The Adverse Effects of Underwater Sound upon Fishes and Invertebrates
Abstract
Underwater sound is very important to most fishes and invertebrates. Underwater sounds from both natural and human sources can have adverse effects upon aquatic animals, and especially fishes, and invertebrates. It is important to examine the effects of sound upon them, and especially the effects of sounds derived from human sources (anthropogenic sounds). It may be possible to introduce protective regulations to reduce their effects. Fishes and invertebrates can detect underwater sounds, and they use sound to obtain key information about the environment around them. They can also make sounds themselves, especially during their spawning. Sounds travel rapidly over great distances in water and can provide detailed information to these animals on the presence of prey, predators, and related species, while the overall acoustic scene provides them with key information about their environment. Anthropogenic sounds can be very harmful, and it is therefore important to deal with them. A succession of reports and scientific papers have emphasised the risks to these animals from exposure to man-made sounds or noise and will be mentioned in this paper, which also deals with the Criteria and Metrics for assessing the effects of underwater sound on fishes and invertebrates.
Author Contributions
Academic Editor: Anubha Bajaj, India
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2023 Anthony D Hawkins
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
The expansion of offshore industrial activities in recent years has led to concern about the impact of man-made (anthropogenic) sounds upon aquatic animals and the aquatic environment. There are current problems for all aquatic animals, including mammals (seals, dolphins & whales); birds, fishes, and invertebrates. Many of them are now in a very poor state. They use the detection of sounds during their everyday lives to track down prey, avoid predators, navigate, and examine the marine environment. Many fishes and invertebrates also make sounds themselves, that they use to communicate with one another, and in some cases their sounds are especially important during their spawning behavior.
However, their detection of sounds may be adversely affected by the sounds generated during human aquatic activities. The effects of sound on Fishes and Invertebrates may assess the pitch of the sound with relevance to frequency of sound that may alter the expression of survival genes that are critical to the well being of the fish. Low, middle and high frequency sounds may determine gene expression in various species of fish that determine the survival and aging of the species 14. In recent years, criteria and metrics have been established for assessing the effects of underwater noise upon fishes and invertebrates. A succession of reports and scientific papers have emphasised the risks to these animals from exposure to man-made sounds or noise 21.
There is much concern about the aquatic changes made by human activities, including Climate Change; Chemical Pollution; Fishing; and other Human Activities, especially those that generate sounds. These noisy activities include offshore oil and gas developments, wind farm construction, wave and tidal power resources, as well as attendant increases in shipping and fishing. It is especially animals that live within the water that are adversely influenced by these sounds, although birds that move over the water may also be affected, mainly because of the effects of sounds upon their food organisms. The generation of anthropogenic sounds in water is very common, but it’s effects are often not examined. The sea, rivers and lakes are not normally silent as there are some natural sound sources. Sound travels almost 5 times faster through the water than through the air, and low frequency sounds can travel many hundreds of kilometres under most normal conditions, with little loss of energy.
There is a need for more work on the impact of human- made underwater noise upon the fitness of aquatic animals. This paper considers the gaps in information that must be resolved. The effects that need to be considered include death and injuries, physiological effects, and changes in behavior.
Natural sound sources
Natural sounds are generated in the sea, rivers, and lakes, by rain, snow, wind, surface waves &
turbulence, and also by the animals living there. Many fishes, and invertebrates make sounds themselves, and so do marine mammals. There are also natural substrate vibration sounds, caused by waves breaking on the shore. Vision, taste, and smell senses are relatively poor in the aquatic environment, and sound is therefore especially important to aquatic animals.
Anthropogenic sound sources
Although some of the background noise in the water is generated by natural sources, many underwater sounds now come from anthropogenic sources (Figure 1). The expansion of offshore industrial activities in recent years has led to greater concern about the impact of anthropogenic sounds upon marine animals and the marine environment. The main activities include offshore oil and gas developments, and their operation; the seismic surveys carried out to examine the seabed, and to decide upon locations for oil and gas installations; wind farm construction and operation and especially the pile driving construction work associated with this, and also and the dredging and installation of pipelines & cables.
Figure 1.Sounds may be generated in water (from vessel engines and propellers, seismic air guns,operational wind turbines, or construction work). However, they may also be generated on the bottom, and in the substrate. The Anthropogenic sources include Ships (and even their trawls if they have them); Sonar and seismic survey systems towed by the ships; Pile driving construction work based upon the seabed (on the right, generating substrate vibration as well as sound in the water); Offshore wind turbines (on the left, also generating substrate vibration and sound in the water); While most sounds arise from in-water operations, it is well known that sounds on land, such as from auto traffic, may get into the water through the substrate. Thus, the underwater acoustic environment, especially near the shore, can be very complex. Some of the animals within the water can also make sounds.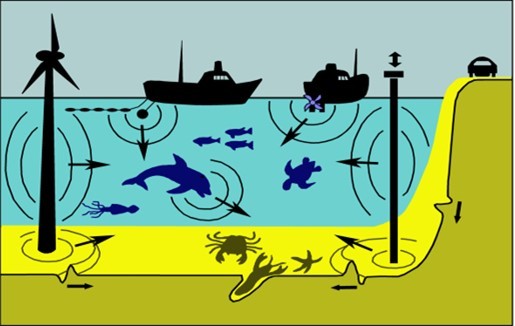
The seismic sonar systems used for seabed examination by humans are especially noisy; so are wave and tidal power resources; as well as attendant increases in shipping and trawling. Seismic sonar systems are used to explore the geological structure of the seabed, using an array of air guns that are slowly towed up and down parallel track lines. The guns are especially noisy, generating intense, low-frequency acoustic signals through the instantaneous release of highly compressed air. Such surveys can operate continuously over 24 hours a day, and can last from weeks and months, covering large areas. The sounds generated by ships propagate along the surface, and pass down into the water column, and get close to and in interaction with the sea bed, so that they will yield variable sound levels and spectra at different distances and depths. The use of fishing trawls may also generate sound close to the seabed. The hearing ability and also also the depth of the fishes and invertebrates will affect the probability and extent of their exposure to the sounds. Low, middle and high frequency sounds may determine gene expression in various species of fish that determine the survival and aging of the species. Thehuman-made sounds that are audible to fishes, and which potentially disturb or damage them or mask other relevant sounds, have been reviewed by 19, 8.
