Evaluation of SterculiaUrens Gum as Novel Carrier for Oral Colon Targeted Drug Delivery System
Abstract
The purpose of the research is to evaluate Sterculiaurens gum as a carrier for oral colon targeted drug delivery system. Sterculia gum has been reported to have wide pharmaceutical applications such as tablet binder, disintegrant, gelling agent and as a controlled release polymer, but it has not been exploited as colon targeting carrier. For evaluation as a carrier for colonic delivery of drugs characterization of gum was done. Microflora degradation studies of gum were conducted in phosphate buffer solution (PBS) pH 7.4 containing rat caecal content under anaerobic environment. Solubility, swelling index, viscosity and pH of the polymer solution were determined. Different formulation aspects considered were: gum concentration (10–40%), concentration of citric acid (10–30 %) on swelling index and in-vitro drug release. The results of the isothermal stress testing (IST) shows no degradation of samples of model drug, azathioprine, in the drug polymer mixture and the core tablet excipients. DSC and FT-IR study has proved the compatibility of the drug with Sterculia gum and other tablet excipients. Microflora degradation study revealed that Sterculia gum can be used as tablet excipient for drug release in the colonic region by utilizing the action of enterobacteria. Sterculia gum exhibits premature drug release in the upper GIT without enteric coating and may not reach to the colonic region. From the study, Sterculia gum as colon targeting carrier is possible via coating with chitosan/Eudragit mixed blend polymers which would provide acid as well as intestinal resistance; but undergo enzymatic degradation once it reaches the colon.
Author Contributions
Academic Editor: Sateesh Kumar Sathigari, Scientist, Formulation and Process Development ,Patheon Inc.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2013 Bipul Nath, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
In the recent times, technologies for development of colon targeted drug delivery system have utilized one or two (in combination) of the following primary approaches, with varying degrees of success: (1) pH-dependent systems, (2) time-dependent systems, (3) pro-drugs, and (4) colonic microflora-activated systems 1, 2. Among the different approaches utilized to achieve colon specific drug delivery system, the use of polymers specifically degraded by colonic bacterial enzymes (such as β-glucoronidase, β-xylosidase, β-galactosidase, azoreductase etc.) holds promise 3, 4.Most of these systems are based on the fact that anaerobic bacteria in the colon are able to recognize the various substrates and degrade them with their enzymes 5. Natural gums are often preferred to synthetic materials due to their low-toxicity, low-cost, and easy availability. A number of colon-targeted delivery systems based both on combination of pH-dependent system and polysaccharide degradation have been designed and developed by various research groups for successful delivery of drugs to the colonic region. Sterculia gum has not yet been exploited as drug carrier specifically to the colon 6. It is insoluble in water, but hydrates quickly and swells to form a homogeneous hydrogel which poses difficulty for its use as polysaccharide coat. But, it seemed to be an interesting polymer for the preparation of hydrophilic matrix tablets. However, Sterculia gum in the form of hydrophilic matrix cannot protect the drug from being released in stomach and small intestine 7. Besides, Sterculia gum is expected to retard drug release due to its higher swelling index, and at the same time it undergoes degradation by the colonic microflora which made it ideal to deliver drugs in the colon. As, it has higher swelling index large surface area is being created for microbial action and the swelling property of Sterculia gum can be used to produce hydrostatic pressure in the design of Microflora Triggered Colon Targeted Drug Delivery System 8. Therefore, it seemed reasonable to design a system consisting of osmotic tablet core (containing drug and Sterculia gum with pH modifier i.e. citric acid), an inner semi permeable coating composed of the mixture of Eudragit RLPO and chitosan to provide intestinal resistance but permeable to intestinal fluid, and which could be further over-coated with enteric polymers (Eudragit L100) to provide acid resistance in the stomach. In these systems, the hydrostatic pressure is produced by polymer swelling, which concurrently drives the drug out of the system through the pores created by the pore-forming agent (chitosan) in the inner coating after exposure of the system to the colonic fluid 9. The objective of the study is to (1) investigate the in-vitro degradation of Sterculia gum by colonic microflora and its suitability as carrier for colon targeted drug delivery system, (2) quantify the swelling, influences of formulation variables on in-vitro drug release and (3) drug delivery behavior of Sterculia gum based tablet in the simulated in-vivo physiological. AZA used in the treatment of inflammatory bowel disease was selected as the model drug to study the Sterculia gum based formulations 10.
Experimental procedure:
Materials and Methods
Commercially available Sterculia gum powder was obtained from Krystal Colloids, Mumbai. Azathioprine was obtained as gift sample from RPG Life Sciences Pvt. Ltd, India. Citric acid monohydrate, anhydrous lactose, magnesium Stearate was purchased from Ranbaxy fine Chemicals Ltd, Mumbai. Disodium hydrogen phosphate (Na2HPO4) anhydrous and potassium dihydrogen phosphate (KH2PO4) were purchased from Rankem, Mumbai, India.
Characterization of Sterculia Gum
Sterculia gum passed through 160# sieve was used in the characterization study. The powder form of the Sterculia gum was observed for its appearance and identified as per the British Pharmacopoeial specifications 11. The pharmacopoeial test for its solubility was done by adding Sterculia gum in water and 60 % ethanol to distinguish it from other gums. The color reaction test was carried out by boiling 1.0 g of the sample with water and hydrochloric acid. Precipitate formation test was done by shaking 1.0 g of the sample with water until mucilage is formed, and the resulting mucilage is mixed with concentrated hydrochloric acid and 5.0 M sodium hydroxide, which is then filtered. To the filtrate added potassium cupric tartrate solution and then heated. The purity of the gum was ascertained by carrying out the tests of loss on drying, total ash value, acid insoluble matter, starch content. The pH of 1.0 %w/v aqueous solution of the polymer was determined using a pH meter. The viscosity of 1.0 %w/v solution of the polymer in phosphate buffer, pH 7.4 was determined by using Brookfield viscometer. Particle size distribution of the finely powdered Sterculia gum was determined by passing it through series of B.I.S. stainless steel sieves (mesh size 80, 100, 120, 160, and 200). Powdered gum passed through mesh 160 and retained on 200 mesh sieve was assigned an arithmetic mean diameter. Swelling factor was determined by taking 1.0 gm. of the Sterculia gum in 25 mL of water in a measuring cylinder, shaken every 10 minutes for one hour and allowed to stand for 4.0 hr. 12. The volume occupied by the gum was measured in mL.
