 42.0 ± 11.12 respectively. While the triglyceride of group (3) (fennel) indicated in Table 6 as 31.57 ± 2.70, 32.57 ± 4.58, 33.57 ± 4.72, 36.43 ± 7.09, 37.71 ± 6.68, 37.57 ± 8.54 and 38.57 ± 7.76 as compared to the group (4) (ator group) 33.86 ± 3.29, 34.29 ± 5.68, 35.57 ± 4.43, 37.0 ± 3.51, 38.14 ± 9.14, 39.29 ± 3.30 and 39.57 ± 1.62 showed no significant form each other but significant increase than group(2) (obesity group) which the high density lipoprotein (HDL) 37.0 ± 6.32, 31.29 ± 4.35, 28.57 ± 7.0, 28.29 ± 6.78, 27.71 ± 5.15, 26.29 ± 4.61 and 24.14 ± 2.54 decrease than the control group. Table 7 and Table 8.
Table 6. Effect of Fennel and Ator on high density lipoprotein (HDL) of obese rats| Parameters: High density lipoprotein (HDL)(mg/dl) | Control (gp1) | Obesity (gp2) | Fennel (gp3) | Ator (gp4) | Fennel+Ator (gp5) |
| After two week form high fat diet | 46.86a± 8.28 | 37.0b±6.3 | 31.57b± 2.70 | 33.86b±3.29 | 34.29b± 5.99 |
| One week form treatment | 46.29a± 9.01 | 31.29b±4.3 | 32.57b± 4.58 | 34.29b±5.68 | 35.86b± 4.45 |
| Two weeks from treatment | 44.57a±10.91 | 28.57c±7.0 | 33.57bc± 4.72 | 35.57bc±4.43 | 36.86b± 3.44 |
| three weeks from treatment | 43.29a± 7.67 | 28.29b±6.78 | 36.43a± 7.09 | 37.0a± 3.51 | 37.71a± 6.13 |
| Four weeks from treatment | 42.43a± 8.81 | 27.71b±5.15 | 37.71a± 6.68 | 38.14a± 9.14 | 39.14a± 7.40 |
| Fiveweeks from treatment | 42.0a± 11.96 | 26.29b±4.61 | 37.57a± 8.54 | 39.29a± 3.30 | 40.29a± 7.72 |
| Sixweeks from treatment | 42.0a± 11.12 | 24.14b±2.54 | 38.57a± 7.76 | 39.57a± 1.62 | 42.0a± 6.53 |
| Parameters | Control (gp1) | Obesity(gp2) | Fenunel (gp3) | Ator (gp4) | Fennel +Ator (gp5) |
| Malondialdehyde (MDA) | 19.50d±2.08 | 52.75a±6.80 | 44.0b±5.35 | 41.75b±2.50 | 35.0c±3.74 |
| Myeloperoxidase (MPO) | 28.50c±3.11 | 55.50a±6.19 | 43.75b±2.63 | 42.75b±4.99 | 39.75b±1.71 |
| Parameters | Control (gp1) | Obesity(gp2) | Fennel (gp3) | Ator (gp4) | Fennel +Ator(gp5) |
| Catalase (CAT) | 49.25a±3.30 | 29.50d±2.08 | 38.50c±1.29 | 36.50c±3.70 | 43.0b±3.56 |
| Superoxide dismutase (SOD) | 63.50a±3.87 | 31.0c±3.56 | 43.25b±2.75 | 46.50b±5.80 | 49.75b±4.92 |
| Glutathione peroxide (GPx) | 57.25a±6.60 | 28.50c±2.65 | 38.75b±4.27 | 41.0b±2.83 | 44.0b±3.74 |
Obesity administration was associated with a highly significant increase in MDA and MPO concentration reaching (52.75± 6.80) and (55.50±6.19) respectively as compared to the control group (19.50±2.08) and (28.50±3.11). Treatment with (fennel), (ator) following obesity administration exhibited a drop in this elevation to (44.0±5.35), 43.75±2.63), (41.75±2.50), (42.75±4.99); respectively as compared to the obesity group .But the treatment with fennel and ator exhibited a high drop in this elevation to (35.0±3.74) and (39.75±1.71) as compared to group (3) and group (4).
A significant decrease was observed in liver CAT, SOD and GPx level in obesity rats reaching a value of (29.50±2.08), (31.0±3.56) and (28.50±2.65); respectively compared to the control group (49.25±3.30), (63.50±3.87) and (57.25±6.60); respectively. Treatment with fennel in combination with obesity group partially improved this decrease in CAT, SOD and GPx level to be (38.50±1.29), (43.25±2.75) and (38.75±4.27); respectively and treatment with ator drug showed a significant increase in these enzymes (36.50±3.70), (46.50±5.80) and (41.0±2.83) as compared to the obesity group. While the group which treated with fennel and ator together showed significantly more increase in these enzymes(43.0±3.56), (49.75±4.92) and (44.0±3.74) as compared with group (3) and group(4) but not reach to the control value.
