Abstract
Background.
Emotional deterioration is frequently found in patients with chronic kidney disease, but some patients are affected by depressed mood without fulfilling the criteria of a depressive disorder. Those patients might rather suffer from an apathy symptom. Apathy as a symptom of a medical disease is accompanied by loss of motivation and interest, cognitive impairments, and emotional distress. Our study tested how groups of apathetic and depressed chronic kidney disease patients responded to a single haemodialysis session on measures of mood.
Methods.
21 haemodialysis patients were assigned to subgroups (depressed, apathetic only, without depression and apathy) according to clinical relevant cut-offs. Sensitive questionnaires were administered to monitor mood state in general and mood changes before and after a single haemodialysis session in the chronic kidney disease patients. The results were compared to 20 age-matched healthy controls receiving no treatment.
Results.
Fortheen dialysis patients had eighter apathy or depressen and seven had neighter apathy nor depression. Mood state was seriously affected in all haemodialysis patients with depressed patients showing the largest effect compared to healthy controls. Patients with apathy and patients without apathy and depression were comparable in their mood state. We observed a positive mood change after the haemodialysis only in patients without apathy and depression. Their mood state improved significantly and reached nearly the level of healthy controls.
Conclusions.
The absence of a short-term mood change in apathetic dialysis patients makes them comparable to dialysis patients with depression. We argue that apathetic patients lack the probable mood brightening effect of the haemodialysis. The lack of emotional improvement by dialysis sessions could also lead to decreased adherence of the patients. Hence, apathy seems to be a serious symptom in chronic kidney disease that is worth to be considered at least in the diagnostic process. Accompanying psychotherapeutic care for these patients would be desirable
Author Contributions
Academic Editor: Addo Boafo, Royal Institute of mental health research
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 R. Schoenfeld,et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Depression has been identified with a prevalence of 21% and as the most common psychiatric disorder in chronic kidney disease (CKD)1, 2, 3. It is an important risk factor of mortality in these patients4, 5. 10-12% of stage 5 CKD patients, i.e. patients with an end-stage renal failure requiring chronic dialysis, have an increased suicidal risk6 and an 84% higher rate of suicide than the general population7. Furthermore symptoms of depression limit the patients’ quality of life and amplify the progression of the underlying renal disease8. A clinical relevant depression decreases also the patients’ adherence on the requirements of the dialysis session such as restriction on fluid intake.9. Depression is characterized by somatic, cognitive, and emotional symptoms. In CKD sleep disturbances, pain, and fatigue are common symptoms of the uraemia. The similarity to the somatic symptoms of a depression makes it difficult to determinate and contributes to false-positive diagnoses of depressions. Studies observing depressive disorders in CKD reported biased depression scores when self-report questionnaires were administered10. These studies turned out cognitive-emotional symptoms of the depression and suicidal thoughts as the best discriminators between CKD patients with and without depression9.
Beside depressive symptoms in CKD patients the clinical impression is often marked by loss of motivation and interest and emotional distress. A simultaneous depletion of behaviour, cognition, and emotion is observed although no pronounced depression exists in these patients. We argue that apathy can describe this state of non-depressed but affective impaired CKD patients. Apathy has received different definitions in the literature. Marin defined apathy as loss of motivation and interest and differentiated between apathy as a syndrome and apathy as symptom of diseases and disorders associated with cognitive impairment, diminished level of consciousness, or disturbance of emotion 22, 23. Whereas the syndrome of apathy is not attributed to states of diminished level of intellect and emotion, apathy as a symptom is related to many psychiatric, neurological, and somatic disorders. In particular apathy is symptomatic in most patients with major depression. Their apathy scores are high although a number of these patients show no symptoms of apathy24. Research on apathy in individuals with Alzheimer’s disease25, traumatic brain injury 26, and HIV-infection27, 28, 29 revealed out the same dissociation between depression and apathy. Subgroups of patients with depression without apathy, with depression and apathy, and with apathy only coexist among these populations. Symptoms of apathy were frequently associated with the cognitive impairment inherent to the disorder and expressed as a lack of motivation and the absence of self-initiated behaviour in patients’ everyday life.
