Acute Effects of White Button and Shiitake Mushroom Powder Supplementation on Postprandial Lipemia and Glycemia Following a High-Fat Meal
Abstract
Background
To determine the acute effects on postprandial lipemia and glycemia by supplementing a high-fat meal with either white button (WB) or shiitake (SH) mushroom powder.
Methods
Nine healthy participants (4-male, 5-female, 23.3±1.3 years, 17.8±6% body fat, 56.2±11.4kg fat free mass) consumed a control hamburger. At one-week intervals, after consumption of a control meal, participants consumed hamburgers in random order, supplemented with 14g of either WB or SH mushroom powder. Peripheral blood for lipids (triglycerides, high-density lipoprotein HDL, low-density lipoprotein LDL), and glucose was obtained at baseline (t=0 hours) and postprandially every two hours for six hours. Data were analyzed using linear mixed effects models.
Results
Lower LDL levels were observed for both SH and WB burgers compared to the control burger (p=0.0007) over the six-hour period. Mushroom powder content did not alter triglyceride, HDL, or glucose levels. Gender affected triglyceride and HDL levels over the treatment course. Triglyceride levels were higher in males (p=0.0084), and HDL levels were lower in females (p=0.0005). Triglyceride and glucose levels were higher, (p< 0.001 and p< 0.0001 respectively), during the postprandial time course (t=0, 2, 4, 6 hours).
Conclusions
Supplementing SH or WB mushrooms during a high-fat meal may lower serum LDL levels.
Author Contributions
Academic Editor: Ian James Martins, PRINCIPAL RESEARCH FELLOW Edith Cowan University
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2023 Lillian A. Talal, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Citation:
Introduction
The current trends of rising obesity levels and poor dietary choices have led to an increased incidence of cardiovascular diseases (CVD), dyslipidemia, and metabolic syndrome 1. Nonmedicinal, cost-effective therapeutic approaches for dyslipidemia that would be readily accepted by individuals who are reluctant to make drastic dietary changes, such as eliminating consumption of red meat, are needed. As opposed to the use of supplements, such as omega-3 fatty acids, niacin, and plant sterols, whole foods may have greater appeal 2, 3, 4. Research into medicinal uses and therapeutic applications of mushrooms is increasing; however, there has been little, if any, investigation into the acute effects of mushrooms on postprandial lipemia (PPL) or postprandial glycemia (PPG), especially in humans 5, 6. There is currently no widely agreed upon technical definition of PPL 7, however certain characteristics of identifying PPL have been acknowledged. Generally, PPL is defined as a complex condition that is characterized by non-fasting hypertriglyceridemia, which is associated with an increased risk of cardiovascular events 7. This pilot study investigated the acute effects of selected mushroom powder supplementation on PPL and PPG following a high-fat meal using the indicators of serum triglycerides, serum high-density lipoprotein (HDL), serum low-density lipoprotein (LDL), and serum glucose.
Mushrooms are a widely available commodity and are heavily consumed for their health-promoting properties, in addition to their palatable and culinary appeal 8. Mushrooms contain high levels of bioactive compounds, such as ergothioneine, a powerful antioxidant, and ergosterol, a hypercholesterolemic agent and a Vitamin D precursor 6. These bioactive compounds are thought to be potential mechanisms for reducing triglyceride and cholesterol absorption from the bloodstream, which could be beneficial for individuals who suffer from metabolic syndrome that includes diabetes, obesity, and cardiovascular disease 9. Soluble dietary fiber (e.g., β-glucans found in certain types of mushrooms) may reduce the rate of nutrient absorption, which has been associated with reduced postprandial blood glucose and higher satiety levels 10. Ergothioneine may also have a role in the reduction of postprandial triglyceride levels 11. Given the versatile uses of mushrooms, mushroom supplementation of high-fat foods may be a promising therapeutic application to lower PPL and PPG, and thus conditions associated with metabolic syndrome or syndrome X, such as obesity, diabetes, and CVD.
Besides their favorable biochemical properties, mushrooms also have a fleshy texture akin to meat products, such as soy 12, that might be used as a meat substitute as an approach to treat dyslipidemia 13, 14. In this study, we utilized shiitake (SH) and white button (WB) mushroom powder as supplements, since they are widely commercially available, and SH is the second most commonly produced edible mushroom in the world 8. In addition, several important anti-inflammatory, lipid-lowering, and antimicrobial compounds have been isolated from SH mushrooms including lentinan, a polysaccharide, which is well studied 8. β-glucan is the major bioactive component in lentinan 15. Eritadenine, a compound isolated from SH mushrooms, is thought to be the major mediator of the lipid-lowering effect 6, 16. Both SH and WB are thought to have cardioprotective effects 11. WB mushrooms, part of the genus Agaricus, have been demonstrated to reduce total daily energy and fat intake 11. They also have been shown to have antidiabetic properties including lowered index of insulin resistance, increased plasma adiponectin, improved hepatic glucose uptake, and reduced oxidative stress 11.
