RETRACTED: Profile of Children with Covid 19 Infection in our Set Up
Abstract
This article has been retracted on July 20, 2022. VIEW THE RETRACTION NOTICE (https://doi.org/10.14302/issn.2692-1537.ijcv-25-5866)
In December 2019, a novel coronavirus, now named as SARS-CoV-2, caused a series of acute atypical respiratory diseases in Wuhan, Hubei Province, China. The disease caused by this virus was termed COVID-19. The virus is transmittable between humans and has caused pandemic worldwide. The number of death tolls continues to rise and a large number of countries have been forced to do social distancing and lockdown. In humans, COVIDs mostly cause respiratory and gastrointestinal symptoms. Clinical manifestations range from a common cold to more severe disease such as bronchitis, pneumonia, severe acute respiratory distress syndrome, multi-organ failure and even death. Preliminary evidence suggests children are just as likely as adults to become infected with SARS-CoV-2 but are less likely to be symptomatic or develop severe symptoms. in our study , we consider the symptomatology , complications and mortality patterns of this disease in children.
Author Contributions
Academic Editor: Raul Isea, Fundación Instituto de Estudios Avanzados - IDEA, Hoyo de la Puerta, Baruta.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2021 Baljinder kaur, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Coronavirus is one of the major pathogens that primarily targets the human respiratory system. Previous outbreaks of coronaviruses (CoVs) include the severe acute respiratory syndrome (SARS-CoV) and the Middle East respiratory syndrome (MERS-CoV) which have been previously characterized as agents that are a great public health threat. In late December 2019, a cluster of patients was admitted to hospitals with an initial diagnosis of pneumonia of an unknown etiology. These patients were epidemiologically linked to a seafood and wet animal wholesale market in Wuhan, Hubei Province, China. 1,2
Early reports predicted the onset of a potential Coronavirus outbreak given the estimate of a reproduction number for the 2019 Novel Coronavirus (COVID-19, as named by WHO on Feb 11, 2020) which was deemed to be significantly larger than 1 (ranges from 2.24 to 3.58) 3
Coronaviruses are enveloped, positive-sense, single stranded RNA viruses of ~30kb. They infect a wide variety of host species.4
They are largely divided into four genera: a, p, y, and 5 based on their genomic structure. a and p coronaviruses infect only mammals.5 Human corona viruses such as 229E and NL63 are responsible for common cold and croup and belong to a coronavirus. In contrast, SARSCoV, MERS-CoV and SARS-CoV-2 are classified to p coronaviruses Figure 1
Figure 1.Human Coronavirus – Structure (Source ncbi.nih.gov bhoopti etall)
Pathophysiology
Coronaviruses consist of four structural proteins; Spike (S), membrane (M), envelop (E) and nucleocapsid (N). Spike is composed of a trans-membrane trimetric glycoprotein protruding from the viral surface, which determines the diversity of coronaviruses and host tropism. Spike comprises two functional subunits: S1 and S2 S1 subunit is responsible for binding to the host cell receptor and S2 subunit is for the fusion of the viral and cellular membranes.6
Angiotensin converting enzyme 2 (ACE2) was identified as a functional receptor for SARS-CoV . Structural and functional analysis showed that the spike for SARS-CoV-2 also bound to ACE2. ACE2 expression was high in lung, heart, ileum, kidney and bladder.7,8,9,10,11
In lung, ACE2 was highly expressed on lung epithelial cells. Whether or not SARS-CoV-2 binds to an additional target needs further investigation. Following the binding of SARS-CoV-2 to the host protein, the spike protein undergoes protease cleavage.12,13,14 After the cleavage at the S1/S2 cleavage site, S1 and S2 subunits remain non-covalently bound and the distal S1 subunit contributes to the stabilization of the membraneanchored S2 subunit at the prefusion state.9
The coronavirus spike is unusual among viruses because a range of different proteases can cleave and activate it .15 The characteristics unique to SARS-CoV-2 among corona viruses is the existence of furin cleavage site (“RPPA” sequence) at the S1/S2 site. 9 Although the S1/S2 site was also subjected to cleavage by other proteases such as transmembrane protease serine 2 (TMPRSS2) and cathepsin L. 14,16 , The ubiquitous expression of furin likely makes this virus very pathogenic.
