Some Aspects of Regulatory Criteria for Metal-ligand Homeostasis in Epidermal Cells
Abstract
The work is dedicated to the problem of the norm in the quantitative evaluation of metal content in the epidermal cells (hair) obtained by the method of spectrometry. Authors have analyzed the hair samples for Zn, Cu, and Fe content, which were obtained from 10000 healthy subjects (5000 males and 5000 females aged 20 to 45).
The definition of the norm, in the authors’ opinion, is closely related to the basic positions of the theory of self-organized criticality (SC). The observed shifts in the homeostasis of essential metals are local and therefore cannot serve as a criterion of sufficient (or insufficient) metal content throughout the body.
Author Contributions
Academic Editor: Jie Yin, Institute of Subtropical Agriculture & University of Chinese Academy of Sciences, China, Email: [email protected]
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2018 V.I.Petukhov, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Despite the huge number of publications dedicated to the diagnosis of metal-ligand homeostasis (MLH) disorders, reliable detection of latent forms of these disorders remains an unresolved task for most essential metals. The only exception is iron (Fe), whose hidden deficit can be diagnosed by the level of serum ferritin and by well-known pathognomonic clinical signs. The lack of other essential metals, in particular, copper (Cu) and zinc (Zn) in hidden (latent) form, does not have reliable clinical symptoms and reliable serum or any other markers.
Although high-precision diagnostic equipment (atomic emission spectrometry, mass spectrometry) and a non-invasive method for obtaining biosubstrate (hair) were introduced into research practice, a rapid solution to the diagnostic problem has not yet been achieved. The main question has remained unanswered: What do the numerical parameters of metal spectrometry mean in a certain substrate (especially, in hair) and how do they relate to the whole body levels of essential metals (Ca, Na, K, Fe, Zn, Cu, etc.)?
The greatest difficulty lies in revealing the latent lack of essential metals when clinical signs are meager or totally absent, while the diagnosis of extreme forms of deficit (or oversupply) of metals usually does not cause problems.
Speaking of epidermis (hair), one cannot regard it as a proven fact that metal content in epidermis enables the diagnosis of a latent form of metal deficiency. Besides, understanding is needed of spectrometric parameters as such and of how they help resolve the main task: the identification of latent deficiency of essential metals.
Conspicuous is the fact that there was a considerable individual variation in metal concentrations when hair was used as the substrate: the coefficient of variation (CV), according to our data, ranged from 34% to 226.5% 1. Moreover, when verifying the hypothesis of normal distribution of spectrometric data, it was found that this distribution did not comply with the normal law and, as it became clear later, it was of a fractal nature 2.
Because of the impossibility to confirm the normal distribution hypothesis, one has to abandon the use of standard methods of estimating the mean value and use alternative approaches to this purpose: the bootstrap method 3, finding the median (Me) 4. But when interpreting the results of hair spectrometry, it can be difficult to resist the illusion that the findings reflect the "elemental status" of the whole body 4, and not the features of the transmembrane metal traffic at the level of epidermocytes – a dynamic process which is susceptible to sharp numerical oscillations (judging by the CV). That is why extrapolation of these spectrometry data to the whole body does not seem to be justified.
In our opinion, it would be more correct to relate the changes of MLH which were observed in the epidermis to the given tissue only and avoid any unproven generalizations leaving open to further research the possible connection of the MLH in the epidermis with similar processes in other tissues.
When studying MLH in the epidermis, it is of interest not only and not so much the content of a particular metal as the features of regulatory control over MLH at the cell level. This control can occur in accordance with the recently discovered universal natural law of self-organized criticality (SC) 5, since (from the point of view of bioenergetics) a living cell is an open dynamic system in which for the counteraction of entropy the energy exchange at the cellular membrane level takes an appreciable place. Such processes include ATP-dependent transmembrane traffic of metal ions (primarily, Na+, K+, Ca2+, Zn2+, etc.), in which the ATPase family (P-type) takes part.