In this pilot study, we sought to determine the acute effects on PPL and PPG by supplementing a high-fat meal, a hamburger, with either WB or SH mushroom powder. We hypothesized that mushroom powder supplementation of a high-fat meal will result in decreased serum lipid concentrations and follow a standard postprandial glucose pattern.
Materials and methods
Participants were enrolled in a prospective, double-blind, randomized study approved by the University at Buffalo (UB) Institutional Review Board. All participants provided written informed consent prior to study entry. Inclusion criteria included ages 18 to 35 years, overall healthy, non-diabetic, and normal to mildly hypercholesterolemic (total cholesterol (TC): 200 – 239 mg/dL, low density lipoprotein cholesterol (LDL-C): 130 – 159 mg/dL), and body mass index (BMI) of <30 or body fat percentage (% BF) by gender (men: <25%, women: <32%). Participants were also screened for liver and gallbladder disease by self-report, lipid metabolism dysfunction, and normal lipemic responses to a lipid challenge (i.e., control meal) 17, 18. Participants completed three-day diet records to confirm their specific caloric and macronutrient intakes. Caloric and macronutrient intakes were determined using a nutrient analysis software entitled “Nutritionist Pro” (Axxya Systems LLC, Redmond, WA, USA). Participants were excluded if they failed to meet any inclusion criteria, failed any of the screenings, or had a self-reported mushroom allergy.
Study design and procedures
After participant screening and collection of anthropometric data, control burgers were used as the chosen method for the lipid challenge to assess baseline PPL. All participants consumed three meals: 1) plain hamburger with bun (control burger), 2) hamburger with WB powder and bun (WB burger), and 3) hamburger with SH powder and bun (SH burger). The control meal consisted of 368.5 grams (g) of irradiated ground beef patty (80/20 lean mass to fat ratio) and a Wegmans Food Market Brand Big Hawaiian bun (Rochester, NY, USA). Mushroom powder, derived from both SH and WB mushrooms, was generously donated by Oakshire Farms (Kennett Square, PA, USA), and was incorporated into the two types of test hamburgers before cooking. The use of mushroom powder allowed for the blinding of participants for all three meals and for investigators after the control burger, which was used to verify that each participant had a normal lipemic response. Each burger was approximately 1000 kcal consisting of 45g carbohydrate, 51g fat, and 72g protein. The nutritional composition of each burger was calculated using Nutritionist Pro software.
Both the measured beef and mushroom powder were added to a mixing bowl, hand kneaded into a uniform mixture, and added to a hamburger press to form patties of similar shape, size, and dimension (4.45 cm in depth and 12.1 cm in diameter). The hamburgers were then cooked (260oC for 15 minutes) in an industrial facility, turned, and removed upon reaching an internal temperature of 71.1oC, the minimum internal USDA cooking temperature for ground meat 19. The hamburgers were then bagged, labeled, and stored at -40oC until participant administration. Prior to each visit, the test hamburger was thawed by placement in cold running water, reheated in an Oster (Intertek) toaster oven (Boca Raton, FL, USA) until the internal temperature of the hamburger was 73.8oC, in accordance with USDA safety protocols for reheating ground meat products 19. The temperature was measured using a digital meat thermometer (Taylor Trutemp Pocket Test Thermometer, Rochester, NY, USA), and the hamburgers and buns’ weights were then recorded using a free-standing electronic food scale (Ohaus model GT8000 scale, Buffalo, NY, USA).
During the initial visit, each participant’s baseline lipemic response was assessed in response to the control burger. After the initial visit, at one-week intervals, participants consumed burgers supplemented with 14g of either WB or SH mushroom powder, mixed into the beef patty, until both mushroom powder burgers were completed. Participants received mushroom-powder supplemented burgers in a random order and were blinded to the contents of all three meals. Each participant completed a total of three visits. Participants maintained normal dietary habits throughout the study period; they were requested not to ingest caffeine or alcohol for 24 hours prior to their visits as verified by a 24-hour diet recall and nutrient analysis using Nutritionist Pro software. They were also asked to fast for 12 hours prior to control or mushroom-supplemented burger consumption. Participants were also asked not to participate in vigorous exercise for 24 hours prior to test meal consumption. Prior to the participant’s first test meal, anthropometric data were obtained including weight, BMI, BF%, and fat free mass (FFM) via the BodPod body composition system (Cosmed/Axxya Systems; Concord, CA, USA).
At the beginning of each visit, a 22-gauge indwelling catheter (Becton Dickinson Infusion Therapy Systems Inc., Sandy, UT, USA) was placed in an antecubital vein by a trained phlebotomist. A 7-inch microbore extension set (B. Braun Medical Inc. Bethlehem, PA, USA) was attached to the indwelling catheter and secured by sterile Tegaderm™ dressing (3M Health Care, St. Paul MN, USA). The catheter remained in the vein for the entire 6-hour study visit. A disposable 10 mL syringe (Nipro Corporation, Miami, FL, USA) was attached to the extension set, and a 10 mL sample of blood was obtained. Approximately 5 mL of blood was placed into each of 2 vacutainers in the following order: 1) serum (BD Vacutainer, Franklin Lakes, NJ, USA), which was allowed to clot at room temperature for 20 minutes and 2) EDTA (BD Vacutainer), which was immediately placed on ice for 20 minutes. Five mL of a 0.9% saline solution (B. Braun Medical Inc., Bethlehem, PA, USA) was used to flush lines between blood draws to prevent coagulation. Five mL of the saline solution was also injected into the participant’s vein, withdrawn, and discarded as a saline-blood mixture. All samples were centrifuged at 3000 RPM for 30 minutes at 4oC (Hermle Labnet Z383 Refrigerated Centrifuge, Labnet, Edison, NJ, USA). Samples were further aliquoted into 3 mL cryovials as serum, plasma, and whole red blood cells. Samples were then placed at -80°C for subsequent analysis. This process was repeated during all subsequent blood draws and study visits.