Whereas the occurrence of depression in CKD and its negative impact on cognition and quality of life has been widely studied, symptomatic apathy has never been considered so far. It remains unclear whether apathy and depression could be differentiated also in CKD patients. We assume that apathetic and depressed patients independently exist among the CKD population. Apathy and depression should have different effects on the quality of life, in particular on the long-term mood state of the patients. Furthermore, symptoms of apathy and symptoms of depression should moderate the emotional reactivity to the haemodialysis treatment to a different extent. The hypothesis was tested by means of a pre-post control group design evaluating changes of mood before and after a single dialysis session.
Material and Methods
Sample
This is a secondary analysis of a study that had been published previously 30. Here, we report data on depression and apathy of, twenty-one CKD patients with age ranging from 37-79 years and twenty-two age matched healthy controls. All patients were treated at the dialysis unit of the two participating tertiary care centres in Hannover and Halle. Controls were recruited in the regions close to the two participating hospitals. Exclusion criteria employed at the recruitment stage were: impaired uncorrected vision, a history of head trauma, neurological signs and symptoms, substance abuse disorder, or psychiatric conditions other than depressive or anxiety disorders. Inclusion criteria for the CKD patients were an end-stage renal failure (CKD stage 5) and a routinely dialyses treatment for at least one month.
All participants were tested twice within a 24-hour retest interval. CKD patients were tested at two time points, immediately before dialysis as well as 24 hours after dialysis to assess the potential effect of dialysis avoiding circadian changes. Tests were performed in a quiet room. Controls were tested in their home settings. Demographic data, depression and apathy were assessed from all participants. From dialysis patients, specific medical history, routine laboratory tests and a complete list of medication were obtained.
The study was approved by the local Ethics Committee (reference #2012-30) and conducted according to the ethical principles regarding human experimentation as laid down in the Declaration of Helsinki. After all participants were provided with detailed information about the purpose, course, and possible risks of the study, they gave their writtenconsent.
Assessment of Apathy and Depression
Apathy was assessed with a German version of the Apathy Evaluation Scale (AES). The AES is a self-assessment syndrome-independent scale to evaluate symptoms of a lack of motivation in several disorders and diseases11. The questionnaire consists of 18 items with a 4-point Likert scale scoring categories “strongly agree”, “agree”, “disagree”, and “strongly disagree”. Each item is scored with 1 up to 4 points indicating increasing symptoms of apathy. A total sum score is computed with 18 points indicating no symptoms of apathy. The AES is missing an appropriated criterion to decide for a clinical relevant apathy so far. We computed a cut-off value ≥34 (2 SD) derived from the normative data provided in Lueken et al.11. Participants with AES scores exceeding this value were classified as apathetic.
The Beck Depression Inventory (BDI) was used to assess the severity of a probable mood disorder12. As a self-assessment questionnaire the BDI consists of 21 items related to affective, cognitive, and somatic symptoms. A sum score is computed and compared to a cut-off value. CKD patients scoring ≥15 are considered with clinical relevant symptoms of a depression13. BDI subscales were also computed to further analyse the underlying structure of the depression14. Items 10, 11, 16, 17, 18, 19, 20, and 21 ask for somatic symptoms and were summarized in a sub score. The items 3, 5, 7, and 8 are associated with cognitive symptoms. Items 1 and 2 were related to depressed mood. All together were summarized into a score for cognitive-emotional symptoms. Suicidal thoughts were asked only with item 9. Items 4, 6, 12, 13, 14, and 15 asses lack of satisfaction, sense of punishment, body image, social withdrawal, and indecisiveness, but do not represent a single sub score in the underlying factorial structure of the BDI14.
Mood State
To assess persistent deteriorations in mood a questionnaire of a German health survey of Quality of Life was administered15. This questionnaire asks for prevailing mood, mood state, life orientation, somatic complaints, and social support within a time period of the last four weeks. We used a short form of the questionnaire with 28 items in total. Each item was measured on a 5-point rating scale and recoded such that higher values indicate a better mood state. The sum score of all items was conducted to the statistical analysis. This score was reported as consistent (Cronbach alpha = 0.88) and reliable (rt= 0.70) by the authors15.