We already reported on the facts indicating the belonging of the MLH in the epidermis to the SC phenomena for sodium (Na), potassium (K) and calcium (Ca) 2. The possible evidence of such affiliation for iron (Fe), zinc (Zn) and copper (Cu) is presented in this work.
Material and Methods
In order to diminish possible environmental contamination the hair samples were washed with acetone and then rinsed thrice with deionized water with subsequent drying at 60°C on air. Further preparation of the samples was performed using microwave degradation. In particular, 50 mg hair samples were introduced into Teflon container and added with 5 ml of concentrated analytical grade HNO3 (Sigma-Aldrich Co, St. Louis, MO, USA). Decomposition was performed in Berghof speedwave four system (Berghof Products & Instruments, Germany) during 20 min at 170-180°C. After decomposition deionized water was added to a final volume of 15 ml.
Hair metal content was assessed by inductively coupled plasma mass spectrometry (ICP-MS) with NexION 300D (Perkin Elmer Inc., Shelton, CT, USA) using Dynamic Reaction Cell technology removing the majority of interferences with little or no loss of analyte sensitivity and equipped with ESI SC-2 DX4 autosampler (Elemental Scientific Inc., Omaha, NE, USA).
Using mathematical statistics, we have analyzed the results of atomic emission spectrometry of hair samples for Zn, Cu, and Fe content, which were obtained from 10000 healthy subjects – Moscow residents (5000 males and 5000 females) aged 20 to 45.
In order to reveal a power-law relationship between the content of metals in the epidermis and the number of subjects (the criterion of belonging to the SC-phenomenon) we estimated the probability density function (PDF) and made a fit the power-law distribution. A similar mathematical approach was used by us in the previous study 2.
The Matlab software tool was used for statistical data processing.
Results and Discussion
It is known that the power-law dependence takes the form of a straight line in a double logarithmic scale. Therefore, the corresponding graphs were constructed to determine the power-law relationship between the metal content (Zn, Cu and Fe) in the epidermis and the number of subjects.
The probability density distribution of the power is defined by the following formula:
To make it easier to judge the quality of fit, an ‘ideal’ case of random number matching with the Pareto distribution (RND) is shown (See Figure 1).