Histopathological Studies
An important organ that is affected directly by the obesity which is the heart: The heart is the main important organ in circulatory system, the heart wall has three basic layers; the tunica intima of the heart is called endocardium, the tunica media of the heart is called the myocardium, the tunica adventitia of the heart the epicardium. The histological examination of the hearts of control rats feeding standard diet showing normal cardiac tissue with elongated, unbranched multinucleated muscle fibers (arrow), (Figure 1). The rats are feeding (HFD) for 2 weeks (obese rats) showing large number of inflammatory cells (star), degeneration muscles fiber ,circle and congested Blood vessels (arrow) (Figure 2). By comparison hearts of rats are treated by fennel after obesity and control rats observed show improved cardiac tissue with less infiltration (star) and well organized elongated muscle fibers with peripheral nuclei (arrow) (Figure 3). While examination of rats heart that treated by ator after obesity appears show less improved cardiac tissue with few inflammatory cells between disorganized muscle fiber (star), (Figure 4). While combined hearts of rats are treated by fennel and ator after obesity and control mice are showing nearly cardiac tissue with well-organized muscles fibers (arrow), (Figure 5).
Figure 1.Photomicrograph of heart section of control rat showing normal cardiac tissue with elongated, un branched multinucleated muscle fibers (arrow), (H&E) (400X).
Figure 2.Photomicrograph of heart section of an obese rat showing large number of inflammatory cells (star), degeneration muscles fiber, circle and congested Blood vessels (arrow), (H&E) (400X).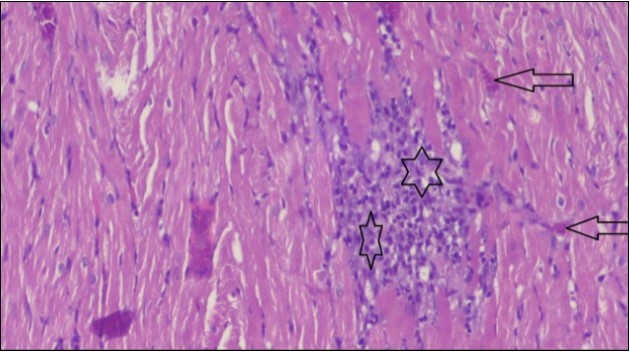
Figure 3.Photomicrograph of heart section of treated rat with fennel herb showing improved cardiac tissue with less infiltration (star) and well organized elongated muscle fibers with peripheral nuclei (arrow), (H&E) (400X).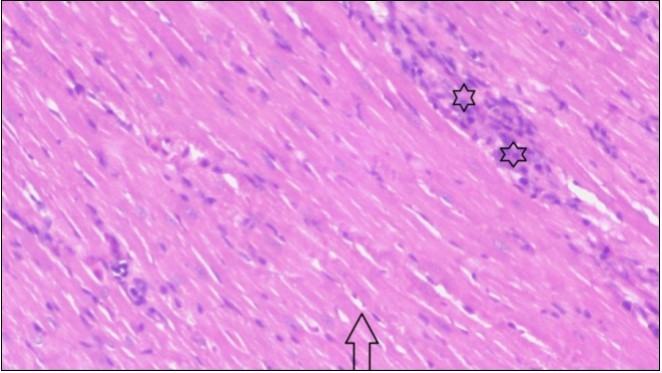
Figure 4.Photomicrograph of heart section of treated rat with Ator drug showing less improved cardiac tissue with few inflammatory cells between disorganized muscle fiber (star), (H&E) (400X).
Figure 5.Photomicrogragh of heart section of treated rat with both fennel herb and Ator drug showing nearly normal cardiac tissue with well-organized muscles fibers (arrow), (H&E) (400X).
Discussion
Obesity is caused by various environmental factors. One of the main environmental factors causing obesity is the intake of a high fat diet 37, 18. Consumption of high fat diet led to obesity and overweight because it facilitates the development of a positive energy balance leading to an increase in visceral fat deposition, this lead to abdominal obesity in particular 38, 18. Weight loss Fennel increases the metabolism of fats and sugars in liver and pancreas. It dissolves fat deposits in bloodstream also and allows it to be used as an energy source. These aspects combined with its natural diuretic effect and a reputation as an appetite suppressant makes it an excellent remedy for weight loss 39, 18.
In the current study, the rats which treated with atorvastatin significantly decrease the protein. In this study it was observed that combination of fennel and atorvastatin showed significant decrease in the protein compared to the other groups and reach to the normal. In this study, the high fat diet (HFD) caused a significant increase in the albumin of the obese rats. In this study, the albumin of the group which was treated with fennel herb was significantly decreased. In the current study, the rats which were treated with atorvastatin significantly decreased the albumin. In this study, it was observed that combination of fennel and atorvastatin showed significant decrease in the albumin compared to the other groups reaching the normal. In the present study, the HFD caused a significant decrease in the RBCs, hematocrit and hemoglobin and that were in agreement with Harishankar et al.40 who observed that higher body condition score would have decreased in RBCs, hematocrit and hemoglobin due to the chronic inflammatory state. Similar to our findings, it was found that there is no relationship between anemia and obesity in rodents while in obese humans there was a higher risk of the development of anemia 41, 18.