Mood Changes
In order to measure short-term changes in mood we needed a method that is sensitive enough to detect subtle differences within a very narrow time interval (24h retest interval). The multi-dimensional state questionnaire is a standardized instrument which is available in two forms (A and B) and met our requirements of short retest intervals16. Form A and B were developed as parallel version and absolutely comparable. We chose both forms for the purposes of a repeated measure in our pre-post design. Form A was administered at the pre-dialysis test session, form B at the post dialysis session. Each form consists of 12 5-point Likert scale items that were summarized into a sum score. The scale measures good momentary mood: a person scoring high on that scale is feeling well, satisfied, and joyful. Internal consistency of form A and B was reported as good (Cronbach alpha =0.7-0.8). Retest reliability between form A and B was reported with rtt=0.8-0.9.
Statistics
First, BDI and AES scores were computed for each participant and compared with the cut-offs. Based on this decision CKD patients were assigned into three subgroups: patients without apathy and depression (CKD─), patients with apathy only (CKD+A), and patients with depression (CKD+D). Healthy controls who meet the apathy or depression criteria were excluded from further analyses. The control group and the three CKD subgroups form the independent variable.
Second, descriptive statistics (group means, standard deviations, standard error of means, or frequencies) were computed for each dependent variable. Hypotheses’ testing was performed as group mean testing and relies on one-way ANOVAs and orthogonal linear contrast in case of comparing healthy controls and CKD patients. Comparisons of CKD patients only rely on one-way non-parametric ANOVAs by Ranks (H test), because of low group numbers. The general level of significance was set to p<0.05. Measurements of effect size were Cohen’s d for between-group comparisons and Hedges g for pre-post comparisons. Both are comparable in its possible size. A general guideline for interpretation of effect sizes is small (0.3), moderate (0.5), and large (0.8). Statistical calculations were performed with SPSS 20 software.
Results
Apathy and Depression in CKD
Seven CKD patients were identified with clinical relevant symptoms of depression (BDI≥15). Clinically relevant symptoms of apathy were detected in eleven out of 21 CKD patients and one control person (AES≥34). The diagnosis of apathy overlapped with the diagnosis of depression in four CKD patients. Seven CKD patients did show neither depression nor apathy symptoms. Figure 1 shows the assignment of CKD patients to the subgroups CKD─, CKD+A, and CKD+D, respectively. Note that we combined depressed patients with and without apathy in the CKD+D group. CKD+A represents patients with apathy only, free of symptoms of depression. Two participants of the control group were identified with depression or apathy and excluded from further analyses, such that the control group contains 20 participants. Table 1 illustrates the characteristics of the CKD subgroups and the control group. Patients and controls were comparable in formal education, but not in their state of employment. No significant differences were obtained in the mean dialysis vintage (length of time on dialysis in month) between the CKD subgroups. However, the range of the dialysis vintage was very wide in all three groups.
Nine of the CKD patients were under medications with psychopharmacological effect: antidepressant and anticonvulsant drugs were most frequently used in the CKD+D group. Sedatives and analgesics were more frequent in CKD with apathy further indicating the subtle complaints in this group of patients.
Apathy and depression symptoms were further characterized by means of an analysis of the BDI and AES subscales, respectively. Somatic and cognitive-emotional symptoms and the BDI sum score were significantly increased in CKD patients compared to healthy controls. CKD+D patients differed significantly to the CKD+A and the CKD─ patients in these scores. However, an average BDI score of 17.4 in CKD+D patients indicated no more than a mild depression. CKD+A and CKD─ showed small but no significant differences compared to healthy controls. The subscale suicidal thoughts could not differentiate the CKD groups.
Behavioural and cognitive-emotional symptoms of apathy and the AES sum score were significantly increased in the CKD+A as well as in the CKD+D group compared to healthy controls, indicating that depression shares nearly all symptoms of apathy. CKD─ patients obtained an AES profile that was comparable to those of the control group.
