The Agronomy of Mauka (Mirabilis expansa (Ruíz & Pav.) Standl.) - A Review
Abstract
The Andean region is the centre of origin and domestication of at least 9 species of native root and tuber crops in addition to several species of native potatoes. Within this group, Mauka – also known as Miso or Taso ‒ (Mirabilis expansa Ruíz & Pav.) Standl. ‒ Nyctaginaceae) is one of the least well known, despite having much potential. It is cultivated at high altitudes (2300 to 3500 m a.s.l.) in Ecuador, Peru and Bolivia and is thought to be of pre-Inca origin. Mauka is characterized by its high nutritional value due to substantial levels of protein, calcium and phosphorus, as well as secondary metabolites with nutraceutical properties of varied application. It also has good potential as a forage plant. Based on ethnobotanical knowledge and scientific investigations, this review presents advances in the agronomic understanding of Mauka since its discovery five decades ago in several isolated rural Bolivian communities. The information presented covers both Andean and non-Andean countries. It includes results from journals on natural resources, botany, agronomy, and the congress minutes from botanical, agronomic and phytogenetic resources conferences. Theses on Mauka specifically and on phytogenetic resources in general were also reviewed. Books and manuals were reviewed in the libraries of the International Potato Center, INIAP-Ecuador, INIA-Peru and universities.
The plant is described with emphasis on its agronomic traits and according to its propagation forms (seed or vegetative); in terms of its agroecology, phenology, growth dynamics and their indices, crop management, harvest and post-harvest processes. It is concluded that important advances in the understanding of the agronomy of Mauka have been accomplished. Furthermore, the review highlights aspects requiring further research, in order to develop improved production technologies to ensure its future use and conservation.
Author Contributions
Academic Editor: Morad Mokhtar, Agricultural Genetic Engineering Research Institute, Genome Mapping Research, Giza, Egypt.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Juan F. Seminario, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
In 1965, Julio Rea and Jorge León reported on the discovery, in a remote community in Bolivia (Yokarguaya, Italaque, Camacho province), of a new cultivated plant unknown to science: ‘Mauka’ (Mirabilis expansa (Ruíz & Pav.) Standl.), Nyctaginaceae 1. This plant is part of a complex of at least 9 tuberous species of Andean origin, which share domestication history, cultivation methods and uses 2, 3. As a wild plant it was originally described in 1798 by Ruíz and Pavon as Calyxhymeniaexpansa, in Flora Peruviana et Chilensis ‒ recorded from the arid hills of Chancay and Lima-Amancaes 4. In his 1931 revision of the genus Mirabilis L., Paul C. Standley 5, assigned it the current name, and registered it; most likely based on wild specimen as found in Venezuela (Mucurubá), Ecuador (Alausí-Chimborazo, Ambato), Chile (Valparaiso) and Peru ‒ without specifying the locality 5. Subsequent studies indicate that it is only cultivated in communities at high altitude in Ecuador, Peru and Bolivia.
The tuberous root of Mauka is an important food source ‒ high-yielding and rich in nutrients, particularly calcium (table 1).
Table 1. The nutritional contents of Mauka| Nutrient | Amount | Source | |
| fwb | dwb | ||
| Carbohydrate | 10.4‒20.2% | 70% | 6 7 |
| Carbohydrate ‒ amylose | 21% | 6 | |
| Protein | 4.4% | 8 | |
| Calcium | 111 mg 100 mg-1 edible part | 8 | |
| Phosphorus | 283 mg 100 mg-1 edible part | 8 | |
It has promising potential as a forage species 9, 10 and as a biological controller against bacteria, fungi and viruses, due to its content of ribosome-inactivating proteins (RIPs) 11, 12, 13, 14. Furthermore, steroidal saponins, coumarins and catechin-type tannins have been identified in ethanolic extracts of Mauka root 15. These compounds are secondary metabolites of very various applications for the maintenance of wellbeing – both as a food, through their functional and nutraceutical properties, and as a medicine, through their potential to mitigate certain non-communicable chronic diseases 16, 17. A preliminary test on the effect of ethanolic Mauka extract on osteoporosis induced by dexamethasone in ovariectomized rats indicated that it has a mitigating effect in the face of bone mass loss. This effect is being confirmed and is to be analysed through complete phytochemical studies, as a doctoral thesis project conducted at the National University of San Marcos (Nancy Rojas, pers. comm.).
The National Research Council (NRC) 18 describes Mauka as one of the ‘lost crops of the Incas’ and states that ‒ in terms of research ‒ almost everything remains to be done in order to fully understand the potential of the crop. The first statement is misleading, because the accumulated evidence indicates that Mauka is grown and used as part of the broad set of food crops that confers variety and balance to the diet of certain disaggregated rural farming communities in the Ecuadorian, Peruvian and Bolivian Andes; i.e. it is not universally distributed throughout the region in the way that other native root crops such as the arracacha (Arracaciaxanthorrhiza Bancr.) are. Mauka has only ever been recorded at relatively small scales of cultivation; farmers usually maintain just a few plants, although on several occasions between 20 and 40 plants have been observed. Nevertheless, Mauka may have had more significance historically; with some farmers in Ancash, Huánuco and Puno reporting that Mauka played a more central role in the diets of, and was cultivated at a larger scale by, their parents and grandparents 19.
Twenty-nine years later, we can conclude that the second statement made by the NRC 18 is still valid, as Mauka remains considerably understudied. During the mid-2000s, the Peruvian government classified Mauka as an almost endangered species 20. Whilst Mauka cultivation is clearly scarce, its conservation status cannot be fully concluded until full assessments of explorations carried out in recent years 9, 21, along with reviews of those currently in progress [Saturnino Castillo, Miguel Valderrama and Milka Tello, pers. comm.) and further work in underexplored areas of Peru - namely Junin, Huancavelica, Ayacucho and Apurimac - are made.
Although existing studies demonstrate that Mauka has many beneficial properties, its full potential has yet to be realised. In a pilot experiment facilitated by Gendall in 2016 18, chefs from the renown Peruvian restaurant ‘Central’ trialled Mauka as a gastronomic ingredient, meeting with and purchasing roots from farmers in Ancash. Since 2018 they have begun to source it more regularly. For a crop which previously had no market value or recognition beyond the traditional Andean kitchen, this demonstrates that there is commercial interest in the crop and is a notable step towards raising its profile.
Fifty-three years after its discovery as a cultivated plant, we present a synthesis of the agronomy of the Mauka, incorporating insights from ethnobotanical studies and the results of scientific research advances that have taken place on the subject to date.
The Mauka Plant
The cultivated Mauka is a herbaceous plant, perennial, erect when young and decumbent or prostrate and open, to maturity. Propagated from seed, the primary root is axonomorphic, thickened, branched from an early age, with small fine rootlets on its surface that have an absorption function 22, 23. In contrast, the plants originated from vegetative propagation ‒ basal stems and cuttings ‒ have a fascicle of roots (without primary root differentiation) thickened that emerge from the crown or base of the plant 24. At maturity, regardless of the origin, the roots show abundant lenticels grouped in circular rows.
The thin stems (Ø = 0.5 to 3 cm at the base) are green or purple-green and can reach lengths of 80 to 140 cm, with ramifications of the first, second and third order. Their number per plant varies from 5 to 17, depending on whether the plant originates from seed or vegetative propagation. The leaf nodes are prominent and the internodes up to 16 cm long. Generally, only one of the two axillary buds at each node (opposite leaves) develops while the other atrophies. Just below ground level the base of the stems thicken and form short, bulging and globose corm-like internodes with depressed nodes.
These corms constitute the principal source of propagules for vegetative propagation as typically used by the farmers. At the base of the stems (neck of the plant) a crown is formed from which the roots emerge downwards and the stems upwards. The opposite, cordate-ovate or elliptical leaves are light green or dark green with or without purple pigmentation 25. Before flowering the leaves can reach 12 cm in length and 9 cm in width 1, 24, 26.