Figure 1.An ‘ideal’ case of random number matching with Pareto distribution (RND). The blue curve is the density estimate from spectrometry data. The red curve is a fitting of the power distribution by the linearization method. RMSE is the mean square error; Cthris the smallest concentration at which the fit begins.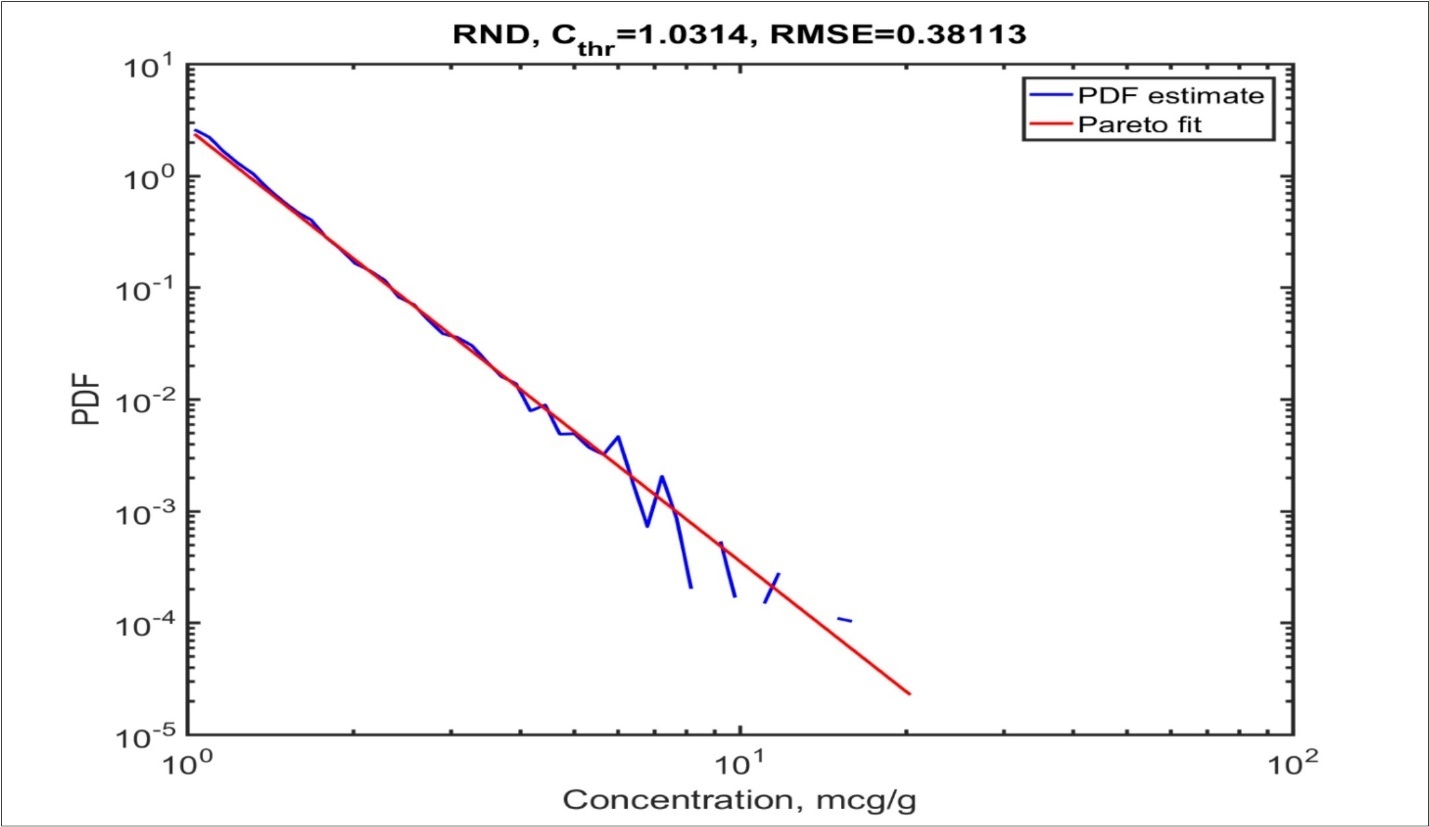
Figure 2, Figure 3 and Figure 4 show estimates of the distribution density from spectrometry data (blue curve) and linearization of the power distribution (red curve) for zinc (Figure 2), copper (Figure 3), and iron (Figure 4).
Figure 2.Estimate of the density and linearization of power distribution for Zn (the notation is the same as in Figure 1)
Figure 3.Estimate of the density and linearization of power distribution for Cu (the notation is the same as in Figure 1).
Figure 4.Estimate of the density and linearization of power distribution for Fe (the notation is the same as in Figure 1).
To obtain a normalized histogram, the boundaries of the columns were chosen uniformly on a logarithmic scale (30 bars for 10,000 measurements). The above indications are: the name of metal, where the dovetailing begins with the smallest concentration (the data corresponding to the concentrations that are less than Cthrnot taken into account), and the RMSE is the root-mean-square error. Calculation of the distribution parameters is presented in Table 1.
Table 1. Calculated power distribution parameters (PDF)| Metal | k | m |
|---|---|---|
| Zn | 4.9268 | 18.2564 |
| Cu | 3.8834 | 2.5106 |
| Fe | 3.4374 | 1.77 |
| RND | 5.0046 | 0.90352 |
Figure 2, Figure 3 and Figure 4 show segments of different sizes that practically coincide with a straight line, which, according to the theory of SC 5, may indicate the existence (within a straight line) of the power-law relation between the level of a specific metal in the epidermis and the number of individuals. These sections of linear approximation testify to the critical state of MLH, in which the branching parameter σ is equal to 1. The fragments of the curves with no such approximation (before and after the straight line) should be referred to the subcritical (σ < 1) and supercritical (σ > 1) state of MLH 2. In the case of zinc (Figure 2) and copper (Figure 3), curves belonging to the supercritical state are to the left of the straight line segment, and subcritical to the right (along the abscissa axis). In the case of iron (Figure 4), the localization of these fragments of the curve acquires the opposite view: the subcritical state on the left, and the supercritical state on the right of the straight line along the abscissa axis.
