Abstract
Diabetes mellitus (DM) is a chronic metabolic disorder. Streptozotocin is a naturally occurring cytotoxic chemical, particularly toxic to the pancreas and insulin producing beta cells in mammals and induces diabetes. Glimepiride is a second generation sulfonylurea, used as second-line or add-on treatment options for type 2 diabetes. Fenugreek (Trigonella foenum graecum) seeds have been documented as a traditional plant treatment for diabetes. Soluble dietary fiber of Fenugreek significantly improved oral glucose tolerance in diabetic rats. It also exerts anti-diabetic effects mediated through the inhibition of carbohydrate digestion and absorption and the enhancement of peripheral insulin action. Most herbal remedies can interact with allopathic drugs resulting in altered activity and toxicity. At the same time, herbal remedies might produce the same kind of effects as the drug produce. Current published research information on herb-drug interactions is scanty. So, the aim of this study was to investigate the possible interaction between conventional drug used for the management of diabetes; (Glimepiride) and a traditional herbal remedy; Fenugreek aqueous extract in Streptozotocin induced diabetic male albino rats. In conclusion, combination therapy induces better hematological, biochemical effects and improves the oxidative stress biomarkers and antioxidant enzymes. Histological studies showed better results on some organ functions. The results emphasize the benefit of using the combination of Fenugreek seeds aqueous extracts as supportive complementary anti-diabetic therapy.
Author Contributions
Academic Editor: Farah Yousef, Damascus University, Damascus (Syria), Syrian Arab Republic.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Nawal A. Elghazaly, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Diabetes mellitus (DM) is a chronic metabolic disorder featured by hyperglycemia and disturbances in protein, carbohydrate and fat metabolism. These metabolic abnormalities result either from a deficiency of the blood sugar-lowering hormone; insulin or from insulin resistance; a defect in the body’s capacity to respond to insulin. Streptozotocin (N-Nitro derivative of Glucosamine) is a naturally occurring broad spectrum antibiotic and cytotoxic chemical. It is produced by the bacterium Streptomyces achromogenes and it is particularly toxic to the pancreas and has been used widely to create animal models of diabetes 1. Amaryl® (Glimepiride) is a sulfonylurea used as the drug of choice in the treatment of type 2 diabetes. The hypoglycemic activity of Glimepiride relied on its ability to enhance insulin release and action 2. Trigonella foenum graecum (Fenugreek) is a medicinal plant that grows annually and used daily in Africa and Asia. Fenugreek is known to have several pharmacological effects. One of them is the hypoglycemic effect. The seeds have been documented as a traditional plant treatment for diabetes. Soluble dietary fiber (SDF fraction) of Fenugreek significantly improved oral glucose tolerance in diabetic rats 3. The aim of this study is to investigate the possible interaction between Amaryl® tablets (Glimepiride) and Fenugreek seed aqueous extract on Streptozotocin induced diabetes in male albino rats.
Materials and Methods
Experimental Animals
Male albino rats, weighing between 150-160 g, were housed in a clean and well ventilated animal house, maintained at room temperatures, with a constant 12 hr light and 12 hr dark cycle, with access to tap water and standard diet (SCF; Ewu Feed Mill, Edo State, Nigeria) for a 2-week acclimatization period. The handling of the animals was in accordance with the standard principles of laboratory animal care of the United States National Institutes of Health 4.
Chemicals
Streptozotocin (STZ) was purchased from Sigma – Aldrich chemical, USA). Amaryl® (Glimepiride) was obtained from local pharmacies, Egypt. It was grinded using a mortar. The drugs were administered orally through tube once a day.
Herbal Samples
Trigonella foenum –Graecum (Fenugreek) water extract was made from 100g fresh seed. Seeds were grinded and mixed with 2000 ml distilled water 5. The water extract was lyophilized and collected as powder.
Experimental Design
Diabetes was induced in the experimental rats by intraperitoneal injection with Streptozotocin at single dose (40 mg/kg body weight). After 72 hours, rats had more than 250 mg/dl random blood glucose concentration. Treatment with Fenugreek and Amaryl® started 3 days after Streptozotocin injection and continued for eight weeks.
Animal Model
Thirty-five Male Albino rats were randomly divided into five groups (7 rats/group) as follows:
Group1 (Control Group)
Rats of this group were injected with 0.1 ml of citrate buffer (pH; 4.5).
Group 2 (Diabetic Group)
Rats of this group were injected intra-peritoneally with Streptozotocin (STZ).
Group 3 (Glimepiride; Amaryl® Group)
Rats of this group were injected intra-peritoneally with STZ and after 3 days, they received powdered Amaryl® tablet (Glimepiride) at 800µg/kg body weight orally once a day.
Group 4 (Fenugreek Group)
Rats of this group were injected intra-peritoneally with STZ and after 3 days they received the dried aqueous extract of Fenugreek at a dose rate of 500mg/kg.
Group 5 (Combination of Fenugreek and Glimepiride Group)
Rats of this group were injected intra-peritoneal with STZ at single dose (40 mg/kg body weight) and after 3 days they received aqueous extract of Fenugreek at a dose rate of 500mg/kg and powdered Amaryl® tablet (Glimepiride) at 800µg/kg body weight daily.
During the experiment, weights and random blood sugar levels of the rats were measured. At the end of the experiment, rats were sacrificed for the histological investigation of kidney, pancreas and testes. Hematological and biochemical assessments were studied.
Statistical Analysis
The data, expressed as Mean ± SD. were analyzed statistically using column statistics and one way ANOVA with Newman-Keuls Multiple Comparison Test as a post- test using the computer statistics Data were fed to the computer and analyzed using IBM SPSS software package version 20.0. (Armonk, NY: IBM Corp).The Kolmogorov-Smirnov test was used to verify the normality of distribution Quantitative data was described using mean and standard deviation. Significance of the obtained results was judged at the 5% level. The minimum level of statistical significance was set at P < 0.05.
Results and Discussion
Body Weight Through Eight Weeks of Treatment
Results showed a reduction in the body weight in STZ injected rats. This reduction may be attributed to insulin depletion provoking a loss of adipose tissues and the changes in carbohydrates and protein metabolism that occur in rats with Streptozotocin-induced diabetes 6. The increase in the body weight of group (5) exceeds that of group (3) and group (4). It reached the same value of the control group at the eighth week. The body weight of group (3) and group (4) improved during the experiment (Figure 1). Fenugreek aqueous extract significantly increase the body weight. It exhibited antioxidant property which protects the functional organs and increase the body weight 7. Glimepiride increases the body weight significantly 8. Combination of Fenugreek and Glimepiride showed more increase in the body weight compared to other groups.