The inflorescences are aggregated cymes with the terminal inflorescence unit a cyme with 3 to 5 densely grouped flowers. The pentamerous and sympetalous flower buds are yellow, brown or purple, 3‒4 mm long while 5‒6 mm in diameter at anthesis. The synsepalous calyx consists of five plicate, green, sepals; they are persistent covering the gynoecium at it grows and at fruit maturity almost completely, i.e. anthocarpous. The perianth is formed by five sympetalous tepals, lilac, white or white coloured with slight violaceous pigmentation. It has three free stamens and anthers with two reniform thecae containing 25‒30 pollen grains each. The style is longer than the stamens and curved at the apex. The stigma is capitate-papillate, and the ovary is superior, monocarpellary, unilocular, central placentation, single ovule.
The flower opens only once, and in conditions of the valley of Cajamarca, the anthesis starts between 05:30 and 06:00 and terminates at midday between 12:00 and 13:00s. Its reproduction type is fundamentally autogamous, with 6‒25% cross-pollination. Cross-pollination is entomophilous, mainly carried out by species of the genera Syrphus Fabricius, 1775, Allograpta Osten Sacken, 1875, Apis Linnaeus, 1758 and a Microlepidoptera of the Cosmopterigidae family 27.
The fruit is an elliptical or ovoid achene, 3 mm long × 1.7 mm Ø; grey, black or brown in colour, wrapped by the fleshy anthocarp of the persistent calyx and covered by glandular gum containing hairs most abundant at the apex. The gum allows the fruit to adhere to any surface when detached from the plant and be transported by animals and humans, i.e. an adaptation to epizoic dispersal. At maturation, it is common to find insects, and even birds or mice trapped between the plants. The achenes are the unit of dispersal. The seeds have white or crystalline endosperm and have epigeal germination. One hundred seeds weigh 0.6‒1.2 g 1, 24, 26, 28.
Agroecology
The Mauka plant, a perennial geophyte that – in a similar way to other tuberous species – has evolved in the Andean environment, marked by a wet-temperate season alternating with a dry and cold season. In its natural environment, the abundant reserves stored in its roots and the base of its stems enables it to survive under adverse growth conditions. In addition, the thickened basal stems, i.e. corms, serve as a means of regeneration 29, 30.
Its cultivation in Bolivia, Ecuador and Peru probably dates from the pre-Inca period. Findings and collections, since 1965, indicate that in the three countries it is cultivated at altitudes of 2300–3500 m a.s.l. 1, 20, 21, 31, 32, 33. In Peru, these altitudes correspond to the Quechua region 33. In this region, the average daily temperature is 13.5 °C, with maximum temperatures of 25 °C and minimums of 5 °C, and the average annual rainfall is 680 mm. However, the area of greatest distribution is the same as that of starchy maize (Zea mays L.) cultivars (above 2200 m a.s.l. and below 3000 m a.s.l.), with which it is frequently associated.
It responds better to soils of frank and sandy-loam texture, with a pH between 6.8 to 7.2, with a good proportion of organic matter (≥ 3%). Cultivated in association with crops such as potatoes (Solanum spp. incl. S. tuberosum L.), maize and vegetables it is competitive. Its foliage grows and expands rapidly, due to its decumbent nature, thus compromising the growth potential other species. Due to this behaviour, it is common for small-scale farmers to plant Mauka along the edge of the main crops 7, 26, 35. Nevertheless, it does not behave like an invasive plant 36, 37, 38. It produces better in full sun than in shade, as seen in a study carried out by INIAP (Instituto Nacional de Investigaciones Agropecuarias), Ecuador, which showed that experimental plots under shade trees yielded up to 35% less than plots in full sun 39.
Five distinct landraces (I, II, III, IV and V) have been characterized by Seminario and Valderrama 21; one originating in Southern Peru (Puno) and the rest from northern Peru (Cajamarca and La Libertad) ‒ with one originating from a cross occurring ex-situ. Another landrace was observed by Gendall 19 in Puno, distinguished by the vivid magenta colour of its subterranean stem cortex. In an unpublished thesis by Alvarez-Mamani 33, two landraces were registered in Chullín (La Paz Department, northern Bolivia) ‒ Yuraq Mauk’a (white) and Kellu Mauk’a (yellow). It is not yet known whether these are distinct from those landraces seen in Peru, because the descriptors used are not identical and, since the material is not conserved ex-situ, no direct comparison has yet been made.
The plant has shown high adaptability to environments outside the Andes, and in recent decades has been successfully introduced in the Czech Republic and Belgium 40 and the state of Illinois, USA 10, 38 and in the UK (Owen Glyn Smith, pers. comm.).
Propagation
Mauka is, as mentioned above, propagated both by seeds and vegetatively (Figure 1). The easiest method of vegetative propagation is by using the basal corms. These corms are thickened ‒ with abundant reserves and with two buds at each node ‒ and can be planted directly in the field. This is the most common approach used by traditional Andean farmers. In Puno and Cajamarca, farmers usually separate the propagules and leave them out in the sun for a period of time, anywhere between a few days and two weeks, before planting. It has been suggested that this practice mitigates against fungal attack from soil residues and may accelerate sprouting 19. The plant is also propagated by aerial stem cuttings, rooted in a suitable substrate before being transplanted to the field. Whether the stem cuttings originate from the base, the middle section or the terminal section makes no difference to quality or yield. In general, these cuttings are ready to be transplanted to the field following 45‒60 days 28. During the rainy season the direct planting of cuttings in the field, i.e. without previous rooting, can be successful. Franco and Rodríguez 41 reported that cuttings planted directly in the field had a 71% to 97% survival rate.
Figure 1.Three methods for the propagation of the Mauka. Left: Thickened underground stems (corms, i.e. the most common means of vegetative propagation). Centre: Rooted stem cutting (60 days after planting in sand and under shade). Right: Seeds.
The advantage of propagation by cuttings is that the multiplication rate is higher than with underground stems. An evaluation carried out using the purple landrace, at seven months of age, found that each mother plant produces 266 cuttings, with four nodes each (eight buds); meanwhile 100 plants are needed to plant one hectare at the density of 0.90 m × 0.50 m, i.e. 22 222 plants ha-17. However, the results do vary depending on the substrate used and the time of year.
Under greenhouse conditions, Alva 42 studied the effect of three substrates on the propagation of the Mauka by stem cuttings. The substrates were tested using three landraces (I, II and III), in completely randomised block design and with three repetitions. The test lasted for 60 days, at which time the cuttings were ready to be transplanted to the field. The following traits were evaluated: number of cuttings with shoot, number of rooted cuttings with shoot, height of shoot (cm), number of roots per cutting, length of root (cm) and number of leaf pairs per cutting. No significant statistical differences were found for the landrace interaction by substrate type, for the variables studied. The best landrace was light green (III), in which 83% of rooted shoots were obtained and the best substrate was sand with 72% of rooted cuttings. Each shoot produced 12 to 20 leaves (Table 2a–b).Sexual propagation implies regeneration from seed, which is produced in abundance by the plant 7, 43. This seed is non-dormant, orthodox, and retains its viability for several years. Tests with seeds kept in a closed container at 18 °C showed an 88.5% germination rate after three years 28. The characteristics of seed lots of three Mauka landraces and the germination (in Petri dish on paper) at six months post-harvest were similar; all showed high germination rates. Similarly, planting seeds of the same lots in soil, sand and humus (at the ratio 1: 1: 1) showed similar seedling emergence and number of days to first pair of true leaves (Table 3). This uniformity in morphological and physiological characteristics (absence of dormancy, uniform and rapid germination, although the crop has retained the auto-dispersal of their seeds) indicate that these landraces have been strongly affected by the domestication syndrome in that they no longer behave as wild material 44.