The explanation for this lies in the opposite effect of oxidative/nitrosative stress on the content of these metals in the epidermocytes. It is known that increased production of reactive oxygen species (ROS) and reactive nitrogen species (RNS) can activate the membrane ATPases due to oxidative modification and/or nitrosylation of their molecules, which, according to our data, was accompanied by a significant decrease of Zn and Cu level in the cell, but by an increase of Fe 3, 6.
Therefore, the direction (sequence) of SC-periods (subcritical → critical → supercritical) by the content of metals and the increase in ATPase activity should be for zinc and copper - from higher concentration values to lower values, and for iron – from smaller to larger values.
The resultant visual (and therefore with a certain approximation) numerical ranges of fragments approximated by a straight line (a critical state) were as follows: for zinc – 200-1050 mcg/g, for copper 13-65 mcg/g, and for iron – 15-50 mcg/g (Figure 2, Figure 4).
It should be noted that the branching parameter σ in the oscillator system (in this case, membrane ATPases) is equal to the average number of the nearest ‘neighbors’ to which each of the oscillators can transmit the energy impulse (information). In the critical state, σ is equal to 1, that is, the transfer of information (energy) in such a system will occur almost immediately to all its members and, very importantly, over considerable distances. This circumstance is one of the basic features for the critical (synchronous) functioning of oscillatory systems. Moreover, the already mentioned power-law connection (as a sign of criticality) being fractal (independent of the scale of the system) can be detected not only at the cell level, but also at the whole body level.
The presence of a specific metal in the epidermal cell within the critical state means that within these boundaries the cell is able to control (regulate) the MLH of the given metal. The belonging of MLH of essential metals to the critical range can obviously use as a normative criterion of the provision of the epidermal cell with these metals. The normative indices of electrogenic metals (EM) content in the hair – calcium (Ca), potassium (K) and sodium (Na) – found before by the SC-criterion were as follows: Cafemale - 400 to 3000 μg/g; Camale- 400 to 2000 μg/g; K - 50 to 1000 μg/g; Na - 80 to 1000 μg/g 7. The presented numerical values of the EM level in epidermis correspond to the critical state, which, in all likelihood, should be attributed to the cell’s ability of controlling (regulating) EM homeostasis. This ability can serve as a kind of criterion for normal functioning of the cell.
The criticality of the operation of membrane pumps for EM should be understood as the synchronous nature of their functioning, since synchronization is a specific instance of the critical state. Thus, if the Na+/K+-ATPases of the outer membrane work synchronously (in the critical mode), then the K+ and Na+ levels in the cell should show a correlation relationship.It must be remembered that each Na+/K+-pump transfers three Na+ ions outward and two K+ ions inside the cell. It is significant that this very statement, which is often found in modern literature, presents an a priori acknowledgment of the existing proportionality of the intracellular content of Na+ and K+ ions.Meanwhile, such proportionality can only occur when the membrane Na+/K+-pump operation is synchronous.
The existence of direct correlation between Na and K could be clarified by measuring the content of these metals directly in the epidermal cells if it were not for the methodological difficulties related to such an approach. However, if EM homeostasis is indeed a
SC- phenomenon, then the Na - K correlation should have fractality (independence from the scale of the system) and be detected not only at the cell level but also at the body level. In most study subjects (in a given range of numerical EM values) one can detect not only the power-law relationship between the results of spectrometry and the number of individuals but also a direct correlation between the level of Na and K in epidermis by correlation analysis (Pearson) at the level of individuals.
The linear positive Na-K bond (rNa-K=0.6-0.8; p<0.05) was detected by us both in healthy individuals (n=947) and in the liquidators of the Chernobyl accident (n=954), in which the signs of oxidative/nitrosative stress have been found 3.