Damage to the animals
The human-made (anthropogenic) sounds may be audible to fishes and invertebrates and they can potentially disturb or deter them, or mask other sounds that are relevant to the animals17, 9. Exposure to man-made sounds can also have physiological and behavioral effects that may be detrimental to the animals. Some of these human-made sounds can kill or injure fishes and other aquatic animals, also impairing their hearing, and altering their behavior. Death can occur as a result of body damage taking place during sound exposure. Lower damage to body tissues can also take place, including: internal hemorrhaging; disruption of gas-filled organs like the swim bladder, and consequent damage to surrounding tissues. Single Gene Inactivation can occur with Implications to Diabetes and Multiple Organ Dysfunction Syndrome2. The animal may also receive injuries to its auditory system, with the ears themselves being damaged. When the animals are very close to sound sources they may be temporarily deafened by loud noise, and with fishes this may be the result of damage to the sensory hair cells in the inner ear. At even lower sound levels their hearing abilities may be affected. Low, middle and high frequency sounds may determine gene expression in various species of fishes that determine the survival and aging of the species. This may not cause immediate effects but may have longer-term consequences in terms of affecting their communication, reducing their avoidance of predators, or preventing them from capturing prey. The accelerated aging in fish by the frequency of sound may be an important factor in their survival. Lower levels still may also affect the behavior of the animals: for instance, driving an animal away from, or perhaps attracting it towards, an area. Animals may especially be excluded from key habitats, and this may occur at a ‘bad’ time in terms of their migrations or breeding.
The nature of underwater sounds
In water, sound is generated by the movement or vibration of any immersed object and results from the inherent elasticity of the surrounding medium. As the source moves, kinetic energy is imparted to the medium and is passed on as a travelling acoustic wave, within which the component particles of the medium are alternately forced together and then apart. The particles of the medium oscillate back and forth along the line of transmission in waves of compression and rarefaction. The disturbance propagates away from the source at a speed that depends on the density and elasticity of the medium. The combined effects of sound on fish survival may be assessed with relevance to the temperature and xenobiotic content of the water. The Underwater sound is essentially made up of two elements. Sound is generated by the movement or vibration of some immersed object and results from the inherent elasticity of the surrounding medium. There are waves of compression and rarefaction – termed the Sound Pressure. These are monitored by conventional aquatic hydrophones. However, in addition, as a result of the motion of sound sources in water, particles of the water are alternately forced together and then apart (Figure 2). This is termed the Particle Motion, which travels along a line of passage, and is a vector quantity. Particle Motion levels are much higher in the near field, close to the source, especially at low frequencies.
The Sound Pressure acts in all directions (it is a scalar quantity). It is generally measured as levels of micro Pascal (μPa). In a free or ideal sound field, especially in the open sea, the Particle Motion can be calculated from the measurement of the sound pressure, using wave equations; the plane wave equation or the spherical wave equation. In the far-field, distant from the source, the Particle Motion is directly proportional to the Sound Pressure. Closer to the source the Particle Motion is higher for a given Sound Pressure – the Near Field Effect. Sound is reflected and refracted at boundaries with different media: Close to the water surface, and in shallow water, the Particle Motion increases, and is vertical, as a result of pressure release into the air. Under many aquatic circumstances it can be quite difficult to estimate the Particle Motion by measuring the Sound Pressure. Most fishes and invertebrates are primarily sensitive to the Particle Motion, which enables them to determine the direction from which the sound is coming, and only a few fish species are sensitive to the Sound Pressure. Even those which can detect Sound Pressure may rely on detecting Particle Motion to determine the direction of a sound source through vector weighing. The fishes and invertebrates are moved back and forth by the Particle Motion within the water, and are very sensitive to it.
