Identification and Expression Analysis of Fragaria Vesca MLO Genes Involved in Interaction with Powdery Mildew (Podosphaera Aphanis)
Abstract
Strawberry powdery mildew, caused by Podosphaeraaphanis is a major fungal disease that affects strawberry yield and quality. In the model plant species Arabidopsis and the crop plants barley, tomato and pea, the Mildew resistance locus O (MLO) proteins have been found to be required for powdery mildew susceptibility. The present study, based on the sequence of a wild plum (Prunus americana) MLO protein, identified 16 MLO genes within the genome of woodland strawberry, Fragaria vesca and examined their expression pattern in response to powdery mildew infection in three diploid strawberry cultivars. Phylogenetic analysis showed that the FvMLO genes can be classified into six clades. Four FvMLO genes were grouped into clade III, which comprises MLO genes from Arabidopsis, tomato and grapevine that mediate powdery mildew susceptibility. A RNA-seq analysis of two diploid strawberry cultivars, F. vescassp. vesca accession Hawaii 4 (HW) and F. vesca f. semperflorens line “Yellow Wonder 5AF7” (YW) at 1 d (1 DAI) and 8 d (8 DAI) after infection showed the expression of 12 out of the 16 FvMLO genes. The comparison of Fragments Per Kilobase of transcript per Million mapped reads (FPKM values) detected by RNA-seq and expression values of qRT-PCR for FvMLO genes showed substantial agreement. The FvMLO3 gene, which was grouped in clade III and orthologous to the Arabidopsis,tomato and grapevine genes, was highly expressed in YW compared to other FvMLO genes across varieties. The results showed that FvMLO genes can be used as potential candidates to engineer powdery mildew resistance in strawberry based on MLO suppression or genome editing.
Author Contributions
Academic Editor: Cumali Keskin, Artuklu University,School of Health
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Shridhar Jambagi, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
The mildew resistance locus o (MLO) genes encode a specific class of plant-specific proteins that possess seven transmembrane helices and a C-terminal calmodulin-binding domain 1, 2. An MLO gene was first identified in barley 3 and currently, a varying number of MLO genes have been identified in Arabidopsis, Medicago truncatula, chickpea, rice, maize, wheat, soybean, cucumber, tomato, rose, grapevine, peach, apple, sweet orange, cultivated Solanaceae, Brachypodium, and Cucurbitaceae species respectively 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18. Although the biological functions of MLO genes are not yet completely known, they act as suppressors of defense responses.
The mutant MLO genes confer durable and broad spectrum powdery mildew resistance in monocots and dicots and the functions of these genes are highly conserved in the plant powdery mildew interactions. In barley, the presence of HvMLO is an absolute requirement for the powdery mildew fungi to penetrate the host cell wall successfully 19, whereas the recessive loss-of-function alleles of MLO gene showed durable and broad spectrum resistance to these pathogens by forming cell-wall appositions 20, 21, 22, 23. Recently, Acevedo Garcia et al. 24 identified simultaneous mutations in the three wheat homologues of TaMlo, TaMlo-A1, TaMlo-B1 and TaMlo-D1 and produced homozygous triple and double mutant lines that showed increased resistance to powdery mildew. In another study, the knock-down of grapevine MLO genes (VvMLO7 in combination with VvMLO6 and VvMLO11) decreased powdery mildew severity up to 77 %25. Like barley mlo-mutants, the A. thaliana mlo2 mutants exhibited resistance to multiple powdery mildew species 26 and also the same defense response was identified in the tomato Slmlo1 mutant 27. Antisense down-regulation of RhMLO1 enhances resistance to powdery mildew in Rosa multiflora28, and RNAi methods have been used to knock-down expression of selected MLO genes and reduce susceptibility to powdery mildew in strawberry 29, apple 30, and petunia 31. Previously, the loss-of-function mutations in PsMLO1 showed durable and broad-spectrum resistance to powdery mildew in pea plants 32, and a transposable element insertion in the susceptibility gene CsMLO8 results in hypocotyl resistance to powdery mildew in cucumber 33. In recent studies, genome editing technologies, TALEN (Transcription activator-like effector nucleases) and CRISPR-Cas9 (Clustered regularly interspaced short palindromic repeats-associated
protein-9 nuclease), were used in hexaploid bread wheat to introduce targeted mutations in three TaMLO homoeoalleles which resulted in heritable resistance to powdery mildew 34. The MLO genes therefore serve as good candidate genes for genetic engineering strategies to impart resistance against powdery mildew pathogens.
Podosphaeraaphanis (syn. Sphaerothecamacularis f. sp. fragariae) infection of the cultivated strawberry is a serious problem in strawberry production worldwide, and affects yield and quality. About 50% of strawberries are grown under protected conditions in most parts of the Europe; this leads to persistent powdery mildew problems, since the humid conditions in green/glasshouse is greatly favourable to the pathogen35. The application of systemic fungicides to control powdery mildew is expensive and is limited by legal restrictions in most parts of the world. For this reason, genetic strategies offer an alternative for controlling disease by the identification of desired genes, and their introgression and pyramiding in new genotypes. The genetic source of powdery mildew resistance in strawberry is not well studied and the introduction of new resistant cultivars has been limited 36. These authors used SSR markers for screening of resistant and susceptible seedlings, but none of the markers co-segregated with powdery mildew resistance. The results underlined the difficulty to investigate the allelic linkage for powdery mildew resistance and suggested that efforts should be made to determine linkages in the diploid genome and then further adapt to the octoploid genome.