Table 2a. The effect of three substrates on the propagation by cuttings of three Mauka landraces.| Treatment | Cuttings with root and bud (%) | Height of shoot (cm) | No. roots | Root size(cm) | No. Leaf pairs |
| Landrace III, soil + humus | 78 | 10.3 | 6 | 4.8 | 17 |
| Landrace III, soil + sand + humus | 80 | 9.3 | 5.4 | 3.9 | 18 |
| Landrace III, sand | 93 | 13.3 | 7.2 | 5.0 | 20 |
| Landrace I, soil + humus | 65 | 10.3 | 3.8 | 3.8 | 16 |
| Landrace I, soil + sand + humus | 66 | 7.4 | 4 | 4.2 | 14 |
| Landrace I, sand | 73 | 11.5 | 4.3 | 4.1 | 19 |
| Landrace IV, soil + humus | 45 | 7.5 | 3.3 | 3.5 | 15 |
| Landrace IV, soil + sand + humus | 42 | 6.0 | 3.4 | 4.8 | 12 |
| Landrace IV, sand | 53 | 9.6 | 5.4 | 4.3 | 15 |
| Average | 66 | 9 | 5 | 4 | 16 |
| SD | 17.0 | 2.2 | 1.3 | 0.5 | 2.5 |
| Component | Soil | Lumbricus humus | River sand |
| PH | 6.9 | 8.2 | 7.40 |
| Organic matter (%) | 4.0 | 25.0 | 0.22 |
| Phosphorus (mg kg-1) | 11.8 | 10 000.0 | 1.90 |
| Potassium (mg kg-1) | 239.5 | 3222.0 | 350.00 |
Productive Characteristics of the Plants According to the Type of Propagation Material
A first noticeable difference between plants raised from seed and vegetative propagule (corms or stem cuttings), refers to the origin of the tuberose root, which is the harvestable part. In the first case, the tuberous root is the result of the lengthening and thickening of the radicle and hypocotyl, which form a single tuberous axis (Figure 2). In this aspect, it is different from other roots such as beetroot (Beta vulgaris L.), where the reserve organ corresponds to the hypocotyl only 45. Its thickening due to accumulation of storage reserves (Figure 3) occurs due to the activity of secondary changes that produce vascular tissues and reserve parenchyma that are located successively, in concentric rings, at a rate of approximately one per month 23, similar to what happens in turnip (Brassica rapa L.) and beetroot 45, 46. In each primary root thus described, four to five secondary roots are equally thickened. At 270 days this set of roots has accumulated 88% of their reserves and 330 days have accumulated the total reserves (Figure 4).
Figure 2.Left: Mauka seedling 15 days after sowing in the field. Centre: Mauka seedling 29 days after emergence, with cotyledons and two pairs of true leaves. Note: the radicle and hypocotyl have formed a unit, below the cotyledons, and the thickening has begun. Right: Mauka seedlings, 60 days after emergence. Note: the cotyledonal knot has thickened and an outbreak has arisen, beginning the formation of the crown and the system of secondary stems.
Figure 3.Cross section of the tuberous Mauka root and detail of the growth and reserve storage tissues.
Figure 4.Dry weight% of Mauka tuberous roots from plants raised from seed and according to the age of the plant.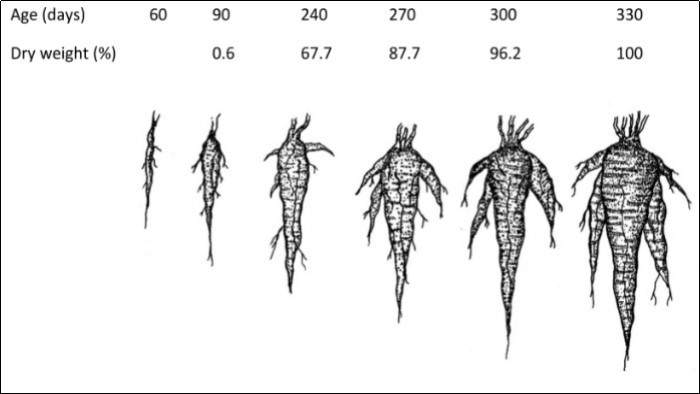
On the other hand, in plants from propagule, the tuberous roots are adventitious and numerous, originating from the corm, i.e. the crown or base of the stems. Whether from seed or propagule, in plants with this type of tuberous roots, i.e. without independent fibrous roots, of which the primary function is to absorb the photosynthates which are destined to the apical meristems of the fibrous roots and the stems are translocated through the tuberous roots. In the same way, both water and mineral nutrients must be transferred through the tuberous root to the leaves 46.
The Cajamarca field experiments were carried out at the National University of Cajamarca UNC, located at 7° 29'45'' S and 78° 10'12'' W, at 2 656 m a.s.l. with an average daily temperature of 14 °C, 65% of RH and an annual rainfall of 650 mm. These experiments involved the comparison of three landraces, raised from both seeds and propagules (Table 4). The results indicated that these plants are similar with respect to growth period, maximum leaf area index and harvest index. However, they differ in terms of the type and linear measurements of the tuberous roots, number and weight of tuberous roots, number of stems, maximum leaf area, crown weight and root yield. The lower yield exhibited by plants raised from seed is notable, and might be explained by the lower number of stems and lower leaf area. These characteristics indicate that Mauka propagated from seed should be planted at higher densities than plants propagated vegetatively, in order to maximize yield per ha-1.
Table 4. Characteristics of Mauka plants propagated by seed and vegetatively| Characteristics | Type of propagation material | |
| Seed* | Vegetative** | |
| Growth period (days) | 326.00 | 312.00 |
| Type of root | Axonomorph | Fasciculate |
| Root length (cm) | 26.00 | 20.00 |
| Greatest root diameter (cm) | 6.00 | 4.00 |
| No. of roots plant-1 | 7.00 | 17.00 |
| Root weight plant-1 (kg) | 1.35 | 2.50 |
| No. of stems plant-1 | 4.50 | 17.00 |
| Maximum leaf area (dm2 plant-1) | 287.00 | 327.00 |
| Maximum IAF | 8.00 | 8.00 |
| Weight of above ground part (kg plant-1) | 0.17 | 0.67 |
| Root yield (kg ha-1) | 31,433.00 | 59,133.00 |
| Harvest index (%) | 52.00 | 49.00 |
Crop Husbandry
Planting
Soil is prepared for planting Mauka in much the same way as when it is prepared for planting maize ‒ with which it is frequently intercropped. This process generally involves ploughing using a tractor, yoke or ‒ at the smaller scale ‒ using a hand tool such as a pick-axe. If planting seed it is recommended to raise plantlets in a seedbed and to transplant these to the planting site once they have developed one to two pairs of green leaves. If using basal stems as propagules these are separated from the mother plant and either planted directly or exposed to the sun for a period of time, as previously mentioned, and then planted. Aerial stem cuttings are best planted after they have been rooted in a nursery.
Planting Density
On average Mauka produces five stems if propagated by seed and up to 17 stems per plant if vegetatively propagated. In both cases, the stems of the decumbent plants branch profusely and can measure up to 1 m in length. These characteristics should be taken into account when considering planting density. Seed should be sown on ridges spaced at 0.8 m, with 0.4 ‒ 0.5 m distance between plants, i.e. 31 250 ‒ 25 000 plants ha-1.
Meanwhile, if using propagules, these should be planted at 0.9 ‒ 1 m between ridges and 0.5 ‒ 0.6 m between plants, i.e. 16 666 ‒ 22 222 plants ha-1. Further study is required to assess the yield of tuberous roots, foliage and corms, in relation to landraces and crop husbandry factors. Also, to harvest roots and foliage most efficiently, it will be necessary to identify the point of equilibrium between the production of underground and aerial parts. Because local farmers typically cultivate and are familiar with no more than one landrace of Mauka, and are accustomed to planting the crop on a relatively small scale, these aspects can not be assessed through ethnobotanical inquiry alone.
Fertilisation
In traditional Andean agriculture, livestock manure is used to fertilise crops. However, due to the scarcity of this resource, it is primarily applied to the main crops, e.g. potatoes and maize. Marginal crops such as Mauka are usually not fertilised. Albeit, there are only few studies of Mauka’s response to the application of manure, compost or chemical fertiliser.
