Record of Aggregation of Alien Tropical Schyphozoan Rhopilema nomadica Galil, 1990 in the Mediterranean Coast of Egypt
Abstract
Recently, annual swarm of invasive Erythrean schyphozoan RhopilemanomadicaGalil, 1990 appeared along Egyptian Mediterranean coasts causing beach closures and fishing problems. The present study conducted survey and field monitoring on R. nomadica during blooming season in the Egyptian Mediterranean coast throughout three consecutive years (2015-2017). Three main features of R. nomadica bloom were addressed; viz starting date, duration and maximum density of aggregation. In 2015, the bloom started on 28 July, and over the following two years the bloom starting date shifted earlier being 19 July in 2016 and 15 June in 2017. The duration of the bloom varied yearly giving the longest duration in 2017 when the bloom continued in high density for a month. The highest density of R. nomadica was about 896 medusae/1000 m3 in 2017. The medusae diameter ranged between 21 to 112 cm. The average bell diameter for each year displayed gradual increasing values over the years. The consistent annual R. nomadica blooming was attributed to the high level of eutrophication and ecosystem degradation occurred along the Mediterranean coast since last decades. The shifting in the annual bloom starting date and duration may reflect the adaptation of R. nomadica to the climate change effect on the Mediterranean Sea temperature.
Author Contributions
Academic Editor: Aurelie Moulins, CIMA research Foundation, Italy.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Fedekar Fadel Madkour, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
After a decade of the first recorded specimen of Rhopilemanomadica from lsraeli coast in 1976 1 and since the mid-1980's, many studies documented the progressive spreading of this species in the Mediterranean basin either as few specimens or massive swarms. Throughout the last forty years, R. nomadica stretched its range sequentially from eastern Levantine Sea off Israeli, Lebanon and Syrian coasts 2, 3, 4, 5, 6, 7, 8 to northern-east Levantine at Marmara and Aegean seas off Turkey 9, 10, 11, 12, 13, 14, passing through Greece and Malta 15, 16 and ending at the westernmost Mediterranean of the Italian island of Sardinia and Tunisia 17, 18.
Since the first appearance of R. nomadica in the eastern Mediterranean Sea in 1976 1, and even though R. nomadica purportedly entered to the Mediterranean via Suez Canal 2, no scientific publication documented the blooming of R. nomadica in the Egyptian coast. In 2016, a short article pointed to the presence of R. nomadica bloom in the Egyptian coast during summer 2015 19. Otherwise, Avian et al. 7 in their study on nematocysts of R. nomadica in the Eastern Mediterranean stated, "large aggregations have become ubiquitous of R. nomadica in the summer and winter months along the Levantine coasts from Egypt to Lebanon". Nevertheless, the presence of R. nomadica in the Egyptian coast as mentioned in Avian et al. study 7 lack evidence. Although the first scientific record of R. nomadica blooms from the Egyptian coast was in 2015 19, it has actually occurred before.
However, during last two decades, great complains from increasing jellyfish appeared within bathers and fishermen along Egyptian Mediterranean coast in summer. The nuisance effect of these blooms and severe stings from similar-looking jellyfish along the Egyptian coast were reported in a local newspaper in 22 August 2001 (http://www.ahram.org.eg/Archive/2001/8/22/INVE4.HTM). Although R. nomadica bloom is known to have both environmental and economic consequences, including injury to bathers causing beach closures and tourism impact, and reduced fishing harvests due to clogging of fishing net 5, no attention was paid to study the blooming density, causes and implications of R. nomadica along Egyptian coast. The present study conducted survey and field monitoring on R. nomadica during blooming season in the Egyptian Mediterranean coast throughout three consecutive years. It aimed to give an overview on the main features of the bloom, represented in the maximum density, the starting date and duration of the aggregation.
