Abstract
Four strains of Arbuscular Mycorrhizal (AM) biofertilizer fungi, combined with two potato cultivars, were in-field tested in a four-replicate arrangement in a factorial experiment. As far as general combinability is concerned, cv. Agria was more responsive to different inocula (yield +5.56%, P 0.02) and to two strains in particular (+8%). On the other hand, the results with Innovator, a cultivar that yields 33% less than Agria, showed a significant reduction in the number of tubers for three AM strains, thus proving a clear genetic Biofertilizer * Cultivar interaction. The study of hay litter-bags has shown a high NIR spectral fingerprint for the Cultivar factor (81%), while the Inoculation factor showed a higher spectral fingerprint in Agria (76%) than in Innovator (65%). The Substrate Induced Respiration predicted from the NIR-SCiO spectra of the litter-bags was significantly increased after inoculation (+6.3%, P 0.04), but appeared lower for Agria (-5.4%) vs. Innovator (P 0.05), with a non-significant interaction. The obtained results show that the adaptation of the AM strains to the genetics of potato cultivars is a first step toward reducing chemical inputs, with consequent benefits for the environment, but without an excessive reduction in yield. The litter-bag technique can therefore be recommended for a simplified monitoring of the complicated plant-mycorrhizosphere relationship.
Author Contributions
Academic Editor: Abubaker Haroun Mohamed Adam, Department of Crop Science (Agronomy), College of Agriculture, Bahri University- Alkadaru- Khartoum -Sudan.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Silvia Volpato, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
In 1990, Reganold et al.1 pointed out that sustainability in agriculture was afflicted by serious problems: “high energy costs, groundwater contamination, soil erosion, loss of productivity, depletion of fossil resources, low farm incomes and risks to human health and wildlife habitats”. Thirty years later, progress in agriculture has increased crop yields to face the rapidly growing human population, which is increasing at a rate of 70 million per year. However, the greater use of chemical fertilizers and pesticides, as a result of the spread of animal husbandry and therefore of the increased leaching of excreta, has led to a greater pollution of the environment, through poisoning of the air, soil and water, which in turn has led to the accumulation of toxic residues in food, as well as the development of the resistance of pests. As a result, less invasive remedies, such as reduced tillage, organic farming and botanical insecticides have been introduced2. Moreover, since much of humanity is fed inadequately and many food productive systems have been pushed beyond safe boundaries, a radical transformation in modern agriculture, evolving from the BAU paradigm3, is being pursued. Sustainability in intensive crop production requires a reduction in chemical inputs, and biofertilizers are therefore being encouraged, with benefits for the environment, but without an excessive reduction in yield, in the search for an optimum economic vs. a maximum system4, 5.
Many experiments have shown that Arbuscular Mycorrhizal (AM) biofertilizer fungi can overcome the nutrient limitations on plant growth by enhancing nutrient acquisition. Furthermore, their benefits ranges from stress alleviation to bioremediation in soils polluted with heavy metals6. Biofertilizers may also enhance the protection of plants against pathogens and increase plant diversity7.
Potato plants are known to be sensitive to water stress and have a low P uptake, due to their rarefied root hair system. Therefore, AM fungi can alleviate abiotic stress caused by low levels of P and/or a partially localized water deficit8. An economic potato yield can only be achieved through a suitable irrigation and through fertilization, provided that a critical Olsen-P concentration of 46 mg/kg is attained to achieve a maximum production of 90%9.
The objective of the current study has been to study the influence of AM on the growth of potato plants of different cultivars, without limited water and P resources. Since functional diversity is present in AM symbiosis, with different combinations of plants and AM species10, and since potato plants usually have a modest AM root colonization, compared with other crops11, four AM species were introduced to test for any differences in general and to establish the specific combinability of each species with two widely used potato cultivars.
The litter-bag technique, coupled with NIR spectra, has recently been developed as a simplified monitoring system of the complicated plant -mycorrhizosphere relationship in biofertilizer experiments 12.
Experimental Procedures
In the framework of biofertilizer studies, four types of AM fungi have been tested their general and specific combinability with two potato cultivars. The productive traits were determined, in a testing station, through the NIRS litter-bag technique12, a method that was developed as a rapid analysis resulting from the availability of the NIR-SCiO smart device. A general holistic model has recently been applied to Olive orchards13 suffering from Xylella attacks and to a corn yield model14, which included scanning the leaves and determining the foliar pH, two methods that have not been applied in the present study because of the excessive distance of the fields from the test station, which has prevented the green perishable material, such as the leaves, from being analyzed precisely.