Zapana et al. 43 studied the effect of cattle manure and chemical fertiliser on the production of seed and tuberous roots. The composition of the manure was 0.8% nitrogen, 0.3% phosphorus and 0.4% potassium and the chemical fertilisers applied were urea (46% N) and triple calcium superphosphate (46% P2O5). The experiment consisted of three treatments: (a) 7.5 t ha-1 of cow manure; (b) 60 and 40 units of nitrogen and phosphorus, respectively; (c) control. A complete randomised block design was used, with three repetitions, evaluating plant height and yields of seed and tuberous roots (t ha-1) for each trial. Significant differences were found for the production of seed and tuberous roots and, in both cases, the treatment with the chemical fertiliser produced the highest yields (Table 5).
Table 5. Plant height, seed and tuberous root yields of Mauka using either cattle manure or chemical fertiliser.| Treatments | Plant height (cm) | Seed yield (t ha-1) | Root yield (t ha-1) |
| 7.5 t ha-1 manure* | 50.7 | 7.2 | 46.4 |
| 60-40-0 N, P, K | 66.3 | 11.3 | 78.5 |
| Control | 39.6 | 4.6 | 31.6 |
In the study by Zapana et al. 43, an application of 7.5 t ha-1 of cow manure produced just 59% of the yield achieved with chemical fertilisers. However, it is important to note that despite its lower output, organic fertiliser may still be preferred by the farmer. In contrast to chemical applications, manure promotes the conditions of the soil in terms of its physicality (structure, porosity, heat absorption, water retention), chemical composition (cation exchange and nutrient retention) and biological health (microbial populations), which in turn improve the development of the plants. Organic methods might also be preferred from the point of view of the consumer.
Weeding, Hilling and Watering
Weeding is necessary during the first months following planting. The frequency of weed removal depends on the conditions during the cropping season, i.e. wet or dry, and the conditions at the site. Removing weeds as they appear, before they become competitive to the crop, is generally recommended. After four to five months the Mauka plants should cover the furrows between ridges and outcompete the weeds on their own accord.
Observing Mauka’s growth habits under various experimental planting arrangements since the late 1980s (again conducted at the UNC experimental fields) indicates that hilling, i.e. the placement of additional soil around the base of each plant several months into its growth cycle, is not of fundamental importance for the production of Mauka roots, although local farmers have reported that the practice is beneficial to cultivation 19. Because the roots develop in a vertical or slightly inclined direction – as seen other Andean root crops, e.g. maca (Lepidium meyenii Walp.), yacon (SmallanthussonchifoliusPoepp. H.Rob.) and arracacha (Arracaciaxanthorrhiza Bancr.) ‒ it appears not to influence how the tuberous roots originate and grow around the crown or main stem. In this sense, they differ from crops classified as tubers, such as oca (Oxalis tuberosa Molina) or mashua (Tropaeolum tuberosum Ruíz & Pav.), where hilling is an indispensable task because it ensures that the stolons develops tubers, as opposed to just producing aerial stems.
Hilling up soil around the base of the Mauka plants instead appears to stimulate the growth in number and thickness of the underground parts, at the expense of the growth and thickening of the roots themselves ‒ which are the principal product. This effect is seen in the first drawing of a Mauka plant cultivated in Bolivia, published by Rea and León 1, which illustrates both the underground corms and a single tuberous root (Figure 5). Hilling is therefore recommended for maximizing the production of underground vegetative propagules, i.e. corms, and to protect plants during the cold season, but it is not advised if root production is the priority (Figure 5). As already indicated, in places with abundant rainfall and flat land good results are obtained by planting on ridges, raised 20–30 cm.
Figure 5.The Mauka plant and the production of roots and tuberous corms. A: First drawing of the cultivated Mauka plant, with numerous and prominently thickened underground corms ‒ probably as a result of a prominent hilling 1. B: A Mauka plant cultivated without hilling, where the tuberous roots predominate, as opposed to the basal corms 26.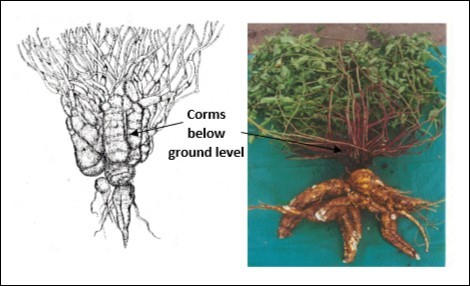
Mauka is a crop grown by smallholder farmers at high altitudes in the Andes, where its cultivation is subject to and synchronised with the rainy season, i.e. September to April. As such it tends to be irrigated only occasionally, as and when the main crops are irrigated in those areas that have access to water for this purpose.
Mauka's water consumption behavior has not yet been compared across different climatic and soil conditions. Further inquiry into these aspects would be highly relevant for the development of the crop.
Pests, Diseases and other Sources of Stress
With regards to stress, the greatest risk to Mauka plants is probably excess water, which can cause root rot. This can occur as a result of abundant rainfall combined with poorly drained soils 7, 10. Albeit, Kritzer et al. 38 observed that in field trials conducted in southern Illinois the two landraces tested did respond differently. Local farmers describe Mauka as a naturally healthy plant which tends not to succumb to pests and diseases 18. A field trial at the UNC ‒ where four Mauka landraces were planted in monoculture plots of 2 000 m2 ‒ found that, whilst stem-boring and leaf feeding insects were observable on leaves and shoots, they did not damage the plants in a way that was economically significant 7. The Mauka mosaic virus (Mirabilis potyvirusMir), transmitted by aphids whose indicator plant is quinoa (Chenopodium quinoa Willd.), has been identified by Lizárraga 48. However, only one case of probable attack by this virus has ever been recorded ‒ during the agricultural season 1994–1995. This manifested as chlorotic stains on the leaves, but only in a few plants of the Mauka germplasm collection maintained ex-situ at the UNC 7. Mauka is a host to nematodes of the genera Pratylenchus and Paratylenchus. In pre and post-culture evaluations, an index of population growth greater than 1 was found 39.
Low temperatures can also induce stress in Mauka plants. In the Cajamarca Valley, where temperatures fall below -2 °C, frost has been known to damage the entire leaf area. However, sustained by its underground reserve and abundance of buds originating from the underground corms ‒ with the addition of weekly irrigation ‒ it can typically recover within 30 days.
Phenology and Analysis of Plant Growth
Phenology
The phenology of three landraces (I, II and III) propagated by both corms and seed (Table 6) in the UNC experimental fields did not show significant phenological differences in terms of duration of the growth stages. When propagated by seed, the total cycle of plant growth – from planting through to physiological maturity – was two weeks longer than that of plants generated from corms. Again we found that landraces exhibited uniformity concerning their phenological behaviour and the duration of their growth cycle, which may imply genetic homogeneity, as a result of manipulation by generations of farmers.
Table 6. Duration of the phenological stages of three Mauka landraces raised from corms.| Landrace | VegetativeStage (days)* | Reproductive stage (days) | Planting to physiological maturity (days) | |
| Sub-stage early** | Sub-stage late*** | |||
| I | 206 | 52 | 54 | 312 |
| II | 213 | 52 | 55 | 305 |
| III | 192 | 53 | 60 | 305 |
| Average | 204 | 52 | 56 | 312 |
| SD | 10.7 | 0.6 | 3.2 | 7.5 |
Growth Dynamics
In a field trial at the UNC, the growth dynamics of Mauka plants and their organs were evaluated and compared across three different landraces, originated both from corms 46 and from seed 23. In the first case evaluations were made every 30 days and up to 330 days and, in the second case, evaluations were made every 60 days and up to 360 days. Although growth patterns were similar for each landrace, differences were observed according to the type of propagation material used.
For example in landrace III (light green), when the vegetative propagule was used the maximum leaf area was recorded at 210 days (Figure 6A), whereas in plants raised from seed the maximum leaf area was recorded at 240 days (Figure 6B). Furthermore, the maximum leaf area for plants from seed was 36% less than the leaf area of the plants propagated vegetatively. This is because the plants originating from corms have a higher number of stems and subsequently a greater number of leaves, hence a larger maximum foliar area (Table 7). The larger foliar area of these plants amounted to a higher weight of the plant and its organs. In both cases, the maximum dry overall weight of the roots was reached approx. 300 days after planting, with roots from vegetatively propagated plants weighing in heavier than the roots from seed-raised plants, i.e. 2.4 kg plant-1 vs 1.1 kg plant-1, respectively (Figure 6A and 6B).