Microflora Degradation Studies of Sterculia Gum in Rat Caecal Contents
Microflora degradation studiesof sterculia gum were conducted in phosphate buffer solution (PBS) pH 7.4 containing rat caecal medium 13, 14. The caecal PBS was further bubbled with CO2 gas. Caecal contents were collected from male Wistar rats weighing 250-300 g each. The caecal contents were dispersed in PBS under anaerobic environment and the concentration of the caecal contents was adjusted to 4.0, 8.0 and 12.0 % (w/v) in the PBS. Finely grounded Sterculia gum powder 100 mg was added into 10 mL of caecal PBS and incubated at 37 ⁰C under anaerobic condition. The pH of caecal PBS was measured at 2 h interval up to 8 h using a pH meter.
Isothermal Stress Testing (IST)
The samples of pure drug and homogenous mixture of drug with each excipient were kept at accelerated conditions of 35 ⁰C and 45 ⁰C in sealed glass vials at 75% ± 5% relative humidity 15. Water (10.0 %w/w) was added in each of the vials and the drug-excipient blends were further mixed with a glass capillary tube (both the ends of which were heat sealed). To prevent the loss of material, capillary was broken and left inside the vial. Each vial was sealed using a Teflon-lined screw cap and stored at 35 ⁰C and 45 ⁰C in hot air oven. Drug-excipient blends mixed with water and kept at 25 ⁰C was served as control. The drug-excipient blends were periodically examined for any unusual color change. The samples were quantitatively analyzed in UV visible spectrophotometer (Shimadzu, Japan) at 281 nm after 3 weeks of storage at above conditions. For sample preparation, 2.0 mL of methanol was added to each vial. The mixture was vortexed for 3 minutes and transferred to 100 mL volumetric flask. Vials were rinsed twice with methanol and transferred to the volumetric flask. The samples were centrifuged and the supernatant filtered through 0.45 µm nylon membrane filters. After appropriate dilutions, samples were analyzed in UV-visible spectrophotometer at 281 nm and drug content was determined from the calibration curve prepared within the expected range.
Differential Scanning Calorimetry (DSC) of AZA with Tablet Excipients
The DSC analysis of physical mixture of pure drug with Sterculia gum and other tablet excipients selected were carried out using a differential scanning calorimeter to evaluate any possible drug-excipient interaction 15, 16. Each of the samples as well as the physical mixture of drug and selected excipients (which was passed through 60 mesh sieve) were weighed directly in the DSC sample pan and the analysis was performed at heat flow rate of 10 ºC /min from 50 º to 300 ºC temperature ranges under nitrogen flow rate of 20 mL/min.
Fourier Transform Infrared Spectroscopy (FT-IR) Study
Drug-Sterculia gum physical mixtures as well as tablet excipients were studied by FT-IR spectroscopy (Perkin-Elmer, USA) to determine any possible drug-excipient interactions 17. The samples for FT-IR analysis were prepared in KBr disks (2 mg sample in 200 mg KBr). The scanning range was 400-4000 cm-1 and resolution was 2 cm-1.
Flow Properties of Physical Mixture of Sterculia Gum with Tablet Excipients
The flow properties of the tablet excipients were determined by angle of repose, bulk density/tapped density and compressibility index 18.
Preparation of Core Tablets under 32 Factorial Design
The factorial design batches of core tablets were prepared by direct compression method, after obtaining the acceptable flow property of the physical mixture of the excipients 19. The physical mixture of each batch of factorial design having an average weight of 240 mg were uniformly mixed and compressed in to core tablet using a rotary tablet machine with 8 mm oval punch. The concentration of Sterculia gum and citric acid were selected by using Central Composite Design (CCD) under design expert software (version 8.0). Concentration of Sterculia gum and citric acid were selected as independent variables 20. Swelling index in 2 h and percent (%) dissolution in 6 h, 12 h and18 h in colonic fluid pH 7.4 were selected as dependent variables.
Physical Evaluation of the Core Tablets under Factorial Design
The physical properties of the prepared tablets were evaluated to ensure that there was no capping, lamination, sticking or other defects during compression. The prepared tablets were tested for hardness, thickness, friability, weight uniformity and drug content 21.
Swelling Index of the Factorial Design Batches of Core Tablets
The extent of swelling was measured in terms of % weight gain by the tablet at specific time interval 22. Three tablets from each batch were individually weighed (W1)andplaced on dissolution basket which was dipped in phosphate buffer(pH 7.4) and incubated at 37º C. At the time intervals of 2, 4,6 and 8 h each basket was removed and the weights of swollen tablets were recorded (W2).
Swelling index (SI) was calculated using the following formula
SI (%) = (W2-W1)/W1x 100
In-vitro Dissolution Study
colonic fluid (phosphate buffer pH 7.4 containing rat caecum 4% (w/w), previously bubbled with CO2) for 18 hour 23. Aliquots of dissolution fluid were removed at specified time intervals and analyzed for AZA by UV-visible spectrophotometer at a wavelength of 281 nm.. From the dissolution data, the core tablet was optimized and the optimized core was used to optimize the coating formula.