Figure 1.Effect of Fenugreek and Glimepiride on the body weight of diabetic male albino rats.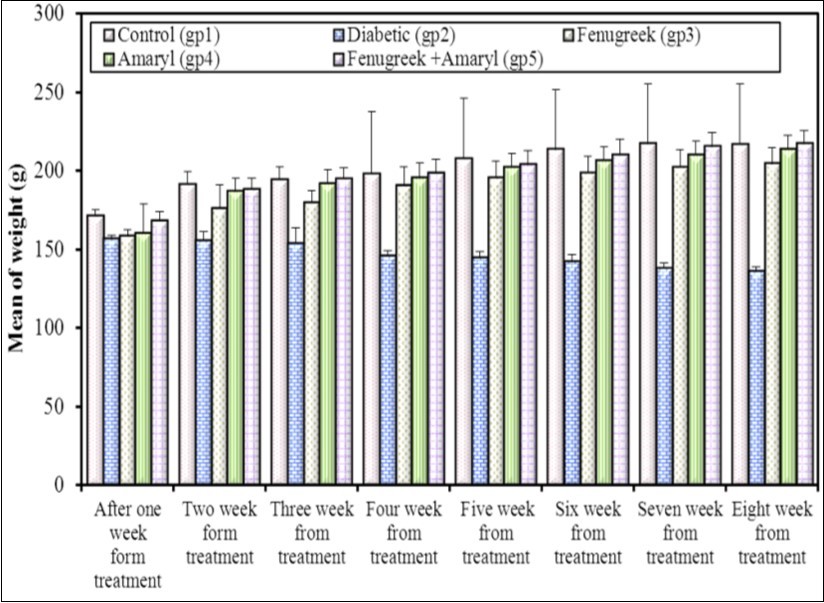
Hematological Parameters
Erythrocyte Count
The alteration induced by Streptozotocin led to some changes in the blood parameters. These changes are exhibited through a decrease in RBCs, Ht, Hb and platelets, on the other side, an increase in HbA1C. These changes could be due to oxidative stress, which lead to lipid peroxidation in RBCs membranes, auto oxidation of hemoglobin. The decrease in Mean Cell Hemoglobin Concentration (MCHC) values observed after administration of STZ is an indication of abnormal hemoglobin synthesis, failure of blood osmoregulation, and plasma osmolality. Oxidation of these proteins and hyper-glycaemia cause an increase in the production of lipid peroxides that lead to haemolysis of RBC 9. The study demonstrated that rats treated with Fenugreek or Glimepiride provided significant protection to the altered hematological variables (Table 1). Treatment with the combination of Fenugreek and Glimepiride were more effective than Fenugreek alone or Glimepiride alone. The anti-oxidant property of Fenugreek 7 inhibits lipid peroxidation of the erythrocytes. Fenugreek contains iron and it can improve anemia conditions. Glimepiride is able to improve the erythrocyte membrane integrity by the mitigation of oxidative damage to the erythrocytes membranes 10. Results showed that combination of Fenugreek and Glimepiride had better RBC, Hb, Ht, and MCHC levels of experimental groups showed no significant difference compared to normal control after eight weeks (Table 1).
Table 1. Effect of Fenugreek and Glimepiride on erythrocytes count of diabetic male rats| Parameters | Control (gp1) | Diabetic (gp2) | Fenugreek (gp3) | Glimepiride (gp4) | Fenugreek + Glimepiride (gp5) | LSD (5%) |
| Red blood cellsRBC(10^6/mm3) | 4.77a+0.29 | 3.67c+ 0.24 | 4.06b+0.10 | 4.12b+0.14 | 4.67a+0.21 | 0.314 |
| Hemoglobin (Hb) (g/dl) | 12.23a+0.3 | 9.90c+0.4 | 11.28b+0.3 | 11.53b+0.5 | 12.08a+0.1 | 0.506 |
| Hematocrit (Ht)(%) | 42.50a+1.7 | 34.48c+1.6 | 39.13b+0.7 | 39.15b+0.7 | 41.13a+0.8 | 1.848 |
| Hemoglobin conc. (MCHC)(g/dl) | 30.75a+0.5 | 28.55c+0.5 | 29.10bc+0.2 | 29.40b+0.5 | 30.15a+0.1 | 0.645 |
| Platelet count (mm3) | 382.75a+33.2 | 230.50c+28.7 | 292.50b+17.0 | 310.0b+18.2 | 357.25a+22.2 | 37.243 |
| Glycosylated hemoglobin (HbA1c)(%) | 5.53c+0.25 | 8.38a+0.49 | 6.38b+0.26 | 6.45b+0.37 | 6.10b+0.22 | 0.500 |
Platelets Count
In diabetes, there is an increased platelet activation and subsequent aggregation favoring thrombosis and also depression of bone marrow stem cells which could be contributory for a reduced platelet count observed. The decrease in circulating platelet number may also means an increase in aggregated platelet typical of atherosclerotic disorders of diabetes mellitus. In Fenugreek group, platelet count increased significantly (p < 0.01). The significant improvement in the levels of circulating platelets in blood could be attributed to inhibitory activity of certain constituents of T. foenum graecum on platelet aggregation 11. Glimepiride group showed an increase of the platelet count. Glimepiride exhibits inhibitory effects on human platelets aggregation through selective suppression of the cyclooxygenase pathway 12. Combination of Fenugreek and Glimepiride showed better platelet count with no significant difference compared with normal control group after eight weeks, (Table 1).
Total Leucocytes Count
Regarding total WBCs, lymphocytes and monocytes; the results showed marked decrease with a distinct increase in the percentage of neutrophils and eosinophil's (Table 2). Fenugreek seeds play a role in protecting the spleen and increasing the lymphocytes. So, they might improve the immunity. These observations were in agreement with those obtained by Sindhu et al.13 and Kandhare et al.14. Fenugreek seeds influenced the hemoglobin and lymphocytes count, improving hematopoietic function and survival. The present study demonstrates that combination of Fenugreek and Glimepiride are more effective than Fenugreek alone or Glimepiride alone in protecting to the altered hematological variables, (Table 2).