Figure 6.Growth dynamics of dry matter) in the Mauka plant (landrace III) and its organs, originated from corms (A) and seed (B). Sources: A. 46, B. 23.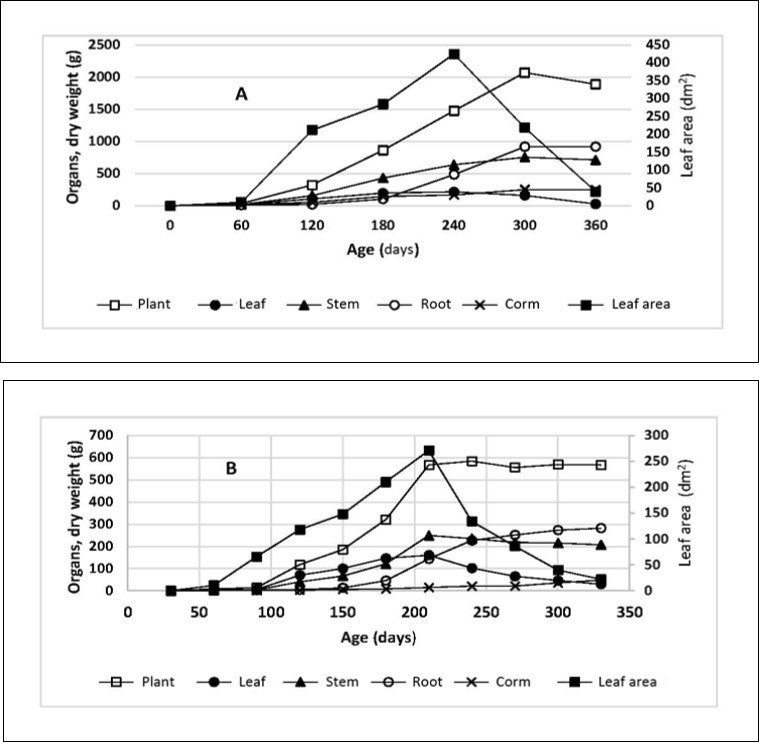
| Landrace | Vegetative stage (days)* | Reproductive stage (days) | Planting to physiological maturity (days) | ||
| Planting till emergence | Emergence till first floral bud | Sub-stage early** | Sub-stage late*** | ||
| I | 18 | 190 | 62 | 63 | 333 |
| II | 15 | 185 | 58 | 60 | 321 |
| III | 13 | 183 | 60 | 62 | 323 |
| Average | 15 | 186 | 60 | 62 | 325 |
| SD | 2.5 | 3.6 | 2.0 | 1.5 | 6.4 |
Growth Rates
In an evaluation of three landraces propagated by corms, conducted at the UNC (Table 8), the main growth indices had similar behaviour 46. These results provide a basis for future productivity studies looking at a wider variety of propagation management factors, against which they may be compared and analysed 49. Besides producing tuberous roots, the various landraces studied behave like forage plants, reaching a leaf area index (LAF) of over 3 after just a few months and increasing up to 8‒10 during its growth cycle; values that are frequently recorded for plants in pastures 50.
Table 8. Growth rates, according to the age of the plant of three Mauka landraces propagated by corms.| Landrace | Age (days) | LAI | Stage (days) | RLGR(dm2 dm-2) | CGR(g m-2 d-1) | RGR(g g-1-d-1) | NAR(g dm-2 d-1) |
| I | 60 | 0.24 | 60‒120 | 0.045 | 7.71 | 0.031 | 0.061 |
| 120 | 3.63 | 120‒180 | 0.006 | 18.57 | 0.018 | 0.042 | |
| 180 | 5.27 | 180‒240 | 0.005 | 5.89 | 0.003 | 0.013 | |
| 240 | 3.79 | 240‒300 | -0.002 | 37.54 | 0.012 | 0.009 | |
| 300 | 3.74 | 300‒360 | -0.027 | -4.08 | 0.000 | -0.022 | |
| 360 | 0.71 | ||||||
| II | 60 | 0.24 | 60‒120 | 0.050 | 11.85 | 0.040 | 0.087 |
| 120 | 4.13 | 120‒180 | 0.008 | 29.88 | 0.019 | 0.056 | |
| 180 | 6.72 | 180‒240 | 0.003 | 8.45 | 0.002 | 0.011 | |
| 240 | 8.20 | 240‒300 | 0.000 | 29.87 | 0.006 | 0.039 | |
| 300 | 5.37 | 300‒360 | -0.02 | -7.79 | -0.001 | -0.029 | |
| 360 | 1.07 | ||||||
| III | 60 | 0.28 | 60‒120 | 0.048 | 12.44 | 0.041 | 0.074 |
| 120 | 5.25 | 120‒180 | 0.006 | 22.66 | 0.016 | 0.035 | |
| 180 | 7.92 | 180‒240 | 0.005 | 25.96 | 0.008 | 0.028 | |
| 240 | 10.57 | 240‒300 | -0.007 | 24.65 | 0.005 | 0.068 | |
| 300 | 6.79 | 300‒360 | -0.028 | -6.95 | -0.001 | -0.026 | |
| 360 | 0.98 |
The allocation of the dry matter to the plant organs was similar for plants vegetatively propagated and those generated from seed, but this aspect did vary according to landrace (Figure 7). The harvest index, i.e. % of dry matter allocated to the roots, varied from 49% to 51% for plants from corms and seed, respectively. In both cases, landrace I produced a greater harvest index than landraces II and III (Figure 6). These values stand out as being higher than the harvest index recorded for yacón (Smallanthussonchifolius) ‒ another tuberous root native to the Andes 51 ‒ and close to those achieved in improved potato varieties 52, 53. These relatively high and stable harvest index levels 54 demonstrate Mauka's potential as a productive crop, whilst the comparison of values for different landraces can be applied to the breeding of potentially even higher yielding cultivars.These relatively high and stable harvest index levels demonstrate Mauka's potential as a productive crop, whilst the comparison of values for different landraces can be applied to the breeding of potentially even higher yielding cultivars. At the end of the vegetative stage (around 200 days into the growth cycle), they have accumulated about 50% of the dry matter and at 300 days they have reached the maximum accumulation of reserves. This information is useful for determining the optimum time to harvest Mauka. Early harvests can be tested to see if fresh produce can be obtained without noticeably compromising total production.
Figure 7.Allocation of assimilates to the plant organs in three Mauka landraces raised from corms (A) and seed (B). Sources: A. 45, B. 23.
Harvest
Farmers typically harvest the Mauka crop after one year of maturation, although some report harvesting it as early as 8 months. Yellowing foliage, shedding leaves and the stickiness of its inflorescences are said to indicate that a Mauka plant is mature enough to harvest. However, as a marginal crop, the harvest of the main crop tends to determine the timing of the Mauka harvest, i.e. Mauka will usually be harvested together with main crops, or just after them. Nevertheless, because Mauka is a subsistence crop with a flexible vegetative period, it is important to note that farmers often harvest it as and when it is required, plant by plant. The practice of digging to remove just a few roots and allowing the plant to continue growing has also been reported. In this sense, Mauka can be conceived of as a sort of ‘living larder’ 19.
From observing the root growth dynamics of the three most common landraces in northern Peru, it is inferred that tender and fresh roots can be harvested from 7.5 months. In the valley of Cajamarca ‒ as indicated above – at 240 days plants have accumulated more than 60% of their total root dry weight; at 270 days they manage to accumulate more than 80% of their dry weight and; at 300 days the growth of these reserves practically ceases (Figure 4 and Figure 6). However, this is a subject that should be studied in more detail. Especially since some farmers have asserted that Mauka roots do in fact continue to increase in size beyond one year 19.