In contrast to our results, studying the impact of obesity on in Hematocrit and hemoglobin levels it was concluded that obese individuals have even higher values compared to normal-weight individuals 42, 18. Classical obesity-associated anemia is characterized by dietary iron deficiency, a higher need for iron because of increased blood volume, physical inactivity and low-grade inflammation 43, 44, 18. The anemia among obese individuals was dietary iron deficiency increased demand for iron due to increased blood volume and physical inactivity and low-grade inflammation 45, 18. In the current study, the treatment with fennel herb showed significant increase in in RBCs, Hematocrit and hemoglobin this result agreed with El ghazaly et al. 18 who reported that medicinal plants show less therapeutic effects compared to synthetic drugs, they have more acceptance than chemical drugs in various communities, because their adverse effects are less common. In recent years, plenty of studies conducted on the fennel plant. The results of this study showed that fennel extract increased the number of RBCs in healthy rats significantly. By their antioxidant properties, plants can neutralize adverse effects of free radicals.
The studies have shown that free radicals cause the destruction of cell membrane of RBC. By increasing the stability of membrane cells due to their antioxidant properties, medicinal plants can be effective in the treatment of anemia. Fennel herb can also increase the RBCs by affecting the liver and kidney as well as increasing the erythropoietin 46, 18. Fennel seeds are concentrated source of minerals like copper, iron, calcium, potassium, manganese, selenium, zinc, and magnesium. Copper is required in the production of red blood cells. Iron is required for red blood cell formation. Zinc is a cofactor in many enzymes that regulate growth and development, sperm generation, digestion and nucleic acid synthesis. Potassium is an important component of cell and body fluids that helps controlling heart rate and blood pressure. Manganese is used by the body as a co-factor for the powerful anti-oxidant enzyme, superoxide dismutase 47.
In this study the treatment with atorvastatin drug showed significant increase in RBCs, Hematocrit and hemoglobin this result was in agreement with Modaresi and Resalatpour, 46, who observed that the deformation of red blood cell (RBC) has been found to be related to ATP release from cells. Membrane cholesterol has been shown to alter the properties of cell membrane such as fluidity and bending stiffness 48, 18 and membrane cholesterol increase has been observed in some CVD. One of the mechanisms underlying the antithrombotic effect of statins may be ascribed to the decrease in erythrocyte membrane lipid composition which increases red blood cell deform-ability, and then decreases RBC aggregation. In the present study we observed that combination of fennel and atorvastatin showed significant effected compared to other group and reach to the normal. In the present study, the HFD caused significant decrease in platelet, this result agreed with Chen et al. 49 who reported that the effects of naturally gained obesity on hematological and biochemical parameters are scarce. Chronic inflammation affects many hematological parametres, including erythrocytes, leukocytes and platelets. It is now well-established that obesity results in a state of chronic low-grade inflammation 50, 18.
In the current study, the rats which treated with fennel herb showed increase in the platelet count, this result was disagreement with Kaur 51 who reported that Fennel seeds are known to have anti-cancerous, anti-inflammatory, antioxidant, anti-platelet, anti-thrombotic, and anti-spasmodic properties. Previous studies have shown that Fennel seeds contain high levels of nitrites and nitrates 52. Nitrites and Nitrates are known to play crucial roles in maintaining vascular and digestive functions 53. In mammals, nitrates are reduced to nitrites and subsequently to nitric oxide (NO), an important vasoactive compound 54, 18 that regulates a wide range of vascular functions 55, 18. Although various studies have reported the beneficial effects of plant derived nitrites, the functional implications of fennel seed derived nitrites on vascular functions is yet unknown. The present study confirms that fennel seeds are a rich source of nitrites and demonstrates that fennel seeds derived-nitrites can modulate vascular functions. In the current study the rats which treated with atorvastatin drug showed increase in the platelet count, this result was in agreement with Sikora et al. 56 who observed that statins have some biological effects independent of their lipid lowering effects, anti-platelet aggregation Statins have anti-platelet activation effect, and can prevent or delay the occurrence or development of CVD in patients with hypertension, coronary heart disease, hyperlipidemia or diabetes 56, 18.
The results of Mahassni and Sebaa 57 reported significant increases in Neu, Ly and total WBCs counts in obese states in humans. Researching obesity in cats, Jaso Friedmann et al.58 found no differences in WBC, Neu or Ly counts between obese and lean cats. As the first immune cells to respond to inflammation, Neu had been recently implicated in obesity 59, 18, but the exact role of pro inflammation in canine overweight and obesity is currently unknown. Adipose tissue dysfunction is found to be associated with systemic low-grade inflammation 60, 18. Having a short life span, NEU indicates the continuous activation of the immune system and chronic low-grade inflammation associated with canine overweight and obesity.