No time constraints were given to participants for completion of the burger, but participants were encouraged to eat the test meal as quickly as possible while not evoking gastrointestinal distress. Water consumption was monitored, and water was offered to the participants ad libitum. After meal ingestion, participants remained sedentary throughout the remainder of the visit. Blood was obtained at two-hour intervals for a total of six hours, resulting in four blood draws per visit. Before participants departed from the clinical research setting, they were given a three-day diet record to be completed at home between visits. Each study visit was completed between 2 and 7 days after the prior visit. The mean time of burger consumption was 14.3 ± 5.2 minutes.
Biochemical assays
Samples were batched and were analyzed for all study participants at all time points at the same time for all markers.
Statistical analysis
We initially computed descriptive statistics consisting of means and standard deviations (SD) and medians and interquartile ranges (IQR) for all measured markers (serum LDL, serum HDL, serum triglycerides, and glucose) for each burger type. We assessed the value of three separate burgers: control, WB-supplemented, and SH-supplemented. We also computed descriptive statistics of the measured markers per burger and stratified by gender (male/female). We performed graphical analysis using boxplots to visualize individual measured markers’ distributional characteristics, such as symmetry of distributions and existence of outliers.
We modeled the multivariate outcome vector Yi= (TRIGi, LDLi, HDLi, GLUCi) T, I = 1, …, 9 using linear mixed effects models. The multivariate analysis did not produce any significant results, possibly due to the very small sample size. Because the effect of the different diets and participant characteristics on each of the collected markers was of interest, we performed individual marker mixed-effects analyses where the outcome variable was each individual marker Yi = (TRIGi), or Yi = (LDLi), or Yi = (HDLi), or Yi = (GLUCi), i=1,…,9. The first analysis has as an outcome a vector acknowledging the fact that these measurements are taken on the same individual, and thus they are correlated. The second analysis treats these measurements as taken independently of each other. Hence, the outcome is the individual marker of interest: serum LDL, serum HDL, serum triglycerides, or glucose.
Mixed effects models are used to model longitudinal data with multiple sources of variation. Covariates in the model include gender (with responses of M or F), time (0, 2, 4, 6, hours), meal type (control, WB, or SH) and time x burger type interaction to account for the diminishing effect of burger type on different markers over time. The models include a random intercept that allows the outcome to vary from participant to participant as well as the covariates. We also include graphical analysis in which the timepoints depicted were 1) adjusted for the value obtained at time 0 (i.e., parameter of interest at time (t) = 0, 2, 4, 6) – time (0) or (2) consecutive differences (value of parameter of interest at time (t) - value at timepoint immediately preceding.
Results
Baseline characteristics of participants
Our sample consisted of 4 (44.4%) male and 5 (55.6%) female participants. The mean age of all 9 participants was 23.3 ± 1.32 years, while the median age was 23 ± 1 years. Female participants were slightly older on average (24.2 + 0.8) than male participants (22.3 ± 1.0) (Table 1). Mean lean body mass (LBM, kg) was also higher in females (65.5 + 6.3) compared to the LBM of the male participants (49.0 + 10.6).
Table 1. Descriptive statistics, stratified by gender, of study participant characteristics.| Variable | Male | Female | All participants | |
|---|---|---|---|---|
| 4 (44.4%) | 5 (55.6%) | 9 | ||
| Age | Mean ± SD | 22.3 ± 1.0 | 24.2 ± 0.8 | 23.3 ± 1.3 |
| Median ± IQR | 22.5 ± 1.8 | 24.0 ± 1.5 | 23 ± 1 | |
| LBM | Mean ± SD | 49.0 ± 10.6 | 65.5 ± 6.3 | 56.3 ± 12.1 |
| Median ± IQR | 49.3 ± 14.3 | 66.2 ± 8.4 | 57.8 ± 13.6 | |
Baseline values of biochemical parameters
Summary results of the four markers of interest (LDL, HDL, triglycerides, and glucose) at baseline, stratified by gender and burger type, are illustrated (Table 2).