Yield of Tuberous Roots and Fodder
Experimental studies in several locations in several locations of Peru, Ecuador and Bolivia (Table 9) indicate that the production potential of tuberous roots is high, but at the same time highly variable (12 to 137 t ha-1) due to the influence of management and environmental conditions. From the data reported, it can be inferred that yields of 30 to 58 t ha-1 are typical for the Andean region.
Table 9. Harvested Mauka yields (experimental) reported in the literature.| Locality, country | Yield (t ha-1) | Plants ha-1 | Source |
| Cajamarca, Peru | 12‒52 | 22 222 | 28 |
| Cajamarca, Peru | 43‒52 | 25 000 | 55 |
| Cajamarca, Peru | 44‒72 | 25 000 | 46 |
| Quito, Ecuador | 2.1‒15.3 | 09 569* | 56 |
| Quito, Ecuador | 16‒25 | 10 000** | 39 |
| Chullina, Saavedra, Bolivia | 32‒40 | Not indicated | 33 |
| Cajamarca, Peru | 30‒37 | 28 571 | 23 |
| Cajamarca, Peru | 31 | 28 571 | 24 |
| Cajamarca, Peru | 45‒137 | 25 000 | 7 |
| Quito, Ecuador | 13‒40 | Not indicated | 57 |
| Cajamarca, Peru | 21‒39 | 20 833 | 20 |
| Tambopata, Puno, Peru | 32‒79 | Not indicated | 42 |
However, the observations of five Peruvian landraces over several plantings and years (Table 10) show important differences in the yield components and the yield of roots and foliage (stems plus leaves), which implies the possibility of selecting materials for specific purposes and the need to test agronomic treatments for crop improvement.
Table 10. Yield components of five landraces (40 entries) of Mauka from Peru.| Landrace | Plant height (cm) | No. Roots | Root length (cm) | Root weight(kg plant-1) | Yield (t ha-1) | Foliage weight(kg pant-1) |
| I | 73.3 | 16.5 | 12.91 | 1.80 | 39.10 | 1.60 |
| II | 77.6 | 14.4 | 15.60 | 0.90 | 20.50 | 1.50 |
| III | 81.2 | 12.7 | 12.90 | 1.60 | 33.80 | 1.70 |
| IV | 47.0 | 10.5 | 13. .0 | 1.40 | 29.10 | 0.60 |
| V | 59.3 | 14.0 | 12.90 | 1.60 | 33.20 | 1.70 |
| Average | 67.68 | 13.62 | 13.46 | 1.46 | 31.14 | 1.42 |
| SD | 14.23 | 2.21 | 1.19 | 0.34 | 6.19 | 0.46 |
In addition to tuberous roots, the Mauka plant also yields leaves and stems (Figure 8), which are traditionally used as feed for both smaller animals, e.g. guinea pigs and rabbits, as well as larger livestock, e.g. cattle and sheep ‒ however, the practice is not universal amongst farmers. 9, 26, 29, 58. Some identify Mauka as an important forage plant, and several farmers have noted that grazing livestock prefer Mauka to other plants. In Corongo (Ancash), the practice of cutting green material from Mirabilis prostrata (Ruíz & Pav.) Heimerl Mauka’s wild relative (proliferating at the border of a field) ‒ as fodder for pigs, has been observed. In Chachapoyas, where the practice of cultivating Mauka now seems to have disappeared, one farmer recounted the historical use of Mauka roots as pig feed 19.
Figure 8.Mauka plant (raised from corm) harvested at 10.5 months. Note the production of many tuberous roots and green material, suitable for use as livestock feed or forage.
An evaluation carried out over three agricultural seasons, which assessed production levels for 40 different accessions of Mauka (5 plants per accession), produced forage yields which averaged 1.6 ± 0.6 kg plant-120 - which is equivalent to 33.3 tons of green fodder ha-1.
In order to assess forage production, Bazan et al. 9 planted a plot of 557 m2 with Mauka plants (purple landrace), with 0.8 m spacing between rows and 0.5 m between plants.. The yield per plant (Table 11) corresponds to 76 t ha-1 of green fodder with a dry matter percentage of 22%. This yield is superior to the yield obtained both in experimental trials with lucerne (Medicago sativa L.) 59, 60 and Napier grass (Pennisetum purpureum Schumach.) from a Taiwanese pasture 61.
Using these studies as a starting point, further research on forage production from Mauka can be carried out compared to the production levels of other forage species, as suggested by Kritzer Van Zant 10, 38, taking into consideration the fact that yields will inevitably vary according to crop husbandry, environmental conditions, the time of harvest and ‒ most likely also ‒ landrace.
Table 11. Number of stems and fresh weight of green forage per Mauka plant| Statistics | No. stems | Forrage fresh weight (kg) |
| Minimum | 4.5 | 0.25 |
| Maximum | 17.5 | 9.25 |
| Average | 9.5 | 1.95 |
| SD | 3.5 | 1.65 |
Postharvest Handling of Tuberous Roots.
The roots, dense clustered with low water content, are resistant to handling. Post-harvest they are seldom attacked by pathogens, and their tissues show no changes of the kind that might inhibit consumption, as with other roots, such as arracacha (Arracaciaxanthorrhiza), yuca/manioc (Manihot esculenta) and yacón (Smallanthussonchifolius). Fresh Mauka roots are naturally astringent; a characteristic which is more pronounced in certain landraces. . This is traditionally removed in two ways: (a) by changing the water once or twice during cooking, and (b) by exposing the roots to the sun for a period of 2‒10 days ‒ a practice known as ‘soleando’, which also sweetens the roots by increasing the concentration of sugars 1, 19.
The roots can be kept outdoors for several weeks without deterioration, and, if they are left to dehydrate in a dry environment, can remain unchanged for many years (Figure 9, left). They can also be preserved using the parboiling technique, following the procedure used to precook arracacha 62 ‒ by washing the root, cooking them for five minutes, slicing, sun drying and packing them (Figure 9, right, top). Another way is through natural drying, used for yuca/manioc, which entails washing and cutting the raw roots, drying them in the sun, and packaging 63. In a modification of this last technique the roots are cut into thin slices (Figure 9, right, bottom).
Both pre-cooked and raw products can be used in savoury and sweet preparations, or they can be ground to obtain flour or starch for various uses.
Farmers wash the roots and peel them either prior to cooking by scraping the thin skin off with a knife, or after cooking ‒ usually by hand. They typically consume the roots boiled as an accompaniment to other food, in the same way that yucca/manioc is eaten Mauka is also served unaccompanied; boiled and washed down with coffee for breakfast or lunch, in place of bread or potatoes. Other dishes made with Mauka mentioned by farmers include ‘mazamorra’ (a sweet pudding), ‘picante de cuy’ (spicy stew made with guinea pig), soups and broths, and ‘pachamanca’ or ‘huatia’ (an earthen oven which entails burying the roots in the ground with hot coals) 19.
Figure 9.Left: A cluster of tuberous roots from a Mauka plant raised from seed, dried outdoors and preserved in a museum (UNC). Upper right: pre-cooked Mauka root pieces. Bottom right: sun-dried raw Mauka root slices.
Conclusions
Over half a century has now passed since the (re-)discovery of the cultivated Mauka plant, and during that time important advances in the understanding of its agronomic traits and potential have been accomplished. The sum of this research forms an empirical and theoretical basis for the promotion and development of Mauka as a commercial crop. So far, studies have focused mainly on the biology of the Mauka plant in agricultural systems, its propagation methods, phenology, and analysis of its growth.
The less studied aspects are its response to organic and chemical fertilisers, variation in productivity across different environments, recommended planting densities ‒ especially in association with other crops ‒ the timing of harvest and the simultaneous optimisation of root and fodder production. Field trials assessing these qualities could further improve the productivity of Mauka, while taste trials examining the variation in astringency and sweetness of various landraces could improve its culinary prospects.