Material and Methods
In 2018, four types of Arbuscular Mycorrhizae (AM) (Table 1) were multiplied in Sorghum sudanensis plants at the CCS-Aosta farm-factory. The whole mycorrhizosphere, including the substrate, was carefully ground using an industrial meat grinder. In 2019, the resulting material was delivered to the Rusthoeve station (Molenweg, 4675 RB Colijnsplaat, The Netherlands, 51.589589, 3.847216) in a soil characterized by a high sand content (39%), N-tot 3300 kg ha-1, C/N ratio 10, OC 1%, OC/Organic matter ratio 0.5, P (available\stock) 3.8\810 kg ha-1, CEC 154 cmolc kg -1, pH 7.2. The four AM were distributed by hand, near the tubers, at a dose of 4000 g ha-1 and at a density of 5555 plants ha-1. The cultivation started on April 23, with an irrigation on July 16, using 200 kg N ha-1, up to the harvest on October 10. The four AM fungi (A, B, C, D) strains in the factorial experiment plus the Control (K) were test-field studied and combined with two potato cultivars, that is, Agria and Innovator, in a four replicate arrangement, with 20 plant seeds per plot-1. One week after seeding, 60 litterbags (3 plot-1) were buried near the tubers, were extracted on July 16, and were then returned to Italy, where they were dried at 65°C for 36 h and scanned in triple.
Table 1. Results of the average productive traits for the two cultivars, according the biofertilizers and effect of size (Ln A÷D/K) and general probability as obtained from Friedman’s test.| Biofertilizer | Cultivar | |||||
| Agria | Innovator | |||||
| Yield | #Tubers | Mean tuber weight | Yield | #Tubers | Mean tuber weight | |
| t ha-1 | k ha-1 | g | t ha-1 | k ha-1 | g | |
| A) Funneliformus mosseae IT201 | 52.500 b | 325 b | 162 | 31.389 | 300 b | 104 |
| B) Gigaspora gigantea PA125 | 54.625 ab | 344 ab | 160 | 33.028 | 347 ab | 96 |
| C) Septoglomus constrictum FL328 | 55.917 a | 372 a | 152 | 33.292 | 322 ab | 109 |
| D) Scutellospora pellucida MN408A | 56.000 ab | 349 ab | 161 | 32.222 | 285 b | 116 |
| K) Control | 51.875 b | 347 ab | 154 | 34.417 | 356 a | 99 |
| Effect Size | ||||||
| Ln A/K | 1.2% | -6.6% | 7.8% | -9.2% | -17.0% | 7.8% |
| Ln B/K | 5.2% | -0.8% | 6.0% | -4.1% | -2.4% | -1.7% |
| Ln C/K | 7.5% | 7.0% | 0.5% | -3.3% | -9.8% | 6.5% |
| Ln D/K | 7.7% | 0.4% | 7.3% | -6.6% | -22.2% | 15.6% |
| Ln Mean A÷D / K | 5.56% | 0.10% | 3.42% | -5.62% | -11.82% | 7.60% |
| P general (Biofertilizer) | 0.02 | 0.36 | 0.45 | 0.07 | 0.01 | 0.65 |
The chemometric elaborations were carried out by means of SCiOTM-Lab software, using the random forest classificatory method. The method used for the two cultivars considered the Spectral Fingerprint of the biofertilized litterbags (SF_A-D), compared to the Control (SF_K) cells, and the cultivars were tested for probability as one proportion, using MedCalc online software.
An NIR-SCiO equation, taken from an experimental trial on tomato plants14, was used to obtain an indirect estimate of the substrate induced respiration (SIR) capacity. The latter was measured according to Anderson and Domsch15. The correlation between the estimated and measured data resulted to be sufficiently high to be considered reliable under comparable conditions.
Individual data of the SIR capacity were analyzed, by means of a bi-factorial linear model16, with the Biofertilizer and Cultivar factors, completed with their interaction.
Yield data from the Control and biofertilized sub-plots and their size effects were analyzed using Friedman’s test for paired comparisons17.