Figure 2.Underwater sound is made up of two elements: There are waves of compression and rarefaction – the Hydrostatic Sound Pressure. But in addition, Particles of the water are alternately forced together and then apart – the Particle Motion. Marine Mammals are sensitive to the Sound Pressure. However, Fishes and Invertebrates mostly detect the Particle Motion, although a few fishes are able to detect the Sound Pressure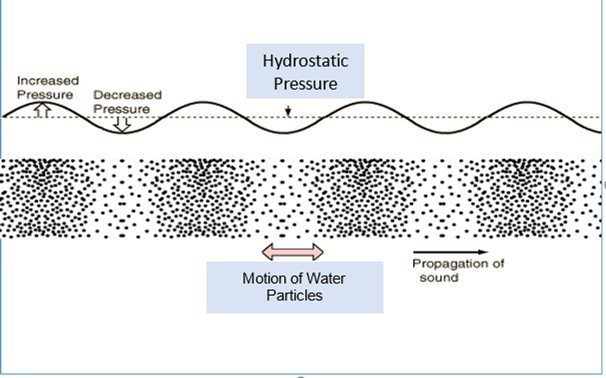
It is possible for us to monitor the Particle Motion by detecting the Particle Displacement or its time derivatives, the Particle Velocity and Particle Acceleration, and it is important to specify its direction. It is possible to monitor the Particle Motion and its direction both in the water and on the substrate using accelerometers, which can be orientated in 3 orthogonal directions, but often this is not done. It is more common to monitor only the Sound Pressure using conventional hydrophones, and that is Insufficient.
Substrate vibration
Seismic interface waves may travel along the surface of the substrate generating high levels of particle motion9. One form of substrate vibration is the seismic surface wave (or ground roll) generated by a pile driver (Figure 3), that not only propagates along the surface of the substrate, but also produces particle motion that enters the water column18, 12, 27.
Figure 3.A seismic interface wave (ground roll), is created here by the impact of a driven pile. The impact energy excites vibration waves traveling radially outward from a central point source. A pile driver, struck by a vertical hammer creates sound pressure waves in water and vibrational waves within the substrate. The motions of some particles, above and below the seismic interface. waves, are shown using hodographs.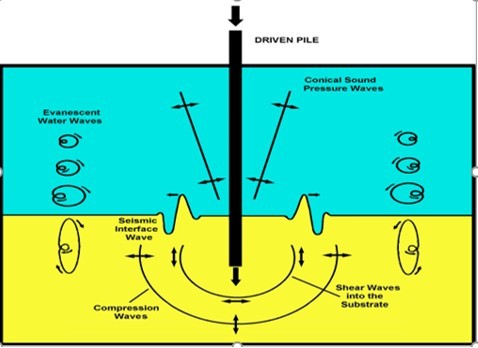
Human activities that can generate vibration of the substrate underwater include: pile drivers; explosives; offshore wind-driven electric turbines that are fixed to the seabed rather than floating at the surface; dredging and trawling activities; aircraft generated sonic booms; air guns used for seismic surveys; and even subsurface transportation tunnels and onshore vehicles on roads close to the water’s edge or on bridges with in-water piling13, 20. Natural sources of substrate vibration include volcanos, earthquakes, and breaking waves, also animal movements/interactions, and falling and rolling objects onto/on the seabed. There is, however, little data on the ambient levels of particle motion close to the seabed and within the substrates of lakes and rivers. Nor is there information on the levels and the characteristics of the particle motion generated by anthropogenic sources in and on the substrate, which may have major effects upon fishes and invertebrates, all of which primarily detect particle motion. Very little is currently known about the sensitivity of aquatic animals to the energy that is generated within and close to the substrate23, 24, 25. However, However, some information is available on the sensitivity of terrestrial invertebrates and vertebrates to substrate vibration28, 15, 11, 27.
Sounds made by fishes and invertebrates
Many marine mammals, fishes, and invertebrates make sounds themselves. Amongst the vocal fishes are some of the most abundant and commercially important fish species, including the cod & haddock (Gadidae). Most of the gadoid fishes make underwater sounds, and the sounds differ between species. Some produce knocking sounds, while others produce grunts. In all cases the sounds are pulsed, with each pulse generated by the contraction of a pair of Drumming Muscles attached to the gas-filled swim bladder (Figure 4). The muscles show very fast contraction.
Figure 4.The drumming muscles of the haddock, that compress the swim bladder and generate sounds.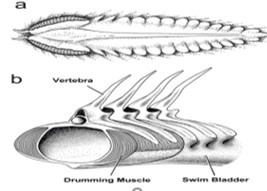
The sounds are made quite often and are especially made during spawning, especially by the male fishes. A good example is the haddock (Figure 5), and their sounds during courtship and mating, have been recorded and analyzed in the aquarium and in the sea5, 6.