In the present study, the treatment with fennel herb showed significant increase in WBC, this result agreed with Cherng et al.61 who reported that the immune modulatory effects of fennel on peripheral blood mononuclear cells and indicated that the extract of fennel has a strong stimulating effect on PBMC cells experimental models. In this study, the treatment with Ator drug showed significant increase in WBC in the test groups compared to the control. A significant depletion in serum HDL level was recorded in obese rats as compared to lean control rats, this result was in agreement with Sohn et al. 62 who reported that hypercholesterolemia and hypertriglyceridemia have been previously detected in obese rats. The reduction in serum triglycerides leads to decreased adipose tissue mass 63, 18. The increased serum LDL level in obese rats has been also recorded in high fat diet supplemented rats 64, 18. This was explained by the decreased HDL level, as recorded in our study, thus decreasing the reverse cholesterol transport from the blood stream to the liver 65, 18. Different fennel extracts seeds used in the present study could reduce serum cholesterol level in obese rats. Non appreciable reduction in serum triglycerides level was recorded in obese rats treated with either one of fennel extracts seeds.
In current study, the rats which treated with fennel significant decrease in serum cholesterol and LDL levels accompanied with insignificant decrease in serum triglycerides level. While, significant increase in serum HDL level, this result was in agreement with Choi and Hwang 15 who observed that fennel methanolic extract seeds treatment could significantly decrease serum LDL level and increase serum HDL level in obese rats. The other fennel extracts could insignificantly reduce LDL level and rise HDL level in obese rats. These results indicated that the constitution of fennel seeds plays an important role in improving blood lipid profile. This could be explained as fennel methanolic extract could significantly increase HDL level. This type of lipoprotein could stimulate the reverse cholesterol transport from the blood stream to the liver 65, 18. Furthermore, it has been shown that fennel, involved in herbal formulation, could delay upper gastrointestinal transit which promotes a decrease in fat and sugar absorption 66, 18. In the present study treatment with atorvastatin significant decrease in serum cholesterol and LDL levels accompanied with insignificant decrease in serum triglycerides level. While, significant increase in serum HDL level
In the current study indicates that the HFD had increase in oxidative stress, these results was agree with Diniz et al. 67, who reported that oxidative stress was found to be associated with obesity. There is a growing awareness that obesity is a prime risk factor for the development of dyslipidemia profile and that oxidative stress may play a role in various adverse effects of obesity. The present data revealed that the lipid peroxidation product (MDA) recorded significant elevation in obese rats when compared with the lean control one. This result agrees with that of Prasanna and Purnima 68. In the present study the rats which treated with fennel herb indicate significant decrease in the oxidative stress, this result agreed with Choi and Hwang 15, who reported that the significant reduction in serum MDA level observed in obese rats treated with either one of the fennel extracts could be attributed to the anti-lipid per oxidative capacity of fennel constituents in its methanolic extract. Augmentation of the antioxidant defense system and the anti-lipid per oxidative activity of aqueous fennel extract has been also reported 2, 18. The efficacy of fennel oil extract to decrease the perceptual feed intake in pigs 69 and in rats as shown in our study indicated that fennel oil could reduce the production of ROS and in turn oxidative stress indirectly via decreasing the caloric intake. Fennel seeds and extract reduced oxidative stress and improve antioxidant defense by reducing MDA level and increasing plasma SOD as well as CAT activities. Foeniculum vulgare fruits and their essential oil are used as flavoring agents in food products such as liqueurs, bread, pickles, pastries and cheese. They are also used as a constituent of cosmetic and pharmaceutical products 18.
The anti-inflammatory and antioxidant activities of fennel have been reported 15, 18. Anti-oxidant Fennel contains flavonoid anti-oxidants compounds like kaempferol and quercetin which helps eliminate harmful free radicals from the body, thus protecting from infection and aging. Rutin bolster inadequate circulation due to its ability to strengthen blood capillaries 70. Fennel seeds are generally eaten for the taste but also very healthy owing to the nutrition value attached to it. Fennel is also used for various health benefits that are derived from its anti-oxidants. These anti-oxidants are essential to curb unwanted free radical reactions in the body to prevent many diseases. In the present study, the rats which treated with atorvastatin indicates significant decrease in the oxidative stress, this results is in agreement with Koter et al.71, who reported that atorvastatin showed a significant reduction in the MDA level. The reduction of MDA by atorvastatin in our study was attributed to the antioxidant mediated effect of atorvastatin which result from inhibition of mevalonate pathway leading to the reduction in the synthesis of important intermediates including iso-prenoids which serve as lipid attachments for in-tracellular signaling molecules in particular inhibition of small GTPase binding proteins whose proper membrane localization and function are dependent on isoprenylation.