Materials and Methods
The outbreak of R.nomadica in the eastern Egyptian Mediterranean off Port Said coast (31° 16' N, 32° 19' E) (Figure 1) was monitored during blooming period (summer season) of the three consecutive years (2015 to 2017). Dispersed medusaeof R.nomadica were estimated every couple of days at a distance of 1 km inshore using fishermen beach trawling net, with estimate volume of sampled seawater of 1000 m3 at each haul. The density of jellyfish was calculated from the total number of medusae counted in hauls and expressed as number of medusae/1000 m3. Each year, the bell diameter of randomly selected specimens of R. nomadica was measured to the nearest cm (the sample size, N, was 505, 97 and 270 specimens for the three consecutive years, respectively). All measured specimens were investigated for the presence/absence of gonads. Surface water temperature was measured in situ using thermometer during sample collection.
Results
Jellyfish specimens observed in the water (Figure 2) and stranded medusa on the beach (Figure 3) were inspected for species identification and was found to be identical with Galil et al.'s description of R. nomadica. The survey was conducted at the onset of R. nomadica bloom along Port Said coast in the southeastern Mediterranean of Egypt during the three consecutive years (2015-2017). To evaluate and compare the aggregation of R. nomadica per time, three main features were addressed; they are starting date, duration and maximum density of aggregation. There were marked changes over the years in the starting date and duration of the aggregation, but little variation in maximum density of jellyfish (Figure 4). Considering each of the starting date and duration of the aggregation, in 2015 the bloom started on 28 July, and over the following two years the bloom starting date shifted earlier being 19 July in 2016 and 15 June in 2017. The duration of the bloom varied yearly giving the longest duration in 2017 when the bloom continued with high density for a month, while during the two preceding years (2015 and 2016), the bloom persisted lower period (11 and 7days, respectively), with longer duration in 2015. Considering R. nomadica density, the aggregation of jellyfish during these years showed little variation in the maximum density. In 2015, the aggregation peak (866 medusae/1000 m3) observed on 29th July. The value of this peak decreased insignificantly throughout the following year 2016, giving about 768 medusae/1000 m3 on 21st July. Then, increased again in 2017 reaching the highest peak (896 medusae/1000 m3) on 17th June (Figure 4).
Figure 2.Adult Rhopilema nomadica in the Mediterranean coast of Egypt. Photo by
Figure 3.Collecting dispersed medusae of R. nomadica from Port Said coast by beach trawling net.
Figure 4.Density of R. nomadica in Egyptian coast off Port Said during bloom period throughout three consecutive years (2015-2017).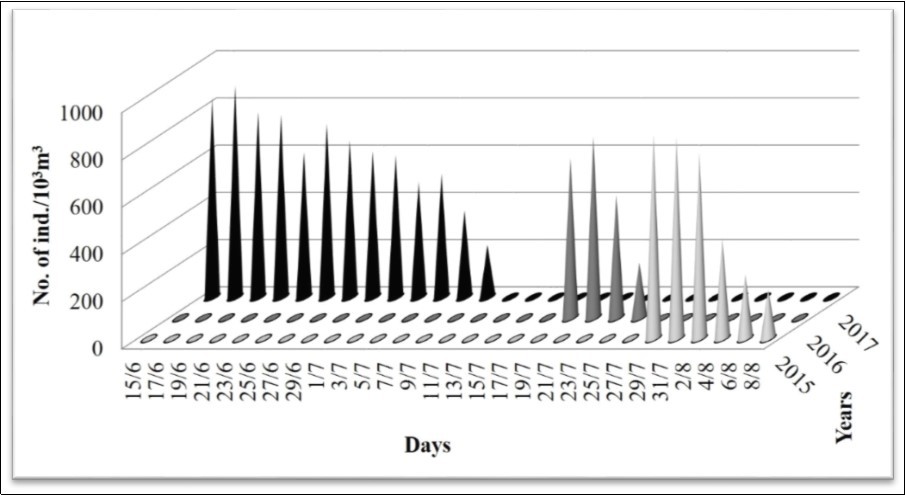
Bell diameter of the measured specimens ranged between the minimum of 21 cm and the maximum of 112 cm. The average bell diameter for each year displayed gradual increasing values over the years from the lowest average of 42.1+7.78 cm in 2015 and the highest average of 46.26+13.03 cm in 2017 (Table 1). In 2015, most of individuals released their gonads (76%), this was inverted in 2017 when only 30% of the investigated medusae shed their gonads, while in 2016 about half of medusae (48%) were empty from gonads (Table 1). Sea surface temperature (SST) ranges throughout three years were 25-31 °C in 2015, 24-33°C in 2016 and 29-38°C in 2017 (Table 1).