A reliable Na-K correlation, indicating synchronous operation of membrane Na+/K+-ATPase, was independent of the sample size, but was closely related to the current mode of membrane Na+/K+-ATPase functioning (critical, sub- and supercritical) or from the synchronous (critical status) and asynchronous (sub- and supercritical state) operation of these pumps. This is well illustrated by the values of rNa-K in the general group (n = 10012) and at the concentration values of K and Na, which correspond to the sub- and supercritical state (see Table 2) 7.
Table 2. The correlation coefficient rK-Na (Pearson): the link between the potassium (K) and sodium (Na) levels in epidermis 7| Study groups | n | r K-Na |
| The general group | 10012 | 0.61 (p <0.05) |
| Subcritical state: Na < 80 μg/g | 424 | 0.1 |
| Supercritical state: Na> 1500 μg/g | 260 | 0.17 |
As shown in Table 2, the correlation coefficient rK-Na (Pearson) in the subcritical and supercritical states is reduced sharply in comparison with the general group, which, in our opinion, reflects the asynchronous operation of membrane Na+/K+-ATPase inherent in these states.
The production ROS/RNS, in which the density of activated ATPase on the cell membrane is sufficient to trigger the synchronization process, will cause the critical state of the system. If the density of the activated membrane ATPases is insufficient, such a start-up can be difficult or impossible (the subcritical state). Hyperproduction of ROS/RNS is able to cause the supercritical state of the system. Synchronization in the functioning of membrane pumps (criticality) is also inherent, to a certain extent, in other membrane pumps from the superfamily ATPase (P-type). This is evidenced by the tightness of the connection (Pearson) between the concentration values of K and Zn in healthy individuals and liquidators of the Chernobyl accident (in the latter, an increase in the production of ROS and RNS) was revealed 2.
For the majority of the liquidators of the Chernobyl accident (88%), the K-Zn correlation was negative and significant (in 205 people, K-Zn = -0.62; p <0.05; for 634 people, K-Zn = -0.41; p<0.05). In 12% of liquidators (115 people) it was not detected (r = -0.03). K-Zn communication was absent (r = -0.01) in 253 healthy individuals (26.7%), in 523 people. (55.2%) was weakly expressed (rK-Zn = -0.22, p<0.05) and was clearly identified (rK-Zn = -0.43, p<0.05) in 171 people only (18.1%). Moreover, the K-Zn bond for the largest |r| was combined with significantly higher levels of K and Na and lower - Zn, than those with a lack of K-Zn correlation (as in liquidators and in healthy subjects).
Table 3, Table 4 and Table 5 show the main parameters of SC-periods obtained for Zn, Cu, and Fe.
As follows from Table 3, the number of individuals with the critical state MLHZn was the largest (n = 5090), slightly exceeding the same index for the supercritical state (n = 4876) and, to a large extent, for the subcritical state (n = 34). The level of Zn in the epidermis was also unequal in different SC periods: the highest in the subcritical (Me = 1,346.8 μg/g) and the smallest (Me = 162.7 μg/g) in the supercritical period. The Me values of the critical phase occupied an intermediate position (Me = 254.6 μg/g). Sex differences were not detected, except for the subcritical period, where the sample size (n = 34) was not sufficient for reliable conclusion.
Table 3. Median (Me) and other parameters of SC-periods in Zn| The SC-periodof MLHZn | n | Median (Me) in different SC-periods (μg/g) | Distribution by sex | |
| men | women | |||
| Subcritical (σ <1) | 34 (0.3%) | 1,346.8 | 6 (17.7%) | 28 (82.3%) |
| Critical (σ =1) | 5090 (50.9%) | 254.6 | 2233 (43.9%) | 2857 (56.1%) |
| Supercritical (σ >1) | 4876 (48.8%) | 162.7 | 2761 (56.6%) | 2115 (43.4%) |
Essential, in our opinion, is the very fact that Zn content (Me) in the critical phase (254.6 μg/g) is different from the median of Zn content in the general group (Me = 201.9 μg/g). It should be noted that many researchers judge by the value of the last parameter about the ‘elemental status’ of the whole body 4.