In the current study indicates that the HFD had decrease in antioxidant, this results was in agreement with El ghazaly et al. 18 who reported that strategies to overcome the undesirable consequences of obesity and HFD on male reproductive system is necessary. Restricted high-fat diet (RHFD) and antioxidant consumption may be appropriate ways to decline the negative effects of obesity in obese patients and also to prevent obesity-induced complications without changes in dietary pattern. Caloric restriction is one of the most efficient ways to promote weight loss and is known to activate protective metabolic pathways. In the present study, the rats which treated with fennel herb indicates significant increase in the antioxidant, this results agreed with Kooti et al.72 reported that medicinal herbs have fewer side effects than synthetic drugs and due to their antioxidant properties they reduce drugs toxicity. The natural effective ingredients cause biological balance and prevent drug accumulation in body 73, 18. Medicinal plants can be used in the treatment of various diseases 72, 18. Fennel seeds contain antioxidants as kaemoferol and quercetin that prevent degenerative reactions 74, 18. Essential oils of the fruits of fennel constituents possess antimicrobial and antioxidant activities 75, 18. Antioxidant defense mechanisms are important for the protection of cells and tissues against oxidative damage. The major endogenous antioxidant enzyme-systems include superoxide dismutase, catalase, selenium-dependent glutathione peroxidase, glutathione peroxidase, and glutathione reductase. The major non-enzymatic endogenous antioxidants include glutathione and vitamin E 76. This effect may be due to fennel content of phenolic and flavonoid compounds by scavenging or quenching free radicals, by chelating metal ions, or by inhibiting enzymatic systems responsible for the generation of free radical and enhance the activity of antioxidant system. D-limonene compound presented in fennel increased the concentration of liver glutathione which is used by several enzymes that participate in the formation of the correct disulfide bonds of many proteins 18. From these results, it is concluded that fennel has potent antioxidant activity. In the present study, the rats which were treated with atorvastatin indicate a significant increase in the antioxidant activities, this results agreed with Wang et al. 77, who observed that statins had anti-proliferative, anti-inflammatory, antithrombotic and antioxidant effects in previous studies.
These results were confirmed with histological changes of feeding rats heart tissues with high fat diet only, which showed vaculation of tunica media and narrowing in the lumen of aorta sections as well as congestion of cardiac blood vessel and hyalinosis of its wall. This result was confirmed by Szilvassy et al., 78, who indicated that although hyperlipidemia increases oxidative stress in the cardiovascular system, it renders the heart and the vasculature more susceptible to stress. Ouwens et al. 79 identified that development of hyper-cholestremia, which is one of the risk factors for cardio vascular diseases is associated with increased blood levels of TC, LDL-C and VLDL-C as well as lowered levels of HDL in rats fed on high fat-diet. This result was confirmed by histological study, which revealed apparent normal histological structure of heart in all treated rats with fennel seeds. Epidemiologic studies have shown an inverse correlation between HDL-C level and the risk of cardiovascular disease. Increasing the HDL cholesterol level by 1mg may reduce the risk of cardiovascular disease by 2 to 3 percent 80. The present data agreed with Fatiha et al. 81, who reported that hyper-lipidemic rats treated with fennel extract had significant decrease in plasma levels of TL, TG, TC, LDL-C and VLDL, and significant increase in HDL-C level. The current study demonstrates that the alteration induced by high fat diet causing changes in blood parameters. These changes are exhibited through a decrease in RBCs, Ht, Hb and platelet and these changes could be due to oxidative stress, which lead to lipid peroxidation in RBCs membranes, auto oxidation of hemoglobin. As regards the total WBCs, lymphocytes and monocytes showed a marked decrease. While a distinct increase in the percentage of neutrophils and esinophils. The present, demonstrates that rats treated with (fennel), (ator) and (fennel with ator) exposure provided significant protection to the altered haematological variables. The effect of the treatment with fennel and ator is more effective than fennel only or ator only. The current study showed that the serum levels of the total protein and albumin in the obese group were significantly higher than those of the control group. The administration of (fennel) revealed significant decrease in the serum levels of the albumin and total protein as compared to the obesity group, although the serum levels of the albumin and total protein of the Ator group less than the obesity group. Otherwise, the levels of the total protein and albumin in fennel and Ator group were near to the similar value of the control group. The finding of this study indicates that the concentration of Malondialdehyde (MDA) and Myeloperoxidase (MPO) in liver homogenates of the fennel and ator group significant decrease than group (3) and group (4) and obesity group. There is a growing awareness that obesity is a prime risk factor for the development of dyslipidemia profile and that oxidative stress may play a role in various adverse effects of obesity.
References
- 1.Jamshidi E, Ghalavand A, Sefidkon F, Goltaph E. (2012) Effects of different nutrition systems (organic and chemical) on quantitative and qualitative characteristics of Fennel (Foeniculum valgareMill.) under water deficit stress. , Iran J Med Aromat Plants 28(2), 309-23.
- 2.Birdane F M, Cemek M, Birdane Y O, Gulcin I, Buyukokuroglu M E. (2007) Beneficial effects ofFoeniculum vulgareon ethanol-induced acute gastric mucosal injury in rats. , World J. Gastroenterol 13, 607-611.
- 3.Delaram M, Kheiri S, Hodjati M R. (2011) comparing the effects of echinop horaplatyloba, fennel and placebo on pre-menstrual syndrome. , J Reprod Infertil 12(3), 221-6.
- 4.Rather M A, Dar B A, Sofi S N, Bhat B A, Qurishi M A. (2012) a comprehensive review of its traditional use, phytochemistry, pharmacology, and safety. , Arabian Journal of Chemistry
- 5.Miller M, Cannon C P, Murphy S A, Qin J, Ray K K et al. (2008) PROVE IT-TIMI 22 Investigators Impact of triglyceride levels beyond low-density lipoprotein cholesterol after acute coronary syndrome in the PROVE IT-TIMI 22 trial. , J Am Coll Cardiol 51, 724-730.
- 6.Prieur X, Coste H, Rodriguez J C. (2003) The human apolipo-protein AV gene is regulated by peroxisome proliferator-activated receptor-alpha and contains a novel farnesoid X-activated receptor response element. , J Biol Chem 278, 25468-25480.