Figure 5.The male haddock varies its sounds during the spawning behaviour, while the female remains silent. On the seabed (a), the male makes repeated sounds that can keep other males away but they attract a female. The male then rises up through the water (b) towards the female and then speeds up the sounds (c) as she approaches him. He then embraces the female (d) and as they move further up through the water together the male then stops producing sounds (e). The female then releases eggs and the male releases sperm so that they create a new juvenile haddock.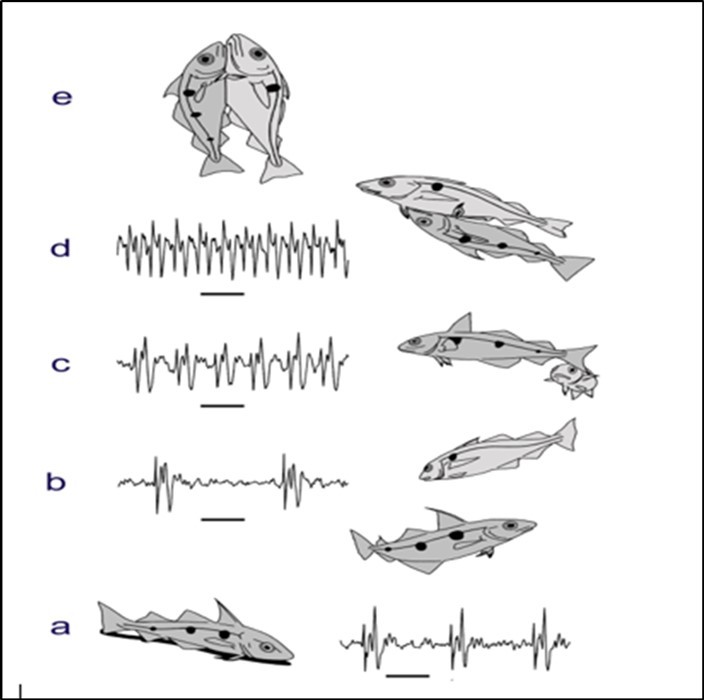
The male haddocks gather together at particular spawning locations close to the seabed, and they use sounds to attract the females, and to advertise their presence at a particular location, that is especially useful for females that wish to engage in spawning with a male. The sounds are often varied during their spawning. The females, that are mainly in midwater, are attracted to the seabed and then select a particular male to spawn with, discriminating males with different characteristics through sound detection. The sounds of individual male haddock can readily be distinguished. The male haddock varies its sounds during the spawning. The male that is chosen by a female moves up into midwater, attracting the female with a new sound, and then mounts the female to spawn, but once it becomes attached to the female it stops making its sounds. The male and female embrace one another and release their eggs and sperm into the water to create juvenile haddock. The female haddock remains silent during spawning, but can make sounds normally. The haddock, including both the males and females, produce sounds when they are feeding, gathering together, or fighting, and they also make sounds associated with their swimming. They produce species-specific sounds, and even individually-specific sounds. Their sounds are often loud, and in areas where haddock are common their sounds may dominate the acoustic environment.
Invertebrates, including squid, lobsters, sea urchins, and some crabs, living close to the seabed, also make sounds.
The hearing abilities of fishes and invertebrates
Fishes and aquatic invertebrates often use sounds to navigate through the water and to detect signals from natural environment sources, and from other animals. Some of the fishes live in mid water, while other fishes and many of the invertebrates live close to the substrate. The vision ability of these animals in the water is generally poor and they use the “acoustic scene” to locate natural underwater features, including preferred habitats, as well as their prey and predators, and they also use sound to set their orientation during migration. Sound travels further and faster in all directions through water than it does through the air and it allows aquatic animals to detect natural sources, and to communicate well with one another. Underwater sound is highly relevant to most aquatic animals. They listen to the “acoustic scene” and use this to orientate and navigate.
Fishes mainly have ears that are sensitive to Particle Motion. There are two fish ears, within the skull, each containing 3 otolith organs, the saccule, utricle and lagena (Figure 6), each of these organs contains a heavy component (the otolith) and it is attached to sensory hair cells that respond to the motion of the otolith. The fish itself, and the sensory tissues have approximately the same density as water and move back and forth in a sound field. However, the otolith is much denser and moves with a different amplitude and phase, resulting in the sensory hair cells that are in contact with the otolith being subjected to a directional shearing force. However, some fishes have their gas-filled swim bladder, that renders them buoyant, close to the ear, where sound pressure causes volume oscillations which are transferred to the inner ear, often via a physical connection, e.g. through paired bladder extensions, additional air cavities or a series of bones. The compression of the swim bladder by Sound Pressure can therefore generate Particle Motion close to the ears, especially at higher frequencies, stimulating the otolith organs and enabling such species to detect the Sound Pressure as well as the Particle Motion.
There is much less information available on the hearing of invertebrates, but it has been pointed out that they have a number of organs which are probably sensitive to particle motion16. Considerably more data are needed on the wide array of invertebrate species that live near to, on, or within the substrate, but there is evidence that at least some species including mussel and hermit crab are sensitive to physical vibration at frequencies of 5 – 410 Hz22, 25, 26. Sensitivity to vibration of water, sand, and the buried animal itself was shown for the North Sea shrimp (Crangoncrangon) and wasmaximal at 170 Hz10. In a series of field-based experiments, Day et. al. investigated the effects upon scallops of exposure to seismic surveys3. The ground roll acceleration was measured using geophones placed on the seabed. It was concluded that the impacts resulted from high seabed ground accelerations driven by the air gun sounds. Thus, while mainly speculative at this point, it is likely that many invertebrates are able to detect substrate vibrations. Aquatic Invertebrates are also sensitive to the Particle Motion. For example, the Lobster has hair fan organs that respond to Particle Motion, giving them low frequency audiograms. Of course, aquatic animals do not hear all frequencies within their functional hearing range. They are more sensitive to some frequencies than others. It is important to examine their audiograms.