The available genome sequence of the diploid strawberry, Fragaria vesca ssp. vesca accession Hawaii 4 37 allows a comprehensive overview of the strawberry MLO gene family. A few studies have previously reported the identification of FvMLO genes in the F. vesca genome sequence 15, 38; however, the level of expression of these genes in response to disease pressure has not been studied. In the present study, we identified FvMLO genes in the F. vescagenome based on the sequence information of the Prunus americana MLO protein and analyzed their expression levels in response to powdery mildew pathogen in three diploid strawberry cultivars, F. vesca f. vesca (Fv), F. vesca ssp. vesca accession Hawaii 4 (HW), and a highly inbred line, “Yellow Wonder 5AF7 (YW5AF7)” F. vesca f. semperflorens (YW), and Eluica, a susceptible variety of the octoploid cultivated octoploid strawberry Fragaria × ananassa. The resistance level of Fv and HW to powdery mildew is not known, whereas YW is reported to be susceptible to powdery mildew 39. This paper describes the identification and phylogenetic relationship of FvMLO genes, and transcript analysis of these genes in response to P. aphanis infection using RNA-seq data and qRT-PCR. In future studies, FvMLO genes will serve as potential candidates to produce strawberry varieties resistant to powdery mildew.
Materials and Methods
In Silico Identification of MLO Predicted Proteins in Strawberry and Primer Design
MLO protein sequences in F. vesca were identified by using a MLO sequence from the related Rosaceous species Prunus americana by using the Basic Local Alignment Search Tool (BLAST) of the strawberry genome tools (https://www.rosaceae.org/blast). Multiple sequence alignment was carried out with ClustalW to search for conserved regions and primers were designed using primer3 software. Primer sequences are listed in Table S1.
Phylogenetic Analysis
For phylogenetic analysis of MLO proteins, a selection of known monocot and dicot MLO protein sequences downloaded from NCBI GenBank were aligned with the FvMLO protein sequences using ClustalW with default settings 40. The aligned sequences were used to generate a phylogenetic tree using Neighbour-Joining 41 method in MEGA4 42. All positions containing gaps and missing data were eliminated (Complete deletion option). The bootstrap value was calculated with 500 replicates to support the branching arrangements.
Plant Material and Powdery Mildew Infection
Three diploid strawberry cultivars, Fv, HW(originally provided by East Malling Research, UK), and YW (seed provided by Dr Janet Slovin, Beltsville, USA), and two octoploid varieties, Emily and Eluica (plants provided by East Malling Research, UK) were used in the present study. The diploid plants were grown in hydroponic medium as previously described by Bindschedler, et al. 43 and maintained in a Fisons growth chamber at a temperature of 20°C with 65% humidity, and a 10 h photoperiod with a photosynthetic photon flux density of 250-260 μmol/m2/s. The octoploid plants were maintained in a glasshouse under natural day length conditions in pots containing potting compost. Leaf material from an Eluica cultivar infected with powdery mildew were used to infect four week old F.vesca plants with P. aphanis by gently tapping conidia from infected leaves above the experimental plants. The leaves (100 mg) from five randomly selected plants were harvested from fully grown healthy (control) plants and infected plants at 1 d and 8 d after infection (DAI), frozen in liquid nitrogen, and stored at -80˚C until use.
RNA Sequencing and Data Analysis
We used RNA-seq data that was generated previously in our laboratory for two diploid cultivars, HW and YW at control, 1 d and 8 d after powdery mildew infection 44. We manually searched for the expression of FvMLO genes in each library and absolute FPKM values were used to analyze their pattern of expression.
Semiquantitative and Quantitative Real-Time PCR Analysis
To examine the expression of FvMLO genes in different strawberry cultivars, total RNA was isolated from the control and 8 DAI frozen leaf samples using an RNAqueous® Kit (Ambion, UK) and then subjected to DNAse treatment with Turbo DNA-free kit (Ambion, UK). First-strand cDNA was synthesized from total RNA using SuperScript™ III First-Strand Synthesis SuperMix for qRT-PCR Kit (Invitrogen, UK). For qRT-PCR analysis, the first experiment was conducted using a semiquantitative RT-PCR evaluating the expression of all the identified FvMLO genes at 8 DAI in all strawberry cultivars. The qRT-PCR was performed on a Rotor-Gene 6000 (Corbet Life Science, UK) by using the sensiMix™ SYBR No-ROX Kit (Bioline, UK) and gene specific primers. The strawberry gene FaGAPDH2 (AF421145) was used as a reference gene, the primer,
forward: 5’-CAGACTTGAGAAGAAGGCCACCTA-3’ reverse: 5’- GATACCCTTCATCTTTCCCTCAGA-3’. PCR mixtures (20 µL total volume) contained 10 µL 2x SensiMix™ SYBR® No-ROX, 0.5 µL each forward and reverse primer (10 µM), 1 µL cDNA template and 8 µL Molecular Biology Grade water (Accugene, UK). A negative control reaction without cDNA template was included to monitor the non-specific binding. The thermal cycling parameters were one cycle at 95˚C for 10 min, 39 cycles of 95˚C for 15 s, 60˚C for 30 s, and 72˚C for 1 min. A standard curve was generated for each run using a cDNA template dilution series and output data were analyzed using the software provided. A Two standard curve method was employed for the analysis of real-time quantitative PCR results. The melting curve analysis was carried out from 50 to 90 °C with a hold of 1 min for every 1 °C. The expression level of FvMLO genes in control leaves was used as a control to compare the relative expressions of the genes in 8 d infected leaves. The relative expression levels of the FvMLO genes were calculated using the 2− CT method 45.
Results
In Silico Identification of F. Vesca MLO Genes
Homology search of the published genome sequence of the strawberry against the known MLO sequence from P. americana was performed to identify the number of MLO genes in the F. vesca genome. Initially, seventeen genes were identified as putative members of the MLO gene family; these were designated as FvMLO genes (FvMLO1 to FvMLO17). However, based on the chromosomal location of FvMLO genes, FvMLO4 was found to be an isoform of the FvMLO3 gene and therefore, the former was excluded in the phylogenetic analysis (see next paragraph). The information of FvMLOs, including accession numbers, and their chromosomal locations are listed in Table 1.