Table 2 . Baseline descriptive statistics (by gender) for four biomarkers of interest| Biomarker | Gender | Control | White Button Mushroom Supplement | Shiitake Mushroom Supplement | |||
|---|---|---|---|---|---|---|---|
| Mean(SD) | Median(IQR) | Mean(SD) | Median(IQR) | Mean(SD) | Median(IQR) | ||
| LDL | F | 110.03 | 108.56 | 123.42 | 128.18 | 126.70 | 121.34 |
| (14.885) | (10.790) | (18.399) | (3.180) | (12.304) | (8.790) | ||
| M | 118.16 | 119.17 | 108.03 | 105.78 | 117.62 | 116.96 | |
| (25.399) | (42.358) | (17.132) | (22.893) | (17.779) | (28.708) | ||
| HDL | F | 69.74 | 71.00 | 71.63 | 74.00 | 70.95 | 72.00 |
| (12.173) | (18.520) | (5.184) | (1.390) | (9.683) | (11.110) | ||
| M | 49.70 | 46.00 | 47.04 | 45.00 | 46.33 | 45.00 | |
| (10.366) | (8.698) | (9.669) | (10.043) | (9.167) | (9.828) | ||
| TRIG | F | 58.20 | 55.91 | 57.62 | 53.45 | 77.91 | 78.66 |
| (16.481) | (20.570) | (13.464) | (15.070) | (29.895) | (36.070) | ||
| M | 106.29 | 103.21 | 97.49 | 101.55 | 130.44 | 126.89 | |
| (35.356) | (54.860) | (14.686) | (18.393) | (56.330) | (75.760) | ||
| Glucose | F | 82.60 | 82.00 | 81.40 | 84.00 | 83.40 | 83.00 |
| (2.881) | (4.000) | (5.177) | (4.000) | (2.702) | (1.000) | ||
| M | 83.25 | 85.00 | 71.50 | 73.00 | 86.25 | 82.00 | |
| (11.615) | (17.750) | (17.137) | (19.000) | (9.215) | (6.250) | ||
Table 2 presents descriptive statistics obtained at baseline (time t = 0) of the different markers stratified by gender and by diet. We present means and associated SDs as well as medians and IQR. The table indicates that for all participants, the measurements of the corresponding markers before the meal consumption are within the normal limits and that substantial variation of the measurements is present as indicated by the SD/IQR values.
Individual marker mixed-effects analysis
Individual marker analysis of the outcomes illustrated significant relationships between the outcomes and the covariates of interest. The results of the individual marker analysis of the outcome variables are demonstrated (Table 3). Graphical analysis is presented in Figure 1, Figure 2, Figure 3, Figure 4.
Figure 1.Consecutive Differences in Low Density Lipoprotein (LDL) Levels (mg/dL) stratified by Gender. Graphical illustration of consecutive differences (LDL value at time (t) - value at timepoint immediately preceding) for A) females and b) males.
Figure 2.Consecutive Differences in High Density Lipoprotein (HDL) Levels (mg/dL) stratified by Gender. Graphical illustration of consecutive differences (HDL value at time (t) - value at timepoint immediately preceding) for A) females and B) males.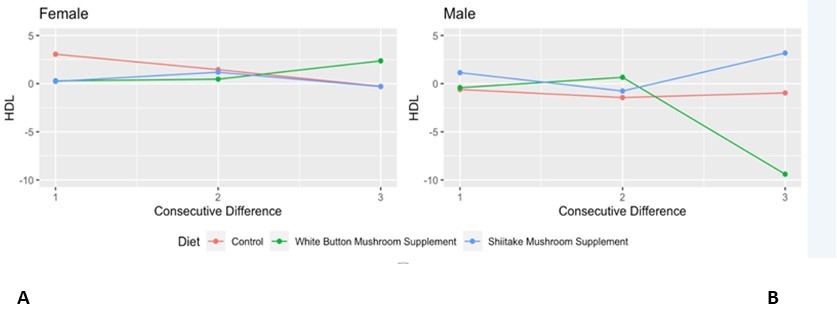
Figure 3.Postprandial Time Period and Consecutive Differences in Serum Glucose Levels (mg/dL) stratified by Gender. Graphical illustration of serum glucose values at baseline and over the six hours postprandially for A) females and B) males and consecutive differences (glucose values at time (t) - values at timepoint immediately preceding) for C) females and D) males.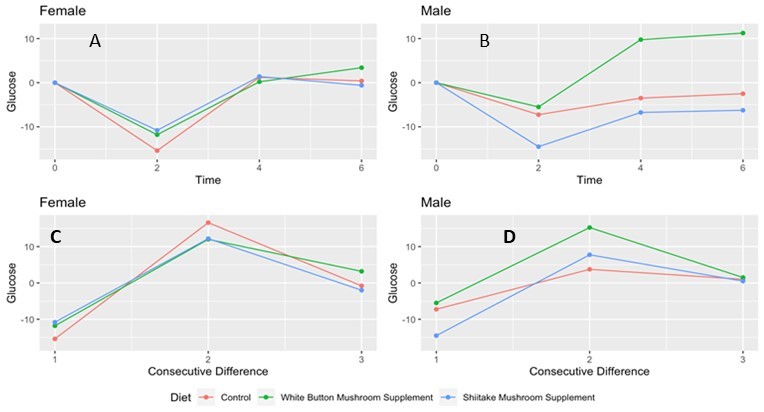
Figure 4.Postprandial Time Period and Consecutive Differences in Serum Triglyceride (TRIG) Levels (mg/dL) stratified by Gender. Graphical illustration of serum triglyceride levels values at baseline and over the six hours postprandially for A) females and B) males and consecutive differences (serum triglyceride values at time (t) - values at timepoint immediately preceding) for C) females and D) males.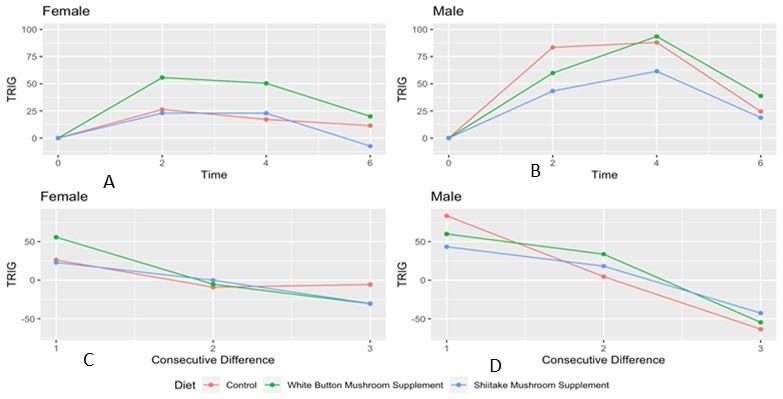
Observed changes in biochemical parameters after experimental meals