Figure 1.Block diagram of the classification of chronic kidney disease (CKD) patients regarding present symptoms of depression and apathy, respectively. Beck-Depression-Inventory (BDI) and Apathy Evaluation Scale (AES) cut-offs, when symptoms have to be considered as clinical relevant (+) or not (─), are shown left and on top of the diagram.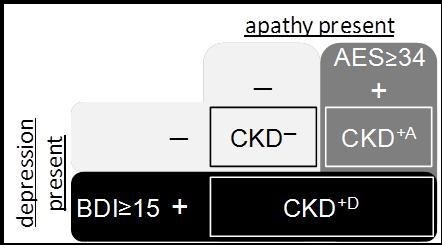
| Controls | CKD─ | CKD+A | CKD+D | P-values | ||
| N=20 | N=7 | N=7 | N=7 | Controls vs. CKD † | CKD only ‡ | |
| Mean age in yrs. (SD) | 56.6 (12.7) {33-80} | 56.9 (18.3) | 57.3 (7.9) | 54.4 (2.8) | 0.925 | 0.833 |
| range | {37-79} | {48-68} | {51-59} | |||
| No. of females | 13 (65%) | 2 (29%) | 1 (14%) | 6 (86%) | 0.155 | 0.042 |
| Employed | 11 (55%) | 3 (43%) | 0 (0%) | 0 (0%) | 0.001 | 0.079 |
| High degree of formal education | 9 (45%) | 4 (57%) | 0 (0%) | 2 (29%) | 0.275 | 0.098 |
| Mean dialysis vintage in month (SD) | - | 35 (36) | 58 (60) | 61 (65) | - | 0.808 |
| range | {1-88} | {4-184} | {3-196} | |||
| Psychopharmacological medication | ||||||
| Antidepressant | 0 (0%) | 0 (0%) | 2 (29%) | 3 (43%) | 0.020 | 0.292 |
| Anticonvulsive | 0 (0%) | 1 (14%) | 0 (0%) | 2 (29%) | 0.079 | 0.742 |
| Sedative, analgesic | 0 (0%) | 0 (0%) | 3 (43%) | 1 (14%) | 0.040 | 0.263 |
| Symptoms of depression (BDI) | ||||||
| Somatic symptoms | 3.5 (2.6) | 4.4 (2.1) | 4.4 (0.8) | 8.0 (2.2) | 0.005 | 0.004 |
| Cognitive-emotional symptoms | 0.4 (0.6) | 0.9 (1.1) | 0.7 (1.5) | 3.3 (1.6) | 0.001 | 0.009 |
| Suicidal thoughts | 0.1 (0.3) | 0.1 (0.4) | 0.1 (0.4) | 0.3 (0.5) | 0.433 | 0.745 |
| Sum score | 5.7 (3.5) | 7.6 (3.7) | 8.0 (2.5) | 17.4 (1.7) | <0.001 | 0.001 |
| Symptoms of apathy (AES) | ||||||
| Behavioural symptoms | 13.0 (2.8) | 13.4 (1.8) | 19.4 (2.1) | 18.9 (5.5) | <0.001 | 0.01 |
| Cognitive-emotional symptoms | 6.5 (1.2) | 6.6 (1.5) | 9.4 (1.1) | 8.4 (2.7) | 0.002 | 0.037 |
| Sum score | 25.5 (4.5) | 26.0 (4.7) | 38.0 (3.8) | 36.1 (10.1) | <0.001 | 0.018 |
Mood State and Mood Changes
Figure 2A shows results of the evaluation of the prevailing mood state in CKD patients and healthy controls by means of the Quality of Life questionnaire. Healthy controls obtained the highest score, which in general indicates a well and stable mood in this group. CKD─ and CKD+A patients together scored significantly below the control group (t(19.3)=2.98, p=0.008, d=1.1). Both CKD groups estimated their overall mood state similar. CKD+D patients answered significantly below all other groups. Group differences were tested against healthy controls (t(8.7)=5.5, p<0.001, d=2.6) and against a combined group of CKD─ and CKD+A (t(9.2)=3.57, p=0.006, d=1.8).