Fishes and invertebrates are often listening to sounds, both the sounds made by their own species, the sounds made by other species, and the natural sounds in the environment. However, background noise levels can sometimes be quite high, over a wide frequency range but especially at the lower frequencies, and they may mask the use of sounds by the species6. Any background noise that exists must be taken into account when determining auditory thresholds.
Dealing with anthropogenic sounds
In the developing world xenobiotic levels have risen and combined effect of anthropogenic sounds, temperature and xenobiotic may determine survival. Body Temperature Regulation determines immune reactions and Species Longevity1. The presence of adverse underwater sounds (often termed “noise”), and also substrate vibration, generated by human activities (anthropogenic sources) can be quite harmful to fishes and aquatic invertebrates, as the detection of sounds of interest to the animals can be adversely influenced by the presence of noises, whether the noises are natural sounds or sounds from human sources. As part of the process of risk assessment it is necessary to know the levels of sounds that may cause potential harm to animals, as well as those levels that are likely to be of no consequence. Developing such criteria for harm resulting from sound exposure is currently of high priority. Criteria are usually provided as a threshold value of an acoustic metric, above which a particular level of damage or effect is likely to occur. The abilities of fishes and invertebrates to use sound to navigate, detect predators and prey, select habitat types, communicate with one another, and take part in spawning, may be strongly affected by exposure to anthropogenic sounds (noise). The noise may cause physical damage to the body tissues; damage to the auditory tissues, including the sensory hair cells; and changes to important behavior patterns, adversely affecting feeding migrations, and spawning. Masking of biologically important sounds, including those from the same species, may also be detrimental.
Noise levels in the sea, lakes and rivers have been changing dramatically as a result of human activities., and the noise that is being generated may have adverse effects upon aquatic animals. The sea, lakes and rivers are relatively dark, low-visibility environments, where it is difficult for animals to view things, and making and detecting sound is especially important.
The effects of sound on animals vary with distance from the source. However, modelling of particle motion levels at different distances from the source is rarely carried out in Impact Assessments. Models have recently been developed for dealing with ground roll generated by seismic surveys, but such models also need to be applied to pile driving and other sources of substrate vibration. Normally, assessments and modelling are only done in terms of sound pressure. Often: no mention is made of particle motion; no account is taken of substrate or interface waves; and sensitivity to Infrasound is ignored. Great efforts are now being made to examine the effects of underwater noise upon marine mammals, but much less attention is being paid to effects upon fishes and invertebrates.
Setting the sound exposure criteria
As part of the process of risk assessment it is necessary to know the levels of sounds that may cause potential harm to animals, as well as those levels that are likely to be of no consequence. Developing such criteria for harm resulting from sound exposure is currently of high priority. Criteria are usually provided as a threshold value of an acoustic metric, above which a particular level of damage or effect is likely to occur. The actual damage should be, but is often not, specified clearly. The Metrics should be, but is not always, specified clearly. They are measures by which sounds can be defined and compared; values that quantify the effects of a sound. The term sound exposure is used in a general sense to describe the dose of sound received by an animal in terms of both its magnitude and its duration. It is critical for regulators to have knowledge of the levels of sounds that may be of potential harm to animals, as well as levels that have few or no consequences. For marine mammals, the UK Joint Nature Conservation Committee currently recommends the use of the 29 criteria for impact assessment. The US Navy is also considering new criteria applicable to its operations based on a more recent paper 4 and has published draft Environmental Impact Statements (EISs) for training and testing activities of its Atlantic USN 2012a, and Hawaii- Southern California USN 2012b fleets, taking into account effects upon a wide range of animals. Separate sound exposure criteria are required for fishes and invertebrates as their hearing
mechanisms and their behavior are very different. Tentative exposure criteria have been
suggested for the onset of direct physical injury in fish exposed to the impact sound associated with pile driving. However, there are few criteria that apply to behavioral responses of fishes or other sub-injurious auditory effects, largely due to the absence of underlying information. Recent papers by Halvorsen et al., (2011, 2012b; 2012c) and Casper et al., (2013) set out to provide quantitative data to define the levels of impulsive sound that could result in the onset of injury to fish. A controlled impedance fluid filled wave tube was used to simulate in the laboratory exposure to high-energy impulsive sound pressures that were characteristic of aquatic far-field, plane-wave acoustic conditions. The sounds used were based upon the impulsive sounds generated by an impact hammer striking a steel shell pile. Neutrally buoyant juvenile Chinook salmon (Oncorhynchus tshawytscha) and other species were exposed to impulsive sounds and the injuries sustained subsequently evaluated for different sound exposure levels. No criteria exist for invertebrates. Knowledge of the hearing abilities of aquatic invertebrates hardly exists. Auditory thresholds and audiograms are required for these species under natural and varied noise conditions.
Actual criteria and metrics for assessing the effects of underwater sound on fishes and invertebrates
Introduction
Initiatives like the EU Marine Strategy Framework Directive and application of the OSPAR Convention (The OSPAR Convention is the current legal instrument guiding international cooperation on the protection of the marine environment of the North-East Atlantic) have recently been aimed at protecting the marine environment from noise. Increasingly, environmental assessments of the impact of offshore developments have had to consider the effects of underwater noise on marine animals.