The overall effects of LBM, gender, burger type, time, and burger type x time interaction are illustrated (Table 3). Serum LDL levels differ significantly (p=0.0007) between the control burger and the two mushroom-supplemented burgers. Graphical analysis of the consecutive differences reveals that in females, serum LDL levels decreased at the last time point (t = 6 hours) in those who received the SH burger. Serum LDL levels decreased at t=6 hours for the control burger for females. Among males, graphical analysis of consecutive differences demonstrated that all three burgers elevated serum LDL levels with the mushroom-supplemented burgers suppressing a greater LDL rise compared to the control burger. Time, gender, and the interaction term (burger type x time) did not significantly affect serum LDL levels (Table 3). In summary, the SH mushroom-supplemented burger resulted in lower LDL levels compared with the control burger after 6 hours in both male and female participants.
Table 3. Type III analysis of the linear mixed effects model with listed covariates| Outcomes | Effect | F-value | P-value |
|---|---|---|---|
| LDL | Lean body mass | 0.14 | 0.78096 |
| Sex | 0.01 | 0.9358 | |
| Diet | 7.94 | 0.0007 | |
| Time | 1.12 | 0.3443 | |
| Diet*Time | 0.93 | 0.4761 | |
| HDL | Lean body mass | 0.07 | 0.7876 |
| Sex | 6.43 | 0.0130 | |
| Diet | 0.38 | 0.6846 | |
| Time | 0.75 | 0.5241 | |
| Diet*Time | 0.17 | 0.9840 | |
| Triglyceride | Lean body mass | 0.28 | 0.6013 |
| Sex | 4.64 | 0.0340 | |
| Diet | 1.28 | 0.2841 | |
| Time | 18.67 | <0.0001 | |
| Diet*Time | 0.54 | 0.7777 | |
| Glucose | Lean body mass | 2.77 | 0.0997 |
| Sex | 0.64 | 0.4261 | |
| Diet | 1.10 | 0.3374 | |
| Time | 14.85 | <0.0001 | |
| Diet*Time | 0.75 | 0.6101 |
Serum HDL levels differed by gender (p=0.0005), while burger type, time, and the interaction term (burger type x time) did not have any significant effect (Table 3). Among females, WB burgers led to an increase in HDL levels while SH burgers did not change HDL levels. Among males, SH burgers slightly increased HDL levels while WB burgers decreased HDL levels at the last time point (t = 6 hours) compared to the immediately preceding time point. Serum HDL levels were unchanged when participants consumed the control burger.
Serum triglyceride values vary significantly by gender (p = 0.0083) and time (p<0.0001) (Table 3). Evaluating the graphs, adjusted for baseline values, in females, the peak in triglyceride levels occurred between t = 2 hours and t = 4 hours postprandially. The highest serum triglyceride peak occurred when participants consumed the WB burger as compared to the consumption of the control or SH burgers. For males, the serum triglyceride peak occurred at t=4 hours, and was highest in those who consumed WB burgers. For females, evaluating the consecutive differences revealed that serum triglyceride levels decrease with the consumption of all three burgers. The most marked differences occurred between the first (t = 0 hours) and second (t = 2 hours) time points and remain relatively stable comparing timepoints 2 (t = 2 hours) and 3 (t = 4 hours). In males, the evaluation of consecutive differences revealed that the serum triglyceride levels decrease over the six-hour period in all three burger types.
Serum glucose levels varied over time (p < 0.0001) (). Graphical analysis revealed that postprandial changes were relatively consistent when comparing females and males. By the third time point (t = 4 hours postprandial), serum glucose levels had returned to baseline. A burger type by gender interaction term included in the modeling was not significant (data not shown).