Ethnobotanical surveys have been carried out, which have significantly enriched our understanding of the versatility of Mauka, but the full distribution range of the crop in its cultivated form is still incomplete and the local significance and history of Mauka across the Andean region remains poorly understood. Efforts to map its current range and collect both cultivated and wild germplasm for conservation should be widened to underexplored areas of Peru and, most urgently, to Bolivia ‒ where no material is conserved ex-situ.
In order to prevent genetic erosion and the conservation of traditional ecological knowledge, it is necessary not only to maintain Mauka in genebanks, but also to promote its continued cultivation in-situ by local farmers. This endeavour can be supported by the application of findings from the agronomic research accumulated so far, and from further research aimed at improving sustainable production techniques. This way, it is hoped that Mauka will be more widely appreciated as both a biological and a cultural resource; and its role as a resilient, nutritious and profitable crop plant secured not only for future generations of Andean farmers but also as an important contributor to the food sovereignty of Andean communities, which merits its study and conservation.
Acknowledgements
The International Potato Center (CIP) and the Swiss Agency for Development and Cooperation (COSUDE) are gratefully acknowledged for funding research via the Andean Roots and Tubers Biodiversity Program on Mauka and other tuber crops during the decade from 1993 to 2003. Thanks to the Universidad Técnica del Norte-Ecuador for the space given for the implementation of conservation fields of miso at the Pradera Biological Knowledge Center. This will allow carrying out future research related to the crop. We also give warm thanks to the farmers who have shared their knowledge with the authors and other researchers over the years.
References
- 1.Rea J, León J. (1965) La mauka (Mirabilis expansa Ruíz y Pavón) un aporte de la agricultura andina prehispánica de Bolivia. Anales Científicos de la Universidad Agraria. 3(1), 38-41.
- 2.Johns T. (1989) A chemical-ecological model of root and tuber domestication in the Andes. In:. Harris DR, Hillman BC (eds.). Foraging and farming. London (UK). Unwin Hyman 504-519.
- 3.Hawkes J G. (1989) The domestication of roots and tubers in the American tropics. In: Harris DR, Hillman BC (eds.). Foraging and farming. London (UK). Unwin Hyman 481-503.
- 5.Standley P C. (1931) . The Nyctaginaceae and Chenopodiaceae of Northwestern South America. Publication 3003. Botanical series XI(3): 71-126.
- 6.Villacrés E, Espín S. (1999) Evaluación del rendimiento, características y propiedades del almidón de algunas raíces y tubérculos andinos. Raíces y tubérculos andinos. Avances de investigación I. Centro Internacional de la Papa y Consorcio para el Desarrollo Sostenible de la Ecorregión Andina. Pp In: Fairlie T, Morales Bermúdez M, Holle M (eds.) 25-36.
- 7.Seminario J. (2004) Aspectos etnobotánicos y productivos del chago, miso o mauka[Mirabilisexpansa(Ruíz & Pavón) Standley].In: Seminario. Raíces andinas. Contribuciones al conocimiento y a la capacitación. Una década de investigación para el desarrollo (1993‒2003).N° 6. Universidad Nacional de Cajamarca, Centro Internacional de la Papa, Agencia Suiza para el Desarrollo y la Cooperación J (ed.) , Lima, Perú. Pp: 367-376.
- 8.Montenegro L F, Franco S. (1988) Evaluación de nutrientes en tres variedades deMirabilisexpansa“chago”. Actas del VI congreso Internacional sobre Cultivos Andinos. , Quito, Ecuador 268-273.
- 9.Bazán M, López J I, Pajares W. (1996) Potencial forrajero del chago (Mirabilisexpansa) en la alimentación de conejos(Oryctolaguscuniculus).Thesis Ing. , Zoot. Cajamarca, Facultad de Zootecnia, Universidad Nacional de Cajamarca 93, p..
- 10.Van Kritzer.Zant M (2016b) History ofMirabilisexpansa(Ruíz and Pav.) Standl.;growth and use in the Andes. , Atlas Journal of Biology 236-248.
- 11.Vivanco J M, Savary B J, Flores H E. (1999) Characterization of two novel type I ribosome-inactivating proteins from the storage roots of the Andean cropMirabilisexpansa. , Plant Physiology 119, 1447-1456.
- 12.Vivanco J M, Flores H E. (2000) Biosynthesis of ribosome-inactivating proteins from callus and cell suspensión cultures ofMirabilisexpansa(Ruíz. , Pavon). Plant Cell Reports 19, 1033-1039.
- 13.Flores H E, Walker T S, Guimarães R L, Bais H P, Vivanco J M. (2003) Andean root and tuber crops: Underground rainbows. , HortScience 38(2), 161-167.
- 14.Vepachedu R, Park S W, Sharma N, Vivanco J M. (2005) Bacterial expression and enzymatic activity analysis of ME1, a ribosome-inactivating protein fromMirabilisexpansa. , Protein Expression & Purification 40, 142-151.
- 15.Rojas N, Flores Y. (2015) Tamizaje fitoquímico preliminar del extracto etanólico deMirabilisexpansa(Ruíz y Pavón) ‘mauka’. Anales de la Facultad de Medicina, Universidad Nacional Mayor de San Marcos. Resúmenes de XIV jornadas científicas sanfernandinas, XVII jornadas de investigación en salud, XXIV jornadas sanfernandinas estudiantiles. 7‒9 setiembre. 22.
- 16.Chandrasekara A, Kumar T J. (2016) Roots and tuber crops as functional foods: A review on phytochemical constituents and their potential health benefits. Article ID 3631647. Visited20thOct2018.Doi.org/10.1155/2016/3631647. 15 , International Journal of Food Science Volume, p..
- 17.Leidi E O, Monteros-Altamirano A, Mercado G C, Rodríguez J P, Ramos A et al. (2018) Andean roots and tubers as sources of functional foods. , Journal of Functional Foods 51, 86-93.
- 18.NRC (National Research Council) (1989) Lost crops of the Incas. Report of anad hocpanel of the advisory committee on technology innovation. Board on Science and technology for international development. National Academy Press. , Washington DC
- 19.Gendall H E. Agricultural Development. Faculty of Sciences, University Copenhagen (2017) Remembering mauka.Biocultural diversity conservation and the case the ‘lost’ Andean cropMirabilisexpansa(Ruíz & Pav.)Standley. MSc. Thesis 218, p..
- 20.D S 043-2006-AG. (2006) Aprueban categorización de especies amenazadas de flora silvestre. El Peruano. , Lima, Peru 323527-323539.
- 21.Seminario J, Valderrama M. (2012) Variabilidad morfológica y evaluación agronómica de maukasMirabilisexpansa(Ruiz & Pav.) Standl. del norte peruano. , Rev. Perú. Biol 19, 249-256.
- 23.Rodríguez L G. (2002) Estudio de la tuberización de chago(MirabilisexpansaR. & P.)a partir de semilla botánica. Thesis Ing. Agr., facultad de Ciencias Agrícolas y Forestales, Universidad Nacional de Cajamarca. 55, p..
- 24.Seminario J, Valderrama M, Rodríguez L. (2002) Estudio de la tuberización de la mauka o chago,Mirabilisexpansa(R. & P.) Standley a partir de semilla sexual. , Caxamarca 10(1), 89-98.
- 26.Rea J. (1982) El Miso: Una contribución de la agricultura Pre-Inca de Ecuador y Bolivia. Desde El Surco 23–26.
- 27.Valderrama M, Seminario J. (2001) Biología floral de cuatro cultivares y tres parientes silvestres de “mauka”Mirabilisexpansa(R. , P.) Standley. Arnaldoa 8(1), 71-86.
- 28.Seminario J. (1993) Aspectos etnobotánicos y morfológicos del cahgo, miso o mauka (MirabilisexpansaR. y P.) en el Perú. , Boletín de Lima 86, 71-79.
- 30.Hartmann H T, Kester D E, Davies F T, Geneve R L. (2014) . Hartmann & Kester's Plant Propagation. Principles and Practices. 8 ed.PearsonEducationLimited, USA 583-615.
