Abstract
The Anthomedusae species, Corymorpha bigelowi Maas, 1905 (Cnidaria: Hydrozoa, Corymorphidae), was recorded in the northern Red Sea fauna, representing it "first invasion" in Egypt. A fact proven by past studies that this species is endemic in the Indo-Pacific region, which is mainly located in southern coast of Japan and Indian Ocean. The specimens were collected from an area located off Hurghada city, and between latitudes 27° 14.427ˊ and 27° 10.816ˊ N, and longitudes 33° 51.085ˊ and 33° 51.603ˊ E using plankton net (350 µm mesh). In total, six mature medausae of C. bigelowi were caught in December 2014 and February 2015 (4 and 2 medusae, respectively). Following this, the captured species medusa of C. bigelowi was photographed and morphological characteristics described in details. As well as, further discussions of biogeographical distribution and morphological speciation of the present species were provided. This work makes a noteworthy contribution to clarify the migration route of C. bigelowi from southern Red Sea to the Mediterranean Sea.
nih.gov/pmc/articles/PMC3155160/
Author Contributions
Academic Editor: Adela J, The Swire Institute of Marine Science and School of Biological Sciences, the University of Hong Kong, Pokfulam, Hong Kong, China
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Fedekar Fadel Madkour, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
A hydromedusae species, Corymorpha bigelowi Maas, 1905 was first described and identified as Euphysora bigelowi from Indonesian waters during the Siboga Expedition 1. C. bigelowi is one of the 20 medusan species of Euphysorai Maas, 1905 that belongs to family Corymorphidae (Cnidaria, Hydrozoa, Anthomedusae) 2, 3, 4. C. bigelowi is widely distributed in the Indo-Pacific region and a common species in the southeastern coast of Japan and Indian Ocean 5, 6, 7, 8, 9.
Although studies on the gelatinous zooplankton of the Red Sea started at the beginning of 20th century 10, our information is still inadequate and scattered. All records of plankton that collected irregularly from various localities of the Red Sea; Gulf of Suez and Gulf of Aqaba earlier to 1969 were reviewed and a list included one species of chondrophores, 25 siphonophores and 15 schyphomedusans was concluded 11. The first intensive study on Hydromedusae of the Red Sea and adjacent water was conducted in 1973 by Schmidt 12. He examined about 500 plankton samples collected from the entire Red Sea and Gulf of Aden by various expeditions and his own collections near Eilat throughout a long period (1956 – 1969). Of them, only 25 samples were taken from the northern Red Sea. He recorded 72 species from the entire Red Sea and found that there is an eightfold increase in the number of species from north to south. He recorded six specimens of C. bigelowi in the southern region only of the Red Sea.
The most recent studies on gelatinous zooplankton in the Egyptian coast of the Red Sea were carried out during a survey on board R/V Meteor in February 1999 13, 14. These studies recorded 16 Hydromedusae, 11 Siphonophorae, two species of Schyphomedusae and one species of Ctenophora from Gulf of Aqaba and the northern Red Sea. A fact proven by all previous studies that C. bigelowi did not record from the northern Red Sea and the two Gulfs, Aqaba and Suez. The present study corroborates the occurrence of a Hydromedusae species, C. bigelowi, for the first time in the northern Red Sea. Also the study clarifies its migration route from southern Red Sea to the Mediterranean Sea.
Materials and Methods
Specimen Collection
Four stations were selected in the coastal region of the Egyptian Red Sea off Hurghada city (1-4 from north to south), between latitudes 27° 14.427ˊ and 27° 10.816ˊ N, and longitudes 33° 51.085ˊ and 33° 51.603ˊ E as shown in Figure 1. The back shore area of station 1 is occupied by a resort. Station 2 represents the main port for sailing boats. Station 3 is rich with seagrasses and well known for hotels, tourism markets and large number of touristic boats. Station 4 is very close to the Island of Magawish. The specimens were collected monthly from four stations during August 2014-July 2015. Horizontal near surface hauls from 2m depth were conducted in the morning, before sunset, using a plankon-net (mesh size: 350 μm, diameter: 100 cm). Gelatinous organism was separated from the catch and fixed in neutralized 4% formalin for later identification. Temperature and salinity were recorded in situ using thermometer and refractometer, respectively.