Table 2. Effect of Fenugreek and Glimepiride on total leucocytes count and the differential count of diabetic male albino rats| Parameters | Control (gp1) | Diabetic (gp2) | Fenugreek (gp3) | Glimepiride (gp4) | Fenugreek + Glimepiride (gp5) | LSD (5%) |
| Total WBC(10^3/mm3)) | 6.58a+ 0.34 | 5.09c+ 0.32 | 5.81b+0.23 | 5.91b+0.25 | 6.33a+0.17 | 0.41 |
| Neutrophils (%) | 60.20c+0.32 | 68.58a+1.57 | 66.05b+0.95 | 66.08b+1.05 | 61.48c+1.18 | 1.644 |
| Esinophils (%) | 1.53c+0.10 | 2.45a+0.24 | 1.98b+0.10 | 2.0b+0.18 | 1.73c+0.15 | 0.244 |
| Lymphocytes (%) | 36.68a+1.98 | 27.15d+1.52 | 30.10c+0.94 | 30.80c+1.36 | 34.03b+0.70 | 2.07 |
| Monocytes (%) | 5.0a+0.18 | 3.90c+0.22 | 4.30b+0.27 | 4.43b+0.36 | 4.83a+0.10 | 0.364 |
Biochemical Parameters
Random Blood Sugar Level
The current study showed some changes in the biochemical measurements due to the injection of Streptozotocin (Figure 2). Group (5); Fenugreek and Glimepiride combination improved the random blood sugar to reach a value near the normal control values. The random blood sugar level of group (3) and group (4) showed no significant difference from each other. Injection with STZ induces activation of poly adenosine diphosphate ribosylation and nitric oxide release. As a result, pancreatic cells are destroyed by necrosis 15. Fenugreek seeds water extract acts as an insulin secretor. Hypoglycemic effect of Fenugreek may be mediated through stimulating insulin synthesis and increasing β pancreatic cells secretion. The primary mechanism of action of Glimepiride in lowering the blood glucose appears to be dependent on stimulating the release of insulin from functioning pancreatic beta-cells 16. Glimepiride binds to sulfonylurea receptors on β-cells leading to blocking of K+ ATP channels, opening of voltage-gated calcium channels and increase in Ca2+ influx leading to insulin release from pancreatic β-cells.
Figure 2.Effect of Effect of Fenugreek and Glimepiride on random blood sugar level and insulin concentration of diabetic male albino rats.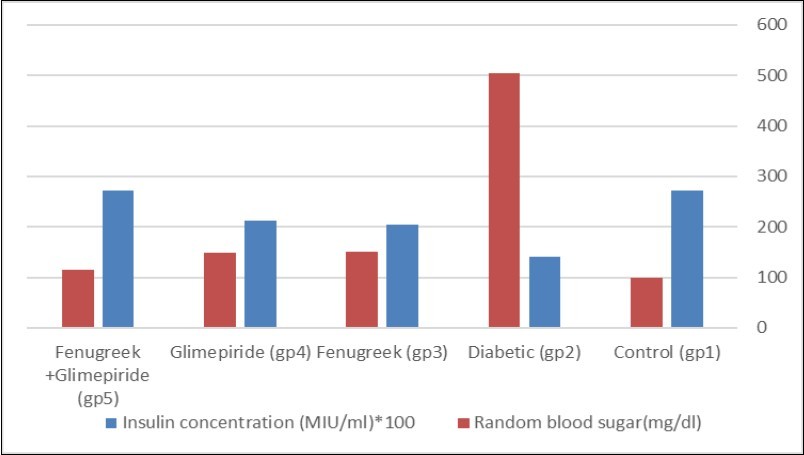
Insulin Concentration
The present study demonstrated that STZ injection induced a significant decrease in the concentration of insulin in serum when compared with the control values. Groups (3 and 4) showed partial improvement in the concentration of insulin when compared with STZ group. However, the insulin concentration of group (5) reached the same value as the control group values (Figure 2).
Serum Total Protein and Albumin
Serum levels of total protein and albumin in the STZ group were significantly lowered while the administration of Fenugreek in group (3) or Glimepiride in group (4) revealed significant increase in the serum levels of albumin and total protein (Table 3). However, their levels in the combination treatment were near to the value of the control group. In Streptozotocin induced diabetes rats, body cells are unable to utilize glucose as a source of energy so proteins are spared as the energy source. The amino acids released from the proteins are used for gluconeogenesis. The decrease in the total protein might be due to micro-proteinuria which was an important clinical marker of diabetic nephropathy and /or might be due to increase in protein catabolism. This leads to a decrease in the protein storage which in turn reduces body weight. Albumin decrease due to its glycation during diabetes may be considered as an important factor responsible for oxidative stress related to diabetes 17.
Following the treatment with Fenugreek or Glimepiride, total protein and albumin level were increased significantly. The elevated serum TP level in Fenugreek groups may be due to high level of protein and other nutrients in T. foenum-graecum L seeds 18.
Table 3. Effect of Fenugreek and Glimepiride serum total protein level and albumin level of diabetic male rats| Parameters | Control (gp1) | Diabetic (gp2) | Fenugreek (gp3) | Glimepiride (gp4) | Fenugreek + Glimepiride (gp5) | LSD (5%) |
| Total protein (g/dl) | 6.85a+ 0.13 | 5.93c+ 0.17 | 6.45b+0.13 | 6.48b+0.22 | 6.78a+0.17 | 0.253 |
| Albumin (g/dl) | 3.83a+0.22 | 2.93c+0.36 | 3.45b+0.13 | 3.43b+0.17 | 3.65ab+0.19 | 0.344 |
Serum Lipid Profile
STZ administration associated with a high significant increase in serum lipid profile (Table 4); total cholesterol, triglyceride, low density lipoproteins LDL and Ratio (cholesterol/HDL) except high density lipoproteins (HDL). Fenugreek treatment improved these elevations. Also, Glimepiride significantly decrease these parameters as compared to the control group. However, group (5) significantly improve all these parameters specially triglycerides which reached a values near the control ones. On the other hand, the high-density lipoproteins (HDL) concentration of diabetic group showed an apparent decrease and groups (3, 4 and 5) improve this decrease.