Such assessments involve evaluation of the effects of noise in causing physical injury, impairment of hearing, behavioural disturbance, and population level impacts in marine animals. The likelihood that an adverse effect upon biological receptors may occur as a result of sound exposure is evaluated. Often a threshold for an adverse effect is sought. A conclusion is subsequently reached about the severity of the effects. Risk assessment can then be used to construct “what-if” scenarios to evaluate new and existing technologies for effective prevention, control, or mitigation of impacts, and to provide a scientific basis for action to reduce risks.
As part of the process of risk assessment it is necessary to know the levels of sounds that may cause potential harm to animals, as well as those levels that are likely to be of no consequence. Developing such criteria for harm resulting from noise exposure is currently of high priority. Criteria are usually provided as a threshold value of an acoustic metric, above which a particular level of damage or effect is likely to occur. The nature of the damage to the animal and the metrics for the sounds should be specified clearly. But often they are not.
The first set of comprehensive underwater sound exposure criteria was defined for marine mammals29. There is much less information available for other aquatic animals including fish and invertebrates, although impacts are currently having to be assessed and interim procedures have been developed to achieve this. This paper reviews attempts to set sound exposure criteria for fish and invertebrates and considers the metrics associated with these criteria.
The term noise is often used to describe unwanted sound, or sound that interferes with detection of any other sound that is of interest. However, noise is also used to describe background sounds in the sea, including the naturally occurring and spatially uniform sounds generated by distributed biological sources, weather events, or other physical phenomena, some of which cannot be assigned to individual sources. In this paper we refer to sounds from identifiable sources, such as vocalising animals, ships or oil and gas platforms, or distinct sources that cannot always be located or identified. We will also use noise to describe unwanted sounds, whatever their source. Where others have used the term ambient noise or background noise to describe naturally occurring sounds from distributed sources, or where noise is used to describe interference with signal detection, then that usage will also be followed.
The assessment process
Environmental statements or assessments are often required for major industrial and other developments proposed for particularly environmentally sensitive or vulnerable locations; or which may have potentially hazardous environmental effects. Any significant effects of development are considered in relation to criteria set out in the guidance, having regard in particular to the geographical extent of the impact; the magnitude and complexity of the impact; the likelihood of impact; and the duration, frequency and reversibility of the impact.
While a careful study of the proposed location is generally required (including environmental survey information), original scientific research is not normally undertaken. There is extensive reliance upon existing scientific papers and reports. However, there is often uncertainty attached to the prediction of environmental effects and this uncertainty is not always given the emphasis it should be. Environmental statements and the supporting documentation receive scrutiny and comment within a legal or quasi-legal context rather than discussed in a scientific forum. Examination is usually not undertaken by independent scientists but by lawyers and admission of uncertainty may be seen as a weakness.
Those preparing environmental statements are usually preparing the statement on behalf of those having particular interests in the outcome. There may be both explicit and subtle pressures to achieve uniformity and consensus. Such statements cannot always be regarded as objective assessments based on the best achievable science. They may reflect the interests of the developer, or alternatively the interests of those opposed to the development. It has become commonplace for such statements rarely to refer to published scientific papers and independent primary sources. Instead they often depend upon other sources including unpublished references and reviews. Documents produced by Governmental agencies may also receive consideration. These documents may state the legal position and present recommendations on what needs to be done to prevent damage to the environment or to protect endangered species. In some cases Government agencies may also present criteria by which damage should be judged, although those criteria do not necessarily reflect the most recent scientific position.
This document reviews present thinking on assessing the effects of noise on fish and invertebrates. It describes the potential effects of noise, and considers those metrics that would allow regulators to assess the likelihood of effects more reliably. It also identifies deficiencies and suggests areas of research that might lead to more consistent and considered evaluation of the effects of sound on fish and invertebrates.
The issues arising with respect to the exposure of fish and invertebrates to noise
As a preliminary it is worth considering the issues that actually arise during the submission of environmental statements with respect to the impact of noise on fishes and invertebrates. We have taken two typical examples in relation to the effects upon protected species of migratory fish of construction noise associated with the installation of inshore wind farms; one in the United Kingdom and the other in the United States of America.
In the UK proposal for an inshore wind farm, the applicant energy company noted that the key cause for concern was the potential for piling noise (created during the installation of turbine foundations) to disrupt the migration of adult Atlantic salmon to local freshwater sites. These sites were designated protected sites under European law, with Atlantic salmon as a particular conservation feature. The applicant was accompanied by specific condition, agreed with regulators, with respect to seasonal restrictions in the piling to cover the sensitive period when juvenile salmon were leaving the rivers and migrating into the sea. The applicant also proposed ‘soft start’ procedures that might act to reduce lethal or non-lethal physical injury or otherwise potentially injurious effects on fish species.
The environmental statement set out a worst-case scenario of large diameter steel piles being installed at 70 locations. The use of a worst-case scenario, followed by a sentence indicating that in reality it is expected that the worst case will never occur, appears to be commonplace in such statements. The underwater noise emissions resulting from this worst-case piling scenario were estimated and supported by site-specific noise propagation modeling. Predicted noise levels were presented using two metrics relevant to fish:
Unweighted peak-to-peak noise levels; for assessing the likelihood of death and physical injury;
Weighted levels; for assessing auditory injury and behavioral effects upon Atlantic salmon.