Statistical Analysis of In-vitro Dissolution Data and Optimization
The response values percent swelling in 2 h (R1), percent drug release in 6 h (R2), 12 h (R3) and 18 h (R4) of factorial batch core tablets were subjected to analysis by response surface reduced quadratic model with the help of Design Expert Software
19, 24. Numerical optimization was used for the core tablet variables to obtain the desired response. An optimized core formula was developed by setting the target (more than 80% drug release in 18 h) on the dependent and independent variables. The software provided optimum concentration of Sterculia gum and citric acid. So, a final batch core tablet was prepared using the concentration provided by the central composite design and its in-vitro drug release studies were carried out in rat caecal medium. The dissolution profile of the optimized batch (F10) is compared with that of the marketed formulations to arrive at the target release.
Stability Studies of Optimized Batch F10 as per ICH Guidelines
The optimized tablet F10 was sealed in aluminium foil packaged container inner coated with polyethylene, and kept separately in humidity chamber at 30 ⁰C/ 65% RH and 40 ⁰C/75% RH for 6 months 26. At the end of the specified period (0, 1, 3 and 6 months), the samples were analyzed for appearance, friability, hardness, drug content and in-vitro dissolution characteristics.
Results and Discussion
Characterization of Sterculia Gum
Powdered Sterculia gum was a light yellow to brown powder. Sterculia gum when treated with water swollen into mucilaginous gel which was acidic to litmus paper. It was insoluble in ethanol, but formed viscous sols in hydro-alcoholic solutions. The sample gave the positive test of gum i.e. development of red colour with HCl and brown colour with sodium hydroxide. Sterculia gum powder produced red precipitation with potassium cupric tartrate solution upon heating. These results confirmed its identity as Sterculia gum. The results of the loss on drying, total ash content, acid insoluble ash content and starch content were found to be 18.7% w/w, 5.81% w/w, 0.56% w/w and 0.00% respectively. Thus, the sample of Sterculia gum has conformed the pharmacopoeial limits for its purity. The pH of 1.0 %w/v aqueous solution of the polymer was found to be 4.6. The viscosity of 1.0 %w/v solution of the polymer in phosphate buffer, pH 7.4 was found to be 1400 cps. The results revealed that highly viscous gel network of the gum has release retarding capacity and would give slower drug release at its increasing concentrations.
The approximate particle size of the powdered gum was found to be 87.5 to 112.5 µm. The results suggested that the gum exudates are uniformly reduced during size reduction process. Hence, reproducible mixtures with other tablet excipients can be obtained at the defined size distribution range.
When Sterculia gum was placed in water, it was immediately hydrated and developed in to a highly viscous mucilaginous gel. Sterculia gum (1.0 g) when treated with 25 mL water gave a swelling factor 15. Sterculia gum also exhibited high swelling index in phosphate buffer, pH 7.4. This property of the gum could be used to produce hydrostatic pressure in the design of Microflora Triggered Colon Targeted Drug Delivery System (MCDDS).
Microflora Degradation Studies of Sterculia Gum
Microflora degradation studies of Sterculia gum revealed that, the pH of caecal-PBS was decreased markedly from pH 7.4 to 5.0 after incubation for 2 h with Sterculia gum
(Table 1). The rate of decrease of pH was depended on the concentration of caecal contents within the 8 h of incubation. The decrease in pH was due to the appearance of degradation products of Sterculia gum such as organic acids by the bacterial enzyme present in rat caecal contents. The rate of decrement of pH of Sterculia gum owing to microflora action at different concentration of the rat caecal contents are shown in Figure 1.
Table 1. Change in pH of caecal-PBS containing Sterculia gum| Sl. No. | Conc. of caecal content in PBS (% w/v) | Conc. of Sterculia gum (% w/v) | pH of caecal -PBS at different time intervals | ||||
| 0.0 h | 2.0 h | 4.0 h | 6.0 h | 8.0 h | |||
| 1 | 4.0 | 0.0 | 7.4 | 7.4 | 7.2 | 7.1 | 7.0 |
| 2 | 4.0 | 1.0 | 7.4 | 6.0 | 5.7 | 5.2 | 4.6 |
| 3 | 8.0 | 1.0 | 7.4 | 5.6 | 5.2 | 4.8 | 4.2 |
| 4 | 12.0 | 1.0 | 7.4 | 5.0 | 4.8 | 4.5 | 3.8 |
Figure 1.Comparative DSC thermogram of drug with the tablet excipients.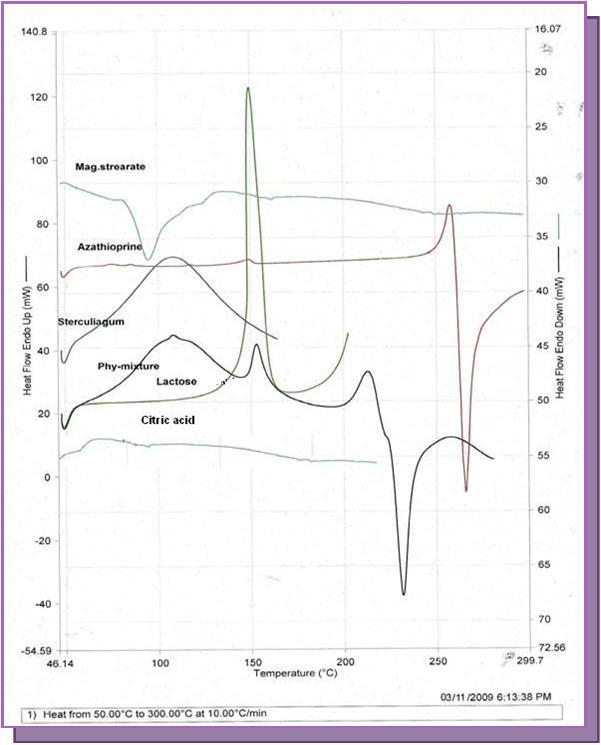
Isothermal Stress Testing (IST)
The study of IST confirmed the physical stability of the drug with Sterculia gum and other excipients at different conditions of temperature and humidity. There was a very negligible amount of loss of drug in the drug excipient mixtures after 3 weeks of storage under stressed conditions, and the residual drug content was found to be within the official limits. The results demonstrated that the model drug was compatible with each of the physical mixture used for prototype formulation. The results of the IST study are presented in Table 2.