Can the observed difference serve as a sign of latent Zn-deficiency? There is no unequivocal answer here. Moreover, it is possible that in the supercritical SC-period a greater number of individuals may be with increased production of ROS and RNS, which leads, as shown by our investigations, to reduce not only the intracellular Zn, but also Cu 2, 3. Thus, the observed shifts are not general but rather local or redistributive.
The critical SC-period of MLHCu was detected by the content of copper in hair (Table 4) in most individuals (54.7%). The nature of changes in the level of Cu in the epidermis, depending on the SC-period, was the same as for Zn: the highest in the subcritical (Me = 90.4 μg/g) and the smallest (Me = 10.6 μg/g) in the supercritical period. At the same time, the distribution of individuals by these metals differed sharply in the subcritical and supercritical periods: Zn in the subcritical period - only 0.4% of the total number of individuals, but copper – 42.3%. The supercritical SC-period for zinc was 48.8%, while for copper – only 3% (seeTable 3 and Table 4). The median (Me) for Cu in the critical phase was 18.4 μg/g with almost equal sex distribution at this phase. Meanwhile the distribution by the sex was markedly different in other SC periods.
Table 4. The median (Me) and other parameters SC-periods in Cu| SC-period MLH Cu | n | Median (Me) in different SC-periods ( μ g/g) | Distribution by sex | |
| men | women | |||
| Subcritical (σ <1) | 4234 (42.3%) | 90.4 | 2651 (62.6%) | 1583 (37.4%) |
| Critical (σ =1) | 5469 (54.7%) | 18.4 | 2267 (43.9%) | 3202 (56.1%) |
| Supercritical (σ >1) | 297 (3%) | 10.6 | 82 (27.6%) | 215 (72.4%) |
The median of the Cu content in the critical phase (18.4 μg/g), like in the case of zinc, exceeded the Me for Cu in the total group (12.8 μg/g). An explanation for this should apparently be sought in the redistributive character of the changes in MLHCu, depending on the level of ROS and RNS in the epidermal cells.
The majority of individuals (59.5%) by the level of iron in hair was in the subcritical (σ < 1) period, the lowest number (5.3%) - in the supercritical period (σ > 1). The SC-critical period (σ=1) was 35.2% of the total number tested. The dynamics of changes in the Fe content depending upon the SC-period was as follows: in the subcritical Me = 9.6 μg/g, in critical Me = 22 μg/g and supercritical period Me = 74 μg/g.
The distribution of individuals across the sex in different SC-periods was essential. Women with the highest iron content predominated (70.9%) in the supercritical period, while the distribution of individuals by gender in the critical and sub-critical SC-period was practically identical (Table 5).The nearlyequal sex distribution in the subcritical period in persons with the lowest Fe content (Me = 9.6 μg/g) requires discussion.
Table 5. Median (Me) and other parameters of SC-periods in Fe| SC-period MLH Fe | n | Median ( Me ) in different SC- periods ( μ g/g) | Distribution by sex | |
| men | women | |||
| Subcritical (σ <1) | 5952 (59.5%) | 9.6 | 3368 (56.6%) | 2584 (43.4%) |
| Critical (σ =1) | 3518 (35.2%) | 22 | 1478 (42%) | 2040 (58%) |
| Supercritical (σ >1) | 530 (5.3%) | 74 | 154 (29.1%) | 376 (70.9%) |
The low level of Fe, according to spectrometry results, is equally likely to be detected in both men and women. This data clearly contradicts the well-known fact of a significant predominance of women (aged 20-45) suffering from an iron deficiency (ID) as compared to men. According to our data, the percentage of men aged 20 to 43 with no evidence of ID was almost five times higher than that of women (60% vs 12.2%, resp.) 8.
All this convinces us that the observed shifts in MLHFe are local, of non-general nature, and therefore cannot serve as a criterion of sufficient (or insufficient) iron content throughout the body. This statement, apparently, can be valid for other metals (Zn, Cu) with the improvement of diagnostic methods of their hidden deficiency.