Table 1. The FvMLO genes as predicted in Fragaria vesca genome sequence| Gene | Chromosome location | Transcript name |
|---|---|---|
| FvMLO1 | LG1: 7017413 - 7022793 | mrna13023.1-v1.0-hybrid |
| FvMLO2 | LG5: 20075140 - 20093212 | mrna31488.1-v1.0-hybrid |
| FvMLO3 | LG6: 8563501 - 8567082 | mrna09653.1-v1.0-hybrid |
| FvMLO4 | LG6: 8563518 - 8566426 | mrna09653.1-v1.0-hybrid |
| FvMLO5 | LG7: 14517490 - 14520807 | mrna23198.1-v1.0-hybrid |
| FvMLO6 | LG3: 10707557 - 10713141 | mrna03210.1-v1.0-hybrid |
| FvMLO7 | LG2: 15724387 - 15725931 | mrna10558.1-v1.0-hybrid |
| FvMLO8 | LG5: 20171268 - 20174872 | mrna31498.1-v1.0-hybrid |
| FvMLO9 | LG3: 17105660 - 17112982 | mrna28541.1-v1.0-hybrid |
| FvMLO10 | LG6: 8541201 - 8546252 | mrna09651.1-v1.0-hybrid |
| FvMLO11 | LG7: 16370664 - 16374969 | mrna26428.1-v1.0-hybrid |
| FvMLO12 | LG5: 19055228 - 19057796 | mrna29285.1-v1.0-hybrid |
| FvMLO13 | LG1: 697457 - 707892 | mrna10166.1-v1.0-hybrid |
| FvMLO14 | LG3: 4660882 - 4663962 | mrna29770.1-v1.0-hybrid |
| FvMLO15 | LG3: 25834627 - 25838931 | mrna10346.1-v1.0-hybrid |
| FvMLO16 | LG3: 9515163 - 9524099 | mrna31264.1-v1.0-hybrid |
| FvMLO17 | LG1: 7820289-7827918 | mrna14592.1-v1.0-hybrid |
Phylogenetic Analysis of the F. vesca MLO Genes
A phylogenetic analysis was performed with the amino acid sequences encoded by the 16 FvMLO genes together with a set of 17 VvMLO protein sequences from grapevine 9, 46 and selected MLO proteins from barley, rice, wheat, maize, tomato, A. thaliana, P. americana and M. toringoides. The analysis identified six distinct clades and also showed that F. vesca MLO family members were distributed between all clades (Figure 1). Clade V comprises the monocot MLO proteins previously characterized to be involved in powdery mildew susceptibility 2, 20, 47. The FvMLO8protein was found to be closely related to VvMLO14 protein and clustered within clade V; this finding is noticeably different from the general conclusion that this clade contains only monocot homologues 8. The barley HvMLO protein involved in powdery mildew susceptibility is orthologous to wheat TaMLO-1 and rice OsMLO2 proteins, induce powdery mildew resistance in barley mlo-mutants 47. Of the 16 FvMLO proteins analyzed, four (FvMLO3, FvMLO5, FvMLO11 and FvMLO14) clustered with the dicot MLO proteins in clade III. This clade is important in terms of putative function as this group contains the MLO proteins from A. thaliana (AtMLO2, AtMLO6, and AtMLO12), tomato (SlMLO1) and grapevine (VvMLO3,VvMLO4andVvMLO17) that have been previously reported to be induced during powdery mildew infection 9, 26, 27. This allowed for the analysis of the F. vesca MLO proteins for domains that are conserved between all proteins responsible for mildew susceptibility. Figure 2 clearly demonstrates that the protein sequences (1 to 100 and 501 to 540) are highly conserved among all members of clade III at the position of the predicted transmembrane domains and the calmodulin-binding sites, which are important features of this protein family.
Figure 1.Phylogenetic relationship of MLO proteins inferred using Neighbouring-Joining method