In later stages of infection, when viral replication accelerates, epithelial-endothelial barrier integrity is compromised. In addition to epithelial cells, SARS-CoV-2 infects pulmonary capillary endothelial cells, accentuating the inflammatory response and triggering an influx of monocytes and neutrophils. Autopsy studies have shown diffuse thickening of the alveolar wall with mononuclear cells and macrophages infiltrating airspaces in addition to endothelialitis. Interstitial mononuclear inflammatory infiltrates and edema develop and appear as ground-glass opacities on computed tomographic imaging. Pulmonary edema filling the alveolar spaces with hyaline membrane formation follows, compatible with early-phase acute respiratory distress syndrome (ARDS).17 Bradykinin-dependent lung angioedema may contribute to disease.18
In severe COVID-19, fulminant activation of coagulation and consumption of clotting factors occur.19,20 A report from Wuhan, China, indicated that 71% of 183 individuals who died of COVID-19 met criteria for diffuse intravascular coagulation.19Inflamed lung tissues and pulmonary endothelial cells may result in microthrombi formation and contribute to the high incidence of thrombotic complications, such as deep venous thrombosis, pulmonary embolism, and thrombotic arterial complications (eg, limb ischemia, ischemic stroke, myocardial infarction) in critically ill patients.21 The development of viral sepsis, defined as life-threatening organ dysfunction caused by a dysregulated host response to infection, may further contribute to multiorgan failure.
The role of the immune response in viral clearance and pathogenesis in the CNS has been well characterized. Both antibody and cell-mediated immune responses are required to protect against coronavirus infections. The CD8+ T and CD4+ T cells are primarily responsible for clearance of the virus during acute infection. Perforin-mediated mechanisms are necessary for clearance of virus from astrocytes and microglia, while gamma interferon (IFN-γ) has been implicated in clearance from oligodendrocytes.22
The life cycle of the virus with the host consists of the following 5 steps: Attachment, Penetration, Biosynthesis, Maturation and Release. Once viruses bind to host receptors (Attachment), they enter host cells through endocytosis or membrane fusion (Penetration). Once viral contents are released inside the host cells, viral RNA enters the nucleus for replication. Viral mRNA is used to make viral proteins (Biosynthesis). Then, new viral particles are made (Maturation) and Released.
Classification
| Asymptomatic | COVID nucleic acid test positive. Without any clinical symptoms and signs and the chest imaging is normal |
| Mild | Symptoms of acute upper respiratory tract infection (fever, fatigue, myalgia, cough, sore throat, runny nose, sneezing) or digestive symptoms (nausea, vomiting, abdominal pain, diarrhea) |
| Moderate | Pneumonia (frequent fever, cough) with no obvious hypoxemia, chest CT with lesions. |
| Severe | Pneumonia with hypoxemia (SpO2 < 92%) |
| Critical | Acute respiratory distress syndrome (ARDS), may have shock, encephalopathy, myocardial injury, heart failure, coagulation dysfunction and acute kidney injury |
Aims and Objectives
To study clinical profile, morbidity, treatment and mortality pattern in children with COVID19 infection.
Type of Study
Observational Study
Study Design
Inclusion Criteria
All children of age between 0 to 18 years of either gender with RT-PCR positive for COVID19 infection
Exclusion Criteria
Children with RT-PCR negative results for COVID19 infection
Materials and Methods
26 children admitted to covid-19 isolation and management unit of government medical college and Rajindra Hospital Patiala. For the purpose of study, children were divided into age group of less than 1 month, 1 month to 1 year, >1 year to 18 years respectively. Name, age, gender, address, clinical examination, treatment profile and morbidity and mortality pattern is recorded on a pre-designed and pre-tested proforma. Data so obtained was subjected to analysis for the purpose of study.