Table 2. Results of IST after 3 weeks of storage at stressed conditions| Samples | Ratio | Drug content of controlledsamples (%) | Residual drug content of stressed samples (%) | ||
| 25°C,75 ± 5%RH | 35°C,75 ± 5%RH | 45°C,75 ± 5%RH | |||
| Pure AZA | ------ | 100.81 ± 0.72 | 101.78± 2.47 | 99.53±1.13 | 98.71± 1.23 |
| AZA+ Sterculia gum | 1:2 | 101.42 ± 1.34 | 102.19±0.49 | 99.68± 1.68 | 99.53± 1.98 |
| AZA+ Anhydrous. lactose | 1:1 | 100.73 ± 1.82 | 99.95±2.31 | 99.96± 0.63 | 99.91± 2.68 |
| AZA+ Citric acid | 1:1 | 101.21± 2.74 | 98.68±1.92 | 98.91± 2.39 | 98.87± 1.39 |
| AZA+ Magnesium. Stearate | 3:1 | 100.84 ± 1.92 | 98.81±1.22 | 99.37± 0.98 | 98.68± 2.41 |
DSC of AZA with Tablet Excipients
The peak temperature, enthalapy values of AZA, excipients and drug-physical mixtures are presented in Table 3. In the DSC thermogram of pure AZA, a sharp endothermic peak was observed at 265.74 ⁰C corresponding to its melting transition point, followed by an immediate exotherm (265.74 ⁰C) indicating drug degradation. In case of Sterculia gum, an endothermic peak was observed at 108.02 ⁰C corresponding to its melting temperature, which suggested that the polymer undergoes thermal degradation. The melting endotherm of the drug was well preserved in the physical mixture of the excipients.. An exothermic peak of AZA was appeared in the thermogram of the physical mixture of AZA with other tablet excipients at 241.6 ⁰C, which was slightly shifted from 265.74 ⁰C to a lower temperature (Table 3). However, there were slight changes in the peak shape with little broadening or shifting towards the lower temperature, which could be attributed due to the mixing process that lowers the purity of each component in the mixture. The retention of the endotherm of AZA in the thermogram of the physical mixture of tablet excipients revealed the compatibility of the drug with the excipients used. Selected DSC scans of drug and drug–excipient mixtures are shown in Figure 2.
Table 3. Peak temperature and enthalpy values of AZA, blank excipients and drug-excipients mixtures| Sample | Tonset (⁰C) | Tpeak (⁰C) | ∆Hfcorr (J/g)a |
| AZA | 253.47 | 265.74 | 385.09 |
| Sterculia gum | 98.14 | 108.02 | 324.94 |
| Anhydrous lactose | 123.48 | 149.43 | 456.81 |
| Citric acid | 89.45 | 92.0 | 128.24 |
| Magnesium stearate | 88.98 | 94.73 | 72.67 |
| Drug-excipient physical mixture | 251.32 | 241.6 | 265.74 |
Figure 2.Changes of pH of phosphate buffer containing different concentrations of caecal contents (-♦-) 4%, (-●-) 8%, (-▲-) 12% and without sterculia gum (-□-). Each point represents the mean±SD.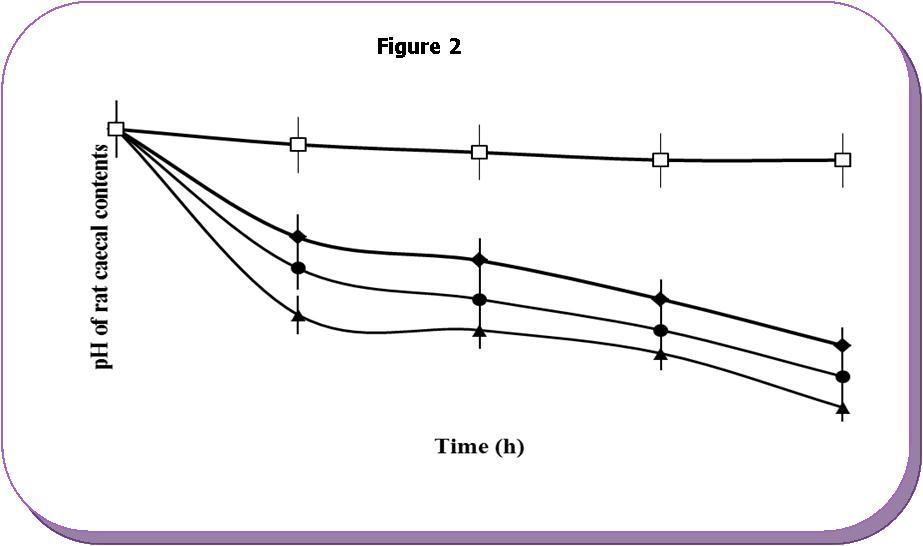
Fourier Transform Infrared Spectroscopy (FT-IR) Study
In the FT-IR study, the principal infra-red absorption peaks of AZA appeared at 3108, 2975 and 2810 cm-1 due to the N-H stretching of the primary amine group (-NH2) and at 1579 cm−1 due to purine ring. However, a sharp peak at 1579 cm-1 appeared due to N-H bending vibrations of the primary amine group. In the IR spectrum the additional peaks at 1531, 1497 and 1439 cm-1 are due to C-N stretching of the -C=NH group. In case of sterculia gum, characteristics peaks were observed due to H- bonded O-H stretching vibration of
hydroxyl groups at 3426 cm−1, C-H stretching vibration of CH2 groups at 2922 cm−1, asymmetric carboxylate anion stretching at 1622 cm−1, symmetric carboxylate anion stretching at 1418 cm−1 and C-O stretching at 1042 cm−1. The identical peaks of N-H stretching, C-N stretching, N-H bending vibrations were also appeared in the spectra of physical mixture of drug with Sterculia gum and other excipients as shown inFigure 3. These observations indicated that no chemical interaction took placed between the drug and the excipients used. So, excipients mixture defined in the prototype formula can be used in formulation development as there was no drug –polymer interactions.