Figure 2.Multiple sequence alignment of FvMLO proteins. Multiple sequence alignment of FvMLO proteins in clade III with selected MLO proteins that have been shown to be involved in powdery mildew susceptibility in Arabidopsis (AtMLO2, AtMLO6 and AtMLO12; Consonni et al., 2006; Consonni et al., 2010), grape (VvMLO3, VvMLO4, VvMLO6, VvMLO9, VvMLO13 and VvMLO17; Feechan et al., 2009) and tomato (SlMLO1; Bai et al., 2008). The alignment was generated by CLC Sequence Viewer 6.6.2. The positions of the seven transmembrane domains (Devoto et al., 1999) are indicated by black arrows and the position of the calmodulin-binding site (Panstruga, 2005) is indicated by a grey bar above the sequences.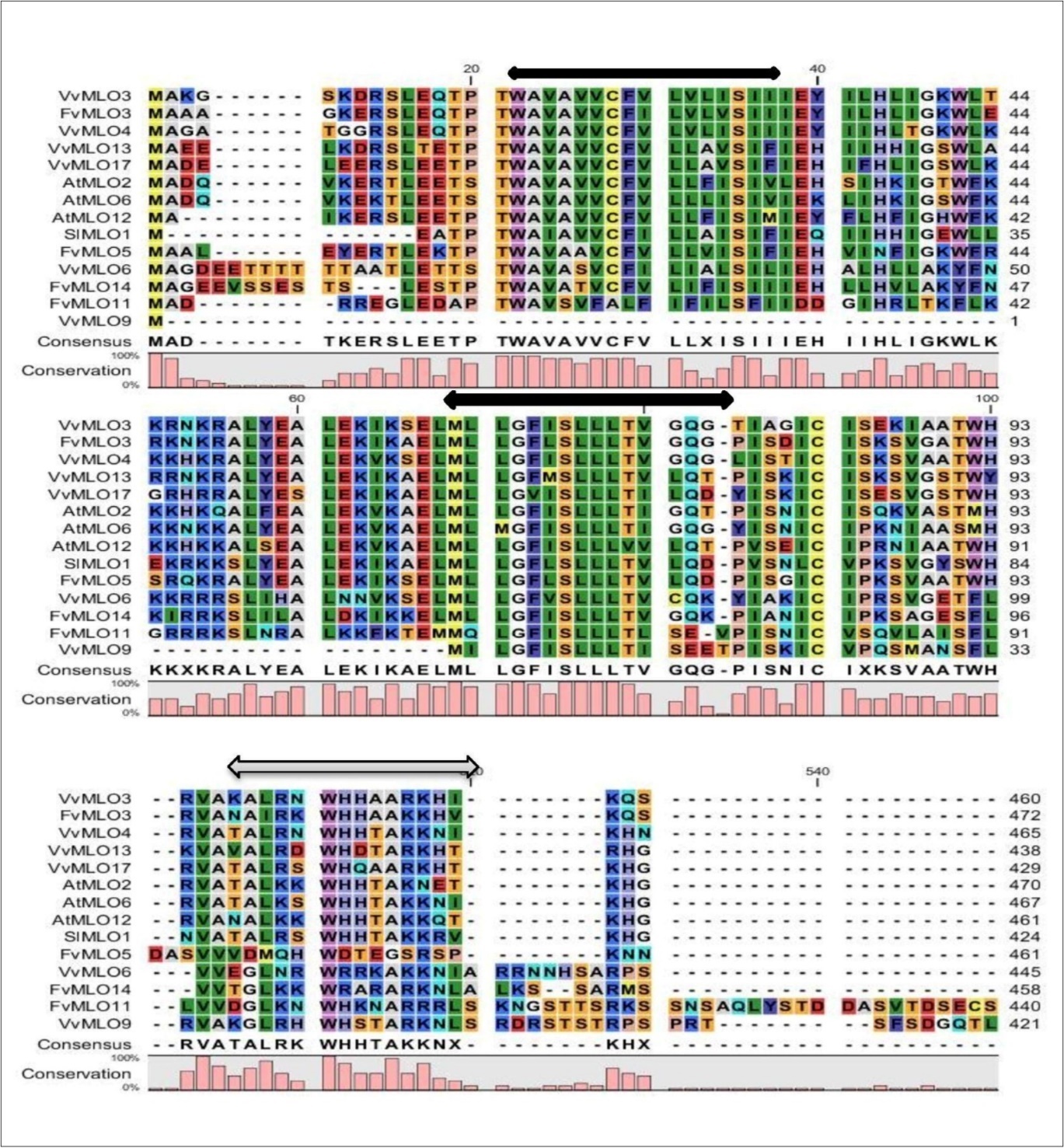
Expression Analysis of FvMLO Transcripts using RNA-Seq Data
In our previous study 44, RNA-seq data was generated for two diploid strawberry cultivars, HW and YW, in response to powdery mildew infection. Global transcriptome analysis of two F. vesca cultivars of healthy (control) and infected leaves (1 DAI and 8 DAI) allowed us to identify the transcriptional changes during P. aphanisinfection. In the present study, RNA-seq data were used to analyze the level of expression of all 16 identified FvMLO genes in HW and YW between control and 1 DAI, and control and 8 DAI. Based on the FPKM values, only 12 FvMLO genes that are grouped into different putative clades in the phylogenetic tree were expressed at all stages; these included gene13023 (FvMLO1), gene09653 (FvMLO3), gene23198 (FvMLO5), gene10558 (FvMLO7), gene28541 (FvMLO9), gene09651 (FvMLO10), gene29285 (FvMLO12), gene10166 (FvMLO13), gene29770 (FvMLO14), gene10346 (FvMLO15), gene31264 (FvMLO16) and gene14592 (FvMLO17). By using the RNA-seq based absolute fold change values, the transcript levels of all 12 FvMLO genes showed a steady increase in response to P. aphanis infection between control and 1 DAI in both HW and YW with fold change values ranging from 0.5 to 3.6 (Figure 3A). Three genes, FvMLO3, FvMLO10 and FvMLO15 showed much higher levels of expression with fold change values of about 30, 4.5 to 7.3, and 13 to 20 between control and 8 DAI in both HW and YW (Figure 3B). The time course between control and 1 DAI, and control and 8 DAI clearly showed an increase in expression of FvMLO genes.
Figure 3.Graph showing the expression of 12 FvMLO genes based on RNA-seq absolute fold change values in HW and YW between control and 1 DAI (A), and control and 8 DAI (B).
Expression Analysis of FvMLO Genes by qRT-PCR
The level of expression of FvMLO genes was analyzed in three biological replicates using qRT-PCR in leaf tissue of the three diploid strawberry cultivars, Fv, HW, and YW, and one octoploid variety, Eluica, between control and 8DAI.
Initial analysis by semiquantitative RT-PCR showed the expression of 12 out of the 17 FvMLO genes (FvMLO1, FvMLO3, FvMLO4, FvMLO5, FvMLO7, FvMLO9, FvMLO10, FvMLO12, FvMLO13, FvMLO14, FvMLO16 and FvMLO17). Among the 12 expressed FvMLO genes, FvMLO3 showed expression only in infected leaves, whereas FvMLO14 showed expression only in control leaves of all strawberry varieties (Figure 4). Expression of the other 10 FvMLO genes varied widely between all varieties in both control and infected leaves.
Figure 4.Semiquantitative RT-PCR products of FvMLO genes and reference gene (FaGAPDH2) in control and infected (8 DAI) leaves of four strawberry varieties. 1-4: Fv, YW, HW and Eluica (control), 5-8: Fv, YW, HW and Eluica (infected).