Table 4. Effect of Fenugreek and Glimepiride on serum lipid profile of diabetic male rats| Parameters | Control (gp1) | Diabetic (gp2) | Fenugreek (gp3) | Glimepiride (gp4) | Fenugreek + Glimepiride (gp5) | LSD (5%) |
| Cholesterol (mg/dl) | 144.25c+11.59 | 192.50a+ 17.08 | 169.75b+8.18 | 171.50b+7.51 | 160.25bc+7.54 | 16.594 |
| Triglyceride (mg/dl) | 89.0c+4.32 | 183.75a+8.54 | 102.50b+14.91 | 97.50bc+2.89 | 91.25bc+2.63 | 12.229 |
| High density lipoproteins (HDL)(mg/dl) | 42.50a+4.36 | 32.25c+2.22 | 38.50b+1.73 | 39.50bc+1.29 | 40.75bc+1.50 | 3.743 |
| Low density lipoproteins (LDL)(md/dl) | 99.75c+9.81 | 158.25a+18.93 | 129.25b+9.54 | 130.0b+7.16 | 117.50b+8.96 | 17.539 |
| Ratio (cholesterol/HDL) | 3.40c+0.32 | 6.03a+0.86 | 4.43b+0.40 | 4.35b+0.21 | 3.95bc+0.33 | 0.725 |
The lipid changes associated with diabetes mellitus are attributed to an increase of free fatty acids into the liver and to insulin deficiency/resistance. This results in excess fatty acid accumulation in the liver. Krishnakumar et al. 19 reported that diabetes-induced hyperlipidemia is attributable to excess mobilization of fat from the adipose due to the underutilization of glucose. Fenugreek extract causes significant decrease in serum triglycerides, total cholesterol (TC) and low-density lipoprotein cholesterol (LDL-c) 20. These effects may be due to sapogenins, which increase biliary cholesterol excretion in liver, leading to lowered serum cholesterol levels. On the other side, oral administration of Glimepiride causes significant decrease in the serum levels of triglycerides, total cholesterol and LDL-cholesterol in contrast to significant elevation in HDL-cholesterol. Glimepiride improved HDL-c level via improvement of plasma adiponectin level as adiponectin could increase HDL-c levels directly via increased lipoprotein lipase and decreased hepatic lipase activity 21. In addition, the anti-lipidemic action of Glimepiride may reside in their ability to stimulate insulin secretion and action.
Serum Kidney Function (Urea, Creatinine and Uric Acid Level)
STZ administration associated with a significant increase in the urea, creatinine and uric acid levels when compared with the control group (Table 5). Treatment in groups (3, 4 and 5) exhibited a high drop in this elevation especially when compared with the diabetic group. Fenugreek and Glimepiride supplement succeeded partially in improving urea, creatinine and uric acid levels when compared with the diabetic group. Group (5) showed a significant decrease in these levels when compared with groups (3 and 4). Raised serum creatinine, urea and uric acid levels in diabetic group may indicate a pre-renal problem such as volume depletion. The high creatinine levels observed in diabetic may be due to impaired function of the nephrons. The increase in urea nitrogen may be accounted for by enhanced catabolism of both liver and plasma proteins that accompany gluconeogenesis 22. Uric acid is an independent predictor of incident type 2 DM. The present data showed that uric acid levels were increased in diabetic rats. Fenugreek seed extract showed decrease in urea, creatinine and uric acid levels, demonstrating renal protective effects of Fenugreek. Nephron protective effect of Fenugreek is due to its antioxidant and hypoglycemic properties and its constructive effects on collagen which prevent ROS induced destruction of filtration and basement membrane of kidneys 23, 24. Fenugreek and Glimepiride supplements succeeded partially in improving the creatinine level in comparing with the diabetic group. The combination of Fenugreek and Glimepiride group showed a significant decrease in the creatinine level (Table 5).
Table 5. Effect of Fenugreek and Glimepiride on serum kidney function of diabetic male rats.| Parameters | Control (gp1) | Diabetic (gp2) | Fenugreek (gp3) | Glimepiride (gp4) | Fenugreek + Glimepiride (gp5) | F(p) | LSD (5%) |
| Urea (mg/dl) | 27.0b+ 2.45 | 40.50a+ 3.11 | 30.50b+3.70 | 30.25b+3.10 | 27.75b+2.22 | 13.394* (<0.001*) | 4.463 |
| Creatinine (mg/dl) | 0.57d+0.05 | 1.35a+0.13 | 0.99b+0.08 | 0.94b+0.07 | 0.78c+0.02 | 52.977* (<0.001*) | 0.12 |
| Uric acid (mg/dl) | 2.0c+0.18 | 3.68a+0.33 | 2.83b+0.17 | 2.90b+0.16 | 2.70b+0.24 | 27.566* (<0.001*) | 0.343 |
Serum Liver Functions (ALT, AST and ALP Values)
The reported present data (Table 6) indicated that administration of Streptozotocin provoked a significant increase in serum ALT, AST and ALP. Treatment with Fenugreek and Glimepiride induced a partial recovery but treatment using combination of Fenugreek and Glimepiride showed significant improvement of the ALT, AST and ALP values. Diabetes is developed due to obstruction of glucose utilization by the tissues through glycolysis and its over-production through excessive hepatic gluconeogenesis. This may be attributed to the relative deficiency of insulin which is responsible for increasing the expression of the glycolytic enzymes in the liver 25. Serum transaminases and alkaline phosphatase activities were generally increased in Streptozotocin-induced diabetic rats. Fenugreek therapy restores the activity of these enzymes. These seeds may inhibit the liver damage induced by Streptozotocin and improved liver function with return of gluconeogenesis toward its normal level 26. Treatment of diabetic rats with sulfonylurea drugs was explained by increased insulin secretion, hepatic uptake of glucogenic amino acids and stimulation of amino acids incorporation into protein 27.
Table 6. Effect of Fenugreek and Glimepiride on serum liver function of diabetic male rats.| Parameters | Control (gp1) | Diabetic (gp2) | Fenugreek (gp3) | Glimepiride (gp4) | Fenugreek + Glimepiride (gp5) | F(p) | LSD (5%) |
| Alanine aminotransferase (ALT) (U/L) | 29.0b+ 2.94 | 58.75a+ 12.50 | 38.50b+2.38 | 35.50b+4.20 | 33.50b+3.70 | 13.194* (<0.001*) | 9.578 |
| Aspartate aminotransferase (AST) (U/L) | 36.75b+3.30 | 52.50a+6.45 | 41.0b+2.71 | 39.50b+1.29 | 37.75b+2.22 | 12.171* (<0.001*) | 5.497 |
| Alkaline phosphatase (ALP) (U/L) | 530.50c+46.77 | 913.50a+71.68 | 736.50b+52.97 | 686.75b+50.34 | 591.0c+14.63 | 33.910* (<0.001*) | 76.495 |
The elevation of biomarker enzymes such as SGOT, SGPT, and ALP was observed in diabetic rats and indicates the hepatocellular damage. Fenugreek and Glimepiride induced a partial recovery and their combination showed a significant improvement. Fenugreek may improve gluconeogenesis toward its normal level. Glimepiride increase insulin secretion, hepatic uptake of glucogenic amino acid and stimulation of amino acid incorporation.