Next we tested changes in mood related to the dialysis treatment. Figure 2B shows group means of the momentary mood estimated immediately before a haemodialysis session (pre dialysis) and 24 hours after the treatment (post dialysis). Pre-test scores of all CKD sub groups were significantly lower than the pre-test scores of the control group (t(37)=4.13, p<0.001, d=1.3). After the dialyses session and the 24h retention interval CKD─ patients stated a significant higher mood score than CKD+A and CKD+D (t(14.7)=2.63, p=0.019, d=1.2). Their post-test score increased significantly compared to the pre-test result (t(6)=2.62, p= 0.039, r=0.185, d=1.3) and reached the level of the mood of healthy controls. Pre-post changes occurred neither in the control group nor in the other CKD sub groups.
Figure 2.Mood state and changes in patients with chronic kidney disease (CKD). Subgroups (+D: with depression; +A with apathy only, -: without depression or apathy) and healthy controls who received no dialysis. A) A quality of life questionnaire served as measure of the prevailing (long-term) mood state. B) Short-term mood changes were assessed by means of a mood questionnaire sensitive for momentary affective state. Bars show group means and errors indicate ±1 SD. Group and pre-post differences that were significant at p<0.05 were labelled with *.
Discussion
In this study we tested how three different groups of CKD patients responded on a measure of short-term mood after a single session of haemodialysis. First, our findings suggest a high rate of apathetic patients (approx. two-third) among the CKD population. Second, a dichotomous distinction between patients with and without clinical relevant depression led to a sub group with symptoms of apathy only. These apathetic CKD patients showed the behavioural and cognitive-emotional symptoms of apathy, but lacked the somatic and cognitive-emotional symptoms of a prevailing depression, which clearly shows that patients were able to differentiate apathy and depression symptoms by means of self-report scales. Depressed mood (cognitive-emotional symptoms) turned out to be the best discriminator between patients with and without a depression. Suicidal thoughts, which were also suggested by former studies3, could not discriminate between CKD groups as well as between patients and healthy controls.
Limitations
Clearly, our sample size is small and not reliable to derive a prevalence rate in general. The study relied on CKD patients receiving haemodialysis treatment only. No conclusions can be thrown on CKD patients receiving other treatments like peritoneal dialysis, which is not limited to a 4-5 hour treatement thrice weekly but is usually done daily over many hours. Symptoms of depression were reported as less frequent and less severe in these patients20, 21. Furthermore, symptoms of apathy strongly overlap with symptoms accompanying a depression, such that all depressed patients scored high on the apathy scale. It seems that patients suffering from a depressive disorder discern their depressiveness also as apathy, when assessed with the AES. Suicidal thoughts could also not discriminate between depression and apathy. This is probably due to a methodological weakness of our subscale analysis where the dimension suicidal thoughts relies on one BDI item only.
To further explore apathy in CKD we focused on the differential effect of apathy and depression on the sustained mood state. CKDs’ quality of life was often reported as severely affected by an additional depression17. Reduced quality of life expresses sustained depressed mood as the prevailing affective state with high complaints, low social integration and coping. We identified a large effect for depression on quality of life measures with depressed CKD patients scoring 2.6 standard deviations below healthy controls. It confirms the tremendous deterioration of the mood state that depressed CKD patients often discern, and it should be a strong concern to treat clinical relevant depressions in patients with renal failure. But also CKD patients without depression and apathy reported restricted quality of life compared to healthy controls. The difference is also large in terms of an effect size (Cohen’s d=1.8), and it seems that CKD patients without apathy symptoms responded equally to the Quality of Life measure compared to patients with apathy. We suggest that the primary somatic complaints accompanying the disease and the dialysis procedure are most likely responsible for the reduced quality of life in these patients. Physicians can meet that challenge with the medical treatment of the renal disease. If patients develop depressive disorders then subjective health complaints and a loss of social integration become more prominent in CKD patients’ daily life. To treat these secondary complaints a psychotherapeutic intervention seems to be recommended18.