Discussion
In this pilot investigation, we evaluated the postprandial effects of WB or SH mushroom supplementation into a high-fat meal regarding serum lipids (LDL, HDL, and triglycerides) and serum glucose amongst nine healthy participants. LDL levels differed significantly for both SH and WB burgers compared to the control burger over six hours. Subsequent statistical analysis demonstrated that SH-supplemented burgers significantly lowered serum LDL levels in both genders. Burger type did not affect triglyceride, HDL, and glucose levels. Triglyceride levels were higher in males and HDL levels were higher in females. Triglyceride and glucose levels were significantly higher during the postprandial timepoints 2 (t = 2 hours) and 3 (t = 4 hours). By timepoint 4 (t = 6 hours), triglyceride and glucose levels had returned to baseline (t = 0 hours) values.
Primarily in industrialized countries, metabolic syndrome is an increasing health condition. The ineffectiveness of and/or the variable adherence of individuals to current therapeutic approaches has promoted searching for novel strategies to address obesity-related conditions. High-fat food supplementation with mushroom powder has been proposed as a potential therapeutic intervention to improve cardiovascular and overall health status 6. In our study, we found that SH mushroom-powder supplementation of a high-fat meal significantly lowered LDL levels in comparison with WB mushroom-powder supplementation or no supplementation (control meal). Mushroom-supplementation of burgers did not significantly affect triglyceride, HDL, and glucose levels in both genders.
Previous literature has documented no significant differences between glucose or triglyceride levels among participants who consumed a control meal and a meal fortified with mushrooms. For example, Dicks et al. did not observe any differences in glucose or triglyceride levels among 22 participants with impaired glucose tolerance when comparing a meal supplemented with 20g of Oyster mushroom powder rich in β-glucans to a control meal (i.e., a meal without mushroom supplementation) 20. β-glucans have been shown to alter lipid and glucose metabolism as well as to reduce cholesterol, which has led to their investigation as potential therapies for metabolic syndrome and obesity 21. Metabolic syndrome is a constellation of conditions associated with an increased risk of developing diabetes, obesity, and CVD that are caused by insulin resistance, including central obesity, hypertension, hyperglycemia, high serum triglyceride levels, and HDL levels 22, 23. The cholesterol-lowering effects of SH mushrooms are attributed to the mycochemical, eritadenine 24. Studies in rats have shown lower serum concentrations of triglycerides, non-HDL cholesterol, and total cholesterol in rats fed SH 16.
Gender is an important modifying factor regarding postprandial lipemia. For example, in response to fat obtained from either butter, coconut oil, olive oil, or canola oil, men exhibited a greater peak triglyceride response than women 25. Among men, those with a greater BMI (>23 kg/m2) compared to men with a lower BMI (<23 kg/m2) exhibit a greater increase in postprandial lipemia in response to a high-fat meal 26. Differences in visceral adipose accumulation sites between men and women may account for the amplified postprandial response observed between the genders 27. Men tend to store fat primarily in the android regions (trunk/abdomen) and to experience visceral tissue fat accumulation. In a rodent study evaluating the lipid lowering effect of SH, female rats had lower triglyceride and higher HDL levels compared with male rats 16. This study also illustrated that, at least in rats, SH’s ability to lower plasma lipids was equivalent between the genders, an effect that had previously been thought to be limited to male rats 28.
Our results are consistent with several prior publications that have demonstrated that HDL levels are elevated in women while triglyceride levels are higher in men. Prior studies have shown that men have almost two-fold greater quantities of visceral adipose tissue compared to premenopausal women (as would be relevant to our participants who were all premenopausal) 29, and the accumulation of abdominal fat is associated strongly with alterations in lipoprotein concentrations. Von Hafe reported that in a study containing 77 men and 84 women, men had higher levels of visceral fat, higher plasma insulin, higher glucose, higher triglycerides, and lower HDL than women 30. Another study by Godsland et al. in 1987 reported that men are characterized by high fasting triglyceride and low HDL-cholesterol levels compared with women 31. Couillard et al. investigated the postprandial triglyceride response to a standardized meal and found that men had a significantly greater plasma triglyceride response compared with women 27. These investigators also found that men had decreased HDL-cholesterol compared with women. Kolovou et al. replicated the same findings in a much larger cohort of participants (n=1385). Among these individuals (40% female), high triglyceride and low HDL-cholesterol were two times more common in men than women 32.
Edible mushrooms have been shown to contain active compounds that exhibit antioxidant 33, anti-atherosclerotic 34, and hypoglycemic 35 activities. An important consideration is specifically how WB or SH mushrooms mediate their lipid-lowering effects. WB mushrooms have been shown to lower serum total cholesterol by upregulating hepatic LDL receptor mRNA expression 36. SH mushrooms have also been shown to reduce plasma cholesterol 37. Eritadenine, a compound isolated from SH mushrooms, is hypothesized to be the major mechanism by which total cholesterol and non-HDL cholesterol are decreased 16. Another potential lipid lowering mechanism is through decreased absorption of lipids from the gastrointestinal tract, such as through β-glucans 7. Further research is needed to clarify the exact pathways for lipid lowering mechanisms for both SH and WB mushrooms. Mushrooms have also been proposed to mediate their anti-obesogenic and antidiabetic effects through a variety of biochemical and anti-inflammatory pathways 15, 38, 39.