- 8.Connecticut Commission on Children. Annual Report (2006).Accessed March19,2009, Available at: http://www.cga.ct.gov/COC/annual_reports.htm.
- 9.Buhl R, Soler M, Matz J. (2005) Omalizumab provides long-term control in patients with moderate-to-severe asthma. , Eur Respir J 20, 73-78.
- 10.Abumrad N A.Klein S (2010).Update on the pathophysiology of obesity. , Curr Opin Clin Nutr Metab Care 13, 357-8.
- 11.Fabbrini E, Mohammed B S, Magkos F. (2008) Alterations in adipose tissue and hepatic lipid kinetics in obese men and women with nonalcoholic fatty liver disease. , Gastroenterology 134, 424-31.
- 12.Brunt E. (2010) Pathology of nonalcoholic fatty liver disease. , Nat. Rev. Gastroenterol Hepatol 7, 195-203.
- 13.K S Jain, M K Kathiravan, R S Somani, C J Shishoo. (2007) The biology and chemistry of hyperlipidemia review. , Bioorgan. Med. Chem 15, 4674-4699.
- 14.Saravana Kumar A.Avijit Mazumder, Saravanan VS (2008). Anti-hyperlipidemic activity of Camellia sinensis leaves in Triton WR-1339 induced albino rats. Pharmacognosy magazine. 4(13), 70-64.
- 15.Choi E M, Hwang J K. (2004) Antiinflammatory, analgesic and antioxidant activities of the fruit ofFoeniculum vulgare. , Fitoterapia 75, 557-565.
- 16.Ofir R, Tamir S, Khatib S, Vaya J. (2003) Inhibition of serotonin reuptake by fennel constituents. , J.Mol. Neurosci 20, 135-140.
- 17.Changchien C, Wang J, Tsai T, Hung C. (2003) Corrélation between fatty liver and lipidemia in Taiwanese. , J. Med. Ultras 11(2), 60-65.
- 19.Roglans N, Sanguino E, Peris C, Alegret M, Vázquez M.Adzet T,et al.(2002). Atorvastatin treatment induced peroxisome proliferator-activated receptor alpha expression and decreased plasma non esterified fatty acids and liver triglyceride in fructose-fed rats. , J Pharmacol Exp Ther 302, 232-239.
- 21.Dacie J V, Lewis S M. (1975) . Practical haematology, 5thed.The English language book society and Churchill,Livingston .
- 22.Oser B L. (1979) Hawk's physiological chemistry, mcgrawhill publishing company. , New 1233-1238.
- 23.Tietz N W, ED. (1990) Clinical guide to laboratory tests. 2nd Ed. Philadelphia: WB Sauners;. 566.
- 24.Miale J B. (1972) . Laboratory medicine. Haematology. 4thed. The C.V. mosby Co., Saint Lueis 913, 588-7963.
- 25.Gornall A J, Bardawill C J, David M M. (1949) Determination of serum proteins by means of the biuret reaction. , J. Biol. Chem 177, 751-66.
- 26.Doumas B T, Waston W A, Biggs H G. (1971) Albumin standard and the measurement of serum albumin with bromocresol green. , Clin. Chim. Acta 31-87.
- 27.Allain C C, Poon L S, Chan R W. (1974) Enzymatic determination of total serum cholesterol:. , Clin. Chem 20, 470-5.
- 28.Fassati P, Prencipe I. (1982) Serum triglyceride determined colorimetrically with an enzyme that produces hydrogen peroxide. , Clin. Chem 28, 2077-80.
- 30.DGKC. (1972) Empfehlungen der Deutschen Gesellschaft fur Klinische Chemie. Standard -Method zur Bestimmung der Aktivitat der alkalischen phosphatase. , Z kin Chem u Klin Biochem 10, 191.
- 31.Ohkawa H, Ohishi W, Yagi K. (1979) Assay for lipid peroxides in animal tissue by thiobarbituric acid reaction. , Anal, Biochem 95(2), 351-8.
- 32.Zhang R. (2001) Association between myeloperoxidase levels and risk of coronary artery disease. Jama286.17: 2136-2142.
- 34.Nishikimi M, Roa N A, Yogi K. (1972) The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. , Biochem. Bioph. Res. Common 46(2), 849-54.
- 35.WN Paglia DE and Valentine. (1967) Studies on the qualitative characterization of erythrocyte glutathione peroxidase. , J. Lab. Clin. Med 70, 158-69.
- 36.Drury R A, Wallington E A. Carleton’s Histological Techniques.5thEdition,Oxford University Press,New York,195 (1980) .
- 37.G A Bray. (2002) Etiology and natural history of obesity. [Electronic version].Clinics in Family Practice,4(2). Retrieved January29,2003 , http://homemdconsult.com/das/article/body/1/jorg=journal&source&sp=12594081&sid=15 .
- 38.K A Amin, M A Nagy. (2009) Effect of carnitine and herbal mixture extract on obesity induced by high fat diet in rats. , Diabetic and Metabolic Syndrome J 1, 1-17.