- 31.Tapia C, Castillo R, Mazon N. (1996) Catálogo de recursos genéticos de raíces y tubérculos andinos en Ecuador. , INIAP. Quito, Ecuador 142-144.
- 32.Rea J. (1999) Conservación y manejo in situ de recursos fitogenéticos agrícolas en Bolivia. Raíces y tubérculos andinos. Avances de investigación I. Centro Internacional de la Papa y Consorcio para el Desarrollo Sostenible de la Ecorregión Andina. Pp In: Fairlie T, Morales Bermúdez M, Holle M (eds.) 61-76.
- 34.Pulgar Vidal J. (1996) Geografía del Perú. Las ocho regiones naturales del Perú. Printer colombiana S.A. , Bogotá, Colombia 302, p..
- 35.Seminario J, Seminario A. (1995) Colección de germoplasma regional de raíces andinas. , Boletín de Lima 98, 27-47.
- 36.QCB Cronk, Fuller J L. (1995) Plant invaders: the threat to natural ecosystems. 'People and plants' conservation manuals Unesco, World Wide Fund for Nature, Royal Botanic Gardens. , Kew 29(241).
- 37.M Kritzer Van Zant. (2016) Review of the economic and ethnobotanical of the family Nyctaginaceae. , Atlas Journal of Biology.Doi-10.5147/ajb2016.0139 249-266.
- 38.M Kritzer Van Zant, Diesburg K. (2018) Lightfoot DA (2018)Mirabilisexpansagrowth, yield, and quality traits: First US field trials for an ancient, endangered, Andean Crop. , Atlas Journal of Biology 551-574.
- 39.Ramos R. (1999) Respuesta de tres raíces andinas: Zanahoria blanca (ArracaciaxantorrhizaB.), Miso (MirabilisexpansaR. & P.) y Jícama (PolymniasonchifoliaP. & E.); dos pastos y una mezcla forrajera, al efecto de tres sistemas agroforestales preestablecidos.Thesis Ingeniero Agrónomo. Universidad de. , Cuenca. Ecuador
- 40.Klásková T, Fernandéz C. Institute of Tropics and Subtropics, Czech University of Life Sciences (2011) Mauka – a promise for fighting with the unbalanced nutrition of high Andean regions. , Prague, Czech Republic, Agricultura Tropica et Subtropica 44(1), 37-40.
- 41.Franco S, Rodríguez J. (1988) Multiplicación por esquejes del chago, miso o mauka(Mirabilisexpansa).Memorias el VI Congreso Internacional sobre Cultivos Andinos. INIAP. , Quito, Ecuador 265-267.
- 42.Alva U. (2005) Efecto de tres sustratos en la propagación del chago (MirabilisexpansaR. y P.) Standley, por esquejes. Cajamarca, Thesis Ing. Agr. Facultad de Ciencias Agrícolas y Forestales, Universidad Nacional de Cajamarca. 39, p..
- 43.Zapana J G, Vilalta P, Mamani M, Escobar F. (2015) Efecto del abono orgánico y fertilizantesquímicos en la producción de semilla botánica y raíz tuberosa de la “mauka”. , Mirabilisexpansa(Ruíz y Pavón) Standley). Rev. Investg. Altoandin 17(1), 73-78.
- 44.Harlan J R. (1975) Crops and man. , Madison, Wisconsin, US, American Society of Agronomy. Crop Science Society of America
- 46.Benjamin L, McGarry A, Gray D. (1997) The root vegetables: beet, carrot, parsnip and turnip. The physiology of vegetables crops. CAB International In H.C. Wien (ed.) , Washington 553-573.
- 47.Seminario J, Ruíz J. (1997) Recursos genéticos de raíces andinas: V. Análisis del crecimiento de tres cultivares de chago (Mirabilisexpansa). Libro de resúmenes IX Congreso Internacional de Cultivos Andinos “Oscar Blanco Galdós”, 22‒25 abril. , Cusco, PE 58-59.
- 48.Lizárraga C. (2004) Virus en raíces andinas. In: Seminario. (1993‒2003).No. 6. Universidad Nacional de Cajamarca, Centro Internacional de la Papa, Agencia Suiza para el Desarrollo y la Cooperación J (ed.) , Lima, Peru. Pp: 123-126.
- 49.Hunt R. (1990) Basic growth analysis. Plant growth analysis for beginners. Unwin Hyman. London England.
- 50.RKM Hay, Walker A J. (1989) An introduction to the physiology of crop yield. , Longman Scientific & Technical 190, p..
- 51.Seminario J, Cruzado A. (2004) Introducción de tres cultivares de llacón en el valle de Condebamba. , Cajamarca. Caxamarca 12(2), 17-24.
- 52.Ñustez C E, Castellanos M S, Segura M. (2009) Acumulación y distribución de materia seca de cuatro variedades de papa(SolanumtuberosumL.)en Zipaquira. , Cundinamarca(Colombia). Rev. Fac. Nal. Agr. Medellín 62(1), 4823-4834.
- 53.Saldaña T M, Patiño J A, Cotes-Torres J M. (2015) Biomass distribution and allocation in diploid potato varieties (SolanumphurejaJuz. et Buk.). , Agronomía Colombiana 33(3), 322-329.
- 54.Gardner F P, RL Pearce RB Mitchell. (1985) Physiology of crop plants. , IowaStateUniversityPress 66-67.
- 55.Franco S, Vejarano J. (1996) El chago o yuca inca (Mirabilisexpansa) raiz andina en peligro de extinción. Instituto Nacional de Investigacion Agraria. , Cajamarca-Peru
- 56.Morillo E. (1998) Análisis de polimorfismo en las colecciones de jícama (PolymniasonchifoliaP. & E.) y miso (MirabilisexpansaR. & P.) del banco de germoplasma del INIAP. Thesis Licenciado en Ciencias Biológicas. Departamento de Ciencias Biológicas, Pontificia Universidad Católica del Ecuador. , Quito, Ecuador 105, p..
- 57.Tapia C, Estrella J, Monteros A, Valverde F, Nieto M et al. (2004) Manejo y Conservación de RTAs in situ en fincas de agricultores y ex situ en el Banco de Germoplasma de INIAP. Raíces y Tubérculos Andinos: Alternativas para la conservación y uso sostenible en In: Barrera V, Tapia C, Monteros A (eds.) 31-74.
- 58.Seminario J. (1988) El chago o mauka (Mirabilisexpansa) en Cajamarca. Resúmenes del VI Congreso Internacional sobre Cultivos Andinos. , Quito, Ecuador 93, p..
- 59.Villegas Y, Hernández A, Martínez P A, Pérez J, Herrera J G et al. (2006) Rendimiento de forraje de variedades de alfalfa en dos calendarios de corte. , Revista Fitotecnia Mexicana 29(4), 369-372.
- 60.Fanlo R, Lloveras J, Chocarro C. (2010) Conveniencia del abonado y la fertilización en el cultivo de alfalfa. Vida rural.Dosier Cultivos forrajeros. Pp. 20‒22. Disponible:https://repositori.udl.cat/bitstream/handle/10459.1/47098/015224.pdf?sequence=1
- 61.Osuna J D, Chávez G, Avalos R, Sánchez M, Leyva J C. (2016) Efecto de la fertilización y la madurez al corte sobre el rendimiento y calidad de. Taiwan.2ed.Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias (INIFAP), MEX. http://apptestrvc.itvara.net/documentos/Folletos/F_efecto_de_la_fertilizacion_nitrogenada.pdf 28, p..
Cited by (6)
This article has been cited by 6 scholarly works according to:
Citing Articles:
(2025) Crossref
Elsevier eBooks (2024) OpenAlex
M. K. V. Zant, K. Triwitayakorn, D. Lightfoot - (2020) Semantic Scholar
M. K. Van Zant, W. Banz, Gary A. Apgar, D. Lightfoot - Atlas Journal of Biology (2019) Semantic Scholar
Economic Botany (2019) Crossref
H. Gendall, J. Seminario, M. Sørensen, I. Theilade - Economic Botany (2019) Semantic Scholar
Economic Botany (2019) OpenAlex