Figure 1.Locations of selected stations in the northern Red Sea of Egypt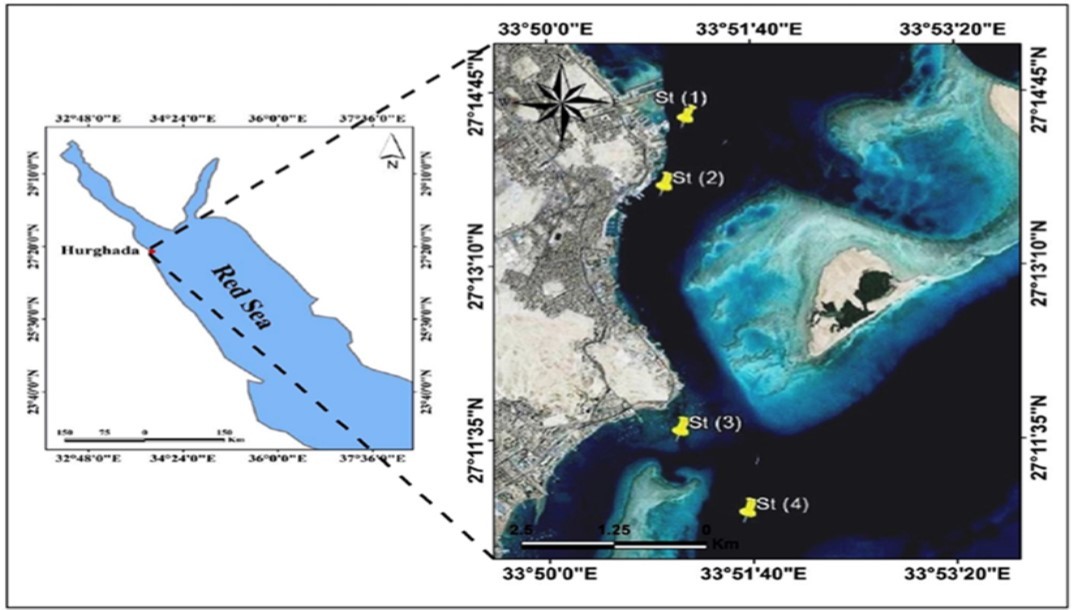
Microscopic Examination
In a preserved state of specimens, the C. bigelowi were examined under stereomicroscope, photographed with Olympus Camera. In order to identify the captured species, the morphological characteristics depicted in detail. The identification was confirmed with regard to main features and measurements as indicated in the literature using different keys 5, 10, 15, 16.
Results and Discussion
Material Examined
Through 48 catches from the northern Red Sea coast off Hurghada at 2m depth during August 2014-July 2015, six mature medusae of C. bigelowi were obtained. Of them four specimens were observed at station 3 in December 2014 and two specimens recorded at station 1 in February 2015.
Systematics
Order ANTHOATHECATA Cornelius, 1992
Suborder CAPITATA Kühn, 1913
Family CORYMORPHIDAE Allman, 1872
Genus Corymorpha Maas, 1905
Corymorpha bigelowi Maas, 1905 (Figure 2a,b)
Since the erection of C. bigelowi by Maas in 1905 as Euphysora and up to the study of Sassaman and Rees 17, this genus had a complex taxonomic history wavering among Euphysora, Euphysa or Corymorpha (as Steenstrupia) 5, 10, 18, 19. The polyp of C. bigelowi was reared in the laboratory and its nomenclature was revised on the basis of life cycle, to be a species of Corymorpha 17. Later on, the living hydroid bearing medusa buds was first collected in the field from coastal region of Akajima Island, Kerama Islands, Okinawa, Japan 20. In samples collected from the sea of Japan at high depths (2545–2555 m and 3340–3347 m), a larva in the form of young polyps (1 mm height) was identified as Euphysora bigelowi 21. All recorded medusae in the present study were adults. However, the immature medusa of this species was recorded in Japan 8, 9.