Table 1. Seawater temperature, medusae diameter and gonads presence/absence of R. nomadica in the Egyptian coast off Port Said, during three consecutive years (2015-2017).| Year | 2015 | 2016 | 2017 | |
| Temperature range (°C) | 25-31 | 24-33 | 29-38 | |
| Medusa diameter (cm) | Min. | 21 | 25 | 28 |
| Max. | 66 | 68 | 112 | |
| Mean | 42.1 ± 7.78 | 43.59±8.42 | 46.26±13.03 | |
| Gonad | Presence (%) | 122 (24 %) | 50 (52 %) | 189 (70 %) |
| Absence (%) | 383 (76 %) | 47 (48 %) | 81 (30 %) | |
Discussion
Of the all studies carried out on the proliferation of R. nomadica along the Mediterranean basin from the southeastern side to the most western side since its appearance in the mid 1970s 1 up to date, few studies documented its massive occurrence on the coasts. Huge swarms of this species started to appear since late 1980s and were restricted to eastern Levantine Sea off Israeli coast 3, 5, Lebanon and Syria coasts 4, and the Turkey coast at Mersin and Iskendurun Bay 9, 10. Afterward, scattered individuals have only been occasionally recorded in the central Mediterranean (Maltese islands, Italian island of Pantelleria and Sardinia), 16, 20, 21, 22. While in the most western Mediterranean, R. nomadica was consistently recorded each summer since 2010 within the Gulf of Tunis 18, then during 2014-2016, outbreaks of R. nomadica started to be established in Bizerte Lagoon, Tunisia 18.
In the present study, the maximum density of R. nomadica outbreak (896 medusae/1000 m3) was close to those recorded in Israeli coast during 1990 (10 medusae/ m33). It was much higher than those recorded in Israeli coast during summer 1989 (160,000 medusae/ km25), Turkey coast during summer 1995 (38,000 medusae per square nautical mile 9 or in Tunisia coast (4.4 medusae/ km218). The rapid proliferation of R. nomadica in the eastern Mediterranean had been related to its high reproductive potential where one settled polyp of this species could produce more than 100 ephyrae within 2 months 5. The strobilation process of the species’ polyps of R. nomadica seems to be temperature dependence where the synchronization of life cycle and annual occurrence of R. nomadica may be controlled by seasonal variations in water temperature regimes 6. In the present study, the recorded ranges (24-38°C) of sea temperature throughout the three years seems to be highly convenient for attaining the highest blooming density of R. nomadica ever recorded before along the Mediterranean. Moreover, decreasing the percentage of individuals that released their gonads with the increasing of sea temperature, suggesting that increasing temperature may induce influx of the R. nomadica swarms from the deep sea to the coast, to be stranded, before releasing their gonads.