Results & Observations
Table 1 showing distribution of children according to their age. Only 7.6% children were under 1 year of age who were COVID19 RT-PCR positive, with no case under 1 month of age while 92.3% were more than 1 year of age. Mean age of COVID19 RTPCR positive children who presented to our setup was 8.9 years
Table 1. Distribution According to the Age and Gender| <1 month of age | nil |
| 1 month to 1 year | 2 |
| > 1 year to 18 years | 24 |
Table 2 showing distribution of children according to gender distribution. Number of male patients outnumbers female patients (61.5% vs 38.4%). Figure 2
Table 2. Distribution of children according to gender distribution. Number of male patients outnumbers female patients (61.5% vs 38.4%)| Males | 16 |
| Females | 10 |
Figure 2.Distribution of Covid -19 RT-PCR positive children according to age and gender distribution.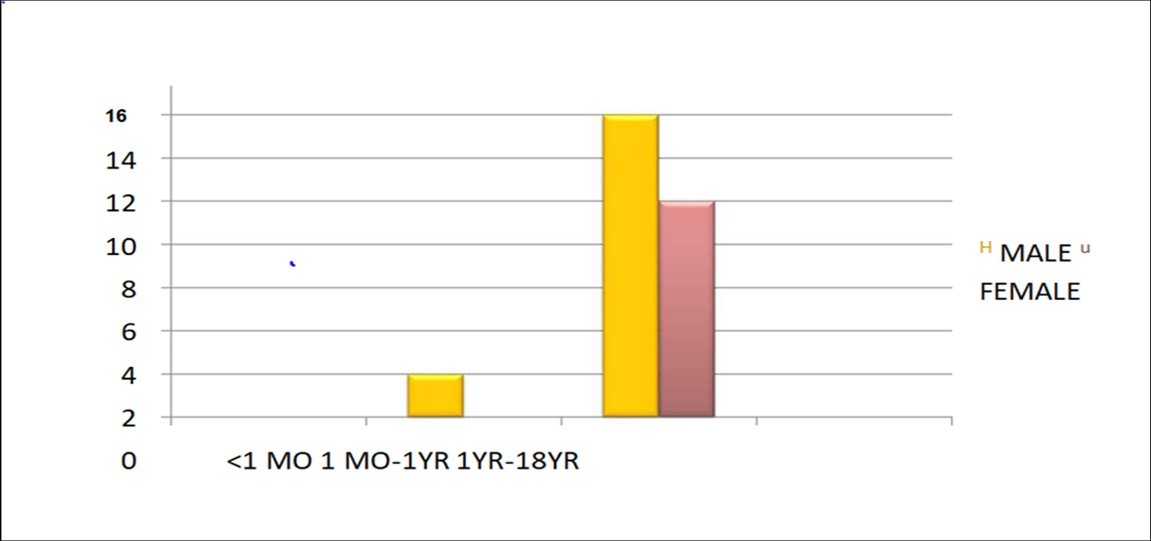
Distribution According to Presenting Complaint
-Fever - 3 -Sore Throat - 4 -Cough - 4
-Respiratory Difficulty - 2 -Diarrhoea - Nil -Rash - 1 -Jaundice - 1 -Seizure -1
Figure 3 showing symptoms with which the child presents to our setup. Most of the children were asymptomatic contacts of COVID19 RT-PCR positive patients and remained asymptomatic throughout the stay while 4 out of 26 patients were having sore throat with cough. 3 children were having fever out of which 2 children presented with respiratory difficulty. 1 child presented with fever, rash, jaundice and respiratory difficulty and seizures.
Figure 3.Symptoms with which the child presents to our setup
Distribution of children According to Oxygen Therapy Provided
No oxygen therapy required - 24 Non invasive ventilation(NIV) - 1 Invasive ventilatory support(IV) - 1
Figure 4 represents number of children requiring oxygen therapy. 1 child was given continuous positive pressure ventilation, which was later tapered and shifted to nasal prongs and then weaned off from oxygen support. Another child was intubated and mechanically ventilated (Child couldnot survive and got expired within 24 hrs of mechanical ventilation). Majority of children (92.3%) did not require any oxygen support.
Figure 4.Represents number of children requiring oxygen therapy.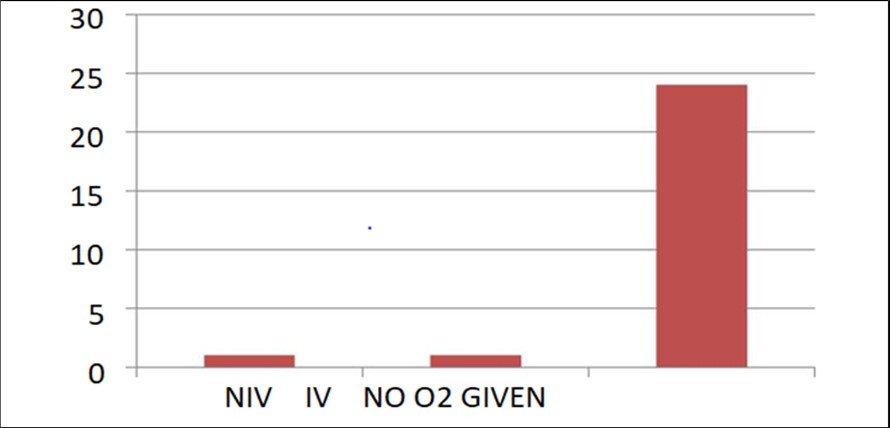
Table 3 shows number of children requiring antibiotic therapy. Antibiotics given to these children include azithromycin. Second line antibiotics added were meropenem and vancomycin in 2 of the cases.