Morphological Description of Adult Medusae
For examined specimens in the present study, the umbrella measured up to 5.5 mm high and 2.5 mm width. The umbrella was dome shaped with apical pointed projection terminating in a patch of small papillae (Figure 2). There were four spherical radial bulbs on the bell end with four tentacles at the bell margin, one long and three short. On the tentacles: the principle one was as long as the bell and differs from others not only in size, but also in structure. It has as many as 10 (possibly nine) unilateral, adaxial nematocyst bulbs along its length and a large distinct terminal bulb. Although the remaining three tentacles were short, pointed and rudimentary, without nematocyst bulbs, the two tentacles adjacent to the principle tentacle, the lateral tentacles, were approximately twice as long as the one opposite (Figure 2a, b). Manubrium is cylindrical and approximately as long as bell cavity, with simple circular mouth. C. bigelowi feed on copepods as shown in Figure 2b.
Figure 2.C. bigelowi in the Red Sea, Egypt; a) Adult medusa showing manubrium (M), principle long tentacle (PT), nematocyst bulbs (NB), lateral short tentacles (LT) and opposite short tentacle (OT), and b) Mature medusa eating copepod (C).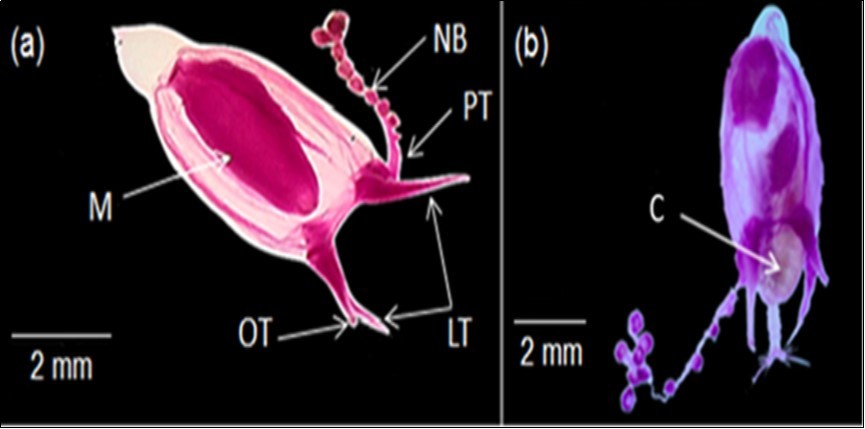
In comparison, the morphological characteristics of C. bigelowi medusae collected from the Egyptian water of the northern Red Sea fitted with the range of variations stated in some studies and varied with others as shown in Table 1. Previous studies proved that there are three quite variable characters in C. bigelowi medusae; i.e the presence or absence of the apical canal, the relative lengths of the three short tentacles, and the relationship between bell height and the number of nematocyst bulbs on the principle tentacle. For the first distinguished character, it would be confused that specimens of C. bigelowi recorded from the Malay Archipelago 1, and from the Sunda Strait 22 included animals with and without apical canals. While other descriptions for specimens from the Indian Ocean and off Japan 19, 23 coincided with the Egyptian material in the complete absence of apical canals. In contrast, all reared California specimens, had well-developed apical canals 17.
Secondly, on the tentacles and the relative length, specimens from Sunda Strait showed that the opposite tentacle is shorter than the other two in small specimens (medusae height: 1.5 mm), but its relative length increases with medusa size and may eventually exceed the lateral tentacles in length (medusae height: 2.25-3 mm) 22. Whereas those noted in small specimens from Indian Ocean off Japan were of equivalent length 23, but that in the larger specimen (4 mm high) the opposite tentacle was much shorter than the lateral tentacles. In the California 17 and Egyptian specimens (present study), the opposite tentacle was substantially shorter than the other two, even in the largest medusa (>5 mm high).