Apathetic CKD patients did not turn out to be at a higher risk than patients without apathy to develop further deteriorations in their prevailing mood state. Hence, in the second part of our study we tried to evaluate this outcome in terms of short-term mood changes. The momentary mood was assessed immediately before a dialysis session and 24 hours later after approximately one day of recovery. Here apathetic CKD patients showed a pattern comparable to that observed in patients with a clinical relevant depression. We could not show a short-term improvement in mood that was associated with the dialysis treatment in the apathetic CKD patients. In CKD patients without apathy we could demonstrated this possible effect. The mood of patients without apathy and depression improved nearly to the level of healthy controls. The effect size (within-subject d=1.3) indicated a large effect on our short-term mood measure.
The latter observation after all indicated apathetic patients as restricted as depressed patients. We argue that apathetic CKD patients are on risk to develop depressions later in the disease process. The absence of a (previously perceived) brightening effect could also mean a loss of positive reinforcement. Loss of positive reinforcement could negatively influence patients’ compliance and is a main factor in the aetiology of a major depression19. Further research is necessary to confirm such an assumption, because our study is small in numbers and restricted to a very short longitudinal observation interval. We could give only a first impression of the importance of the concept of apathy in patients with severe CKD.
Conclusion
Chronic kidney disease patients undergoing haemodialysis frequently show loss of motivation, cognitive impairments, and emotional distress without suffering from a depression. Apathy as a symptom of the renal disease seems to be attributable to this state. Patients with apathy are less impaired in quality of life than patients with depression, but lack a positive emotional reactivity to the dialyses treatment, which was observable in patients without apathy and depression. The clinical practice could consider apathy as a severe symptom in patients with end-stage renal failure. It is possible to discriminate between depression and apathy in those patients with the self-report questionnaires BDI and AES. Treating apathy in CKD could improve the adherence to the requirements of the dialysis treatment and omit the progression of the disease, probable cognitive impairments and an emotional distress.
References
- 1.Hedayati S S, Minhajuddin A T, Toto R D. (2009) Prevalence of major depressive episode in CKD. , Am J Kidney Dis 54, 424-432.
- 2.Hedayati S S, Minhajuddin A T, Toto R D. (2009) Validation of depression screening scales in patients with CKD. , Am J Kidney Dis 54, 433-439.
- 3.Kimmel P L, Peterson R A, Weihs K L. (2000) Multiple measurements of depression predict mortality in a longitudinal study of chronic hemodialysis patients. Kidney Int. 57, 2093-2098.
- 4.Hedayati S S, Minhajuddin A T, Afshar M. (2010) Association between major depressive episodes in patients with chronic kidney disease and initiation of dialysis, hospitalization, or death. , JAMA 303, 1946-1953.
- 5.Kojima M, Hayano J, Fukuta H. (2008) Loss of fractal heart rate dynamics in depressive hemodialysis patients. Psychosom Med. 70, 177-185.
- 6.Martiny C, AC De Oliveira e Silva, JPS Neto. (2001) Factors associated with risk of suicide in patients with hemodialysis. Compr Psychiatry. 52, 465-468.
- 7.Kurella M, Kimmel P L, Young B S. (2005) Suicide in the United States end-stage renal disease program. , J Am Soc Nephrol 16, 774-81.
- 8.Kutner N G, Brogan D, Hall W D. (2000) Functional impairment, depression, and life satisfaction among older hemodialysis patients and age-matched controls: a prospective study. , Am J Phys Med Rehabil 81, 453-459.
- 9.Kimmel P L, Peterson R A. (2006) Depression in patients with end-stage renal disease treated with dialysis: has the time to treat arrived?. , Clin J Am Soc Nephrol 1, 349-352.
- 10.Chilcot J, Norton S, Wellsted D. (2011) A confirmatory factor analysis of the Beck Depression Inventory-II in end-stage renal disease patients. , J Psychosom Res 71, 148-153.
- 11.Lueken U, Seidl U, Schwarz M. (2000) Psychometric properties of a German version of the Apathy Evaluation Scale]. Advances in Neurology-Psychiatry. 74, 714-722.
- 12.Beck A T, Ward C H, Mendelson M. (1961) An inventory for measuring depression. Arch Gen Psychiatry. 4, 561-571.