Figure 3.FT-IR spectrum of drug, Sterculia gum and physical mixture of tablet excipients.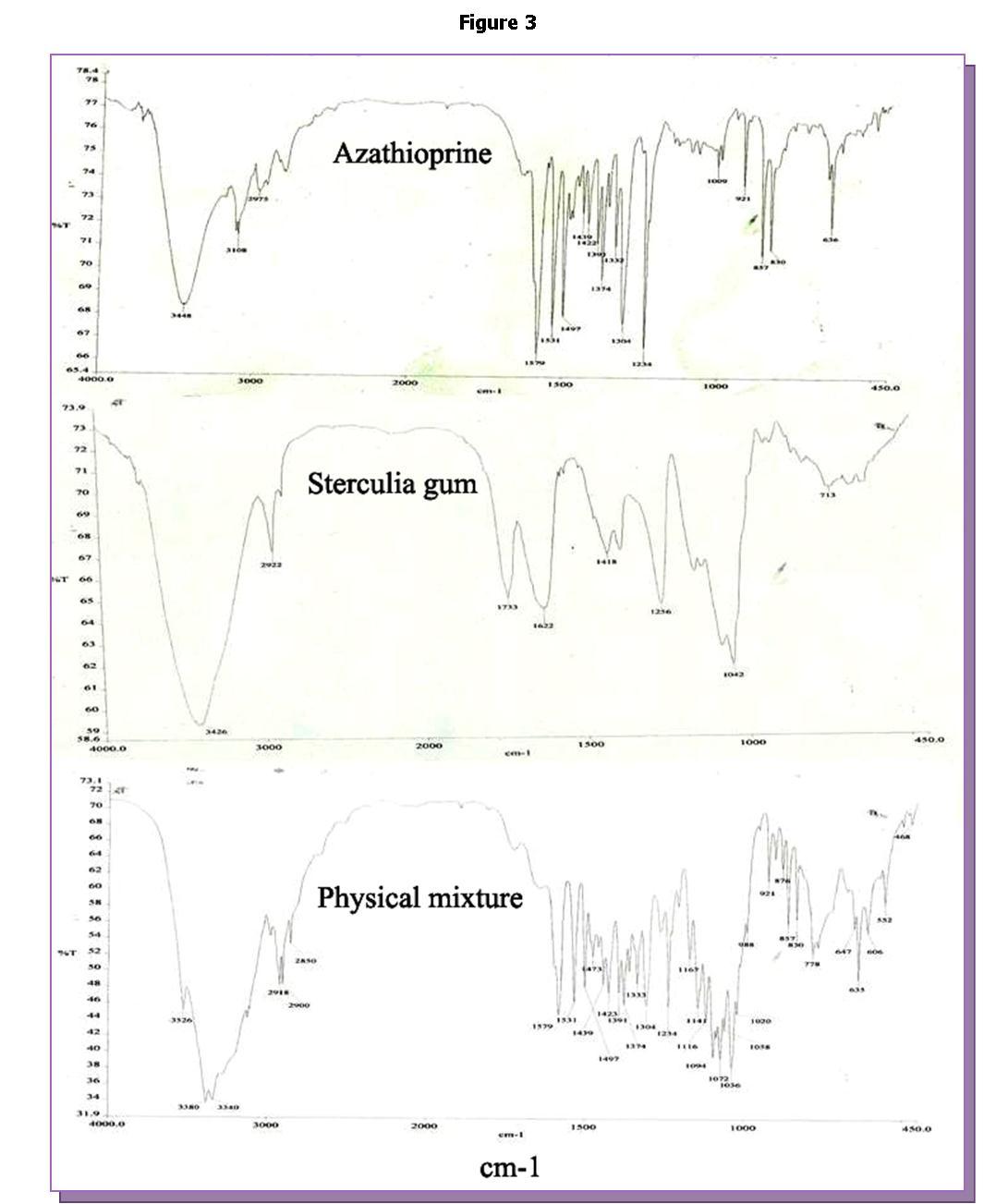
Flow Properties of the Physical Mixture of Different Factorial Batch Tablets
The bulk and tapped densities of the physical mixture of core tablet excipients were found to be in the range from 0.442 to 0.488 g/mL and from 0.531 to 0.614 g/mL respectively. The compressibility index and angle of repose values were found to be in the range from 17.17 to 21.50 % and from 21.96 to 30.37 degree respectively. The results revealed that the physical mixture of the Sterculia gum with tablet excipients has good flow properties for preparation of the tablets by direct compression method.
Formulation Aspects of Core Tablets
The composition of the factorial design batches of core tablets are presented in Table 4. The weight of each tablet was determined to be within the range of 240 ± 5 mg in order to maintain the relatively constant volume and surface area. The core tablet was prepared at average hardness of 4.0 Kg/cm2 and average diameter of 8 mm and thickness 4 mm. The incorporation of citric acid in the core composition increased the hydration of large amount of the gum and expanded its volume to great extent. The weight variation was 240 ± 2.09 and friability was less than 0.5%. Uniformity in drug content was found among different batches of the tablet and the drug content was more than 95% (Table 5).