- 39.Garg.Cet al.(2011). Effect of Foeniculum vulgare Mill. fruits in obesity and associated cardiovascular disorders demonstrated in high fat diet fed albino rats. , J Pharma Biomed Sci 8(19), 1-5.
- 40.Harishankar N, Kummar P U, Sesikeran B, Giridharan N. (2011) Obesity associated pathophysiological and histological changes in WNIN obese mutant rats. , Indian J. Med. Res 134, 330-340.
- 41.Tungtrongchitr R, Pongpaew P, Phonrat B, Tribunyatkul S, Viroonudomphol D et al. (2000) Leptin concentration in relation to body mass index (BMI) and hematological measurements in Thai obese and overweight subjects. , Southeast Asian J. Trop. Med. Public Health 31, 787-794.
- 42.Zhang Y, Ma A, Gong M, Lu Q, Lu M et al. (2010) Red blood cell level is increased in obese but not in non-obese patients with coronary heart disease. , J. Geriatr. Cardiol 7, 143-146.
- 43.Pinhas-Hamiel O, Newfield R S, Koren I, Agmon A, Lilos P et al.M (2003).Greater prevalence of iron deficiency in overweight and obese children and adolescents. , Int. J Obes. Relat. Metab. Disord 27, 416-418.
- 44.Cepeda-Lopez A C, Osendarp S J, Melse-Boonstra A. (2011) Sharply higher rates of iron deficiency in obese Mexican women and children are predicted by obesity-related inflammation rather than by differences in dietary iron intake. , Am. J. Clin. Nutr 93, 975-983.
- 45.Winther S A, Finer N, Sharma A M, Torp-Pedersen C, Andersson C. (2014) Association of anemia with the risk of cardiovascular adverse events in overweight/obese patients. , Int. J. Obes. (Lond.) 38, 432-437.
- 46.Modaresi M, Resalatpour N. (2012) The Effect of Taraxacum officinale Hydroalcoholic Extract on Blood Cells in Mice. Adv Hematol. 653412.
- 47.Singh G, Maurya S, Lampasona M P, Catalan C. (2006) Chemical constituents, antifungal and anti-oxidative potential ofFoeniculum vulgarevolatile oil and its acetone extract. Food Control. 17(9), 745-752.
- 48.Forsyth A M, Braunmuller S, Wan J, Franke T, Stone H A. (2012) The effects of membrane cholesterol and simvastatin on red blood cell deformability and ATP release. , Microvasc Res 83, 347-351.
- 49.Chen Y F, Wu Z M, Xie C, Bai S, Zhao L D. (2013) Expression level of il-6 secreted by bone marrow stromal cells in mice with aplastic anemia. , ISRN Hematol. Article 1-6.
- 50.Solinas G, Karin M. (2010) JNK1 and IKK: molecular links between obesity and metabolic dysfunction. , FASEB J 24, 2596-2611.
- 51.Kaur G J, Arora D S. (2010) Bioactive potential of Anethum graveolens, Foeniculum vulgare and Trachysper mumammi belonging to the family Umbelliferae-Current status.
- 52.Santamaria P. (2006) Nitrate in vegetables: toxicity, content, intake. , and EC regulation, J. Sci Food Agric 86, 10-17.
- 53.Bryan N S, Calvert J W, Elrod J W, Gundewar S, Ji S Y et al. (2007) Dietary nitrite supplementation protects against myocardial ischemia-reperfusion injury. Proc Natl Acad Sci USA 104, 19144-19149.
- 54.Radwan E H, A, Ghonim A Z, elghazaly M M, R El Nagar. (2018) The possibility of using the fresh water bivalve,Spathopsis rubins, in the Nile River, El Mahmoudia water stream as bioindicator for pollution. , International Journal of Limnology 1(1), 1-23.
- 56.Sikora J, Kostka B, Marczyk I, Krajewska U, Chalubinski M et al. (2013) Effect of statins on platelet function in patients with hyperlipidemia. Arch Med Sci.9:. 622-628.
- 57.S H Mahassani, B R.Sebaa (2012): Obesity and the immune system in Saudi Arabian adolescent females. , Int. J. Biochem. & Biotech Sci 1, 1-16.
- 58.Jaso-Friedmann L, J H LEARY, PRAVEEN K, WALDRON M, HOENIG M. (2008) The effects of obesity and fatty acids on the feline immune system Vet. , Immunol. Immunopathol 122, 146-152.
- 59.Chmelar J, Chung K J, Chavakis T. (2013) The role of innate immune cells in obese adipose tissue inflammation and development of insulin resistance. , Thromb. Haemost 109, 399-406.
- 60.Schaffler A, Scholmerich J. (2010) Innate immunity and adipose tissue biology. , Trends Immunol 31, 228-235.
- 61.Cherng J M, Chiang W, Chiang L C. (2008) Immuno-modulatory activities of common vegetables and spices of Umbelliferae and its related coumarins and flavonoids. , Food 106(3), 944-50.
- 62.Sohn C W, Kim H, You B R, Kim M J, Kim H J et al. (2012) High temperature- and high pressure-processed garlic improves lipid profiles in rats fed high cholesterol diets. , J Med 15(5), 435-40.