Table 3. Number of children given antibiotic therapy| <1 MONTH OF AGE | nil |
| 1 MONTH-1YEAR | 1 |
| >1 YEAR-18 YEARS | 3 |
Table 4 shows none of the pediatric COVID19 positive patient were given antivirals as most of our patients were asymptomatic and also there is paucity of data on the safety profile of antivirals in pediatric population.
Table 4. Number of children requiring antiviral therapy.| <1 month of age | nil |
| 1 month to 1 year | nil |
| > 1 year to 18 years | nil |
Table 5 shows that mortality rate in pediatric population in our setup so far is 3.8% with 1 motality out of the 26 COVID19 RT-PCR positive pediatric patients. Distribution according to age shows 0% mortality in children less than 1 year of age in our setup so far.
Table 5. Mortality pattern| <1 month of age | nil |
| 1 month to 1 year | nil |
| >1 year to 18 years | 1 |
In our study, the confirmation of COVID19 infection was done by RT-PCR.
Maximum number of children with COVID19 infection were in the age group of >1 to 18 years (92.3%) followed by age group of 1 month to 1 year of age(7.6%) and no child was found to be COVID19 positive in the age less than 1 month(0%) with mean age of presentation is 8.9 years(n=8.9). Number of male cases outnumbers the number of female cases(61.5 vs 38.4% %). 15.3% children of COVID19 infection presented to us with cough and sore throat as their presenting complaint, 11.5% children presented with fever, 7.6% children presented with respiratory difficulty.
None of the children presented with pain abdomen or diarrhoea. 1 out of 26 children was having fever along with rash, jaundice, seizures and respiratory difficulty. 7.6% children presented with respiratory difficulty requiring supportive oxygen therapy. 15.6% children were given antibiotic therapy with none of them were given antivirals or hydroxychloroquine. Regarding mortality rate and pattern, 1 out of 26 COVID19 RTPCR positive child died due to corona virus disease and its complications (mortality rate 3.8%) and none of the children who died were under 1 year of age. Parental understanding regarding the route of transmission and the ways to prevent transmission of infection like wearing of masks, regular handwashing, restraining onself from going to public places unless unavoidable circumstances, maintaining social distance of minimum 2 meters has worthwhile effect in preventing infection and thus disease spread to children at home and in contact.
Discussion
In our study, a total of 26 patients presenting to the RHP with COVID19 RT-PCR positive. Most of the children were undiagnosed cases and were diagnosed upon admission to RHP. Most of the children were asymptomatic contacts of COVID19 RT-PCR positive patients depicting a high secondary attack rate of the corona virus. Main presenting complaint in symptomatic patients in our set up was cough with sore throat followed by fever. Some were having respiratory difficulty while none of them presented with any GIT complaints.
When interviewed, they knew route of transmission. These people were aware of the high risk behaviour which should be avoided but on further questioning it is found that most of the people were not strictly following the guidelines regarding self protection, wearing of masks, regular hand washing, use of alcohol based hand sanitizers, self hygiene and social distancing at public places.
Regarding management of pediatric patients, asymptomatic patients were kept under observation with children were allowed orally. No parenteral therapy including iv fluids or antibiotic therapy were given. For symptomatic children, treatment were given according to the presenting complaint. Antipyretics were given for fever, antitussives were given for severe cough with advice of saline gargles with mild-moderate cough. Oxygen therapy was provided to children presented with respiratory difficulty along with antibiotics. Non invasive ventilation was provided in the form of oxygen by nasal prongs, face mask, non rebreathing mask, CPAP, BiPAP or invasive ventilation in the form of mechanical ventilation. Suctioning of the secretions and nebulisation were avoided during treatment of COVID19 positive patients as it leads to generation of aerosols and greater spread of virus.