Oxidative Stress Biomarkers and Antioxidant Enzymes
Oxidative stress is enhanced through various sources such as hyperglycemia, dyslipidemia, hyper-insulinemia, insulin resistance, and impaired antioxidant defense. Oxidative stress in DM adversely affects the physiological and biochemical processes of cell, and among those islets cells of pancreas which are more prone to be damaged due to the lowest intrinsic antioxidant defense mechanism. Chronic hyperglycemia and oxidative stress might be resulted due to the multiple biochemical pathways. The concentration of Malondialdehyde (MDA) andMyeloperoxidase (MPO) significantly decreased in pancreas homogenates of group (5) compared with group (3), group (4) and diabetic group (Table 7). The increase induced by Strepotozotocin is attributed to its ability to generate ROS, which induce oxidative damage in several tissues by enhancing lipid peroxidation (LPO) and also decrease of the antioxidant enzymes. Fenugreek led to decrement in MDA level in treated rats, which could be due to the consumption of the extracellular antioxidant, ceruloplasmin, by the oxidants to combat oxidative stress 28. (Table 8) Fenugreek has been reported to have antioxidant and free radical scavenging activity. Fenugreek combats oxidative stress and the antioxidant activity of Glimepiride could be explained through normalization of elevated hyperglycemia as glucose auto oxidation is the major source of free radicals and peroxides. Rats which treated with Glimepiride showed significant increase in the antioxidant enzymes.
Table 7. Effect of Fenugreek and Glimepiride on Oxidative stress biomarkers of diabetic male rats| Parameters | Control (gp1) | Diabetic (gp2) | Fenugreek (gp3) | Glimepiride (gp4) | Fenugreek + Glimepiride (gp5) | F(p) | LSD (5%) |
| Malondialdehyde (MDA) (nmol/mg) | 17.95d+ 2.68 | 52.50a+ 6.45 | 43.25b+4.65 | 39.75bc+3.10 | 34.75c+3.69 | 34.874* (<0.001*) | 6.521 |
| Myeloperoxidase (MPO) (nmol/mg) | 29.50c+4.43 | 56.25a+6.02 | 43.25b+2.50 | 38.0b+5.48 | 37.50b+3.11 | 19.202* (<0.001*) | 6.802 |
| Parameters | Control (gp1) | Diabetic (gp2) | Fenugreek (gp3) | Glimepiride (gp4) | Fenugreek + Glimepiride (gp5) | F(p) | LSD (5%) |
| Catalase (CAT)(U/mg) | 49.50a+ 4.20 | 28.50d+ 2.65 | 37.50c+2.08 | 39.0c+3.74 | 44.25b+2.99 | 23.836* (<0.001*) | 4.857 |
| Superoxide dismutase (SOD)(U/mg) | 63.0a+ 4.97 | 33.0c+4.97 | 44.0b+4.55 | 45.75b+6.50 | 49.50b+4.20 | 18.042* (<0.001*) | 7.68 |
| Glutathione peroxide (GPx)(U/mg) | 56.25a+4.79 | 29.0c+2.94 | 39.25b+3.30 | 40.0b+3.56 | 44.25b+3.86 | 27.708* (<0.001*) | 5.643 |
Oxidative stress in diabetes is coupled with a decrease in the antioxidant status, which can increase the deleterious effects of free radicals. The SOD and CAT are the two major scavenging enzymes that remove free radicals. The antioxidant property of Fenugreek is due to the presence of many active phytochemicals including vitamins, flavonoids, terpenoids, carotenoids, lignin plant sterol 29. Antioxidants exert their mode of action by suppressing the formation of reactive oxygen species either by the inhibition of enzymes or by chelating trace elements. Glimepiride possesses antioxidant activity against the STZ-induced oxidative stress 30. This antioxidant effect of Glimepiride may be attributed to its activation of the redox sensitive transcription factor NF (Kappa) B, activation of antioxidant enzymes such as SOD which is responsible for dismutation of superoxide ion into oxygen and hydrogen peroxide. Thus, it protects the cell from damage caused by superoxide activity.
Histopathological Studies
Testis Histopathological Observations
Histological examination of control rats' testis showed normal histological structure of the seminiferous tubules with an increase in the spermatogonia and other spermatocytes compared with the diabetic rats which showed an obvious decrease in the count of germinal cells generally and sloughing of germ cell in the lumen of seminiferous tubules in addition to absence of the mature sperms (Figure 3 and Figure 4). Rats tested of Fenugreek group and those treated with Glimepiride (Figure 5 and Figure 6) presented an increase in the number of spermatogenic cells and mature sperms with few focal necrotic cells. Whereas the combination therapy group showed normal histological structure of the seminiferous tubules with normal spermatogonia, presence of ledying cells and more mature of sperms (Figure 7). STZ reduced testosterone production, suggesting a decrease in the function of both Leydig (testosterone producing cell) and Sertoli (spermatogenesis) cells, which might be caused by a reduction in insulin secretion. These changes are probably due to increased Reactive Oxygen Species (ROS) production by accelerated Advanced Glycation End Products (AGE) formation 31 hexosamine and Protein Kinase C pathway. The administration of Fenugreek seeds to diabetic rats significantly decrease of sperm shape abnormality and improve the sperm count 32. Glimepiride reduced sperm abnormality and increased testis weights and sperm count by its antioxidant action 30. The potential protective efficacy of Fenugreek seed extract when added to Glimepiride was observed on reproductive systems.
Figure 3.Photomicrogragh of testis section of a control rat showing testicular tissue with seminiferous tubules separated by interstitial tissue with Ledying cells (arrow). Seminiferous tubules with complete spermatogenesis, Sertoli cell (dashed-arrow) and spermatozoa . (H&E) (40X).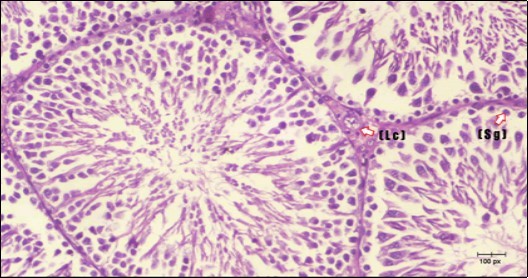
Figure 4.Photomicrogragh of testis section of diabetic rat showing the sloughing of germ cells towards lumen of seminiferous tubules, absence of spermatozoa with low height of germinal epithelium and congested wide interstitial tissue (star). (H&E) (40x).