Concerning the third morphological feature, most observations concluded that the number of nematocyst bulbs on the principal tentacle often increase with decreasing medusa height. For example, the highest medusa recorded from the Indian Ocean (4 mm) 23, California (5 mm) 17, and the present study (5.5 mm), had 11 nematocyst bulbs, whereas a 2.25 mm medusa from the Sunda Strait had 21 nematocyst bulbs, and one individual of 1.5 mm high had 31 22. In other study, a 3.5 mm medusa with 26 subterminal bulbs was recorded 19.
In conclusion, a common view was that the three characters do not appear to be correlated. The observations revealed that the Egyptian specimens are more similar to those from the Indian Ocean and California with regard to the lengths of the secondary tentacles and the number of nematocyst bulbs on the primary tentacle, but resemble those from the Indian Ocean off Japan in lacking the apical canal.
Table 1. Comparing morphological characters of C. bigelowi recorded in the present study with those recorded in previous studies.| Reference | Location | Apical canal | Relative lengths of opposite a and lateral b tentacles | Relationship between medusa height and no. of nematocyst bulbs | ||||
| present | absent | a<b | a>b | a=b | Medusa height (mm) | No. of bulbs | ||
| Maas 1905 | Malay Archipelago | + | + | |||||
| Browne 1916 | Indian Ocean off Japan | + | in large medusa | in small medusa | 4 | 11 | ||
| Uchida 1927 | Japan | + | 3.5 | 26 | ||||
| Kramp 1928 | Sundan Strait | + | + | in small medusa | in large medusa | 2.251.5 | 2131 | |
| Sassaman & Rees 1978 | Reared California specimens | + | in large medusa | 5 | 11 | |||
| Present study | Northern Red Sea | + | in large medusa | 5.5 | 11 | |||
Biogeographic Distribution
This species is originally described from Malay Archipelago, Indonesia 1. It is widely distributed in the Pacific and Indo-Malayan region, also in coastal areas of Japan and China. Additional Indo-Pacific materials are originated from Hong Kong 24, Sunda strait, Philippines 22, Amakusa and Tanabe Bay 25, 26, China 27, 28, and off Pacific Ocean; Palao Islands 29, Chile and Australia 30, 31, Indo-Malayan region 5, Kerama Islands, Okinawa 20, Nansei Islands 32. Also, it was recorded from different localities off Indian Ocean such as; Alphonse Island 23, Bombay 33, Indian coasts 34, 35, Arabian Sea and Bay of Bengal 36. It is extended to Gulf of Aden and southern Red Sea at Dahlak Archipelago 12.
Although C. bigelowi is a widespread species in the Indo-Pacific, and was frequently recorded from many locations in the Pacific and Indian oceans since its first record in 1905 1 up to 2006 32, its occurrence in the Red Sea was restricted to southern end. Curiously, Schmidt 12 mentioned finding specimens of Hydromedusae in the Red Sea and Gulf of Aden. He divided the area of investigation into 6 regions (Bay of Eilat (E); Gulf of Aqaba (A); northern Red Sea proper (S); southern Red Sea (R); Dahlak Archipelago (D) and Gulf of Aden (N)). He reported five specimens of C. bigelowi at five sites in the southern Red Sea (R) (16° 32ˊ N, 41° 06ˊ E) in 1958 and 1964, and one specimen in Dahlak Archipelago (D) in 1962.
Nevertheless, C. bigelowi was not hitherto recorded from the northern Red Sea whereas it was recorded in the eastern Mediterranean 37. It is worth mentioned that the presence of C. bigelowi in the Mediterranean is solely based on two preserved medusae from Israeli coast, without any figure or description 37. Thus, this finding is somewhat doubtful and needs further confirmation since the identification lacks some credibility. Additionally, a limitation of Schmidt findings from samples collected in 1968 up to hitherto, no more records for this species was appeared in the Mediterranean. Despite suggestion of transporting C. bigelowi through the Suez Canal from the Red Sea to the Mediterranean 37, we could not confirm his suggestion due to the lack of information on the presence of this species in Suez Canal and no more findings in the Mediterranean. However, this work could help to figure the route of migration via Suez Canal, in case that his presence in the Mediterranean Sea is confirmed. Further research on tracing this species, which take genetic data into account as molecular evidence, needs to be undertaken to better understand of the introduction route of C. bigelowi to the Mediterranean.