Table 4. Composition of the core tablets under 32 factorial design| Ingredients (mg) | Formulation Code | ||||||||
| F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | F9 | |
| Azathioprine | 50.0 | 50.0 | 50.0 | 50.0 | 50.0 | 50.0 | 50.0 | 50.0 | 50.0 |
| Sterculia gum | 60.0 | 60.0 | 60.0 | 84.0 | 84.0 | 84.0 | 108.0 | 108.0 | 108.0 |
| Citric acid | 24.0 | 48.0 | 72.0 | 24.0 | 48.0 | 72.0 | 24.0 | 48.0 | 72.0 |
| Anhydrous lactose | 79.6 | 55.6 | 31.6 | 67.6 | 43.6 | 19.6 | 55.6 | 31.6 | 7.6 |
| Magnesium Stearate | 2.4 | 2.4 | 2.4 | 2.4 | 2.4 | 2.4 | 2.4 | 2.4 | 2.4 |
| Total weight | 240 | 240 | 240 | 240 | 240 | 240 | 240 | 240 | 240 |
| Formulation Code | Weight variation (mg) | Hardness (Kg/cm2) | Friability (%) | Thickness (mm) | Drug content (%) |
| F1 | 242±2.70 | 3.2 | 0.42 | 4.01±0.010 | 98.33 |
| F2 | 241±1.35 | 3.4 | 0.35 | 4.03±0.015 | 98.46 |
| F3 | 242±1.68 | 3.2 | 0.29 | 4.01±0.016 | 98.19 |
| F4 | 243±1.74 | 3.7 | 0.22 | 4.0±0.026 | 98.65 |
| F5 | 241±2.62 | 3.8 | 0.18 | 4.03±0.019 | 101.06 |
| F6 | 242±1.98 | 4.1 | 0.15 | 4.07±0.010 | 99.60 |
| F7 | 240±1.42 | 4.1 | 0.13 | 4.08±0.013 | 102.12 |
| F8 | 241±2.62 | 4.3 | 0.11 | 4.0±0.010 | 99.01 |
| F9 | 241±1.98 | 4.6 | 0.09 | 4.03±0.019 | 103.07 |
Swelling Index of the Factorial Design Batches of Core Tablets
Direct relationship was observed between swelling index and gum concentration i.e. as gum concentration was increased, swelling index was also increased. It was observed that the weight gained by the tablets was increased proportionally with time up to 6 h, after which it was decreased gradually due to erosion of the outermost gelled layer of tablet into dissolution medium up to 8 h. It was also observed that as the concentration of citric acid was increased the swelling index was also increased; this was due to the rapid solubility of citric acid which in turn increased the hydration and gelling of Sterculia gum in the tablet core, which expanded its volume contributing to the increase in swelling. The SI was found to be increased in the following order: F9>F8>F7>F6>F5>F4>F3>F2>F1. This was due to the fact that formulation containing higher concentration of Sterculia gum absorbed more volume of dissolution fluid and increased the weight of the swollen tablet.The swelling index of various factorial designed batches of tablets is presented in Table 6.
Table 6. Swelling index of factorial design batches of core tablets| Formulation code | Swelling Index | |||
| 2 h | 4 h | 6 h | 8 h | |
| F1 | 125±3.05 | 140±2.12 | 152±1.11 | 145±3.13 |
| F2 | 136±2.42 | 150±1.19 | 160±2.43 | 152±4.31 |
| F3 | 147±3.41 | 163±2.08 | 174±4.17 | 169±5.12 |
| F4 | 165±7.63 | 177±3.21 | 183±4.32 | 175±6.42 |
| F5 | 174±5.61 | 186±5.31 | 191±3.16 | 183±4.56 |
| F6 | 183±3.12 | 192±7.12 | 204±4.18 | 190± 2.34 |
| F7 | 196±5.43 | 207±4.12 | 214±7.13 | 196±1.19 |
| F8 | 205±2.31 | 215±1.31 | 224±6.12 | 205±4.43 |
| F9 | 215±1.19 | 230±2.46 | 241±1.14 | 214±1.12 |
Effects of Core Formulation Variables on In-Vitro Drug Release
In-vitro dissolution data revealed that concentration of citric acid in the core tablets has marked effect on drug release. Citric acid was incorporated as pH regulating excipient in the developed formulations and was expected to decrease the pH of the core tablets to a suitable level. Incorporation of citric acid also determines the rate at which Sterculia gum absorbs dissolution fluid and expands its volume, which pushes the drug out of the core tablet through rapid hydration and gel formation. It was evident from the results that as the concentration of citric acid was increased the drug release was also increased, due to rapid solubility of citric acid by colonic fluid which allowed the hydration of Sterculia gum, and in turn increased the swelling. In addition, incorporation of citric acid increased the ionization of basic amino groups in AZA and increased its release in dissolution media. It was observed that the release of drug was higher from the tablets of batches F2 and F3 as compared to the batch F1, which contained lesser quantity of citric acid than the batches F2 and F3. Figure 4 shows the effect of increasing concentration of citric acid on drug release from the core tablets of batch F1, F2 and F3.