The expression levels of all of the 12 FvMLO genes were further investigated using the qRT-PCR method. The genes that did not show expression in the initial analysis of semiquantitative RT-PCR method were not included in the qRT-PCR expression analysis. In measuring the relative expression level, the PCR efficiencies and coefficients of correlation (R2 value) of reference (FaGAPDH2) and target (FvMLO) genes ranged from 0.89 to 0.97 and from 0.98 to 0.99, respectively (Figure S1). The cDNA synthesized from total RNA extracted from Emily (resistant) octoploid plant was used as a standard. The transcript levels of individual FvMLOgenes were normalized against the transcript levels of FaGAPDH2 in each cDNA sample. The absolute expressions of FvMLO genes in control and infected leaves of all strawberry varieties are shown in Table S2. During powdery mildew infection, two genes FvMLO7 and FvMLO14 showed low level of expression among all varieties, whereas six genes FvMLO1, FvMLO3, FvMLO9, FvMLO10, FvMLO13 and FvMLO16 were upregulated but the level of relative expression across varieties was significantly different (Figure 5). The expression level of FvMLO1, FvMLO3 and FvMLO13 genes were higher in YW and Eluica, whereas FvMLO9 showed higher expression in Eluica, FvMLO10 in YW and HW, FvMLO16 in HW and Eluica. The level of expression of four genes, FvMLO4, FvMLO5, FvMLO12 and FvMLO17 varied widely across varieties in response to infection. The FvMLO4 gene, which was found to be an isoform of FvMLO3, was downregulated in Fv but highly expressed in YW, HW and Eluica. The expression of FvMLO5 was lower in Fv and HW, whereas it was higher in YW and Eluica. FvMLO12 and FvMLO17 showed low levels of expression in Fv and YW but were highly expressed in HW and Eluica. Among all the genes expressed across varieties in response to infection, FvMLO3 showed a very high level of expression (164 fold) in YW. As far as inter-varietal response to infection is concerned, the genotypes exhibited a differential expression pattern of FvMLOs. In the Fv genotype, all FvMLO genes except FvMLO9 were down-regulated under infected conditions, whereas in HW (except FvMLO5, FvMLO7 and FvMLO14), YW (except FvMLO7, FvMLO12, FvMLO14 and FvMLO17) and Eluica (except FvMLO7 and FvMLO14) all FvMLOs showed higher expression in infected plants compared to control.
Figure 5.Bar chart showing relative expression levels of 12 FvMLO genes in four strawberry varieties in response to P. aphanis infection. Graph shows fold increase/decrease in expression under infection over control. All values are compared with control value=1. Values are shown as mean ± standard deviation (n=2). Fv: F. vesca f. vesca, YW: F. vesca f. semperflorens line “Yellow Wonder”, HW: F. vesca ssp. vesca accession Hawaii 4 and Eluica: Susceptible octoploid strawberry variety. Different letters (a, b, c, d) denote significant difference in level of expression across varieties.
Comparison of RNA-Seq and qRT-PCR Based FvMLO Genes Expression
RNA-seq based expression of FvMLO genes in response to P. aphanisinfection was compared with the qRT-PCR based expression levels of FvMLO genes in two F. vescacultivars, HW and YW. The comparison showed that the FvMLO4 gene was expressed in semiquantitative RT-PCR but was not expressed in the RNA-seq experiment. Additionally, RNA-seq data showed the expression of FvMLO15, which was not expressed in semiquantitative RT-PCR. Overall, the transcripts FPKM values detected by RNA-seq and qRT-PCR analysis absolute expression values in HW and YW revealed substantial agreement in the extent of the powdery mildew-induced variation in transcript accumulation for the FvMLO genes (Table 2, Figure 3 and Figure 5).
Table 2. Comparisons of transcripts FPKM values detected by RNA-seq and expression values (mean ± standard error) of qRT-PCR analysis for the FvMLO genes in HW and YW| Gene | HW | YW | ||||||||
| RNA-seq (FPKM) | qRT-PCR (Mean ± SE) | RNA-seq (FPKM) | qRT-PCR (Mean ± SE) | |||||||
| Control | 1 DAI | 8 DAI | Control | 8 DAI | Control | 1 DAI | 8 DAI | Control | 8 DAI | |
| gene13023 (FvMLO1) | 7.53 | 6.52 | 10.07 | 0.57±0.14 | 1.05±0.11 | 6.67 | 7.03 | 11.64 | 0.66±0.10 | 1.78±0.19 |
| gene09653 (FvMLO3) | 0.12 | 0.38 | 3.96 | 15.80±3.13 | 55.76±7.55 | 0.16 | 0.57 | 4.77 | 0.76±0.13 | 123.49±21.33 |
| gene23198 (FvMLO5) | 14.12 | 16.76 | 13.08 | 12.48±4.99 | 10.84±1.67 | 23.02 | 17.56 | 17.95 | 3.35±1.12 | 16.21±1.88 |
| gene10558 (FvMLO7) | 63.49 | 47.46 | 45.17 | 0.84±0.10 | 0.44±0.14 | 78.01 | 57.98 | 45.54 | 1.83±0.37 | 0.57±0.14 |
| gene28541 (FvMLO9) | 12.91 | 15.39 | 32.31 | 1.67±0.55 | 2.90±0.81 | 11.24 | 12.16 | 22.35 | 0.83±0.11 | 2.18±0.37 |
| gene09651 (FvMLO10) | 1.69 | 4.79 | 12.47 | 2.37±0.45 | 144.30±10.94 | 1.86 | 2.85 | 8.49 | 1.37±0.27 | 78.77±11.34 |
| gene29285 (FvMLO12) | 87.71 | 46.49 | 41.05 | 0.14±0.03 | 0.98±0.24 | 86.62 | 52.78 | 42.94 | 0.33±0.06 | 0.15±0.01 |
| gene10166 (FvMLO13) | 10.00 | 12.63 | 31.42 | 17.33±4.03 | 20.53±1.66 | 10.12 | 10.91 | 26.31 | 2.86±0.09 | 19.37±2.63 |
| gene29770 (FvMLO14) | 0.30 | 0.38 | 0.37 | 1.18±0.29 | 0.06±0.04 | 0.27 | 0.49 | 0.67 | 0.31±0.03 | 0.14±0.01 |
| gene10346 (FvMLO15) | 0.27 | 0.16 | 5.55 | - | - | 0.33 | 0.30 | 4.58 | - | - |
| gene31264 (FvMLO16) | 3.09 | 3.11 | 2.69 | 0.25±0.07 | 5.33±0.81 | 2.92 | 3.74 | 3.00 | 1.65±0.23 | 1.69±0.31 |
| gene14592 (FvMLO17) | 21.53 | 21.92 | 21.49 | 0.08±0.03 | 0.25±0.03 | 21.12 | 22.64 | 19.66 | 0.17±0.00 | 0.09±0.01 |
Discussion
Our study focused on identification of MLO genes in sequenced Fragaria genome by BLAST search using sequence information of a Prunus americana MLO protein and expression analysis of FvMLO genes in three diploid (Fv, HW and YW) and one octoploid (Eluica) strawberry cultivars. The in silico analysis allowed the identification of 16 FvMLO genes and the phylogenetic analysis of FvMLO proteins performed with MLO family members of grapevine, Arabidopsis, and selected monocot and dicot MLO proteins revealed that the proteins were grouped into six distinct clades, two of which (III and V) comprise members that are associated with powdery mildew susceptibility in dicots and monocots, respectively. In this study, it was observed that the number of FvMLO genes and clade numbering of the FvMLOgene family were in contrast to the previously categorized FvMLO genes. Pessina, et al. 15 identified 17 FvMLO genes (FvMLO1-FvMLO17) based on sequence information of tomato SlMLO1 amino acid and HMMER programme, whereas Miao, et al. 38 reported 20 MLO genes in the strawberry genome (FvMLO01-FvMLO20) using Arabidopsis thaliana and rice MLO protein sequences. Further, the phylogenetic analysis in their studies showed that MLOs were grouped into seven distinct clades. These results may possibly indicated that the number of FvMLO genes and clades varied according to the query type, target database type, and stringency used for the blast search to identify MLO genes, and also the specific MLO family members of selected crop species used for phylogenetic analysis.