Environmental Assessments
The environmental factors are driving the geographical fauna distribution. In this attempt, the temperature and salinity were assessed. In the present study, C. bigelowi was recorded in the northern Red Sea in winter (December and February) when surface water temperature and salinity were ~18.4 °C and ~40 ‰, respectively. Earlier records indicated that C. bigelowi is a high temperature species being widely distributed in equatorial and tropical areas of the Pacific, Indo-Pacific and Indian Oceans 5, giving a tolerant temperature range between 24 and 28 °C 36. Moreover, C. bigelowi is considered a euryhaline species with an optimal salinity of ~ 33‰, and a tolerate range of 31.9-36‰ 36. The presence of C. bigelowi in the temperate-subtropical conditions of the northern Red Sea could be explained according to one of two assumptions. One of these could be that C. bigelowi broadened its tolerance range for temperature to the extent that it succeeded to occur in such lower temperature than its tolerance range. The second could be related to the climate change which makes the conditions in the northern Red Sea more suitable for C. bigelowi. Finally, it could likely reflect the scarcity of knowledge of Hydrozoa in the Red Sea due to lack of study rather than absence of species.
References
- 1.Maas O. (1905) Die Craspedoten Medusen der Siboga-Expeditie. , Siboga Expedition Monograph 10, 1-84.
- 2.Huang J. (1999) Three new species of genusEuphysorafrom China Seas (Hydrozoa: Anthomedusae. , Corymorphidae). Acta Oceanologica Sinica 18, 435-441.
- 3.Bouillon J, Boero F. (2000) Synopsis of the families and genera of the Hydromedusae of the world, with a list of the worldwide species. , Thalassia Salentina 24, 47-296.
- 4.Xu Z, Huang J. (2003) On new species and records ofEuphysorain Taiwan Strait and its adjacent waters (Cnidaria. , Hydrozoa, Hydroidomedusa, Anthomedusae, Capitata, Corymorphidae), J ournal of Oceanography in Taiwan Strait 22(2), 136-144.
- 5.P L Kramp. (1961) Synopsis of the medusae of the world. , Journal of the Marine Biological Association of the United Kingdom 40, 469.
- 6.Vannucci M, Navas-Pereira D. (1973) Distribution of Hydromedusae in the Indian Ocean. In: B. Zeitschel (Eds.), The biology of the Indian Ocean (pp 273-281). , Berlin-Heidelberg-New York 555.
- 7.Kubota S. (1997) Order Anthomedusae.In:M.Chihara&M.Murano(Eds.), An illustrated guide to marine plankton in Japan Tokyo,TokaiUniversityPress,1568. 485-494.
- 8.Kubota S. (2003) A list of medusae of Anthomedusae and Leptomedusae (Cnidaria. , Hydrozoa) in Japan. Nankiseibutu 45(1), 27-32.
- 9.Kubota S. (2003) A checklist of the Medusozoa and Ctenophora recorded from Tanabe Bay and its vicinities, Wakayama Prefecture, Japan-connection of polyp and medusa, if present, in the life history. , Annual Report of the Seto Marine Biological Laboratory 16, 30-35.
- 10.A G Mayer. (1910) Medusae of the world. , Hydromedusae, Vols. I & II. Scyphomedusae, Vol. III. Washington, Carnegie Institution 735, pp..
- 12.H E Schmidt. (1973) Die Hydromedusen (Hydrozoa: Coelenterata) des Roten Meeres und seiner angrenzenden Gebiete. Meteor Forsch.-Ergebn. 15, 1-35.
- 13.MDowidar M. (2003) Distribution and abundance of Cnidaria community in the Gulf of Aqaba and northern Red Sea. , Egypt. J Egypt Acad Soc Environ Develop 4(3), 119-136.