Figure 5.Photomicrogragh of testis section of treated rat with Fenugreek showing noticeable improvement with increased number of spermatogenic cells included moderate amount of spermatozoa (star) and organized Sertoli cells (arrow). Note liquid infiltration (dashed arrow) in wide interstitial tissue (H&E) (40x).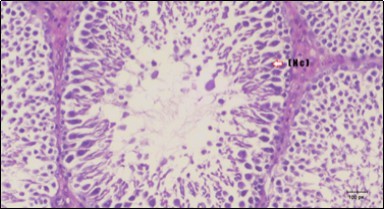
Figure 6.Photomicrogragh of testis section of treated rat showing increased number of spermatogenic cells from spermatogonia till spermatozoa (star). Few liquid infiltration in wide interstitial tissue (H&E) (40X).
Figure 7.Photomicrogragh of testis section of treated rat with combination of Fenugreek and Glimepiride showing nearly normal appearance of seminiferous tubules with organized spermatogenesis end with large number of spermatozoa (star). Note, Sertoli cells (arrows). (H&E) (40X).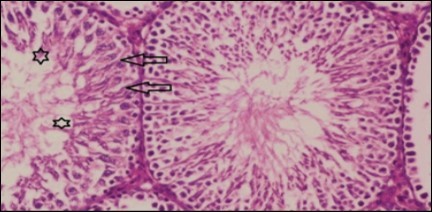
Pancreas Histopathological Observations
Histological examination of pancreas of the control rats showed normal sized islets of pancreas (Ip) compared to the diabetic rats which showed markedly atrophic islets of pancreas (I.p.) (Figure 8 and Figure 9). Pancreas of in groups (3) and (4) rats showed mild atrophic islets of pancreas (I.p.) with inflammatory cellular infiltrate (Figure 10 and Figure 11). The pancreas of rats in group (5) showed nearly normal islets of pancreas (I.p.) with few inflammatory cellular infiltrate (I.c.) (Figure 12).
Figure 8.Photomicrogragh of pancreas section of control rat showing the pale islets of Langerhans contained spindle shape cells (arrow) &polygonal one (dashed- arrow). Strom contained blood vessels (BV) and pancreatic acini (circle) with pyramidal cells have basophilic nucleus at the base. (H&E) (40X).
Figure 9.Photomicrogragh of pancreas section of diabetic rat showing the markedly atrophic islets of Langerhans. (arrow) (H&E) (40X).
Figure 10.Photomicrogragh of pancreas section of treated rat with Fenugreek showing less improved islets of Langerhans with more cells (star). (H&E) (40X).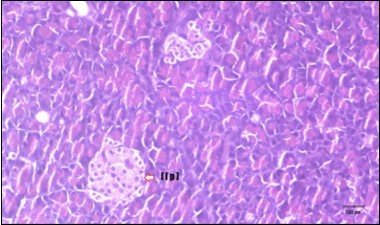
Figure 11.Photomicrogragh of pancreas section of treated rat with Glimepiride showing mildly improved of isled of Langerhans with large number of cells (dashed-arrow). (H&E) (40X).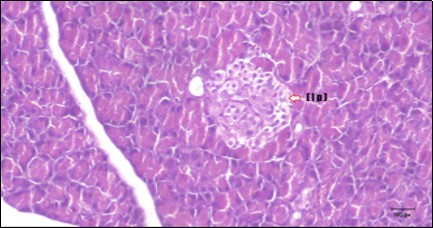
Figure 12.Photomicrogragh of pancreas section of treated rat with both Fenugreek and Glimepiride showing nearly normal islets of Langerhans with spindle (arrow) & polygonal cells (dashed- arrow). (H&E) (40x).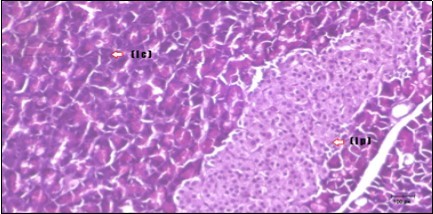
Histopathological observation in diabetic control showed degenerative changes in both endocrine and exocrine pancreases. A probable explanation may be related to oxidative stress resulting from hyperglycemia which decreases the antioxidants levels and increases ROS 33 and the activities of antioxidant enzyme were altered in diabetic rats. These effects further exacerbate the development and progression of diabetes complications and these may represent the causes of degeneration revealed in histological sections. Fenugreek improved blood glucose levels and insulin, lowered pancreatic islet and β-cells damage which may be attributed to its immune modulatory activity and insulin stimulation action along with its antioxidant potential 32. Glimepiride caused restoration of morphology of beta cell of diabetic and it increased percentage of beta cells 34. This protective effect could be attributed to the antioxidant properties of Glimepiride. Combination therapy improves the protective effect of both therapies alone.
Kidney Histopathological Observations
Kidney sections of diabetic rats showed an increase in mesangial cell and matrix of glomeruli with increase in glycogen deposition and hyalinization of arterioles with thickened basement membranes of proximal and distal convoluted tubules. These changes will lead to progressive reduction in the filtration surface of the glomeruli 35. Histological examination of the kidney of the control rats showed normal structure of renal glomerular. The proximal and distal tubules were lined with normal epithelium (Figure 13). The diabetic rats showed tubular casts, inflammatory cellular infiltration and glomerular atrophy (Figure 14). Kidney of rats in groups (3 and 4) showed some glomeruli return to be normal (G). Focal tubules casts (T) in Fenugreek group and some normal tubules in the Glimepiride group with no inflammatory cellular infiltrate in both groups (Figure 15 and Figure 16). However, kidney of rats in group (5) showed normal glomerular (G), normal tubules (T) with no tubular casts (Figure 17).