A previous study comparing MLO sequences from grapevine with those from monocots and dicots supported our phylogenetic results 9. Among four FvMLOs of clade III, the FvMLO3 protein sequence is highly conserved in the positions of transmembrane domains 8 and calmodulin-binding site 19 with other members in the clade and therefore this gene serves as a potential candidate gene in modulating antifungal defense response. Further, FvMLO8 was clustered with monocot MLO proteins in clade V, which includes HvMLO, TaMLO1 and OsMLO2 that have been demonstrated to be required for powdery mildew susceptibility 47. However, the finding that FvMLO8 was not expressed in RNA-seq and semiquantitative RT-PCR analysis suggests that this protein is not likely to be involved in powdery mildew pathogenicity. The grouping of FvMLO8, a dicot MLO, into a clade that only contains monocot MLOs, raises a question about the accuracy of the published sequence for FvMLO8 (Transcript name: mrna31498.1-v1.0-hybrid). Similar conclusions were made by Feechan, et al. 9 for the clustering of the VvMLO14 protein into the same clade. Furthermore, the FvMLO11 grouped in clade III was not expressed in the RNA-seq data and semiquantitative RT-PCR.
Although, FvMLO genes have been identified previously, their expression levels were not examined in strawberry in response to powdery mildew infection. In our previous study, transcriptome analysis of two diploid strawberry varieties (HW and YW) upon P. aphanis infection provided access to a large data set and enabled new insights into the identification of genes expressed during powdery mildew interaction 44. RNA-seq analysis of FvMLO genes showed the expression of 12 FvMLO genes in HW and YW that are distributed in different clades. Comparison of RNA-seq based FvMLO genes expression with qRT-PCR exhibited significant correlation between the two different techniques except for FvMLO4 and FvMLO15 for which more detailed analysis is needed. Two genes, FvMLO3 and FvMLO10, which showed high expression levels in RNA-seq data analysis, were in considerable agreement with the qRT-PCR based expression pattern for these two genes. The FvMLO3 gene is particularly important because it is grouped in clade III along with the AtMLO2, AtMLO6, AtMLO12, SlMLO1, VvMLO3, VvMLO4 and VvMLO17 genes that have been previously reported to be induced by powdery mildew infection 9, 26, 27. Two other genes, FvMLO5 and FvMLO14 that are also grouped in clade III showed the same pattern of expression in HW and YW upon infection compared between RNA-seq data and qRT-PCR results. The FvMLO5 expression level was lower in HW but higher in YW, whereas FvMLO14 was found to be lowly expressed in both HW and YW in response to infection.
The level of relative expression of FvMLOs in all the genotypes varied significantly. In Eluica and YW, most of the FvMLO genes were highly expressed compared to Fv and HW (based on qRT-PCR and RNA-seq data). These results indicated that the expression of FvMLOs could possibly be responsible for the genotypes Eluica and YW being more susceptible to powdery mildew infection, as reported in previous studies. The results for YW in this study were in agreement with the report published by Slovin, et al. 39 that YW is susceptible to powdery mildew, thrips and aphids in a greenhouse environment. Similarly, Eluica is known to be a susceptible octoploid variety as confirmed by East Malling Research, UK. However, the resistance level of the other Fragaria spp. is not known [36] and further functional analysis should be conducted to test the role of these FvMLO genes in the susceptibility of strawberry varieties to P. aphanis.
Conclusion
The results of our study will allow for future strategies to generate powdery mildew resistant strawberry varieties. Previously, it has been reported that loss-of-function mlo-mutants powdery mildew resistance in A. thaliana and tomato 27, 48. Similarly, Jiwan, et al. 29 reported that the antisense expression of peach MLO gene PpMlo1 in transgenic strawberry plants reduced susceptibility to powdery mildew. Therefore, FvMLO genes serve as potential candidates to produce strawberry varieties resistant to powdery mildew. If resistance in strawberry can be achieved through silencing a single FvMLO gene, as in tomato, it would then be possible to look for naturally occurring mutant alleles within F. vesca germplasm collections that could be used in marker-assisted selection to produce progeny that are homozygous recessive at this locus. Conversely, if complete powdery mildew resistance in strawberry is only attained through the silencing of more than one FvMLO gene, then this is practically only amenable totransgenic methods using constructs designed to concurrently silence multiple FvMLO genes, an approach that has already been achieved in A. thaliana. Moreover, recent genome editing technologies, TALEN and CRISPR-Cas9, or other genome editing techniques, can be used to introduce targeted mutations in FvMLOgenes that result in heritable resistance to powdery mildew.