The proposed weighted metric adjusted the measured noise levels to reflect the hearing abilities of Atlantic salmon. It was argued that a particular level of this metric was important in relation to potential impacts on migratory fish. A level was selected, based on initial observations of a range of fish species exposed to swept tonal sounds (rather different from those generated by pile driving strikes). At the selected level it was said that a large proportion of the individuals might react to the noise. However, the effect might be limited in duration and repeated presentation of the noise at this level might result in a decrease in the level of the response or in the proportion of the fish responding. Noise level contours were then drawn for the particular area, showing the area where the noise would exceed the selected level. It was emphasized that the contour should not be regarded as constituting a fixed boundary, within which all fish were likely to be adversely affected, but rather as an approximate indicator that within the area bounded by the contour a large proportion of fish might respond to sound to some degree. The nature of the response itself was not specified.
The noise contour for salmon came close to parts of the coast adjacent to the mouths of the salmon rivers concerned. It was suggested, however, there would be corridors for migration of salmon outside the contour plots at higher tidal heights. Indeed, it was proposed that clear corridors existed between the modeled contours and the coastline. These corridors could potentially allow the passage of returning adult salmon along the coastline and into the river estuaries at all or most states of the tide. It was also argued that in the UK a number of salmon rivers are subject to almost constant or intermittent noise exposure from busy harbors and associated shipping activities. Nevertheless, the salmon stocks (and dependent fisheries) of such rivers appeared to display resilience to such disturbance.
Based on the assessment undertaken and the suggested conditions, the applicant suggested that there would be no adverse effect upon breeding salmon populations. There was no significant risk of instantaneous mortality/injury, and the area within which noise levels might provide a potential disturbance to fish was not sufficient extensive to conclusively alter the migratory and breeding behaviour of the fish.
Those opposed to the development opined that it could not currently be concluded that the project would not have a likely significant effect on Atlantic salmon returning to their protected river systems. It was stated that the weighted metric employed had not been peer reviewed and the value of the hearing thresholds used in the prediction of underwater noise impacts had recently been questioned by others. The data and conclusions should be presented in internationally recognised metrics. However, the level chosen as likely to invoke a behavioral response appeared to have been accepted. It was agreed with the applicant that that not all of the salmon within the specified contour would be prevented from reaching their breeding grounds; and that there was some evidence that migratory salmon might follow the coastline and might therefore avoid entering the area specified as likely to invoke responses. It was also agreed that piling for a period of up to six hours followed by a six-hour period of no piling would to some extent reduce the impact of the operations.
The main argument in opposing the project was that there was a risk that fish delayed in their migration through a response to the noise generated might be prone to additional mortality. Evidence from salmon tracking studies had shown that fish tagged in the lower estuaries of rivers in low flow conditions during warm summers left the river system and were not detected again (i.e. they were effectively lost from the breeding population). Delayed entry to an estuary linked to disturbance from piling noise, resulting in a missed opportunity for in-river migration, could have similar consequences. There was a particular risk that delayed fish might be subject to increased levels of predation from seal colonies present in the area. Given the uncertainties about migration routes and the behavioral responses of salmon to piling noise it was unclear what proportion of the population might be affected by the development and what the implications were in terms of any additional mortality. The evidence available could not put an adverse effect beyond reasonable scientific doubt. Accordingly, the authority charged with nature conservation advised that there was sufficient uncertainty as to the likely effects on migratory salmon that an adverse effect on site integrity could not be excluded in respect of the worst-case piling scenario.
It was acknowledged by opponents that steps taken to reduce the exposure of fish to piling noise would reduce the risk of any adverse affect. Reduction in either the size of the piles or in the number of piling operations would significantly reduce the potential noise exposure of salmon, perhaps preventing any adverse effects on the integrity of the protected sites. If the applicant was able to accept suggestions for reducing the noise levels, then the conservation advisers did not see the need for any further ‘fish science’ being necessary.
The outcome of these discussions depended upon the definition of a particular contour as a significant threshold for a behavioral response by a large proportion of the exposed salmon. The contours for this metric also depended upon assumptions made in the modeling of sound propagation at the particular site. In practice there was considerable uncertainty in relation to both these sets of assumptions. The main focus of argument, however, was the likelihood of fish falling within the estimated contours, the behavior they might subsequently show in terms of delayed migration, and the consequences of this delay in terms of subsequent survival to breed.
Summary
It is evident that there are major gaps in our understanding of the effects of anthropogenic sounds on individual fishes and invertebrates as well as the potential impacts of such sounds upon fish and invertebrate populations and aquatic ecosystems. Much of the research and literature has limited applicability, as many of the experiments until now have been carried out on captive fishes and invertebrates under laboratory conditions where animals do not behave in the same way as they do in the wild. Additionally, there is a lack of information on their responses to particle motion rather than to sound pressure. Thus, to develop better guidelines and criteria for fishes and invertebrates, it will be critical to fill many knowledge gaps on the potential impacts of sounds upon them. The goal must be to increase knowledge so that sound exposure criteria can be revised and improved. Such quantifiable indicators of impact or response need to be integrated into environmental impact assessments to ensure that fishes and invertebrates are protected. It is important to assess the sound levels at which the animals are at risk of death or sustaining serious injury to internal organs, including damage to hearing. Further research is also needed on the behavioral responses of a range of aquatic fish and invertebrate species to different sound sources and under different conditions. As well as investigating responses to sounds of short duration, information is also required on responses to continuous or repeated exposure. What are the immediate effects of sound exposure, what are the longer-term effects in terms of fitness, and what are the likely impacts on populations?