Figure 13.photomicrogragh of kidney section of control rat showing normal rounded Bowman,s capsules with normal glomeruli (arrow) ,round proximal tubules (dash-arrow) and elongated distal tubules with high cuboidal cells (star) . (H&E) (40X)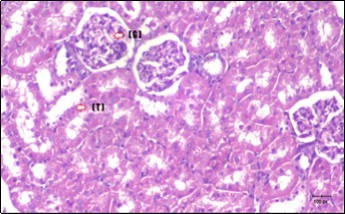
Figure 14.Photomicrogragh of kidney section of diabetic rat showing a trophy of Bowman,s capsules and damage of glomeruli (arrow) and damage of distal and proximal tubules with congested blood vessels (star) . (H&E) (100Px).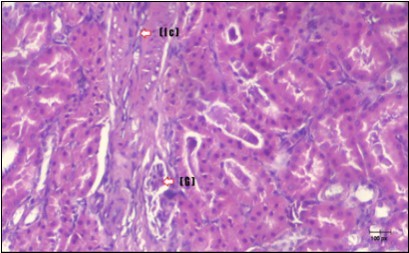
Figure 15.Photomicrogragh of kidney section of treated rat with Fenugreek showing improvement of Bowman,s capsules with normal glomerular (arrow) and nearly return of distal tubules (dashed –arrow) and proximal tubules (star) (H&E) (40xX).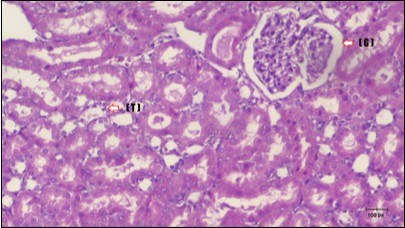
Figure 16.Photomicrogragh of kidney section of treated rat with Glimepiride showing improvement of Bowman,s capsules and partial improvement of glomerulir (arrow) and completely return of distal tubules and proximal tubules to the normal shape with wide urinary space. (H&E) (40X).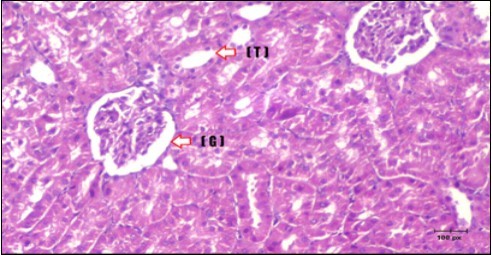
Figure 17.Photomicrogragh of kidney section of treated rat with both Fenugreek and Glimepiride showing improved cortical tissue with most of both Bowman,s capsules (arrow) and proximal tubules (dash-arrow) with nearly normal structure .Note distal tubules still suffering (star). (H&E) (40X).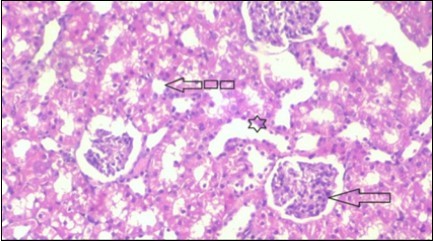
Conclusion
Fenugreek is a natural therapy that has many beneficial health effects while Glimepiride is an efficient conventional anti-diabetic drug. The combination of aqueous extract of Fenugreek seeds and Glimepiride gives better results in monitoring body weights and hyperglycemia in STZ induced diabetic rats. It also improves all hematological, biochemical and histological parameters and decrease the incidence of diabetes complications. So, addition of Fenugreek as a supportive supplement to Glimepiride can improve all parameters on diabetic individuals than each one alone. Moreover, there is no need to increase the dose of Glimepiride which may lead to increase the probability of consequences of side effects on diabetic patients.
References
- 1.Gupta S, Kataria M, Gupta P K, Murganandan S, Yashroy R C. (2004) Protective role of extracts of neem seeds in diabetes caused by streptozotocin in rats. , J. Ethnopharmacol 90, 185-189.
- 2.Kakadiya J, Shah M, Shah N. (2010) Glimepiride reduces on experimentally induced ischemia/reperfusion in diabetic rats. , Int. J. Appl. Biol. Pharm. Technol 2, 276-285.
- 3.Hannan JMA, Ali L, Rokeya B, Khaleque J, Akhter M et al. (2007) Soluble dietary fibre fraction ofT. foenum graecum(fenugreek) seed improves glucose homeostasis in animal models of type 1 and type 2 diabetes by delaying carbohydrate digestion and absorption and enhancing insulin action. , Br. J. Nutr 97(3), 514-521.
- 5.Rafiq K, Sherajee S J, Sufiun M A, Mostofa M, Alam AKMR et al. (2011) Comparative efficacy of stevia leaf (Stevia rebaudiana bertoni), methi seeds (Trigonella foenum-graecum) and glimepiride in streptozotocin induced rats. , Int. J. of Phytopharmacology 2(1), 9-14.
- 6.Pepato M T, Keller E H, Baviera A M, Kettelhut I C, Vendramini R C et al. (2002) Anti-diabetic activity of Bauhinia fortificata decoction in streptozotocin-diabetic rats. , J Ethnopharmacol 81, 191-97.
- 7.Thirunavukkarasu V, Anuradha C, Viswanathan P. (2003) Protective effect of fenugreek (Trigonella Foenum graecum) seeds in experimental ethanol toxicity.Phytother Res. 17(7), 737-43.
- 8.Weitgasser R, Lechleitner M, Luger A, Klingler A. (2003) Effects of glimepiride on HbA(1c) and body weight in Type 2 diabetes: results of a 1.5-year follow-up study. Diabetes Res. and Clinical Practice. 61, 13-19.
- 9.Arun G S, Ramesh K G. (2002) Improvement of insulin sensitivity by perindopril in spontaneously hypertensive and Streptozotocin diabetic rats. , Indian J Pharmacol 34, 156-64.
- 10.Takagi S, Murata H, Goto T, Hatate H, Endo M et al. (2011) Role of taurine deficiency in inducing green liver symptom and effect of dietary taurine supplementation in improving growth in juvenile red sea bream (Pragrus major) fed non-fish meal diets based on soy protein concentrate. , Fisheries Science 77, 235-244.
- 11.Lawson S R, Gabra B H, Guerin B, Neugebauer W, Nantel F et al. (2005) Enhanced dermal and retinal vascular permeability in streptozotocin-induced type 1 diabetes in Wistar rats: blockade with an elective bradykininB1 receptor antagonist. , Regul. Pept.124(1-3): 221, 224.
- 12.Hadi N, Al-amran F, MAA Hussein, Rezeg F A. (2012) Evaluation of the effects of glimepiride (Amaryl) and repaglinide (novoNorm) on atherosclerosis progression in high cholesterol-fed male rabbits. , Journal of cardiovascular disease research 3(1), 5-11.