Supplemental data:
Acknowledgements
We would like to thank Felix trust for financial support, and Dr Janet Slovin, Beltsville, USA and Dr Richard Harrison, East Malling Research, UK for providing materials utilised for the study. We acknowledge Dr Ihsan Ullah, University of Reading, UK for the technical help in data analysis.
References
- 1.Devoto A, Piffanelli P, Nilsson I, Wallin E, Panstruga R. (1999) Topology, subcellular localization, and sequence diversity of the Mlo family in plants. , J. Biol. Chem 274, 34993-35004.
- 2.M C Kim, Panstruga R, Elliott C, Müller J, Devoto A. (2002) Calmodulin interacts with MLO protein to regulate defence against mildew in barley. , Nature 416, 447-451.
- 3.Büschges R, Hollricher K, Panstruga R, Simons G, Wolter M. (1997) The barleyMlogene: a novel control element of plant pathogen resistance. , Cell 88, 695-705.
- 4.Appiano M, Pavan S, Catalano D, Zheng Z, Bracuto V. (2015) Identification of candidateMLOpowdery mildew susceptibility genes in cultivated Solanaceae and functional characterization of tobaccoNtMLO1. Transgenic Res. 24, 847-858.
- 5.Ablazov A, Tombuloglu H. (2016) Genome-wide identification of themildew resistance locus O(MLO) gene family in novel cereal model speciesBrachypodiumdistachyon. , Eur. J. Plant Pathol 145, 239-253.
- 6.Chen Y, Wang Y, Zhang H. (2014) Genome-wide analysis of themildew resistance locus O(MLO) gene family in tomato (SolanumlycopersicumL.). , Plant Omics 7(2), 87-93.
- 7.Deshmukh R, V K Singh, B D Singh. (2017) Mining theCicerarietinumgenome for themildew locus O(Mlo) gene family and comparative evolutionary analysis of theMlogenes fromMedicagotruncatulaand some other plant species. , J. Plant Res 130(2), 239-253.
- 8.Devoto A, Hartmann H A, Piffanelli P, Elliott C, Simmons C. (2003) Molecular phylogeny and evolution of the plant-specific seven-transmembrane MLO family. , J. Mol. Evol 56, 77-88.
- 9.Feechan A, A M Jermakow, Torregrosa L, Panstruga R, I B Dry. (2009) Identification of grapevineMLOgene candidates involved in susceptibility to powdery mildew. , Funct. Plant Biol 35, 1255-1266.
- 10.Iovieno P, Andolfo G, Schiavulli A, Catalano D, Ricciardi L. (2015) Structure, evolution and functional inference on theMildew Locus O(MLO) gene family in three cultivated Cucurbitaceae. , BMC Genomics 16, 1112.
- 11.Kaufmann H, Qiu X, Wehmeyer J, Debener T. (2012) Isolation, molecular characterization, and mapping of four rose MLO orthologs. , Front. Plant. Sci 3, 244.
- 12.Konishi S, Sasakuma T, Sasanuma T. (2010) Identification of novelMlofamily members in wheat and their genetic characterization. , Genes Genet. Syst 85, 167-175.
- 13.Liu L P, Qu J W, Yi X Q, Huang H H. (2017) Genome-wide identification, classification and expression analysis of theMildew Resistance Locus O(MLO) gene family in sweet orange (Citrussinensis). , Braz. Arch. Biol. Technol 60, 17160474.
- 14.Liu Q, Zhu H. (2008) Molecular evolution of theMLOgene family inOryza sativaand their functional divergence. , Gene 409, 1-10.
- 15.Pessina S, Pavan S, Catalano D, Gallotta A, R G Visser. (2014) Characterization of theMLOgene family in Rosaceae and gene expression analysis inMalusdomestica. , Bmc Genomics 15, 618.
- 16.Rispail N, Rubiales D. (2016) Genome-wide identification and comparison of legumeMLOgene family. , Sci. Rep 6, 32673.
- 17.Shen Q, Zhao J, Du C, Xiang Y, Cao J et al. (2012) . Genome-scale identification of MLO domain-containing genes in soybean (Glycine maxL. Merr.). Genes Genet. Syst 87, 89-98.
- 18.Zhou S, Jing Z, Shi J. (2013) Genome-wide identification, characterization, and expression analysis of theMLOgene family inCucumis sativus. , Genet. Mol. Res 12, 6565-6578.
- 19.Panstruga R. (2005) Serpentine plant MLO proteins as entry portals for powdery mildew fungi. , Biochem. Soc. Trans 33, 389-392.
- 20.Jørgensen J H. (1992) Discovery, characterization and exploitation of Mlo powdery mildew resistance in barley.InBreeding for disease resistance (Springer). 141-152.
- 21.Wolter M, Hollricher K, Salamini F, Schulze-Lefert P. (1993) Themloresistance alleles to powdery mildew infection in barley trigger a developmentally controlled defence mimic phenotype. , Mol. Gen. Genet 239, 122-128.
- 22.Piffanelli P, Zhou F, Casais C, Orme J, Jarosch B. (2002) The barley MLO modulator of defense and cell death is responsive to biotic and abiotic stress stimuli. , Plant Physiol 129, 1076-1085.
- 23.Piffanelli P, Ramsay L, Waugh R, Benabdelmouna A, D'Hont A. (2004) A barley cultivation-associated polymorphism conveys resistance to powdery mildew. , Nature 430, 887-891.
- 24.AcevedoGarcia J, Spencer D, Thieron H, Reinstädler A, Kosack Hammond et al.(2017)mlobased powdery mildew resistance in hexaploid bread wheat generated by a non transgenic TILLING approach. , Plant Biotechnol. J 15, 367-378.
- 25.Pessina S, Lenzi L, Perazzolli M, Campa M, Costa Dalla et al. (2016) Knockdown ofMLOgenes reduces susceptibility to powdery mildew in grapevine. Horticulture Res. 3, 16016.
- 26.Consonni C, Bednarek P, Humphry M, Francocci F, Ferrari S. (2010) Tryptophan-derived metabolites are required for antifungal defense in theArabidopsismlo2mutant. Plant Physiol. 152, 1544-1561.
- 27.Bai Y, Pavan S, Zheng Z, N F Zappel, Reinstadler A. (2008) Naturally occurring broad-spectrum powdery mildew resistance in a Central American tomato accession is caused by loss ofmlofunction. , Mol. Plant Microbe Interact 21, 30-39.
- 28.Qiu X, Wang Q, Zhang H, Jian H, Zhou N. (2015) AntisenseRhMLO1gene transformation enhances resistance to the powdery mildew pathogen inRosa multiflora. Plant Mol. , Biol. Rep 33, 1659-1665.
- 29.Jiwan D, E H Roalson, Main D, Dhingra A. (2013) Antisense expression of peachmildew resistance locus O(PpMlo1) gene confers cross-species resistance to powdery mildew inFragariaxananassa. Transgenic Res. 22, 1119-1131.
- 30.Pessina S, Angeli D, Martens S, R G Visser, Bai Y. (2016) The knock down of the expression ofMdMLO19reduces susceptibility to powdery mildew (Podosphaeraleucotricha) in apple (Malusdomestica). , Plant Biotech. J 14, 2033-2044.
- 31.Jiang P, Chen Y, H D Wilde. (2016) Reduction ofMLO1expression in petunia increases resistance to powdery mildew. , Sci. Hort 201, 225-229.
- 32.Humphry M, Reinstadler A, Ivanov S, Bisseling T, Panstruga R. (2011) Durable broadspectrum powdery mildew resistance in pea er1 plants is conferred by natural loss of function mutations inPsMLO1. , Mol. Plant Pathol 12, 866-878.
- 33.J A Berg, Appiano M, Martínez M S, Hermans F W, Vriezen W H. (2015) A transposable element insertion in the susceptibility geneCsaMLO8results in hypocotyl resistance to powdery mildew in cucumber. , BMC Plant Biol 15, 243.
- 34.Wang Y, Cheng X, Shan Q, Zhang Y, Liu J. (2014) Simultaneous editing of three homoeoalleles in hexaploid bread wheat confers heritable resistance to powdery mildew. , Nature Biotechnol 32, 947-951.
- 35.Peries O. (1962) Studies on strawberry mildew, caused bySphaerothecamacularis(Wallr. , ex Fries) Jaczewski. Ann. Appl. Biol 50, 211-224.
- 36.Lifshitz C, Shalit N, Slotzky S, Tanami Z, Elad Y et al. (2008) Heritability s studies and DNA markers for powdery mildew resistance in strawberry (Fragaria×ananassaDuchesne). VI International Strawberry Symposium 842 561-564.
- 37.Shulaev V, D J Sargent, R N Crowhurst, T C Mockler, Folkerts O. (2010) The genome of woodland strawberry(Fragariavesca). , Nature Genet 43, 109-116.
- 38.Miao L, Jiang M, Zhang Y, Yang X, Zhang H. (2016) Genomic identification, phylogeny, and expression analysis ofMLOgenes involved in susceptibility to powdery mildew inFragariavesca. , Genet. Mol. Res 15, 15038400.
- 39.J P Slovin, Schmitt K, K M Folta. (2009) An inbred line of the diploid strawberryFragariavescaf.semperflorensfor genomic and molecular genetic studies in the Rosaceae. , Plant Methods 5, 15.
- 40.J D Thompson, D G Higgins, T J Gibson. (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. , Nucleic Acids Res 22, 4673-4680.
- 41.Saitou N, Nei M. (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. , Mol. Biol. Evol 4, 406-425.
- 42.Tamura K, Dudley J, Nei M, Kumar S. (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. , Mol. Biol. Evol 24, 1596-1599.
- 43.L V Bindschedler, Palmblad M, Cramer R. (2008) Hydroponic isotope labelling of entire plants (HILEP) for quantitative plant proteomics; an oxidative stress case study. , Phytochemistry 69, 1962-1972.
- 44.Jambagi S, J M Dunwell. (2015) Global transcriptome analysis and identification of differentially expressed genes after infection ofFragariavescawith powdery mildew (Podosphaeraaphanis). doi: 10.4172/2329-8936.1000106. Transcriptomics: Open Access vol-3issue-1.
- 45.K J Livak, T D Schmittgen. (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCTmethod. , Methods 25, 402-408.
- 46.Winterhagen P, S F Howard, Qiu W, L G Kovacs. (2008) Transcriptional up-regulation of grapevineMLOgenes in response to powdery mildew infection. , Am. J. Enol. Vitic 59, 159-168.
Cited by (9)
This article has been cited by 9 scholarly works according to:
Citing Articles:
Samantha C. Lynn, Jim M. Dunwell, A. Whitehouse, H. Cockerton - Frontiers in Plant Science (2024) Semantic Scholar
BMC Plant Biology (2020) OpenAlex
BMC Plant Biology (2020) Crossref
Jun Feng, Min Zhang, Kang-Ning Yang, C. Zheng - BMC Plant Biology (2020) Semantic Scholar
PLANT BIOTECHNOLOGY AND BREEDING (2020) OpenAlex
И. Э. Храбров, О. Ю. Антонова, Максим Игоревич Шаповалов, Л. Г. Семёнова - (2020) Semantic Scholar
PLoS ONE (2019) OpenAlex
D. Sargent, M. Buti, Nada Šurbanovski, M. Brurberg, M. Alsheikh et al. - PLoS ONE (2019) Semantic Scholar
Jun Feng, Min Zhang, Kangning Yang, Wenting You, C. Zheng - (2019) Semantic Scholar
Journal of Advances in Plant Biology (2017) OpenAlex