References
- 2.J Clin Epigenet. (2017) Single Gene Inactivation with Implications to Diabetes and Multiple Organ Dysfunction. , Syndrome 3(3), 24.
- 3.R D Day, R D McCauley, Q P Fitzgibbon, Hartmann K, J M Semmens. (2017) Exposure to seismic air gun signals causes physiological harm and alters behavior in the scallop Pecten fumatus,”. , Proc. Natl. Acad. Sci. U.S.A 114, 8537-8546.
- 4.J, A K Jenkins. (2012) Criteria and Thresholds for U.S. Navy Acoustic and Explosive Effects Analysis.
- 5.A D Hawkins, C J. (1966) Underwater sounds of the haddockMelanogrammus aeglefinus(L.). , Journal of the Marine Biological Association of the United Kingdom 46, 241-247.
- 6.Hawkins A D, Chapman C J. (1975) Masked auditory thresholds in the cod,GadusmorhuaL. , J Comp Physiol 103, 209-226.
- 7.Hawkins A D, Amorim MCP. (2000) Spawning sounds of the male haddock,Melanogrammus aeglefinus. , Envir. Biol. Fishes59: 29-41.
- 8.Hawkins A D, Pembroke A E.Popper AN (2014a) Information gaps in understanding the effects of noise on fishes and invertebrates. Rev. fish Biol. Fisheries. in press
- 9.Hawkins A D, Hazelwood R A, Popper A N, Macey P C. (2021) Substrate vibrations and their potential effects upon fishes and invertebrates. , Journal of the Acoustical Society of America 149, 2782.
- 10.Heinisch P, Wiese K. (1987) Sensitivity to movement and vibration of water in the North Sea shrimpCrangoncrangonL,”. , J. Crustac. Biol 7, 401-413.
- 12.J M Hovem. (2014) Particle motions caused by seismic interface waves,”. in Proceedings of the 37th Scandinavian Symposium on Physical Acoustics, February 2–5 , Geilo, Norway 1-12.
- 13.S B Martin, A N Popper. (2016) Short-and long-term monitoring of underwater sound levels in the Hudson River. , (New York, USA),”, J. Acoust. Soc. Am 139, 1886-1897.
- 14.Tine Mbaye. (2017) Evidence of the Complexity of Gene Expression Analysis in Fish Wild Populations. Int J Genomics. 1258396.
- 15.P M Narins, E R Lewis, J, O’Riain J. (1997) The use of seismic signals by fossorial southern African mammals: A neuroethological gold mine,” Brain Res. , Bull 44, 641-646.
- 16.A N Popper, Salmon M, K W Horch. (2001) Acoustic detection and communication by decapod crustaceans,”. , J. Comp. Physiol. A 187, 83-89.
- 17.Popper A N, Fewtrell J, Smith M E, McCauley R D. (2003) Anthropogenic sound: Effects onthe behavior and physiology of fishes. , Marine Technol. Soc. J 37, 35-75.
- 18.A N Popper, M C Hastings. (2009) The effects of anthropogenic sources of sound on fishes,”. , J. Fish Biol 75, 455-489.
- 19.Popper A N, Hawkins A D, Fay R R, Mann D A, Bartol S et al. (2014) Sound exposure guidelines for fishes and sea turtles. ASA press (Acoustical Society of America, Springer):. Zeddies DG, Tavolga WN S3/SC1.4 TR-2014 .
- 20.D B Reeder, J E, T A Rago. (2020) Underwater sound generated by motor vehicle traffic in an underwater tunnel,”. , J. Acoust. Soc. Am 148, 215-220.
- 22.Roberts L, Cheesman S, Breithaupt T, Elliott M. (2015) Sensitivity of the musselMytilus edulisto substrate-borne vibration in relation to anthropogenically generated noise,”. , Mar. Ecol. Prog. Ser 538, 185-195.
- 23.Roberts L, Breithaupt T. (2016) Sensitivity of crustaceans to substrate borne vibration,” in The Effects of Noise on Aquatic Life II. edited by , New York) 925-931.
- 24.Roberts L, Cheesman S, Elliott M, Breithaupt T. (2016) Sensitivity ofPagurusbernhardus(L.) to substrate-borne vibration and anthropogenic noise,”. , J. Exp. Mar. Biol. Ecol 474, 185-194.
- 25.Roberts L, Elliott M. (2017) Good or bad vibrations? Impacts of anthropogenic vibration on the marine epibenthos,” Sci. Total Environ. 595, 255-268.
- 26.Roberts L, M E Laidre. (2019) Finding a home in the noise: Crossmodal impact of anthropogenic vibration on animal search behavior,”. , Biol. Open 8, 041988.
- 27.Roberts L, Howard D. (2021) Substrate-borne vibrational noise in the Anthropocene: From land to sea,” in Biotremology: Physiology, Ecology and Evolution, edited by. in press , New York)