Figure 4.Effects of concentration of citric acid on drug release from the core tablets of batches F1 (10% w/w), F2 (20%w/w) and F3 (30%w/w).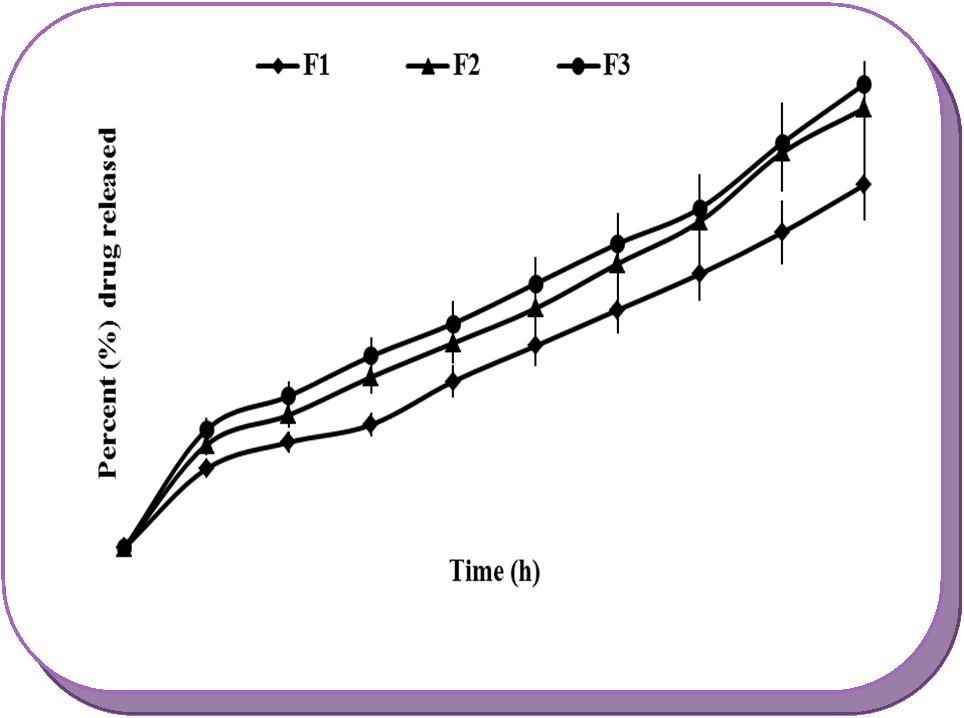
Again, keeping the amount of citric acid constant, when the amount of Sterculia gum was increased, the surprising decrease of the drug release was observed. The release of drug was slower from the tablets of batches F4 and F7 as compared to the batches F1 (F7<F4<F1). The reason for the same was that, large amount of Sterculia gum in the tablets of batches F4 and F7, upon contact with the dissolution fluid, hydrated very slowly and swelled to minimum extent and diffusion of the drug took place at a slower rate. The result revealed that at higher concentration of gum, very thick gel barrier was formed which restricted the penetration of the dissolution fluid into the tablet core to solubilize the citric acid present, which in turn took much longer time to release the drug. Figure 5 shows the effect of increasing concentration of Sterculia gum on drug release from the core tablets of batches F1, F4 and F7.These results were taken into consideration while selecting the constraint for optimization of the core tablet formula.
Figure 5.Effects of increasing concentration of Sterculia gum on drug release from core tablets of batches F1 (25%w/w), F4 (35%w/w) and F7 (45%w/w).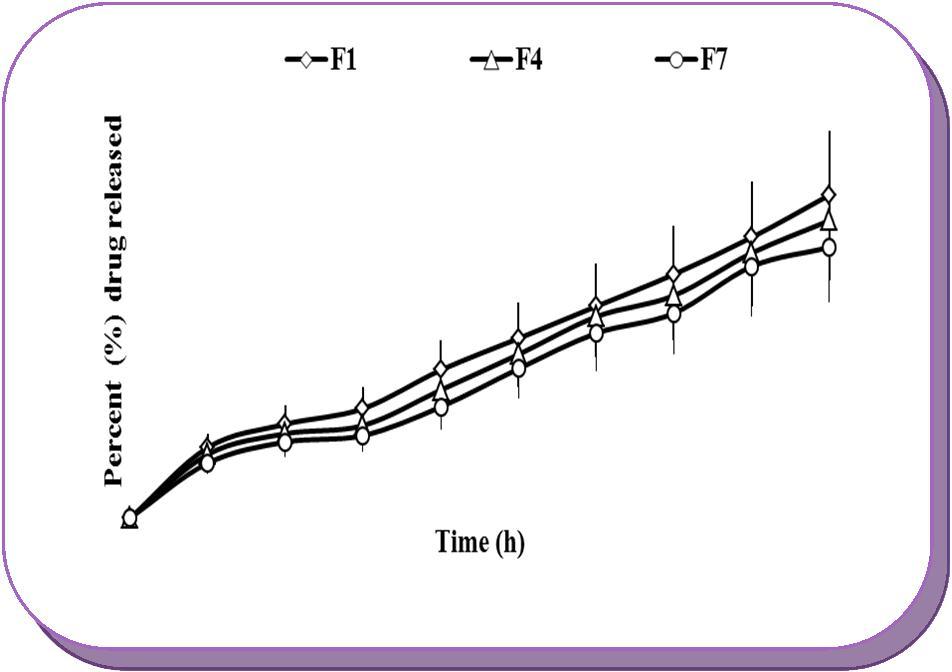
Statistical Analysis of In-Vitro Dissolution Data and Numerical Optimization
The in-vitro release profile of different factorial design batches of core tablets were fitted statistically to obtain the numerical solutions. The target responses (constraints) selected were swelling index in 2 h (more than 120 % w/w) and maximum of 80% drug release in 18 h in dissolution media, which would be the best formulation for the design of MCDDS. The software predicted that Sterculia gum at the concentration of 35.53% and citric acid at 15.15% provided the suitable controlled release property and extended the drug release up to 18 h. So, the optimized tablet (batch F10) with the composition of AZA 50 mg, Sterculia gum 85.3 mg, citric acid 36.4 mg, anhydrous lactose 65.9 mg and magnesium stearate 2.40 mg for a total weight of 240 mg was prepared and its in-vitro release rate was compared with that of the marketed formulation. Response. The observed responses 85.7% of drug release in 18 h and swelling index of 125% w/w in 2 h was found to be in good agreement with the predicted value. The dissolution profile of final optimized batch was also compared with the marketed formulation to arrive at the desired target release. Figure 6 shows the in-vitro drug release profile of the optimized formulation (F10) and marketed tablet of AZA in phosphate buffer medium at pH 7.4 containing rat caecal medium. The marketed tablet of AZA shows immediate release up to 4 hour, and maximum of 85-94% drug released in RCM within 4-6 hour duration, whereas, the optimized core extend the drug release up to 18 hour releasing more than 85% of the drug within 12 hour duration. From the stability data, the percentage drug content (ranged from 98.60 ± 0.05 to 101.92 ± 0.08%) and hardness (4.46 ± 0.28 to 4.54 ± 0.28 kg/cm2) of the core tablet (F10) at different temperature and humidity was found to be within acceptable pharmacopoeial limit. So, the optimized core tablet was found to be stable, because there was no significant change in the percentage of drug content and hardness after six month of stability study. Thus, optimized core tablet of batch F10 could be used in the design of microflora triggered colonic delivery system as the central microflora triggering layer. However, Sterculia gum based core tablet cannot prevent premature drug release in the stomach and small intestine without coating. So, the success of Sterculia gum as colon targeting carrier is achievable by coating with chitosan/Eudragit mixed blend polymers which would provide acid as well as intestinal resistance; but undergo enzymatic degradation once it reaches the colon.