The problem of reliable diagnosis of ID is becoming topical in connection with the likely dependence of the bioavailability of nitric oxide (NO) on the body iron level. The existence of such a dependence is indicated by the modern concept of a three-component system (NO + free thiols + non-heme Fe). In the cell, this biosystem is the main and permanent source of nitrogen monoxide, more precisely, of its most reactogenic forms: nitrosonium (NO+) and nitroxyl (NO– or HNO) 9.Within the framework of this system, S-nitrosothiols and dinitrosyl iron complexes with
thiol-containing ligands (DIC) are continuously synthesized, both in mononuclear [(RS)2Fe(NO)2] – M-DIC, and in binuclear form [(RS)2Fe2(NO)4] – B-DIC.
In this system a constant inter transformation of DIC and S-nitrosothiols (RS-NO) occurs in the self-oscillation mode (according to the type of the Belousov-Zhabotinsky reaction). Thus one of the main conditions for the normal operation of this system is replenishment of the necessary components, primarily, NO and intracellular Fe (in the case of sufficient amount of free SH groups in the cell). As a result, in the conditions of Fe-deficiency, we can expect a decrease in the number of self-oscillating systems and a decrease in the density of their distribution in the cytoplasm, which cannot but affect the bioavailability of NO.
According to modern concepts, RS-NO↔DIC oscillators can generate the most reactogenic forms NO: nitrosonium (NO+) and nitroxyl (HNO) ions during inter conversions of RS-NO and DIC, which have a significantly longer lifetime than nitrogen monoxide (the lifetime of NO is measured in milliseconds). As a result, conditions are created for the stable (long-term) functioning of the short-lived NO molecule as a universal manager 9.
The possible variants of the RS-NO↔DIC-oscillators existence in the cell need discussion. The fact is that, having the ability to self-oscillation, these systems, upon reaching a certain (threshold) density in the cytoplasm, must order (synchronize) their oscillations or, in the terminology of the theory of self-organized criticality, they must ‘self-organize’ themselves critically, go into a critical state.The synchronous (critical) mode of operation of RS-NO↔DIC systems is important and has clear advantages over pre- and supercritical regimes in the transmembrane traffic of ions of electrogenic metals (primarily Ca2+), since synchronized, uninterrupted production of NO+ and HNO under conditions of critical regimen provides the necessary activation of membrane ATPases by nitrosylation of SH-groups in their protein molecules.
In addition, under conditions of critical regimen it is possible to control the entry and exit of the ions of electrogenic metals from the cell that is necessary to maintain (within acceptable limits) the dynamic balance between these parameters.
This can be quite real, if only because the derivatives of self-oscillating RS-NO↔DIC systems (nitrosonium and nitroxyl ions) are acting as activators of membrane pumps belonging to the same family of ATPases (P-type) for the entry and exit of metals. The only condition that mustbe carried out in the normal operation of these systems is their critical state. We note, that the synchronous (‘salvo’) appearance of Ca2+ ions in the hyaloplasm (‘calcium sparks’), which was detected with the help of confocal microscopy 10, illustrates this possibility well enough. The subcritical (σ <1) mode of operation of RS-NO↔DNA-oscillators is most probable for ID and situations with insufficient synthesis of NO by constitutive NO-synthases. At the same time, it cannot be ruled out that the deficit of NO+ and HNO, which is quite expected here, can impede the full-scale activation (nitrosylation) of membrane ATPases. This will inevitably lead to shifts in the homeostasis of electrogenic metals at the cell level and to the possible clinical manifestations of those shifts.
For example, the violation of calcium homeostasis in the myocyte, regardless of the ‘specialization’ of this cell (cardiomyocyte, smooth muscles of the vessels and hollow organs, skeletal muscles), can lead to ‘malfunctions’ in the transmembrane traffic of Ca2+: inadequate replenishment of the Ca2+ depot in the sarcoplasmic reticulum (SR) and the associated reduction in the yield of Ca2+ in the cytosol.As a result it decreases the contractile ability of muscles.