In this pilot study, we sought to investigate the antilipemic effects of a high-fat meal supplemented with WB or SH mushroom powder compared to a control hamburger. The strength of this investigation is the longitudinal sampling and follow-up that enabled us to observe these effects on humans. The limitations of this work are the relatively small numbers of individuals enrolled and the only one-time meal administration (as opposed to assessing repeated measures of the same meal). However, Sciarrillo et al. recently published a study that included ten participants that evaluated the postprandial lipemic responses in triglycerides to four dietary fat sources 25. Our pilot study provides valuable information for how to design, execute, and scale similar dietary interventions using mushroom powder as a functional food in a conventional, Western high-fat meal.
Conclusions
We observed that LDL levels were significantly decreased in individuals who consumed SH burgers compared to WB or control burgers. Specifically, males had significantly higher serum triglyceride and lower serum HDL levels in comparison with female participants. While glucose levels were significantly affected by ingestion of any burger as expected, the levels did not vary significantly when comparing the three burger types, and they followed a standard postprandial glucose curve. Further investigation is required to assess whether SH and WB mushroom supplementation is effective in decreasing serum lipid concentrations in humans and whether these differences vary by gender. Further research is also needed to assess whether the postprandial effects on glucose and on lipids observed with SH and WB powder supplementation are generalizable to other types of mushrooms. Additional investigation is also needed to assess whether the effects of SH and WB mushroom supplementation persist in individuals who have metabolic conditions, such as diabetes or hypercholesterolemia. These studies should also attempt to follow participants for a longer postprandial period (e.g., 12 or 24 hours).
Acknowledgments
Horvath Laboratory Group: Harry Marsales, Theodore J. Robak, Courtney Miller, Alyssa Massaro, Taylor Furst, Zach Lamacchia; Dr. Todd C. Rideout, Associate Professor, UB Department of Exercise and Nutrition Science; Oakshire Farms, who generously donated the mushroom powders; Dedicated to Matthew J. Morris, whose work was crucial to project.
Funding
This research was funded by State University of New York (SUNY), University at Buffalo (UB) Center for Undergraduate Research and Creative Activities (CURCA) Undergraduate Research Award. Oakshire Farms generously donated the shiitake and white button mushroom powders for this research.
References
- 1.Hyson D, Rutledge J C, Berglund L. (2003) Postprandial lipemia and cardiovascular disease Curr Atheroscler Rep. 5(6), 437-44.
- 2.Baumgartner S, Mensink R P, Plat J. (2011) Plant sterols and stanols in the treatment of dyslipidemia: new insights into targets and mechanisms related to cardiovascular risk. 17(9), 922-32.
- 3.Boden W E, Sidhu M S, Toth P P. (2014) The therapeutic role of niacin in dyslipidemia management. , J Cardiovasc Pharmacol Ther 19(2), 141-58.
- 4.Weitz D, Weintraub H, Fisher E, Schwartzbard A Z. (2010) Fish oil for the treatment of cardiovascular disease. , Cardiol 18(5), 258-63.
- 5.Gaitan-Hernandez R, Lopez-Pena D, Esqueda M, Gutierrez A. (2019) Review of Bioactive Molecules Production, Biomass, and Basidiomata of Shiitake Culinary-Medicinal Mushrooms, Lentinus edodes (Agaricomycetes). , Int J Med 21-9.
- 6.Islam T, Ganesan K, Xu B. (2019) New Insight into Mycochemical Profiles and Antioxidant Potential of Edible and Medicinal Mushrooms: A Review Int. , J Med 21(3), 237-251.
- 7.Kolovou G D, Mikhailidis D P, Nordestgaard B G, Bilianou H, Panotopoulos G. (2011) Definition of postprandial lipaemia. , Curr Vasc Pharmacol 9(3), 292-301.
- 9.Shang A, Gan R Y, Xu X Y, Mao Q Q, Zhang P Z. (2021) Effects and mechanisms of edible and medicinal plants on obesity: an updated review. Crit Rev Food Sci Nutr.61(12):. 2061-2077.
- 10. (2011) European Food Safety Authority. Scientific opinion on the substantiation of health claims related to beta-glucans from oats and barley and maintenance of normal blood LDL-cholesterol concentrations (ID 1236, 1299), increase in satiety leading to a reduction in energy intake (ID 851, 852), reduction of postprandial glycaemic responses (ID 821, 824), and “digestive function” (ID 850) pursuant to Article 13(1) of Regulation (EC) No 1924/2006. , 2011.2207.EFSA J 9, 2207.
- 11.Tung Y T, Pan C H, Chien Y W, Huang H Y. (2020) Edible Mushrooms: Novel Medicinal Agents to Combat Metabolic Syndrome and Associated Diseases.Pharm Des.26(39):-4970-4981.
- 12.Pinero M P, Parra K, Huerta-Leidenz N, L Arenas de Moreno, Ferrer M. (2008) Effect of oat's soluble fibre (beta-glucan) as a fat replacer on physical, chemical, microbiological and sensory properties of low-fat beef pattiesMeat Sci.80(3):. 675-8.