Antibiotic were given in severe cases. Azithromycin(@ 10 mg/kg/dose OD) is preferred antibiotic. Other antibiotics being used in our set up so far are meropenem(20 mg/kg/dose TDS), vancomycin(@10 mg/kg/dose QID). Regarding other treatment options like hydroxychloroquine and antivirals, they are not being given due to paucity of safety profile in pediatric population.
Conclusions
As per this study most children fell into asymtomatic category followed by mild disease. PPE,& social distancing by adults& children will help preventing spread of virus
References
- 1.I, Watts A, Thomas-Bachli A, Huber C, Kraemer M et al. (2020) Pneumonia of unknown aetiology in Wuhan, China: potential for international spread via commercial air travel. , Journal of travel medicine 27(2), 008.
- 2.Lu H, Stratton C W, Tang Y W. (2020) Outbreak of pneumonia of unknown etiology in Wuhan, China: The mystery and the miracle. Journal of medical virology. 92(4), 401-2.
- 3.Zhao S, Lin Q, Ran J, Musa S S, Yang G et al. (2020) Preliminary estimation of the basic reproduction number of novel coronavirus (2019-nCoV) in China, from2019to2020: A data-driven analysis in the early phase of the outbreak. International journal of infectious diseases.92:. 214-7.
- 4.Channappanavar R, Zhao J, Perlman S. (2014) Cell-mediated immune response to respiratory coronaviruses. , Immunologic 59(1), 118-28.
- 5.Rabi F A, Al Zoubi MS, Kasasbeh G A, Salameh D M, Al-Nasser A D. (2020) SARS-CoV-2 and coronavirus disease 2019: what we know so far. Pathogens. 9(3), 231.
- 6.Bosch B J, Zee R Van der, De Haan CA, Rottier P J. (2003) The coronavirus spike protein is a class I virus fusion protein: structural and functional characterization of the fusion core complex. Journal of virology. 77(16), 8801-11.
- 7.Li W, Moore M J, Vasilieva N, Sui J, Wong S K et al. (2003) Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 426(6965), 450-4.
- 8.Chen Y, Guo Y, Pan Y, Zhao Z J. (2020) Structure analysis of the receptor binding of 2019-nCoV. Biochemical and biophysical research communications.
- 9.Walls A C, Park Y J, Tortorici M A, Wall A, McGuire A T et al. (2020) Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein.
- 10.Letko M, Marzi A, Munster V. (2020) Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses. Nature microbiology. 5(4), 562-9.
- 11.Zou X, Chen K, Zou J, Han P, Hao J et al. (2020) Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Frontiers of medicine. 12, 1-8.
- 12.Belouzard S, Chu V C, Whittaker G R. (2009) Activation of the SARS coronavirus spike protein via sequential proteolytic cleavage at two distinct sites. Proceedings of the National Academy of Sciences 106(14), 5871-6.
- 13.Millet J K, Whittaker G R. (2014) Host cell entry of Middle East respiratory syndrome coronavirus after two-step, furin-mediated activation of the spike protein. Proceedings of the National Academy of Sciences 111(42), 15214-9.
- 14.Ou X, Liu Y, Lei X, Li P, Mi D et al. (2020) Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nature communications. 11(1), 1-2.
- 15.Belouzard S, Millet J K, Licitra B N, Whittaker G R. (2012) Mechanisms of coronavirus cell entry mediated by the viral spike protein. Viruses. 4(6), 1011-33.
- 16.Hoffmann M, Kleine-Weber H, Schroeder S, Kruger N, Herrler T et al. (2020) SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell.
- 17.Xu Z, Shi L, Wang Y. (2020) Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 8(4), 420-422.
- 18.FL Van de Veerdonk, Netea M G, M Van Deuren.et al.(2020). Kallikrein-kinin blockade in patients with COVID-19 to prevent acute respiratory distress syndrome. Elife.
- 19.Tang N, Li D, Wang X, Sun Z. (2020) Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. , J Thromb Haemost 18(4), 844-847.
- 20.Thachil J, Tang N, Gando S. (2020) ISTH interim guidance on recognition and management of coagulopathy in COVID19. , J Thromb Haemost 18(5), 1023-1026.