This symptom in the clinical picture of ID is universal (not depending on the type of musculature). It is known that the weakness of skeletal muscles is a pathognomonic symptom of Fe-deficient anemia. One can be observe not only coronary blood flow deterioration in such patients (as a result of impaired peristalsis of coronary vessels) but also reduced contractile ability of the myocardium, as well as weakening of tonus and peristaltic contractions of the hollow muscular organs.
It does not seem accidental that within 4-5 days after the start of Fe therapy (time is too short for the recovery of erythropoiesis but apparently long enough for the RS-NO↔DIC system to go from subcritical to critical condition) the number of angina attacks decreased significantly in patients with coronary heart disease and iron deficiency anemia.
A supercritical state (σ > 1), in which the operating RS-NO↔ DIC-oscillators may appear, is most likely due to a sharp increase in NO production when inducible NOS (iNOS) is activated, for example, in sepsis and all kinds of shock.
Thus, the use of hair spectrometry for determination of metal content in epidermal cells has proven to be ineffective in diagnostics of latent ID forms. However, the spectrometric analysis may be suitable for detecting ‘criticality’ (synchronization) as a normative (regulatory) criterion in the operation of membrane ATPases.
References
- 1.V I Petukhov, E V Dmitriev, A P Skesters, A V Skalny. (2006) Problems of the integral evaluation of human elemental status according to the spectrometry of hair. , Mikroelementy v Medicine 7(4), 7-14.
- 2.V I Petukhov, E V Dmitriev, Baumane L Kh, A V Skalny, Lobanova Yu N. (2016) Electrogenic metals in epidermis: relationship with cell bioenergetics. , Insights in Biomed 1(2), 9-14.
- 3.V I Petukhov, E V Dmitriev, Kalvinsh I, Baumane L Kh, E D Reste et al. (2011) Metal-ligand homeostasis in epidermic cells of Chernobyl accident liquidators. , Vitamins & Trace Elements 1(2), 1-8.
- 4.S V Notova, E V Kiyaeva, I V Radysh, I E Laryushina, M L Blagonravov. (2017) Element status of students with different levels of adaptation. , Byulleten` Eksperimental`noi Biologii i Meditsiny 163(5), 541-543.
- 6.V I Petukhov, L K Baumane, Reste E D, Zvagule T, Romanova M A et al. (2013) Diagnosis of nitrosative stress by quantitative EPR-spectroscopy of epidermal stress. , Bull. Exp. Biol. Med 154, 734-736.
- 7.V I Petukhov. (2017) What are the limits, if any, of normal content of electrogenic metals (K, Na, Ca) in epidermis?. , Insights Biomed 2(2), 13-17.
- 8.А Н Щуков, Е Я Быкова, В И Петухов, Л Х Баумане. (2014) Железодефицитные состояния (ЖДС) как предиктор репродуктивных нарушений. , Евразийский Союз Ученых (ЕСУ) # VI, C. 69-70, | Медицинские науки
Cited by (9)
This article has been cited by 9 scholarly works according to:
Citing Articles:
V. T. Surikov - Аналитика и контроль (2025) Semantic Scholar
Аналитика и контроль (2025) Crossref
N. Chemodurova, V. А. Turkina - (2020) Semantic Scholar
Acta Medica Leopoliensia (2020) Crossref
Acta Medica Leopoliensia (2020) OpenAlex
V. Petukhov, Е.V. Dmitriev, L. Baumane, A. Skalny - Biomedical Journal of Scientific & Technical Research (2020) Semantic Scholar
Biomedical Journal of Scientific & Technical Research (2020) OpenAlex
Petukhov Vi - Biomedical Journal of Scientific & Technical Research (2019) Semantic Scholar
Biomedical Journal of Scientific & Technical Research (2019) OpenAlex
V. Petukhov, Е.V.Dmitriev, A.V.Skalny, A.R.Grabeklis - International Journal of Negative Results (2018) Semantic Scholar
International Journal of Negative Results (2018) Crossref
International Journal of Negative Results (2018) OpenAlex