Figure 6.Comparative in-vitro drug release studies of optimized batch F10 and marketed tablet of AZA in simulated colonic fluid.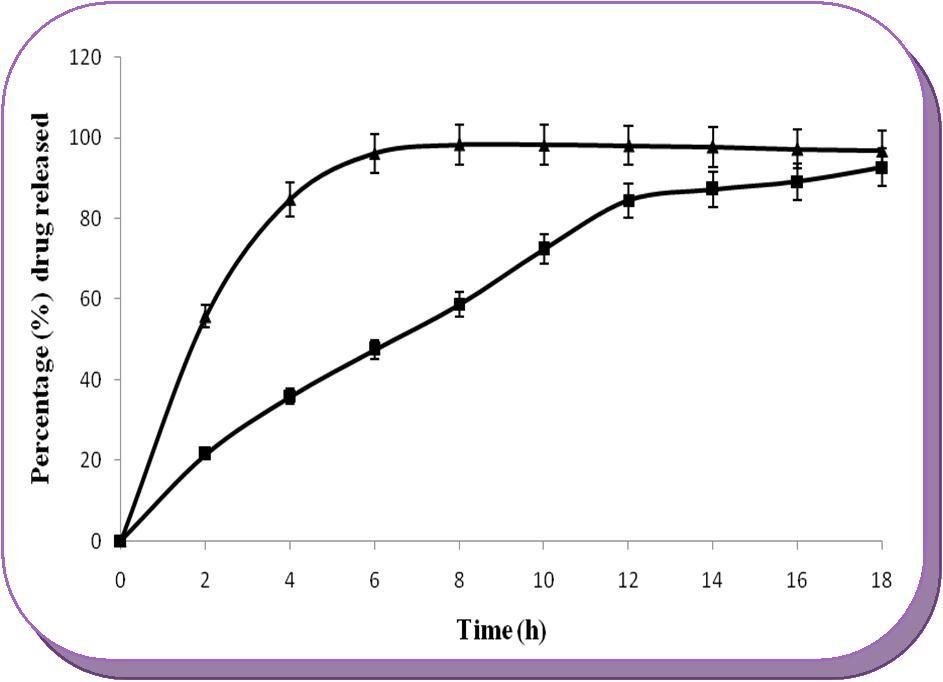
Conclusion
The sample of Sterculia gum used has conformed to the pharmacopoeial limit of its purity. Sterculia gum when treated with water swollen into a mucilaginous gel and gave high value of swelling factor. The uniformity in size distribution of the powdered gum confirmed its usefulness in giving reproducible mixtures with drug and excipients. The gum was slightly acidic in nature and highly viscous even at low concentration of the gum, which makes it suitable for preparation of hydrophilic matrix tablets. The results of microflora degradation studies of the gum in rat caecal medium revealed that Sterculia gum can be used to release drug in the colonic region by utilizing the action of enterobacteria. Results of the IST showed that there was no degradation of samples of pure drug, the drug polymer mixture and other tablet excipients. DSC study has proved the compatibility of the drug with Sterculia gum and other tablet excipients. FT-IR study further confirmed that there was no interaction between drug, Sterculia gum and tablet excipients. Sterculia gum exhibits high swelling index due to its rapid hydration and gelation property in dissolution media, which in turn gives premature drug release in the upper GIT without coating and may not reach to the colonic region. However, the swelling property of the polymer can be used to produce hydrostatic pressure inside the tablet if it is coated with semipermeable membrane and can be used for targeting drugs to the colon. The swelling force of the Sterculia gum could concurrently drive the drug out of the polysaccharide core due to the rupture of the mixed film coating under colonic microflora environment.
Acknowledgements
The authors greatly acknowledge Indian Institute of Technology (IIT), Gauhati, India, to carry out FT-IR and DSC studies. The authors are also thankful to RPG Life Sciences Pvt. Ltd. Mumbai, India for providing azathioprine as a gift sample.
References
- 6.Hocking G M.(1897) Sterculia foetida. A dictionary of natural products, 2nd Ed.Plexus Publishing Inc. , Medford, NJ,1997,pp.754–55
- 11.Pharmacopoeia British. (2009) General Notices and Monograph, Powdered sterculia and sterculia gum, thirty fourth ed. , British Pharmacopoeial Commission, London (U. K)
Cited by (1)
This article has been cited by 1 scholarly work according to:
Citing Articles:
