Serum Vitamin D Level in Oral Lichen Planus Patients of North India- A Case-Control Study.
Abstract
Background:
Studies have unveiled lower levels of serum vitamin D in autoimmune diseases and the role of vitamin D in inhibition of helper T cells proliferation, stimulation of regulatory T cells, diminishing of B-lymphocyte differentiation and inhibition of immunoglobulin secretion has been discussed in the literature. Oral lichen planus (OLP) is an autoimmune mucocutaneous disorder in which cell mediated immunity plays an important part and so it may also be related to serum vitamin D levels.
Aim:
To estimate serum vitamin D in oral lichen planus patients of North India, compare it to the controls and to evaluate various factors influencing vitamin D level in OLP cases such as sun exposure, gender, psychosocial factors, meteorological influences, religion and diet.
Methods and Material:
Venous blood samples of 102 clinically diagnosed oral lichen planus patients and 102 age and sex matched controls were obtained for the study and serum vitamin D levels were estimated using Vitek Immunodiagnostic Assay System, an automated bench-top immuno-analyzer, based on the Enzyme Linked Fluorescent Assay. The data was analyzed using chi-squared test and t-test.
Results:
Mean vitamin D in OLP cases and controls was 20.40 ng/ml and 32.67 ng/ml respectively. Vitamin D3 deficiency was more in OLP cases (70.6%) as compared to controls (34.3%) and insufficiency was more in controls (35.3%) as compared to OLP cases (16.7%). These differences were statistically significant.
Conclusion:
Although vitamin D deficiency was found more in OLP cases pointing towards the possible co-relation of vitamin D and OLP, yet insufficiency was seen more commonly in controls which revealed that this important vitamin was lacking in North Indian population.
Author Contributions
Academic Editor: Anand Rotte, Chronobiology and Sleep Group, Brain and Mind research Institute, the University of Sydney
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Akanksha Gupta, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Oral Lichen planus (OLP) is a chronic, autoimmune, mucocutaneous, psychosocial disease. It is common in middle aged females and clinically present in various forms including reticular, papular, plaque-like, atrophic, erosive and bullous types.1,2 It was described by Erasmus Wilson in 1869.3 In general population, the prevalence of OLP is about 0.5%-2.2%.4World Health Organization (WHO) classifies OLP as a “potentially malignant disorder” with unspecified malignant transformation risk, so OLP patients should stay under close observation.5
OLP probably results from an immunologically induced degeneration of basal layer and is characterized by cytotoxic CD8+ cell response on modified keratinocytes surface antigen. Langerhans cells are increased in OLP lesions and MHC class II expression is upregulated. Langerhans cells probably mediate the MHC class II antigen presentation in OLP. There is MHC class II antigen presentation to CD4+ helper T-cells, followed by keratinocyte apoptosis triggered by CD8+ cytotoxic T-cells. 6,7
Active vitamin D or 1,25-dihydroxycholecalciferol plays a vital role in calcium and phosphorus metabolism. Many studies have reported a role of vitamin D in the regulation of immune responses as well. Proliferation of helper T cells is inhibited, regulatory T cells is stimulated, B-lymphocyte differentiation is diminished and immunoglobulin secretion is inhibited by vitamin D. It also restricts antigen presentation via macrophages and modulates dendritic cells maturation.8
Studies have unraveled lower levels of 25(OH) vitamin D in autoimmune diseases such as Behcet’s disease, Diabetes Mellitus (DM), inflammatory bowel disease, rheumatoid arthritis (RA), Multiple sclerosis, Systemic lupus erythematosus (SLE) and Multiple sclerosis (MS) as compared with healthy individuals.8,9,10,11
Faezi et al (2014) did a case control study and found that about 57% and 17% of Behcet’s patients had vitamin D deficiency and insufficiency respectively.8 Fakharan et all evaluated the relationship between serum level of vitamin D and RA disease activity score. The mean serum levels of vitamin D in high activity group (17.057±7.7 mg/ml) was significantly less than moderate (30.5±11.3 mg/ml) and low (36.7±19.5 mg/ml) activity groups (P<0.001). The outcome of this study showed that serum level of vitamin D is inversely correlated with the activity of RA.9 Ibrahim et al (2014)10 studied the prevalence of vitamin D deficiency among newly diagnosed multiple sclerosis patients. Low total vitamin D level was seen among 65% of patients with MS (13/20); however, this was only 20% of normal controls (4/20). They concluded that hypovitaminosis D was common in MS patients. An analysis of data from the Nurses’ Health Study and Nurses’ Health Study II observed that the risk of developing MS was significantly reduced for women taking ≥400 international units/day of vitamin D (relative risk 0.59, 95% CI 0.38 - 0.91). Schoindre et al (2014)11 studied the relationship between 25(OH)D levels and Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) score, and assessed for the first time the role of vitamin D in predicting SLE flare-ups. They found a low vitamin D status in the majority of patients with SLE, and a modest association between lower 25(OH)D levels and high disease activity. Varma et al (2014) reported a case of a patient who had patient with concurrent vitamin D deficiency and lichen planus both of which showed improvement after vitamin D therapy.12
These studies indicate that vitamin D deficiency is probably contributing to the increase in autoimmune disorders.
Due to increasing prevalence of oral lichen planus and the risk of malignant transformation to oral squamous cell carcinoma (OSCC), we must endeavor to investigate various factors that are associated with OLP.
As several studies have shown significant role of vitamin D in the autoimmune diseases, this study is an effort to evaluate vitamin D levels in patients with OLP.
Materials and Method
The study was conducted in our institute over a period of 2 years. The study protocol was approved by the institutional ethical committee (SDC/CER/2015). All patients enrolled in the study group and the control group submitted informed consent prior to taking part in this investigation. Sample size included 102 patients who were clinically diagnosed with OLP. Serum vitamin D levels were evaluated in the patients with OLP and the levels were compared with 102 controls. Control group included age and sex matched healthy volunteers from the patient population of our college. Cases and controls were from similar social classes, with similar nutritional habits. Serum vitamin D estimation of a selected control was done as soon as the estimation was done in the case to prevent bias due to meteorological influence.1
The clinical diagnostic criteria for oral lesions used in this study was- 1. The presence of keratotic, pinhead-sized, white, slightly elevated papules (papular lichen planus), which may be discrete or arranged in reticular (reticular lichen planus) or plaque-like (plaque-like lichen planus) configurations. 2. Atrophic lichen planus, characterized by thinning of the epithelium leading to the appearance of atrophic red areas within the white lesions. 3. Erosive (ulcerative) lichen planus, characterized by areas of well-defined ulceration within the above mentioned lesions. 4. Bullous lichen planus, characterized by the presence or development of bullous areas within the above mentioned lesions.1,2Representative images of OLP are shown in Figure 1 and Figure 2.
Figure 1.(A) Reticular OLP on right buccal mucosa; (B) Reticular OLP on left buccal mucosa; (C) Erosive and papular OLP on right buccal mucosa and vestibule; (D) Bullous OLP on right buccal mucosa;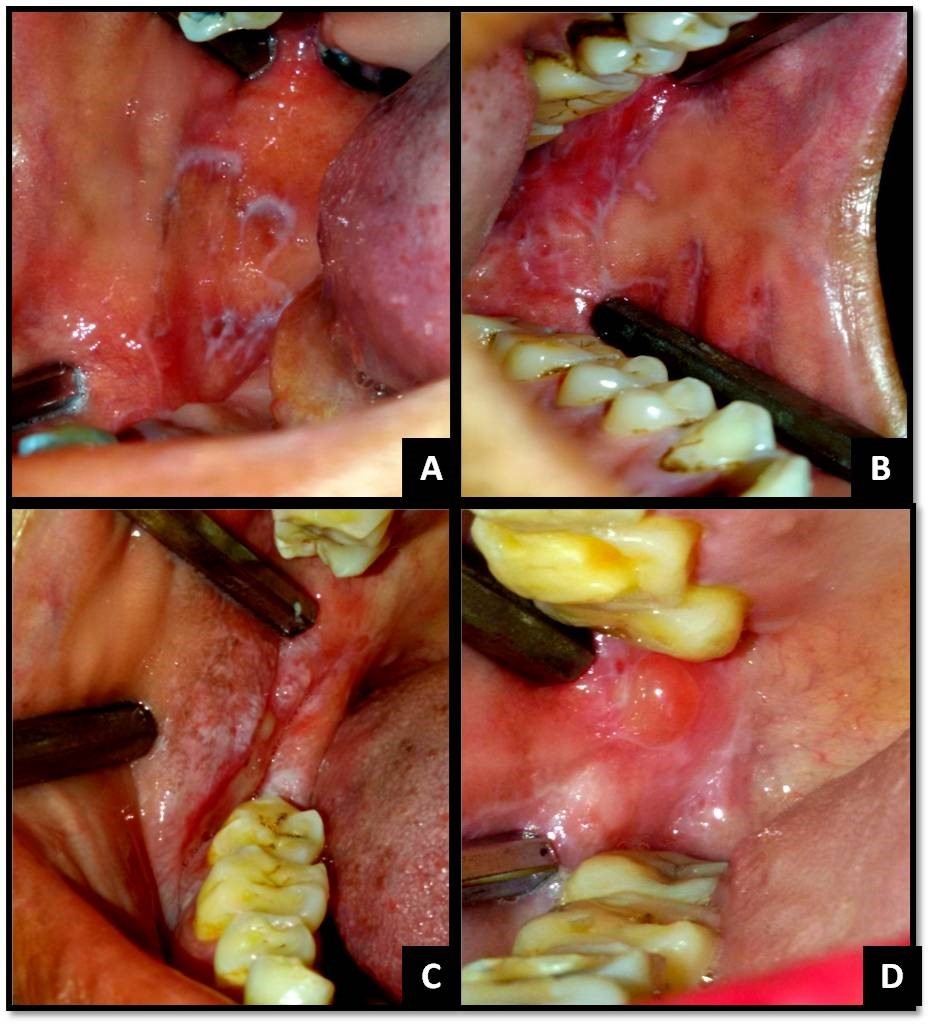
Figure 2.(A) Papular and plaque-like OLP on dorsal aspect of tongue; (B) Reticular and erosive OLP on the palate; (C) Reticular OLP on gingiva with desquamative gingivitis; (D) Reticular and erosive OLP on the tongue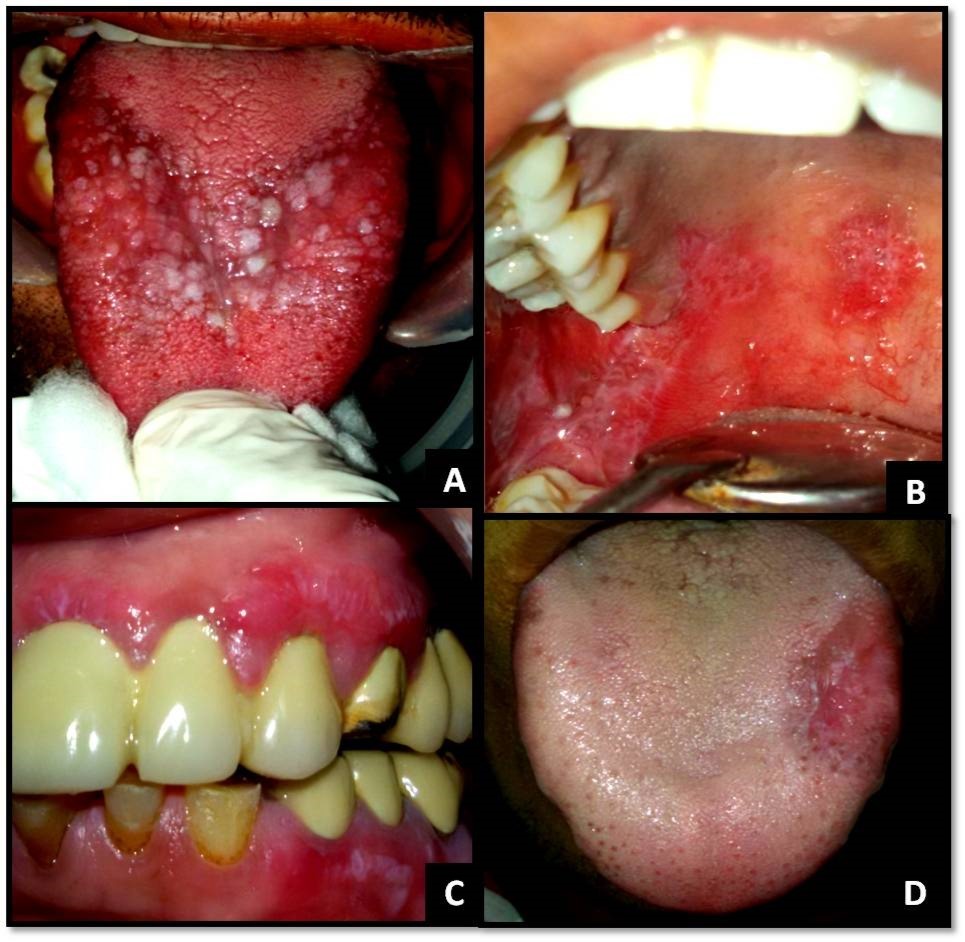
Exclusion criteria included subjects with history of taking any kind of vitamin D supplements, any kind of corticosteroids or cytotoxic drugs, anti-malarial drugs and anti-hypertensive drugs, patients with the habit of betel nut or smokeless tobacco, patients having bilateral amalgam restorations, patients with history of chronic disease including renal or hepatic diseases, bone metabolic disease, malabsorption, type 1 diabetes mellitus, malignancies,8 thyroid or parathyroid disease, hyperparathyroidism, pregnant females.9Controls with history of vitamin D, Multivitamin or any drug affecting bone metabolism (such as phenobarbital or phenytoin) in the last 6 months were excluded8(Flowchart 1).
There was a standard proforma and the elements to evaluate during the questionnaire included demographic features, general status of the patient, disease duration, clinical manifestations, Depression Anxiety and Stress Scales (DASS 21 scale) for psychological evaluation13 and Kuppuswamy’s socioeconomic status scale.14,15
Patients with oral lichen planus who agreed on the terms of the study were referred to the hospital, where the venous blood sample was taken and sent to the Biochemistry department for estimation of vitamin D levels.
Vitamin D measurement was performed using Vitek Immunodiagnostic Assay System (bioMérieux, Marcy-l'Étoile, France), an automated bench-top immuno-analyzer, based on enzyme-linked fluorescent assay (ELFA) technology.
Vitamin D levels lower than 20ng/ml were ascribed to vitamin D deficiency category; 20-30 ng/ml were ascribed to vitamin-D insufficiency category; 30-100 ng/ml were categorized as vitamin-D sufficiency and vitamin D more than 100 ng/ml were categorized as vitamin-D toxicity.10
Statistical analysis of the data was performed using the Statistical Package for Social Sciences (ver. 17.0; SPSS, Inc., Chicago, IL, USA). The t-test and chi square test were used for non-parametric values. A probability (P) value of <0.05 was considered to be significant and P < 0.001 was considered to be highly significant.
Assessment of Immune Profile
Blood T-cell levels were assessed in twenty LP cases who did not take any medication for their treatment and sign and symptoms did not subside, twenty controls and twenty LP patients who took vitamin D supplements. Five milliliters of antecubital vein blood was drawn into an EDTA-containing test tube. The complete blood count was assessed. Then the blood T-cell levels were measured by means of two-color flow cytometry using Partec PAS (Partec, Munster, Germany). Conjugated antibodies with fluorescent material were added to the peripheral blood and incubated for 30 minutes in 4ºC. Then blood cells were incubated with a lysing agent (Uti- Lyse solution number 1 and 2, DAKO Corporation, Denmark) to remove erythrocytes. Lymphocyte phenotype analysis was performed using monoclonal antibodies (DAKO, Denmark) to distinguish cells expressing CD3+, CD4+, and CD8+ antigens. Cells passed the laser beam and scattered the laser light which was detected by the cytometry.
Result
The results were analyzed for 102 (36 males i.e 35.3% and 66 females i.e. 64.7%) age and sex matched OLP cases and controls. Mean age of OLP cases was 37.12 years. The mean duration of symptoms of our study group was 9.32 months. Patient characteristics are shown in Table 1. Statistical analysis was rigorously performed and statistically significant values are mentioned.
Table 1. Showing Patient Characteristics.| Patient characteristics | Percentage of OLP cases |
|---|---|
| Age range (years)15-2425-3435-4445-54≥55 | 20.5823.5223.5218.6213.72 |
| GenderMaleFemale | 35.364.7 |
| Duration of sun exposure (minutes)<2020-6060-120>120 | 5.437.330.427 |
| ResidenceUrbanSub-urbanRural | 47.535.816.7 |
| ReligionHinduMuslim | 70.629.4 |
| DietVegetarianNon-vegetarian | 65.734.3 |
| Socio-economic statusMiddle/lower middle classUpper middle classUpper lower classUpper classLower class | 54.427.511.34.92 |
| Psychological stateDepressionNormalMildModerateSevereVery severeAnxietyNormalMildModerateSevereVery severeStressNormalMildModerateSevereVery severe | 14.727.523.526.57.86.921.632.424.514.75.929.422.532.49.8 |
Serum Vitamin D3 Levels in OLP Cases and Controls
Mean vitamin D in OLP cases and controls was 20.40 ng/ml and 32.67 ng/ml respectively. Vitamin D3 deficiency was more in OLP cases (70.6%) as compared to controls (34.3%), insufficiency was more in controls (35.3%) as compared to OLP cases (16.7%). There were more number of controls with vitamin D sufficiency (26.5%) as compared to OLP cases (8.8%). These differences were statistically significant(P value = 0.000). Equal number of OLP cases as well as controls (3.9%) had vitamin D toxicityTable 2
Table 2. Showing distribution of cases and controls according to their serum vitamin D levels.| Group | Total | ||||
| Cases | Controls | ||||
| Vitamin D group(ng/ml) | <20 (Deficient) | Count | 72 | 35 | 107 |
| % within Group | 70.6% | 34.3% | 52.5% | ||
| 20-30 (Insufficient) | Count | 17 | 36 | 53 | |
| % within Group | 16.7% | 35.3% | 26.0% | ||
| 30-100 (Sufficient) | Count | 9 | 27 | 36 | |
| % within Group | 8.8% | 26.5% | 17.6% | ||
| >100 (Toxicity) | Count | 4 | 4 | 8 | |
| % within Group | 3.9% | 3.9% | 3.9% | ||
| Total | Count | 102 | 102 | 204 | |
| % within Group | 100.0% | 100.0% | 100.0% | ||
Gender of OLP Cases and Vitamin D Levels
More number of female patients (77.3% ) had vitamin D deficiency as compared to male patients (58.3%). Vitamin D insufficiency was more in males (27.8%) as compared to females (10.6%). Vitamin D sufficiency was in 9.1% females and 8.3% males. Vitamin D toxicity was seen in 3% females and 5.6% males.
Season in which the OLP Cases Reported and Vitamin D levels
90% of patients who reported in winters were vitamin D deficient and 10% suffered from insufficiency. 83.3% who reported in pre-monsoon were deficient, 11.1% had vitamin D sufficiency and 5.6% had toxicity. 73.5% patients who reported in summer monsoon were deficient, 14.7% suffered from insufficiency, 8.8% had vitamin D sufficiency and 2.9% had toxicity. 46.7% OLP cases who reported in post monsoon were deficient, 33.3% had insufficiency, 13.3% had sufficiency and 6.7% had toxicity. These differences were statistically significant (P value= 0.05).
Duration of Sun Exposure in OLP Cases and Vitamin D Levels
Among those patients who had a sun exposure of less than 20 minutes, 60% were vitamin D deficient and 40% suffered from insufficiency. Among those patients who had a sun exposure between 20 to 60 minutes, 68.4% were deficient, 7.9% had insufficiency, 21.1% had sufficiency and 2.6% had toxicity. Among those patients who had a sun exposure between 60 to 120 minutes, 80.6% were deficient, 16.1% had insufficiency and 3.2% had toxicity. Among those patients who had a sun exposure more than 120 minutes, 64.3% were deficient, 25% had insufficiency, 3.6% had sufficiency and 7.1% had toxicity.
OLP Cases from Rural, Sub-Urban and Urban Areas and Vitamin D Levels
Among OLP cases from rural areas 60% were vitamin D deficient, 30% suffered from insufficiency and 10% had sufficiency. Among the OLP cases from sub-urban areas, 68.3% had deficiency, 22% had insufficiency, 4.9% had sufficiency and 4.9% had toxicity. Among the OLP cases from urban areas, 78% had deficiency, 4.9% had insufficiency, 12.2% had sufficiency and 4.9% had toxicity.
Religion of OLP Cases and Vitamin D Levels
Among Hindu patients, 72.2% were vitamin D deficient, 12.5% had insufficiency, 9.7% had sufficiency and 5.6% had toxicity. Among Muslim OLP cases, 66.7% were deficient, 26.7% had insufficiency and 6.7% had sufficiency.
Diet of OLP Cases and Vitamin D Levels
Among non-vegetarian OLP cases, 68.6% were vitamin D deficient, 22.9% had insufficiency and 8.6% had sufficiency. Among vegetarian OLP cases, 71.6% had deficiency, 13.4% had insufficiency, 9% had sufficiency and 6% had toxicity.
Socio-Economic Status of OLP Cases and Vitamin D Levels
Among the upper class patients, 80% had vitamin D deficiency and 20% had insufficiency. Among the upper-middle class patients, 71.4% had deficiency, 17.9% has insufficiency and 10.7% had sufficiency. Among middle/lower-middle class OLP cases, 70.9% were deficient, 14.5% had insufficiency, 7.3% had sufficiency and 7.3% had toxicity. Among upper lower class, 75% had deficiency, 8.3% had insufficiency and 16.7% had sufficiency. Among lower class patients, 100% patients had vitamin D insufficiencyTable 3
Table 3. Correlation of serum vitamin D in cases with their socio-economic status.| Group | Vitamin D | Total | ||||||
|---|---|---|---|---|---|---|---|---|
| <20 (Deficient) | 20-30 (Insufficient) | 30-100 (Sufficient) | >100 (Toxicity) | |||||
| Cases | Socio-EconomicStatus | Upper | Count | 4 | 1 | 0 | 0 | 5 |
| % | 80.0% | 20.0% | .0% | .0% | 100.0% | |||
| Upper middle | Count | 20 | 5 | 3 | 0 | 28 | ||
| % | 71.4% | 17.9% | 10.7% | .0% | 100.0% | |||
| Middle/Lower middle | Count | 39 | 8 | 4 | 4 | 55 | ||
| % | 70.9% | 14.5% | 7.3% | 7.3% | 100.0% | |||
| Lower/upper lower | Count | 9 | 1 | 2 | 0 | 12 | ||
| % | 75.0% | 8.3% | 16.7% | .0% | 100.0% | |||
| Lower | Count | 0 | 2 | 0 | 0 | 2 | ||
| % | .0% | 100.0% | .0% | .0% | 100.0% | |||
| Total | Count% | 7270.6% | 1716.7% | 9 8.8% | 43.9% | 102100 | ||
Psychological State of Cases and Vitamin D Levels
Relationship of psychological factors with serum vitamin D has been demonstrated in Table 4, Table 5, Table 6.
Table 4. Correlation of serum vitamin D of cases with depression.| Group | Vitamin D | Total | ||||||
|---|---|---|---|---|---|---|---|---|
| <20 (Deficient) | 20-30 (Insufficient) | 30-100 (Sufficient) | >100 (Toxicity) | |||||
| Cases | Depression | Normal | Count | 11 | 3 | 1 | 0 | 15 |
| % | 73.3% | 20.0% | 6.7% | .0% | 100.0% | |||
| Mild | Count | 22 | 4 | 1 | 1 | 28 | ||
| % | 78.6% | 14.3% | 3.6% | 3.6% | 100.0% | |||
| Moderate | Count | 17 | 3 | 3 | 1 | 24 | ||
| % | 70.8% | 12.5% | 12.5% | 4.2% | 100.0% | |||
| Severe | Count | 17 | 5 | 3 | 2 | 27 | ||
| % | 63.0% | 18.5% | 11.1% | 7.4% | 100.0% | |||
| Very severe | Count | 5 | 2 | 1 | 0 | 8 | ||
| % | 62.5% | 25.0% | 12.5% | .0% | 100.0% | |||
| Total | Count | 72 | 17 | 9 | 4 | 102 | ||
| % | 70.6% | 16.7% | 8.8% | 3.9% | 100.0% | |||
| Group | Vitamin D | ||||||
|---|---|---|---|---|---|---|---|
| <20 (Deficient) | 20-30 (Insufficient) | 30-100 (Sufficient) | >100 (Toxicity) | Total | |||
| Anxiety | Normal | Count | 5 | 1 | 1 | 0 | 7 |
| % | 71.4% | 14.3% | 14.3% | .0% | 100.0% | ||
| Mild | Count | 17 | 2 | 2 | 1 | 22 | |
| % | 77.3% | 9.1% | 9.1% | 4.5% | 100.0% | ||
| Moderate | Count | 25 | 6 | 1 | 1 | 33 | |
| % | 75.8% | 18.2% | 3.0% | 3.0% | 100.0% | ||
| Severe | Count | 17 | 5 | 2 | 1 | 25 | |
| % | 68.0% | 20.0% | 8.0% | 4.0% | 100.0% | ||
| Very severe | Count | 8 | 3 | 3 | 1 | 15 | |
| % | 53.3% | 20.0% | 20.0% | 6.7% | 100.0% | ||
| Total | Count | 72 | 17 | 9 | 4 | 102 | |
| % | 70.6% | 16.7% | 8.8% | 3.9% | 100.0% | ||
| Vitamin D | Total | |||||||
| <20 (Deficient) | 20-30 (Insufficient) | 30-100 (Sufficient) | >100 (Toxicity) | |||||
| Cases | Stress | Normal | Count | 3 | 2 | 1 | 0 | 6 |
| % | 50.0% | 33.3% | 16.7% | .0% | 100.0% | |||
| Mild | Count | 19 | 9 | 1 | 1 | 30 | ||
| % | 63.3% | 30.0% | 3.3% | 3.3% | 100.0% | |||
| Moderate | Count | 15 | 2 | 4 | 2 | 23 | ||
| % | 65.2% | 8.7% | 17.4% | 8.7% | 100.0% | |||
| Severe | Count | 29 | 2 | 1 | 1 | 33 | ||
| % | 87.9% | 6.1% | 3.0% | 3.0% | 100.0% | |||
| Very severe | Count | 6 | 2 | 2 | 0 | 10 | ||
| % | 60.0% | 20.0% | 20.0% | .0% | 100.0% | |||
| Total | Count | 72 | 17 | 9 | 4 | 102 | ||
| % | 70.6% | 16.7% | 8.8% | 3.9% | 100.0% | |||
Difference in Severity of Symptoms Inside Deficiency and Insufficiency Groups
52.77% of vitamin D deficient OLP cases suffered from erosive and atrophic lichen planus which was associated with severe pain and burning sensation. This percentage was reduced to 47.05% in OLPcases suffering from vitamin D insufficiency.
T-Lymphocyte Subsets in Patients with Oral Lichen Planus
Mean percentage of CD4+, CD8+, CD3+ was 40.74, 25.04 and 66.39 respectively.
T-Lymphocyte Subsets in Controls
Mean percentage of CD4+, CD8+, CD3+ was 42.84, 24.22 and 67.25 respectively.
T-Lymphocyte Subsets in Patients with Oral Lichen Planus who took vitamin D Supplements
Mean percentage of CD4+, CD8+, CD3+ was 40.95, 24.04 and 66.92 respectively.
Discussion
Vitamin D deficiency has been reported worldwide, even in countries with low latitude, where it was thought that UVB radiation was adequate enough to prevent vitamin D deficiency.16 Vitamin D deficiency prevails in epidemic proportions all over the Indian subcontinent, with a prevalence of 70%–100% in the general population.17
Innumerable studies have thrown light upon the immunoregulatory role of vitamin D and its deficiency among patients with autoimmune disorders. Therefore, in patients with immunologic aberrancies, vitamin D deficiency should be comprehensively addressed. Our study is an endeavour to evaluate serum vitamin D levels and elucidate its significance in oral lichen planus.
There is agreement that circulating serum 25(OH)D or calcidiol levels (a summation of D3 and D2 forms) are currently the best available indicator of the net incoming contributions from cutaneous synthesis and total intake (foods and supplements).18,19 Therefore, in our study calcidiol levels were estimated in 102 cases of OLP and 102 age and sex matched controls using VIDAS.
Mean vitamin D levels were found to be significantly lower in patients with OLP than the disease free controls. Our results were consistent with other studies that unveiled lower levels of vitamin D in autoimmune diseases.8,9,10,11,12 Although vitamin D deficiency was found more in OLP cases, yet insufficiency was seen more commonly in controls which revealed that this important vitamin was lacking in North Indian population.
Epidemiological studies in humans and experimental studies in mice have deciphered that UVR exposure can suppress the initiation and development of immune-driven models of disease.20 More than 80% of vitamin D3 is obtained by exposure of skin to UVR in sunlight. Solar ultraviolet B photons are absorbed by 7-dehydrocholesterol in the skin, leading to its transformation to previtamin D3, which is rapidly converted to vitamin D3. Once formed, vitamin D3 is metabolized in the liver to 25-hydroxyvitamin D3 and then in the kidney to its biologically active form, 1,25 dihydroxyvitamin D3.20,21 Although chronic excessive exposure to sunlight can increase the risk of skin cancer, the avoidance of all direct sun exposure increases the risk of vitamin D deficiency, which can have serious repercussions.21There are many consequences of 1,25(OH)2D3 on the activity of immune cells but generally they have been defined as the stimulation of innate immunity and suppression of adaptive immunity.20The typical recommendation for “modest” UV exposure is 5–30 minutes exposure to legs and arms three times a week, depending on skin type, location, and season.22 In our study, we also evaluated the average duration of sun exposure per day in the patients of lichen planus and its co-relation with serum vitamin D. We found that increased duration of sun exposure from less than 20 minutes to more than 120 minutes neither significantly reduced the number of lichen planus cases nor increased the serum vitamin D level.
In our study, OLP was seen more in females. Also, more number of female patients had vitamin D deficiency as compared to male patients. This could be the result of cultural and social taboos that often dictate lifestyle patterns such as conservative clothing that may limit sun exposure in females, confinement to family and home and lack of social activities resulting in decreased vitamin D synthesis.
In our study, maximum number of patients reported in summer monsoon season and 73.5% of them had vitamin D deficiency. Minimum number of patients reported in pre-monsoon season and 83.3% of them were vitamin D deficient. Slightly more number of patients reported in winters and 90% of them had vitamin D deficiency. In a study conducted by Mohan et al (2013), maximum patients suffering from OLP reported in summers and minimum in winters.1 A number of studies have examined serum 25(OH)D levels in different seasons. Rapuri et al. (2002) examined women in Omaha and reported mean serum 25(OH)D levels of 68 nmol/L in February and 86 nmol/L in August.23 Gozdik et al. (2009) found that in winter, serum 25(OH)D levels of individuals with East and South Asian backgrounds were significantly lower than those of individuals with European ancestry.24 In the light of previously reported results in literature and our own study, it could be said that although serum vitamin D levels go down in winter in North Indian population yet maximum cases of OLP are reported in summers when serum vitamin D is comparatively high but the possibility of a bias cannot be ruled out as there is a general decrease in OPD in the dental college and hospital during winter due to extreme weather conditions.
In our study, maximum (47.5%) patients belonged to urban areas and 78% of them had vitamin D deficiency; minimum (16.7%) belonged to rural areas and 60% of them were vitamin D deficient. In OLP, psychogenic factors seem to play an important role and increased level of depression, anxiety and stress has been reported in urban areas due to increased stressors and factors such as overcrowded and polluted environment, high levels of violence, and reduced social support which can result in development of more cases of OLP.25,26 The cities are densely populated and people have to live in overcrowded apartments, which are closely packed and many stories high. As a result, direct sunlight does not reach inside most parts of the living quarters, thereby disallowing any sun exposure to an individual in the privacy of one’s home. Moreover, there are limited options for outdoor activities due to lack of space. Additionally atmospheric pollution in the urban areas reduces solar penetration resulting in decreased synthesis of serum vitamin D and thus making an individual prone to autoimmune disorders. Although less than urban population, yet vitamin D deficiency was seen in rural population as well and can be attributed to the phytate rich diet of the villagers in North India. Diet among the socio-economically lower classes consists of phytate as cereals and legumes which are more affordable and easily available than vegetables, milk and other dairy products. Besides, they are sources of protein for the vegetarians. Phytate is the principal storage form of phosphorus in many plant tissues, especially the bran portion of grains and other seeds and cannot be digested. Phytates chelate micronutrients such as calcium and iron, and thus reduce intestinal absorption of these nutrients. The studies in rat models have shown that a low-calcium diet or a high-phytate diet resulted in increased catabolism of 25(OH)D concentrations which leads to the formation of inactive metabolites and results in reduced 25(OH)D concentrations.27
OLP and vitamin D deficiency was more prevalent in vegetarians than non-vegetarians. Most of the food items rich in vitamin D are of animal origin. According to Taylor et al (2014), taking into account the serum 25(OH)D content of beef, pork, chicken, turkey and eggs, they can increase the estimated level of vitamin D in the food from two to eighteen times.28 However, vitamin D deficiency cannot be attributed to the diet itself as there are a number of factors that influence vitamin D.
In our study more number of Hindus were affected by OLP (with vitamin D deficiency in 72.2% of the patients) than Muslims. Among all the Hindu patients in our study, 90% were vegetarians and thus, vitamin D deficiency in our Hindu patients can be attributed to their eating habits. However religion itself cannot influence vitamin D levels.
We found that the majority of OLP patients who reported in our institute, were from the middle/lower middle socio-economic classes and least number of OLP patients reported from lower class (minimum) and upper class. Our result was consistent with the study conducted by Mohan et al1 and the study of Ingafou et al.,29 who concluded that distribution of patients across the socio-economic groups was not equal, ranging from 2.3% of employers or managers to 20% of semi-skilled manual workers. The social and physical conditions of a community, influences the health of its residents. Neighborhoods with low socio-economic status are commonly positioned where options for nutritional variety and food quality are limited.30 Oily fish, mushrooms, and egg yolks, are the sources of food with naturally occurring vitamin D31 but these food items are atypical and can be tougher to access among persons with income limitations and low socio-economic status. Even in individuals belonging to upper class, there are a number of factors that can lead to vitamin D deficiency such as higher usage of sunscreen and umbrella, less time spent on outdoor physical activity and greater indulgence in indoor activities like watching television and other recreational activities. This is in keeping with the fact that the main source of vitamin D is that produced by the action of solar UV B radiation acting on 7-dehydrocholesterol in skin and only small amounts are obtained from dietary sources. Also, it has been proposed that higher body fat leads to increased sequestration of vitamin D in adipose tissue, resulting in lower serum vitamin D levels,32 and in Indian population, upper class individuals have higher body fat as compared to those of lower class.33 In our study, there is possibility of a bias in this aspect. Least number of patients belonged to lower and upper class probably because people from the lower class ignore their health and avoid visiting a doctor. People from the upper class generally prefer a private clinic according to their convenience.
It has long been documented that psychological factors can trigger and exacerbate lichen planus.34 In our study, only 14.7% patients had normal depression, 6.9% patients had normal anxiety and 5.9% patients had normal stress. All the other patients suffered from varying degree of depression, anxiety and stress. Berk et al. (2007) reported that vitamin D deficiency may play a role in depression and possibly other mental disorders.35 Seasonal affective disorder which is characterized by depression, hypomania or mania is prevalent during winter season when serum vitamin D is deficient.36 Armstrong et al. (2007) concluded that vitamin D deficiency is common in fibromyalgia and occurs more frequently in patients with anxiety and depression.37 Chronic stress can lead to increased cortisol production from the adrenal glands which can lead to decreased expression of vitamin D receptor and thus decreased uptake, or activation, of vitamin D.38 So there appears to be a co-relation between psychological factors, vitamin D deficiency and OLP.
Mean percentage of CD3+ and CD4+ T cells was slightly lower in patients with OLP in comparison to the controls group; however, the mean percentage of CD8+ T cells was slightly higher in OLP subjects in contrast to controls. According to Eversole,39 the decreased lymphocyte count in LP patients in the peripheral blood is mainly related to the migration of these cells from the blood vessels and infiltration of the basal skin/mucous membrane layer. Mean percentage of CD4+ and CD3+ cells was slightly higher in LP patients who showed improvement in sign and symptoms after receiving vitamin D supplements.
In our study, very less number of cases had extraoral involvement and majority of them did not have sufficient serum vitamin D. There was slight difference in severity of symptoms inside deficiency and insufficiency groups. More number of vitamin D deficient cases suffered from erosive and atrophic lichen planus associated with severe pain and burning than vitamin D insufficient cases pointing towards the possible correlation of vitamin D levels with severity of symptoms. Regardless of the site and type of lichen planus inside the oral cavity, maximum number of patients required vitamin D supplements to make up for the lack of this vital vitamin in their body.
The limitation of this study was that many cases did not turn up again and follow-up could not be done, as a result we could not evaluate the effect of vitamin D supplements on the patients and whether they showed any improvement after increase in their serum vitamin D level or not.
Also, it is not clear that how toxic vitamin D levels could be reached inside a theoretically normal/deficient population. There is a possibility that patients suffering from vitamin D toxicity did not give correct history regarding intake of vitamin D supplements which lead to bias. This was another limitation of this study.
Despite this limitation, we believe that our study points towards the importance of evaluating serum vitamin D in OLP patients. Further studies are required to elucidate the effect of vitamin D supplements in the treatment of OLP.
Conclusion
This study shows that patients with OLP have significantly lower level of serum vitamin D as compared to healthy controls. Although vitamin D deficiency was seen more in OLP cases, yet insufficiency was more prevalent in controls which points towards the fact that serum vitamin D was lacking in general in the North Indian population.
It is possible that administration of vitamin D in patients with OLP can help in suppressing adverse reactions of immune system activity. Performing an interventional study on two OLP groups with one group receiving vitamin D supplements for the treatment and observing the results may provide better insight towards understanding the correlation between vitamin D and OLP.
References
- 1.M Ravi Prakash S, Ghanta S, Verma S, Agarwal N, Gupta N et al. (2013) Meteorological influences on the incidence of lichen planus in a north Indian population. , J Oral Sci 55, 311-18.
- 2.Gupta A, RPS Mohan, Gupta S, Malik S, Goel S et al. (2017) Roles of serum uric acid, prolactin levels, and psychosocial factors in oral lichen planus. J Oral Sci. 59(1), 139-146.
- 3.Kapoor C, Wadhwan V, Vaidya S, Malik S. (2013) Triology of nitric oxide, mast cell and stress in pathogenesis of oral lichen planus. , J Oral Maxillofac Pathol 17(2), 156-62.
- 5.Georgakopoulou E A, Achtari M D, Achtaris M, Foukas P G, Kotsinas A. (2012) Oral Lichen Planus as a Preneoplastic Inflammatory Model. , J Biomed Biotechnol.2012
- 6.S B Ismail, Kumar S K S.Zain R B.(2007) Oral lichen planus and lichenoid reactions: etiopathogenesis, diagnosis, management and malignant transformation. , J Oral Sci 49, 89-106.
- 8.S T Faezi, Ansari N, Paragomi P, Akhlaghi M, Ghanavat M.. Davatchi F.(2014) Vitamin D deficiency in patients with Behcet’s disease. J Diabetes Metab Disord.13(1): 18.
- 9.Fakharan M, Haghighi A, Arabi M.. Loghman M.(2014) Investigating the Levels of Serum Vitamin D in Patients with Rheumatoid Arthritis Referred To Rasoul-Akram Hospital During 2011-2012. IJMS 39(5), 476-9.
- 10.M H Ibrahim, T K Alloush, Rahim M.K A.(2014) Vitamin D Level in Multiple Sclerosis Patients. Could Vitamin D Level Be Routine Investigation for Multiple Sclerosis Patients? Neurosci Med. 5 :. 201-4.
- 11.Schoindre Y, Jallouli M, Tanguy M-L, Ghillani P, Galicier L et al. (2014) Lower vitamin D levels are associated with higher systemic lupus erythematosus activity, but not predictive of disease flare-up. Lupus Sci Med.1(1):e000027.
- 12.Varma R B, Valappila N J, Pai A, Saddu S C, Mathew N. (2014) Oral lichen planus: Is vitamin D deficiency a predisposing factor? a case report. , IJSS 2(7), 230-232.
- 13.Henry J D, Crawford J R. (2005) The short-form version of the Depression Anxiety Stress Scales (DASS-21): Construct validity and normative data in a large non-clinical sample. , Br J Clin Psychol 44(2), 227-39.
- 14.S R Dudala. (2013) Updated Kuppuswamy's socioeconomic scale for2012. , J NTR Univ Health Sci 2, 201-2.
- 15.Oberoi S S. (2015) Updating income ranges for Kuppuswamy's socio-economic status scale for the year 2014. Indian J Public Health.59:. 156-7.
- 16.Palacios C, Gonzalez L. (2014) Is vitamin D deficiency a major global public health problem?. , J Steroid Biochem Mol Biol.144 Pt A: 138-45.
- 17.Ritu G, Gupta A. (2014) . Vitamin D Deficiency in India: Prevalence, Causalities and Interventions. Nutrients.6: 729-75.
- 18.Brannon P M, Yetley E A, Bailey R L, Picciano M F. (2008) Vitamin D and health in the 21st century: an update. Proceedings of a conference held September2007 in Bethesda , Maryland, USA, Am J Clin 88(2), 483-592.
- 19.Davis C D. (2008) Vitamin D and cancer: current dilemmas and future research needs. , Am J Clin 88(2), 565-9.
- 20.P H Hart.Gorman S.(2013) Exposure to UV wavelengths in sunlight suppresses immunity. To what extent is UV induced Vitamin D3 the mediator responsible? Clin Biochem Rev. 34, 3-13.
- 21.Holick M F. (2004) Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease. , Am J Clin Nutr.80(6 suppl): 1678-88.
- 22.B H Kim, Glanz K.Nehl E J.(2012) Vitamin D beliefs and associations with sunburns, sun exposure and sun protection. , Int J Environ Res Public Health 9, 2386-95.
- 23.P B Rapuri, H K Kinyamu, J C Gallagher.Haynatzka V.(2002) Seasonal changes in calciotropic hormones, bone markers, and bone mineral density in elderly women. , J Clin Endocrinol Metab 87(5), 2024-32.
- 24.Gozdik A, J L Barta, Wu H, Cole D, Vieth R et al. (2009) Seasonal changes in vitamin D status in healthy young adults of different ancestry in the greater Toronto area. Presented at the 14th Workshop on Vitamin D. October 4-8,2009 , Brugge, Belgium .
- 25.Sandhu S V, Sandhu J S, Bansal H, Dua V. (2014) Oral lichen planus and stress: An appraisal. Contemp Clin Dent. 5(3), 352-56.
- 27.Harinarayan C V, Ramalakshmi T, Prasad U V, Sudhakar D, Srinivasarao P et al. (2007) High prevalence of low dietary calcium, high phytate consumption, and vitamin D deficiency in healthy south Indians. , Am J Clin Nutr 85, 1062-7.
- 28.Taylor C L, Patterson K Y, Roseland J M, Wise S A, Merkel J M et al. (2014) . Including Food 25-Hydroxyvitamin D in Intake Estimates May Reduce the Discrepancy between Dietary and Serum Measures of Vitamin D Status.J Nutr 144, 654-59.
- 29.Ingafou M, Leao J C, Porter S R, Scully C. (2006) Oral lichen planus: a retrospective study of 90 British patients. Oral Dis. 12, 463-68.
- 30.Dubowitz T, Heron M, C E Bird, Lurie N, B K Finch et al. (2008) Neighbourhood socioeconomic status and fruit and vegetable intake among whites, blacks and Mexican Americans in the United States. , Am J Clin Nutr 87(6), 1883-91.
- 31.Drewnowski A, Specter S E. (2004) Poverty and obesity: the role of energy density and energy costs. , Am J Clin Nutr 79(1), 6-16.
- 32.Martini L A, Wood R J. (2006) Vitamin D status and the metabolic syndrome. , Nutr Rev 64, 479-86.
- 33.Dinsa G D, Goryakin Y, Fumagalli E, Suhrcke M. (2012) Obesity and socioeconomic status in developing countries: a systematic review. Obes Rev. 13(11), 1067-79.
- 34.Manolache L, Seceleanu-Petrescu D, Benea V. (2008) Lichen planus patients and stressful events. , J Eur Acad Dermatol Venereol 22(4), 437-41.
- 35.Berk M, Sanders K M, Pasco J A, Jacka F N, Williams L J et al. (2007) Vitamin D deficiency may play a role in depression. , Med.Hypotheses 69, 1316-19.
- 36.Roecklein K A, Rohan K J. (2005) Seasonal Affective Disorder-An Overview and Update. , Psychiatry 2(1), 20-26.
- 37.D J Armstrong, G K Meenagh, Bickle I, Lee A S H, E S Curran et al. (2007) Vitamin D deficiency is associated with anxiety and depression in fibromyalgia. , Clin Rheumatol 26(4), 551-54.
Cited by (37)
This article has been cited by 37 scholarly works according to:
Citing Articles:
Clinical and Experimental Dermatology (2025) Crossref
Clinical Oral Investigations (2025) OpenAlex
Clinical Oral Investigations (2025) Crossref
Rania Shalaby, Ghada Nabil, Sally Ibrahim, A. A. Kotb, H. Amer et al. - Clinical Oral Investigations (2025) Semantic Scholar
María García-Pola, Lucía Rodríguez-Fonseca - Nutrients (2024) Semantic Scholar
Annals of Agricultural and Environmental Medicine (2023) OpenAlex
Karolina Thum-Tyzo, B. Tyzo, R. Chałas - Annals of Agricultural and Environmental Medicine (2023) Semantic Scholar
Annals of Medicine & Surgery (2023) Crossref
T. Nosratzehi - Annals of Medicine and Surgery (2023) Semantic Scholar
Annals of Medicine and Surgery (2023) OpenAlex
Journal of Indian Academy of Oral Medicine and Radiology (2022) Crossref
Journal of Indian Academy of Oral Medicine and Radiology (2022) OpenAlex
Indian Journal of Pharmaceutical Sciences (2022) OpenAlex
Mersin Üniversitesi Sağlık Bilimleri Dergisi (2022) Crossref
Mersin üniversitesi sağlık bilimleri dergisi/Mersin üniversitesi sağlık bilimleri dergisi (2022) OpenAlex
Cellular and Molecular Biology (2022) OpenAlex
Cellular and Molecular Biology (2022) OpenAlex
Yuhan Hu - Cellular and Molecular Biology (2022) Semantic Scholar
Biomedicines (2022) Crossref
S. Saeed, Priyadarshini Choudhury, S. Ahmad, T. Alam, Rajat Panigrahi et al. - Biomedicines (2022) Semantic Scholar
Biomedicines (2022) OpenAlex
Lupus (2021) Crossref
Gulhane Medical Journal (2021) OpenAlex
BioMed Research International (2021) OpenAlex
F. Rezazadeh, S. Haghighat - BioMed Research International (2021) Semantic Scholar
The Journal of Contemporary Dental Practice (2020) OpenAlex
Lupus (2020) OpenAlex
Assem Abo-Shanab, S. Kholoussi, Rania Kandil, D. Dorgham - Lupus (2020) Semantic Scholar
Saudi Journal of Oral and Dental Research (2020) OpenAlex
Nilofer J Adhiraja, Deepak Daryani, Chikku Shaila John - Saudi Journal of Oral and Dental Research (2020) Semantic Scholar
N. Gholizadeh, Nafiseh Sheykhbahaei - Biological Trace Element Research (2020) Semantic Scholar
Biological Trace Element Research (2020) OpenAlex
P. Motahari, Fatemeh Pournaghi Azar, Arefeh Rasi - Ethiopian Journal of Health Sciences (2020) Semantic Scholar
Ethiopian Journal of Health Sciences (2020) OpenAlex
Diyala Journal of Medicine (2019) OpenAlex
S. Ahmed - Diyala Journal of Medicine (2019) Semantic Scholar
Journal of Indian Academy of Oral Medicine and Radiology (2019) OpenAlex
Deleted Journal (2017) OpenAlex