- 13.Wong W W, Smith E O, Stuff J E, Hachey D L, Heird W C. (1998) Cholesterol-lowering effect of soy protein in normocholesterolemic and hypercholesterolemic men. , Am J Clin Nutr 68(6), 1385-1389.
- 14.Rebholz C M, Reynolds K, Wofford M R, Chen J, Kelly T N. (2013) Effect of soybean protein on novel cardiovascular disease risk factors: a randomized controlled trial. , Eur J Clin 67(1), 58-63.
- 16.Yu S, Wu X, Ferguson M, Simmen R C, Cleves M A. (2016) Diets Containing Shiitake Mushroom Reduce Serum Lipids and Serum Lipophilic Antioxidant Capacity in Rats. , J 146(12), 2491-2496.
- 17.Ochiai M. (2020) Evaluating the appropriate oral lipid tolerance test model for investigating plasma triglyceride elevation in mice PLoS One.15(10),e0235875.
- 18.Bodell N G. University of Nevada Las Vegas (2019) Triglyceride metabolism following oral fat tolerance tests of varying fat Content in adults.
- 19. (2020) United States Department of Agriculture-Food Safety and Inspection Service. Safe Minimum Internal Temperature Chart.
- 20.Dicks L, Jakobs L, Sari M, Hambitzer R, Ludwig N. (2022) Fortifying a meal with oyster mushroom powder beneficially affects postprandial glucagon-like peptide-1, non-esterified free fatty acids and hunger sensation in adults with impaired glucose tolerance: a double-blindrandomized controlled crossover trial. , Eur J Nutr 61(2), 687-701.
- 21.Murphy E J, Rezoagli E, Major I, Rowan N J, Laffey J G. (2020) beta-Glucan Metabolic and Immunomodulatory Properties and Potential for Clinical Application.J Fungi (Basel). 6(4), 356.
- 23.Moller D E, Kaufman K D. (2005) Metabolic syndrome: a clinical and molecular perspective. , Annu Rev Med 56, 45-62.
- 24.Fukushima M, Ohashi T, Fujiwara Y, Sonoyama K, Nakano M. (2001) Cholesterol-lowering effects of maitake (Grifola frondosa) fiber, shiitake (Lentinus edodes) fiber, and enokitake (Flammulina velutipes) fiber in rats. , Exp Biol Med 226(8), 758-65.
- 25.Sciarrillo C M, Koemel N A, Tomko P M, Bode K B, Emerson S R. (2019) Postprandial Lipemic Responses to Various Sources of Saturated and Monounsaturated Fat in Adults. 11(5), 1089.
- 26.Kasai M, Maki H, Nosaka N, Aoyama T, Ooyama K. (2003) Effect of medium-chain triglycerides on the postprandial triglyceride concentration in healthy men. , Biosci Biotechnol 67(1), 46-53.
- 28.Shimada Y, Morita T, Sugiyama K. (2003) Eritadenine-induced alterations of plasma lipoprotein lipid concentrations and phosphatidylcholine molecular species profile in rats fed cholesterol-free and cholesterol-enriched diets. , Biosci Biotechnol Biochem 67(5), 996-1006.
- 29.Tchernof A, Despres J P. (2013) Pathophysiology of human visceral obesity: an update. , Physiol Rev 93(1), 359-404.
- 30.P von Hafe. (2019) Gender differences in lipid profile and therapy.2019.09.003. Rev Port Cardiol (Engl Ed).38(8):-571-572.
- 31.Godsland I F, Wynn V, Crook D, Miller N E. (1987) Sex, plasma lipoproteins, and atherosclerosis: prevailing assumptions and outstanding questions. , Am Heart 114(6), 1467-503.
- 32.Kolovou G D, Anagnostopoulou K K, Damaskos D S, Bilianou H I, Mihas C. (2009) Gender differences in the lipid profile of dyslipidemic subjects. , Eur J Intern Med 20(2), 145-51.
- 33.Mau J L, Lin H C, Chen C C. (2002) Antioxidant properties of several medicinal mushrooms. , J Agric Food Chem 50(21), 6072-7.
- 34.Yamada T, Oinuma T, Niihashi M, Mitsumata M, Fujioka T. (2002) Effects of Lentinus edodes mycelia on dietary-induced atherosclerotic involvement in rabbit aorta. , J Atheroscler 9(3), 149-56.
- 35.Wasser S P, Weis A L. (1999) Therapeutic effects of substances occurring in higher Basidiomycetes mushrooms: a modern perspective. Crit Rev Immunol.19(1):. 65-96.
- 36.Fukushima M, Nakano M, Morii Y, Ohashi T, Fujiwara Y. (2000) Hepatic LDL receptor mRNA in rats is increased by dietary mushroom (Agaricus bisporus) fiber and sugar beet fiber. , J 130(9), 2151-6.
- 37.Kaneda T, Tokuda S. (1966) Effect of various mushroom preparations on cholesterol levels in rats. , J Nut 90(4), 371-6.