Results
Divergent results were obtained for the two cultivars (Table 1). The average effect of size on the yield of Agria amounted to +5.56% (P 0.02), with differences in favor of the C (Septoglomusconstrictum )biofertilizer, with as much as +7% in the number of tubers. On the other hand, the Innovator cultivar responded negatively to the biofertilizers, and the tubers were reduced (-11.82%, P 0.01), as was the yield (-5.62%, P 0.07).
The two dimensions of the letter-bags are highlighted in Figure 1, namely the Substrate Induced Respiration (SIR) on the abscises and the Spectral Fingerprint (SF) on the ordinate.
Figure 1.Plot of the Substrate Induced Respiration (X axis, SIR, mcg CO2 g-1 vs. the Spectral Fingerprint (Y axis, SF) of the litterbags for the non-inoculated control (K, circle and empty triangle) and biofertilized A,B,C,D (circle and full triangle) for the two cultivars Agria (circle) and Innovator (triangle), with fitted regressions of SF on SIR. The average SIR effects for the biofertilizers (K 223, A-D 237, +6%, P 0.04) and for the cultivar (P<0.001) are reported at the margin of the X axis. The average SF responses for the Agria cultivar ( SF 0.73 for K and 0.79 for A-D, P<0.001) and Innovator cultivar (SF 0.60 for K and 0.69 for A-D, P 0.157 and 0.005, respectively) are reported at the margin of the Y axis.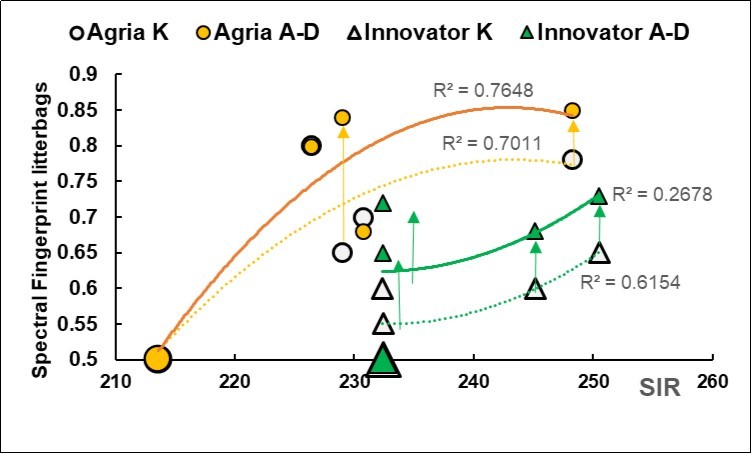
The respiratory parameter of the soil responded positively to the biofertilizers, with an average value of 223 mcg CO2 g-1 in the control, which increased to 237 in the biofertilized samples (+6%, P 0.04). On the other hand, the values of the litter-bags from the Agriacultivarwere significantly below the respiratory values of the Innovator cultivar by about -5% (P 0.05), but there was no interaction between the cultivar and the biofertilizer factors (P 0.37).
Unlike the trend observed for the SIR parameter, the spectral fingerprinting of the litterbags from the Agria cultivar obtained a signature of 0.76, which was significantly higher (P 0.02) than the 0.65 obtained from the Innovatorcultivar. The biofertilized litter-bags showed an average prevalence of +0.08 vs. the Control (P 0.03).
The positive correlation of the respiratory and the fingerprint appears throughout Figure 1 (green arrows facing up), where the polynomial regressions of SF on SIR averaged R2 0.58 for the four subgroups, with a minimum of 0.27 in the biofertilized Innovator cultivar.
Discussion
The preliminary results from this experiment confirm the luxuriating effects albeit of an emersion of a genotype * genotype interaction. The maximum amount of +8% registered for Septoglomusconstrictum (C) in Agria was comparable with the results of a wide range of extension field tests published by Hijri18 involving 231 pairwise field trials on an affirmed biofertilizer in North America and Europe applying a calculated 71 spores of Rhizophagusirregularis (formerly named Glomus intraradices and then Glomus irregulare) per seed. A relevant increase in yield of the biofertilized fields (42.2 t ha-1) vs. the controls (38.3 t ha-1, +10.1% ± 14%, P<0.0001) was found over a period of four years. The Author fixed the break-even point at +1.7%, and the outcome was economically positive in 79% of the cases. When the original data from the supplementary table was considered, the result was only negative in 35 pairwise cases (15%), while when classified by cultivar, it emerged that Innovator excelled as far as the yield is concerned by +14% vs. the control (36.0 t ha-1). However, the positive results of the 19 censed varieties were characterized by a coefficient of variation of 84% (+10% ± 8%). In the present work, the average standard control yield for Innovator obtained in Holland was 34.4 t ha-1, while it was 51.9 t ha-1 for Agria. RhizophagusirregularisDAOM 197198was not present in the current work, but it should be considered that strain variations within that species could lead to different results. For example, Kokkoris et al.19 compared commercial Rhizophagusirregularis DAOM 197198 samples with locally sourced Rhizophagusirregularis GD50 samples inoculated into five crop plants and five wild plants. Pots (3 L) were filled with a 3.5 kg mix of a sterile medium. No effect on the total biomass was detected, but a variable interactive effect was observed for the P content of the leaf, particularly for wild plants. All the commercial strain plants had a lower leaf P. Overall, these data show that the favorable results previously observed for potato cannot be replicated for Zea mays, Linumusitatissimum, Triticum aestivum, Glycine max or Lens culinaris. Moreover, the quality characteristics of the leaves may be affected by different strains and may be involved in a theoretical framework that can predict mutualistic outcomes for AM.
Douds et al.20 tested commercially available inoculum containing Glomus intraradices(nowRhizophagusirregularis)DAOM 197198 in the potatocv. Superior and obtained 370 g plant-1 vs. 270 in the control (+37%) in one experiment and 400 g plant-1 vs. 340 in the control (+18%) in a second experiment. The control consisted of a compost made up of 15 cm3of a 1:9 mixture of yard clippings and vermiculite to provide nutrients and the natural background microorganisms.
Davies et al21, in a test on Glomus intraradicesinPeruvian potato (Solanum tuberosum L.) Yungay found that the size effect for mycorrhizal inoculation, compared to a sterilized medium condition, ranged from +44% to +57%, thus indicating that Yungay was moderately to highly mycorrhizal dependent.
Lone et al.22 studied the compatibility of a mycorrhizal fungus with potato plants under a sterilized soil condition and phenotyped Jyoti and TPS potato plant cultivars inoculated with Glomus intraradices and G. mosseae. The fresh plant weights on the 80th day were 126.8 and 107.4 g for the inoculated Jyoti (+19%) and the control, vs. 26.6 and 22.8 (+17%) for TPS, respectively. The chlorophyll content was also higher in the AM inoculated samples than in the control.
According to Rai 23, inoculation of the KufriLalima and KufriSindhuri with Glomusmosseae and G. fasciculatum potato cultivars increased yields by 4.2 and 5.5%, respectively.
The biofertilizer dose can be under the Mitscherlich law. Three biofertilizers were compared in one potato cv., Kufri Sindhuri24, at doses of 250, 500, 750 g ha-1. One product was identified as the best to increase all the growth, yield and quality parameters. The optimum concentration was found to be the minimum one, that is, 250 g ha-1. Both the mycorrhizal products produced adverse effects on all the growth and yield parameters at much higher concentrations. The Authors suggested that AM should be used as a supplement, along with commonly used fertilizers, to increase the growth and yield.
According to Sakha et al.25, no AM * cultivar interaction was detected in sweet potatoes under natural field conditions when a mix of Rhizophagusirregularis, Funneliformismosseae, Claroideoglomusclaroideum and Claroideoglomusetunicatum was inoculated into two cultivars. The size effect was +30% for the vine length, -4% for the number of branches, +18% for the marketable weight and +29% for the shoot biomass.
A critical point is the choice for the level of mineral fertilization vs. the zero level in the control plots. Mukongo et al.26 investigated how to reduce inorganic P fertilizer in sweet potato, under natural field conditions, considering two soil-season conditions by integrating N and K with a mix of two AM biofertilizers, namely i) Glomus mosseae, G. intraradices, G. etunicatum, G. claroideum and ii) Glomus and Gigaspora spp. with Bacillus megaterium var. phosphaticum. The results of the three-factorial experiment were quite promising when the size effect referred to the zero control. However, no evident effect emerged when the biofertilized plots were compared with the true conventional and fertilized control. Furthermore, biofertilizer ii) enhanced a negative interaction (yield -14% in the zero control and -31% in the fertilized control) for the more productive condition. The results also show that starter nutrients should be included to obtain a significant contribution from AM in nutrient depleted soils.
The results of Ekin et al.27, with reference to the optimization of biofertilizers with mineral elements, indicate that Glomus intraradices inoculation has a great potential to decrease potassium fertilizer levels in the Granola potato cv. On average, the marginal yield was +0.7% kg K-1, which increased to +1.9% kg K-1 with AM, that is, with a size effect of +27%, but, interestingly, an optimum of +40% was found for a dose of 10 kg K-1.
Other effects that can corroborate natural AM sources can be obtained through the use of potassium phosphites (KPhi). Tambascio et al.28, at the McCain Experimental Station, Balcarce, Argentina, observed that the application of KPhi to Shepody and Kennebec cultivars reduced the period between planting and emergence, increased the leaf area and dry matter and increased the incidence of autochtonous AM.
Rhizophagusirregularis is ubiquitous in the natural environment and is reportedly a preferential colonizer of potato plants in agricultural systems throughout Italy29, and the in vitro mass-production of different strains of that species has been found to significantly increase the yields of the globally important crop cassava30. Other works31, 32 favor the on-farm production of the inoculum of indigenous AM biofertilizers.
In this experiment, the litter-bag features were closely correlated with the yield, thus confirming the importance of the respiratory capacity of the control soil and above all the increment that is realized after the inoculation, as clearly outlined in Figure 2. In fact, a multivariate relationships on the averages of the eight sub-groups allowed a model with R2 0.67, thus confirming the effectiveness of the holistic symbiotic models build in the previous experiments for Olive13 and Corn14.
Figure 2.Plot of the measured vs. predicted paired responses of the Yield effect size (ln A-D/K) in the two cultivars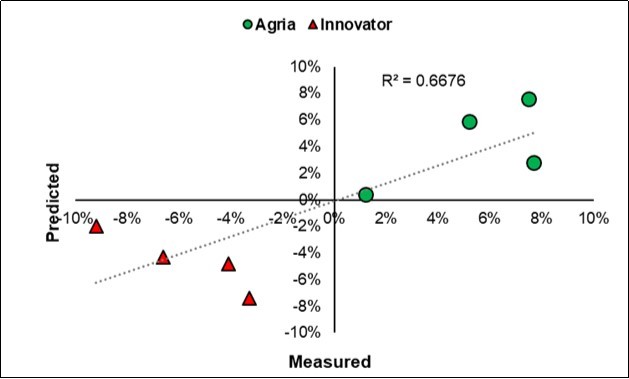
Conclusion
The main result of this experiment is that a possible AM * cultivar interaction, which had previously been shown for corn14, has been confirmed. Thus, the adaptation of AM strains to the genetics of potato cultivars can be considered a first step toward reducing chemical inputs, with benefits for the environment, but without an excessive reduction in yield.
The litter-bag technique may be recommended for a simplified monitoring of the complicated plant-mycorrhizospher relationship, preferably combined with a foliar pH measurement.
Acknowledgments
Thanks to John van Klaren for the valuable support to the symbiotic method in Holland.
References
- 1.Reganold J P, Papendick R I, Parr J F. (1990) . Sustainable agriculture.Scientific American262(6) 112-21.
- 2.Campos E V, Proença P L, Oliveira J L, Bakshi M, Abhilash P C et al. (2019) Use of botanical insecticides for sustainable agriculture: future perspectives.Ecological. Indicators105 483-95.
- 3.Willett W, Rockström J, Loken B, Springmann M.Lang T.et al.(2019) Food in the Anthropocene: the EAT–Lancet Commission on healthy diets from sustainable food systems.The Lancet393(10170). 447-92.
- 4.Avio L, Turrini A, Giovannetti M, Sbrana C. (2018) Designing the ideotype mycorrhizal symbionts for the production of healthy food.Frontiers in plant science9. 1089.
- 5.Limonard T, Ruissen M A. (1989) The significance of VA-mycorrhiza to future arable farming in the Netherlands.Netherlands. , Journal of Plant Pathology 95(1), 129-35.
- 6.Clark R B, Zeto S K. (2000) Mineral acquisition by arbuscular mycorrhizal plants.J. , PlantNutr 23, 867-902.
- 7.Aggarwal A, Kadian N, Tanwar A, Yadav A, Gupta K K. (2011) Role of arbuscular mycorrhizal fungi (AMF). in global sustainable development.Journal of Applied and Natural Science3(2) 340-51.
- 8.Liu C, Ravnskov S, Liu F, Rubæk G H, Andersen M N. (2018) Arbuscular mycorrhizal fungi alleviate abiotic stresses in potato plants caused by low phosphorus and deficit irrigation/partial root-zone drying.The. , Journal of Agricultural 156(1), 46-58.
- 9.Freeman K L, Franz P R, De Jong RW. (1998) Effect of phosphorus on the yield, quality, and petiolar phosphorus concentration of potatoes (cvRusset BurbankandKennebec) grown in the krasnozem and duplex soils of Victoria.Australian. , Journal of Experimental Agriculture38 83-93.
- 10.Feddermann N, Finlay R, Boller T, Elfstrand M. (2010) Functional diversity in arbuscular mycorrhiza–the role of gene expression, phosphorous nutrition and symbiotic efficiency.Fungal Ecology3(1). 1-8.
- 11.Tarkalson D D, Jolley V D, Robbins C W, Terry R E. (1998) Mycorrhizal colonization and nutrient uptake of dry bean in manure and compost manure treated subsoil and untreated topsoil and subsoil.Journal of Plant Nutrition21. 1867-1878.
- 12.Masoero G, Delmastro M, Cugnetto A, Giovannetti G, Nuti M. (2018) NIRS footprint of bio-fertilizers from hay litter bags. , Journal of Agronomy and Research 1(1), 22-33.
- 13.Giovannetti G, Polo F, Nutricato S, Masoero G, Nuti M. (2019) Efficacy of commercial symbiotic bio-fertilizer consortium for mitigating the Olive Quick Decline Syndrome (OQDS). , Journal of Agronomy and Research 2(1), 1-21.
- 14.Volpato S, Masoero G, Mazzinelli G, Balconi C, Locatelli C. (2019) Spectroscopic and foliar pH model for yield prediction in a symbiotic Corn Production. , Journal of Agronomy Research 2(3), 1-18.
- 15.Anderson J, Domsch K. (1978) A physiological method for the quantitative measurement of microbial biomass. in soils.Soil Biology and Biochemistry10 215-221.
- 18.Hijri M. (2016) Analysis of a large dataset of mycorrhiza inoculation field trials on potato shows highly significant increases in yield. , Mycorrhiza 26(3), 209-14.
- 19.Kokkoris V, Hamel C, Hart M M. (2019) . Mycorrhizal response in crop versus wild plants.PloSone14(8)
- 20.Douds Jr DD, Nagahashi G, Reider C, Hepperly P R. (2007) Inoculation with arbuscular mycorrhizal fungi increases the yield of potatoes in a high P soil. , Biological agriculture & horticulture 25(1), 67-78.
- 21.Davies Jr FT, Calderón C M, Huainan Z. (2005) Influence of arbuscular mycorrhizae indigenous to Peru and a flavonoid on growth. yield, and leaf elemental concentration of Yungay potatoes. , HortScience 40, 381-385.
- 22.Lone R, Shuab R, Sharma V, Kumar V, Mir R et al. (2015) Effect of arbuscular mycorrhizal fungi on growth and development of potato(Solanum tuberosum)plant. , Asian Journal of Crop Science 7(3), 233-43.
- 23.Rai R P. (1990) Improved yields in potato through mycorrhizal inoculation. In:Trends in mycorrhizal research.Proceedings of the National Conference on Mycorrhiza, held at Haryana Agricultural University, Hisar, India,Feb.14-16 148-149.
- 24.Kumar N, Kumar-Singh A, Prasad-Moharana D.Kumari A.et al.(2017) Field Evaluation of Arbuscular Mycorrhizal Fungal Formulations on Production Performance of Potato (Solanum tuberosum L.) cv. Kufri Sindhuri. , Current Journal of Applied Science and Technology 24(3), 1-6.
- 25.Sakha M A, Jefwa J, Gweyi-Onyango J P. (2019) . Effects of Arbuscular Mycorrhizal Fungal Inoculation on Growth and Yield of Two Sweet Potato Varieties.Journal of Agriculture and Ecology Research International27 1-8.
- 26.Mukhongo R W, Tumuhairwe J B, Ebanyat P, AbdelGadir A H, Thuita M et al. (2017) Combined application of biofertilizers and inorganic nutrients improves sweet potato yields.Frontiers in plant science8. 219.
- 27.Ekin Z, Demir S, Oğuz F, Yıldırım B. (2013) Effect of Arbuscular Mycorrhizal Fungus (AMF)Glomusintraradiceswith different potassium fertilizer levels on the tuber yield and size distribution of potato (Solanum tuberosumL.).Journal of Agricultural Sciences23(2). 154-63.
- 28.Tambascio C, Covacevich F, Lobato M C, C de Lasa, Caldiz D O et al. (2014) The application of K phosphites to seed tubers enhanced emergence, early growth and mycorrhizal colonization in potato (Solanum tuberosum).American. , Journal of Plant Sciences5 132-137.
- 29.Cesaro P, D van Tuinen, Copetta A, Chatagnier O, Berta G et al. (2008) Preferential colonization ofSolanum tuberosumL. roots by the fungusGlomusintraradicesin arable soil of a potato farming area.Appliedand Environmental. Microbiology74 5776-5783.
- 30.Ceballos I, Ruiz M, Fernandez C, Pena R, Rodriguez A et al. (2013) Thein vitromass-produced model mycorrhizal fungus,Rhizophagusirregularis, significantly increases yields of the globally important food security crop cassava.PLoSONE8. 70633.
Cited by (10)
- 1.Cugnetto Alberto, Lajolo Luciano, Vitaloni Giovanni, Sarasso Giuseppe, Borgogno Mondino Enrico Corrado, et al, 2021, Vineyard Clusters Monitored by Means of Litterbag-NIRS and Foliar-NIRS Spectroscopic Methods, Journal of Agronomy Research, 3(2), 39, 10.14302/issn.2639-3166.jar-20-3676
- 2.Caradonia Federica, Ronga Domenico, Tava Aldo, Francia Enrico, 2022, Plant Biostimulants in Sustainable Potato Production: an Overview, Potato Research, 65(1), 83, 10.1007/s11540-021-09510-3
- 3.Masoero Giorgio, Sarasso Giuseppe, Delmastro Marco, Delmastro Renato, Antonini Massimiliano, et al, 2023, Soluble Biobased Substances in soil or Salicylic Acid on leaves affect the foliar pH and soil biovariability of Grapes-as explained by the NIR Spectroscopy of Litterbags and Teabags, Journal of Agronomy Research, 5(2), 10, 10.14302/issn.2639-3166.jar-23-4648
- 4.Masoero Giorgio, Oggiano Philipp, Migliorini Paola, Taran Aliaksei, Nuti Marco, et al, 2022, Litterbag-NIRS to Forecast Yield: a Horticultural Case with Biofertilizer Effectors, Journal of Soil Science and Plant Nutrition, 22(1), 186, 10.1007/s42729-021-00643-5
- 5.Hett Jonas, Neuhoff Daniel, Döring Thomas F., Masoero Giorgio, Ercole Enrico, et al, 2022, Effects of Multi-Species Microbial Inoculants on Early Wheat Growth and Litterbag Microbial Activity, Agronomy, 12(4), 899, 10.3390/agronomy12040899
- 6.Aguilar-Paredes Ana, Valdés Gabriela, Nuti Marco, 2020, Ecosystem Functions of Microbial Consortia in Sustainable Agriculture, Agronomy, 10(12), 1902, 10.3390/agronomy10121902
- 8.Nuti Marco, Giovannetti Giusto, Scortichini Marco, Pergolese Giovanni, Saracino Michele, et al, 2021, The Olive Quick Decline Syndrome: A Syndemic Outbreak in the Apulia Region, Southern Italy, Journal of Agronomy Research, 3(3), 13, 10.14302/issn.2639-3166.jar-21-3703
- 9.Masoero Giorgio, Mazzinelli Gianfranco, Balconi Carlotta, Locatelli Sabrina, Lanzanova Chiara, et al, 2020, Spectroscopic Kernel Quality from a Symbiotic Corn Production, Journal of Agronomy Research, 2(4), 18, 10.14302/issn.2639-3166.jar-20-3260
- 10.Baldi Elena, Toselli Moreno, Masoero Giorgio, Nuti Marco, Adam Abubaker Haroun Mohamed, 2020, Organic and Symbiotic Fertilization of Tomato Plants Monitored by Litterbag-NIRS and Foliar-NIRS Rapid Spectroscopic Methods, Journal of Agronomy Research, 3(1), 9, 10.14302/issn.2639-3166.jar-20-3363