- 63.Jeong S, Han M, Lee H, Kim M, Kim J. (2004) Effects of fenofibrate on high-fat diet-induced body weight gain and adiposity in female C57BL/6J mice. , Metabolism 53, 1284-1289.
- 64.Novelli E L, Diniz Y S, Galhardi C M.GM Ebaid and HG Rodrigueset al.(2007). Anthropometrical parameters and markers of obesity in rats. , Lab Anim 41, 111-119.
- 65.Raveh O, Pinchuk I, Fainaru M, Lichtenberg D. (2001) Kinetics of lipid peroxidation in mixture of HDL and LDL, mutual effects. Free Radic. , Biol. Med 31, 1486-1497.
- 66.Capasso R, Savino F, Capasso F. (2007) Effects of the herbal formulation ColiMil on upper gastrointestinal transit in mice In vivo. , Phytother. Res 21, 999-1101.
- 67.Diniz Y S, Kkhr Rocha, Souza G A, ELB Novelli, Galhardi C M et al. (2006) Effects of N-acetylcysteine on sucrose-rich diet-induced hyperglycaemia, dyslipidemia and oxidative stress in rats (Report). , Eur J Pharmacol 543(13), 151-7.
- 68.Prasanna G S, Purnima A. (2011) Protective effect of leaf extract of Trichilia connaroides on hypercholesterolemia induced oxidative stress. , Int. J. Pharmacol 7, 106-112.
- 69.Schone F, Vetter A, Hartung H, Bergmann H, Biertumpfel A. (2006) Effects of essential oils from fennel (Foeniculi aetheroleum) and caraway (Carvi aetheroleum) in pigs. , J. Anim. Physiol. Anim. Nutr 90, 500-510.
- 70.Barnes D, Yaffe K. (2011) The projected effect of risk factor reduction on Alzheimer’s disease prevalence. , Lancet Neurol 10, 819-828.
- 71.Koter M, Broncel M, Chojnowsk-Jezierska J, Klikcznska K, Franiak I. (2002) The effect of atorvastatin on erythrocyte membranes and serum lipids in patients with type-2-hypercholesterolemia. , Eur. J. Clin. Pharmacol 58(8), 501-506.
- 72.Kooti W, Hasanzadeh-Noohi Z, Sharafi-Ahvazi N, Asadi-Samani M, Ashtary-Larky D. (2016) Phytochemistry, pharmacology, and therapeutic uses of black seed (Nigella sativa). , Chinese Journal of Natural Medicines 14(10), 732-45.
- 73.El Ghazaly NA, Zaatout H, Radwan E H, Elghazaly M M.and EA ElSheikha (2019).Trigonella foenumGraecum extract benefits on hematological, biochemical and male reproductive system of a complementary therapy with Glimepiride in treating streptozotocin induced diabetic rats. , Journal of bioinformatics and 1(3), 45-59.
- 74.Alexandrovich I, Rakovitskaya O, Kolmo E, Sidorova T, Shushunov S. (2003) The effect of fennel (Foeniculum vulgare) seed oil emulsion in infantile colic: a randomized, placebo-controlled study. , Altern Ther Health Med 9, 58-61.
- 75.Abdelaaty A S, Hanaa H A, M H Faiza, Haider G. (2012) Regulation of obesity and lipid disorders byFoeniculum vulgarextracts andPlantago ovatein high-fat diet-induced obese rats. , American Journal of Food Technology 7, 622-632.
- 76.Abd El-Ghany MA, Ramadan A M, Ghozy S F. (2012) Nutraceutical effects of curcuma, ginger, celery, yeast and honey on side effects of gentamicin induced nephrotoxicity in rats. , World Applied Sciences Journal 16(5), 646-655.
- 77.Wang J, Forsyth A M, Stone H A. (2011) Red blood cell dynamics: from cell deformation to ATP release. , Integr Biol (Camb); 3, 972-981.
- 78.Szilvassy Z, Csont T, Pali T, Droy-Lefaix M T, Ferdinandy P. (2001) Nitric oxide, peroxynitrite, and GMP in atherosclerosis-induced hypertensive rabbits: Beneficial effects of cicletanine. , J. Vasc. Res 38, 39-46.
- 79.Ouwens D M, Boer C, Fodor M, P De Galan, Heine R J et al. (2005) Cardiac dysfunction induced by high-fat diet is associated with altered myocardial insulin signalling in rats. , Diabetologia 48, 1229-1237.
Cited by (2)
- 1.Noreen Sana, Rehman Habib‐ur, Tufail Tabussam, Badar Ul Ain Huma, Awuchi Chinaza Godswill, 2023, Secoisolariciresinol diglucoside and anethole ameliorate lipid abnormalities, oxidative injury, hypercholesterolemia, heart, and liver conditions, Food Science & Nutrition, 11(6), 2620, 10.1002/fsn3.3250
- 2.Barakat A I, Radwan EH, De Patricio, 2020, Effect of Trigonella Foenum against Ethylene Diamine Tetra Acetic Acid induced Nephrotoxicity in Male Albino Rats, Journal of Zoological Research, 1(1), 32, 10.14302/issn.2694-2275.jzr-20-3435