- 14.MDowidar M. (2003) Mesozooplankton communities in the Gulf of Aqaba and northern Red Sea. , Egyptian Journal of Aquatic Biology and Fisheries 7(1), 1-21.
- 15.Bouillon J, Medel Pagès, Gili F, M J Boero, F. (2004) . , Fauna of the Mediterranean Hydrozoa. Scientia Marina 68(2), 1-449.
- 16.Bouillon J, Gravili C, Pagès F, M J Gili, Boero F. (2006) An introduction to Hydrozoa. Muséum national d'Histoire naturelle: Paris. , France 591, pp..
- 17.Sassaman C, J T Rees. (1978) The life cycle ofCorymorpha(=Euphysora)bigelowi(Maas 1905) and its significance in the systematics of corymorphid hydromedusae. , Biol Bull 154, 485-496.
- 18.Hartlaub C. (1907) Craspedote Medusen,Tiel.I,Lief.I.Codoniden und Cladonemiden. , Nordisches Plankton 6, 1-135.
- 19.Uchida T. (1927) Studies on Japanese hydromedusae. , I. Anthomedusae. Journal of the Faculty of Science, Tokyo University, Zoology 1(3), 145-241.
- 20.Kubota S, Iwao K. (2002) Cnidarian medusa collected from coastal region of Akajima Island, Kerama Islands. , Okinawa, Japan, Midoriishi 13, 19-22.
- 21.SStepanjants D. (2013) Deep-water Hydrozoa (Cnidaria: Medusozoa). in the Sea of Japan, collected during the 51st Cruise of R/V Akademik M.A. Lavrentyev, with description Opercularella angelikae, sp. nov. Deep Sea Research II 86 - 87, 231-237.
- 22.P L Kramp. (1928) Papers from Dr. Th. Mortensen's Pacific Expedition 1914-1916. , XLIII. Hydromedusae I. Anthomedusae. Vidensk Medd Dansk Naturh Foren Kbh 85, 27-64.
- 25.Uchida T. (1938) Medusae in the vicinity of the Amakusa Marine Biological Station. , Bulletin of the Biogeographic Society of Japan 8, 143-149.
- 26.Yamazi I. (1958) Preliminary check-list of plankton organisms found in Tanabe ay and its environs. , Publications of the Seto Marine Biological Laboratory 7, 111-163.
- 27.S T Chiu. (1954) Studies on the medusa fauna of south-eastern China coast, with notes on their geographical distribution. , Acta Zoologica Sinica 6(1), 49-57.
- 28.T H Chow, M C Huang. (1958) A study on Hydromedusae of Chefoo. , Acta Zoologica Sinica 10, 173-191.
- 29.Uchida T. (1947) Some medusa from the central Pacific. , Journal of the Faculty of Science Hokkaido Uiversity Series VI. Zoology 9, 297-319.
- 30.P L Kramp. (1952) Reports on the Lund University Chile Expedition 1948-49. 2. Medusae collected by the L. , U. Exp. 1948-49. Acta Universitatis Lundensis 47, 1-19.
- 31.P L Kramp. (1953) . , Hydromedusae. Scientific Reports of the Great Barrier Reef Expedition 6(4), 259-322.
- 32.Kubota S. (2006) Hydromedusan fauna of the Nansei Islands. Proceedings of the 10th International Coral Reef Symposium , Okinawa, Japan.197-201 .
- 33.S H Lele, P B Gae. (1935) Some common Hydromedusae of the Bombay harbor. , Journal of the University of Bombay 3, 90-101.
- 34.NairK K. (1951) Medusae of the Trivandrum coast. Part I. , Systematics. Bull Res Inst Univ Travancore, Ser C Nat Sci 2, 47-75.
- 35.P N Ganapati, Nagabhushanam R. (1958) Seasonal distribution of the Hydromedusae off the Visakhapatnam coast. , Andhra University Memoirs in Oceanography 62(2), 91-99.
Cited by (1)
- 1.Schuchert Peter, Collins Richard, 2021, Hydromedusae observed during night dives in the Gulf Stream, Revue suisse de Zoologie, 128(2), 10.35929/RSZ.0049