- 13.Craven J L, Rodin G M, Littlefield C. (1988) The Beck Depression Inventory as a screening device for major depression in renal dialysis patients. , Int J Psych Med 18, 365-374.
- 14.Keller F, Kempf W. (1993) Stability and validity of latent classes within the Beck depression inventory (BDI)]. Psychiatric practice. 20, 68-71.
- 15.Averbeck M, Leiberich P, M T Grote-Kusch, Olbrich E, Schroeder A et al.. K.(1997). Scales of Assessment of Quality of Life (SEL)]. Harcourt Test Services: , Frankfurt .
- 16.Steyer R, Schwenkmezger P, Notz P, Eid M. (1997) MDBF - Multi-dimensional mood questionnaire]. Hogrefe: Goettingen;.
- 17.Bárány P, Pettersson E, Konarski-Svensson J K. (1993) Long-term effects on quality of life in haemodialysis patients of correction of anaemia with erythropoietin. Nephrol Dial Transplant. 8, 426-432.
- 18.Cukor D, Ver Halen N, Asher D R. (2014) Psychosocial Intervention Improves Depression, Quality of Life, and Fluid Adherence in Hemodialysis. J Am Soc Nephrol. 25, 196-206.
- 19.Der-Avakian A, Markou A. (2012) The neurobiology of anhedonia and other reward-related deficits. Trends Neurosci. 35(1), 68-77.
- 20.Wuerth D, Finkelstein S H, Ciarcia J, Peterson R, Kliger A et al. (2001) Identification and treatment of depression in a cohort of patients maintained on chronic peritoneal dialysis. Am J Kidney Dis. 37(5), 1011-1017.
- 21.Finkelstein F O, Finkelstein S H. (2000) Depression in chronic dialysis patients: Assessment and treatment. Nephrol Dial Transplant. 15, 1911-1913.
- 22.Marin R S. (1990) Differential diagnosis and classification of apathy. , Am J Psychiatry 147(1), 22-30.
- 23.Marin R S. (1991) Apathy: a neuropsychiatric syndrome. , J Neuropsychiatry Clin Neurosci 3(3), 243-254.
- 24.Marin R S, Firinciogullari S, Biedrzycki R C. (1993) The source of convergence between measures of apathy and depression. J Affect Disord. 28(1), 7-14.
- 25.Starkstein S E, Ingram L, Garau M L, Mizrahi R.On the overlap between apathy and depression in dementia. , J Neurol, Neurosurg. & Psychiat 76(8), 1070-1074.
- 26.Marin R S, Wilkosz P A.Disorders of Diminished Motivation Focus on Clinical Practice and Research. , J Head Trauma Rehabil 20(4), 377-388.
- 27.Castellon S A, Hardy D J, Hinkin C H, Satz P, Stenquist P K et al. (2006) Components of depression in HIV-1 infection: their differential relationship to neurocognitive performance. , J Clin & Exp Neuropsych 28(3), 420-437.
- 28.Rabkin J G, Ferrando S J, W van Gorp, Rieppi R, McElhiney M et al. (2000) Relationships Among Apathy, Depression, and Cognitive Impairment in HIV/AIDS. , J Neuropsychia & Clin Neurosc 12(4), 451-457.
Cited by (2)
- 1.Matsunaga Yusuke, Takahashi Hiroki, Suzuki Yuta, Yamamoto Shohei, Imamura Keigo, et al, 2022, Relationship between psychiatric symptoms and activities of daily living in patients undergoing hemodialysis, Renal Replacement Therapy, 8(1), 10.1186/s41100-022-00425-4
- 2.Voorend Carlijn G.N., van Buren Marjolijn, Berkhout-Byrne Noeleen C., Kerckhoffs Angèle P.M., van Oevelen Mathijs, et al, 2024, Apathy Symptoms, Physical and Cognitive Function, Health-Related Quality of Life, and Mortality in Older Patients With CKD: A Longitudinal Observational Study, American Journal of Kidney Diseases, 83(2), 162, 10.1053/j.ajkd.2023.07.021