- 13.Sindhu G, Ratheesh M, Shyni G L, Nambisan B, Helen A. (2012) Anti-inflammatory and antioxidative effects of mucilage ofTrigonella foenum graecum(Fenugreek) on adjuvant induced arthritic rats. , Int. Immunopharmacol 12, 205-211.
- 14.Kandhare A D, Bodhankar S L, Mohan V, Thakurdesai P A. (2015) Effect of glycosides based standardized fenugreek seed extract in bleomycin induced pulmonary fibrosis in rats: Decisive role of Bax. Nrf2, NF- B, Muc5ac, TNF- and IL-1 , Chem. Biol. Interact 237, 151-165.
- 15.Yadav M, Tomar R, GBKS Prasad, Jain S, Yadav H. (2008) Complementary hypoglycemic and anti-hyperglycemic activity of various extracts of Fenugreek seeds in rats. , Asian J Biochem 3, 182-7.
- 16.Muller G. (2005) The mode of action of the antidiabetic drug glimepiride-beyond insulin secretion. , Curr. Med. Chem. - Immun., Endoc. Metab. Agents 5(6), 499-518.
- 17.Jin L, Xue H Y, Jin L J, Li S Y, Xu Y P. (2008) Antioxidant and pancreas-protective effect of aucubin on rats with streptozotocin-induced diabetes. , Eur. J. Pharmacol 582, 162-167.
- 18.Abbas R J. (2010) Effect of using fenugreek, parsley and sweet basil seeds as feed additives on the performance of broiler chickens. , Int. J. of Poultry Sci 9(3), 278-282.
- 19.Krishnakumar K, Augustti K T, Vijayammal P L. (2000) Hypolipidaemic Effect of Salacia oblonga Wall. Root bark in Streptozotocin Diabetic Rats. , Med. Sci 28, 65-67.
- 20.UCS Yadav, Moorthy K, Baquer N Z. (2004) Effects of sodium orthovanadate andTrigonella foenum graecumseeds on hepatic and renal lipogenic enzymes and lipid profile during alloxan diabetes. , J. Biosci 29, 81-91.
- 21.Emoto M, Konishi T, Ikuno Y, Lee E, Teramura M et al. (2009) Glimepiride increases high-density lipoprotein cholesterol via increasing adiponectin levels in type 2 diabetes mellitus. , Metabolism 58(2), 143-8.
- 22.Prakasam A, Sethupathy S, Pugalendi K V. (2004) Influence of Caseariaesculenta root extract on protein metabolism and marker enzymes in streptozotocin-induced diabetic rats. , Pol. J. Pharmacol 56(5), 587-593.
- 23.Kaviarasan S, Viswanathan P, Anuradha C V. (2007) Fenugreek seed (Trigonella foenum graecum) polyphenols inhibit ethanol-induced collagen and lipid accumulation in rat liver. , Cell. Biol. Toxicol 23, 373.
- 24.Belaïd-Nouira Y, Bakhta H, Haouas Z, Flehi-Slim I, Cheikh H B. (2013) Fenugreek seeds reduce aluminum toxicity associated with renal failure in rats. , Nutr. Res. Pract 7, 466.
- 25.Sun Y, Liu S, Ferguson S, Wang L, Klepcyk P et al. (2002) Phosphoenolpyruvate carboxykinase overexpression selectively attenuates insulin signaling and hepatic insulin sensitivity in transgenic mice. , J. Biol. Chem 277, 23301-23307.
- 26.Abdel-Moneim A, EL-zayat E, Mahmoud S. (2002) Effect of some antioxidants on streptozotocin diabetic rats. , J Egypt Ger Soc Zool 38, 213-245.
- 27.Ashour A A, Yassin M M, Elyazji N R. (2004) Improvement of glucose level, lipid profile, some enzyme activities and structure of pancreas and liver in diabetic rats treated with glibenclamide. , J. Islam Univ 12, 139-156.
- 28.Devi R, Nandhini J, Madhavan M, Kumar S.(2012).The effect of fenugreek (Trigonella foecum graecum) intake on the oxidative stress in hyper-cholesterolaemic subjects. , Nat J Lab Med 2(1), 5-7.
- 29.Bukhari S B, Bhanger M I, Memon S. (2008) Antioxidative Activity of Extracts from a Fenugreek Seeds (Trigonella foenum-graecum).J. , Anal. Environ. Chem 9(2), 78-83.
- 30.S I Rabbani, Devi K, Khanam S. (2009) Inhibitory effect of glimepiride on nicotinamide – streptozotocin induced nuclear damage and sperm abnormality in diabetic Wister rats. , Ind. J. of experimental Biol 47, 804-810.
- 31.Mallidis C, Agbaje I, McClure N, Kliesch S. (2011) The influence of diabetes mellitus on male reproductive function: a poorly investigated aspect of male infertility. , Urologe A 50(1), 33-37.
- 32.Hamden K, Jaouadi B, Carreau S, Aouidet A, El-Fazaa S et al. (2010) Potential protective effect on key steroidogenesis and metabolic enzymes and sperm abnormalities by fenugreek steroids in testis and epididymis of surviving diabetic rats. , Arch Physiol Biochem 116, 146-155.
- 33.Al-Kufaishi A M. Babylon University (2012) Study of Hypoglycemic Effect of IraqiTeucrium polium(L.) on Diabetic Rabbits via Oxidant-Antioxidant System. Thesis of M.Sc.
Cited by (3)
- 1.Barakat A I, Radwan EH, De Patricio, 2020, Effect of Trigonella Foenum against Ethylene Diamine Tetra Acetic Acid induced Nephrotoxicity in Male Albino Rats, Journal of Zoological Research, 1(1), 32, 10.14302/issn.2694-2275.jzr-20-3435
- 2.Kashif Muhammad, Nasir Amar, Gulzaman , Rafique Muhammad Kamran, Abbas Mazhar, et al, 2023, Unlocking the anti‐diabetic potential of Gymnema sylvestre, Trigonella foenum‐graecum, and their combination thereof: An in‐vivo evaluation, Food Science & Nutrition, 11(12), 7664, 10.1002/fsn3.3685
- 3.EL Hak Heba Nageh Gad, Metawea Shimaa Ibrahim, Nabil Zohour Ibrahim, 2022, Fenugreek (Trigonella foenum graecum L.) supplementation safeguards male mice from aflatoxin B1-induced liver and kidney damage, Comparative Clinical Pathology, 31(6), 925, 10.1007/s00580-022-03413-6