Swarming of jellyfish in the Mediterranean Sea had been suggested to have positive correlation with pollution 22, 23. These findings relate jellyfish blooms to the ability of medusae to utilize plankton blooms produced by pollution-caused eutrophication, or by their ability to utilize pollutants directly as food. Another positive correlation between swarming of jellyfish and certain climate change was also found 24. For R. nomadica, previously recorded outbreaks in the coastal waters of Israel 5, Turkey 9 and Tunisia 17 were related to degraded ecosystem in addition to climatic changes, in agreement with Purcell’s proposal 25 who stated that jellyfish can be used as indicators of global warming. Also, the establishment of a viable R. nomadica population within the Bizerte lagoon, as opposed to a more ephemeral occurrence within the other central Mediterranean areas, have been attributable to the lagoon trophic status (less oligotrophic) and by virtue of the sheltered nature of the lagoon 18. In the same context, the Egyptian Mediterranean coast had sever pollution impact throughout the last decades because of continuous effluents from the coastal lagoons that connected to the sea. The Egyptian Mediterranean coast receives huge volumes of wastewaters every year loaded by variable amounts and types of pollutants, in addition to great amount of nitrogenous and phosphorous compounds through these lagoons 26. This situation promotes high level of eutrophication and ecosystem degradation along the Mediterranean coast, providing a great opportunity for consistent annual R. nomadica blooming.
The present study revealed that the annual massive occurrence of R. nomadica showed continuous shifting in the starting date with time towards the less warm months experienced in the study area. It shifted over the three years from end of July in 2015 to be during mid-June in 2017. When tracing the past observations on the aggregation dates and duration of R. nomadica bloom in other locations of the Mediterranean basin, a clear change was noticed. In Israeli coast, Mass swarming of R. nomadica used to occur mostly in the summer particularly mid-August and ending between September and November 6. This change in the annual bloom starting date and duration can be explained in the light of the assumption that the R. nomadica adapted itself to the climate change effect on the Mediterranean Sea temperature. Increasing temperature with time was obvious from recorded temperature ranges (Table 1).
The size range for medusae diameter in the present study (21-112 cm) was wider than given by previous observations in other locations in the Mediterranean Sea. The previously recorded ranges of medusa size were 20- ~100 cm, commonly 20-60 cm 3, 50-58 cm 9, 10-85 cm 7, 40-42 cm 9, 13, 15, 16. The largest medusae size recorded in the present study (112 cm) was higher than those recorded before which did not exceed 100 cm 7. It is worth mentioned that the advance in the starting date of aggregation was associated with bigger medusa size.
References
- 2.B S Galil, Spanier E, W. (1990) The Scyphomedusae of the Mediterranean coast of Israel, including two lessepsian migrants new to the Mediterranean. , Zoologische Mededelingen Leiden 64, 95-105.
- 3.Spanier E, Galil B. (1991) Lessepsian migration: a continuous biogeographical process. , Endeavour, New Set 15, 102-106.
- 4.Lakkis S, Zeidane R. (1991) Jellyfish swarm along the Lebanese coast. (Abstract) Lebanese Association for the Advancement of Science,11th Science Meeting AmericanUniversity of Beirut.
- 5.Lotan A, Ben-Hillel R, Loya Y. (1992) Life cycle ofRhopilema nomadica: a new immigrant scyphomedusan in the Mediterranean. , Marine Biology 112, 237-242.
- 6.Lotan A, Fine M, Ben-Hillel R. (1994) Synchronization of life cycle and dispersal pattern of the tropical invader schyphomedusanRhopilemanomadicais temperature dependent. Marine Ecology Progress Series 109, 59-65.
- 7.Avian M, Spanier E, Galil B. (1995) Nematocysts ofRhopilemanomadica(Scyphozoa: Rhizostomae), An immigrant jellyfish in the Eastern Mediterranean. , J. Morphol 224, 221-231.
- 8.Ikhtiyar S, Durgham H, Bakr M. (2002) Contribution to the study of the scyphomedusaRhopilemanomadicain Syrian coastal waters. , Journal of Union of Arab Biologists Cairo A Zoology 18, 227-244.
- 9.A E Kideys, A C Gücü. (1995) A Lessepsian scyphomedusan new to the Mediterranean coast of Turkey. , Israel Journal of Zoology 41, 615-617.
- 10.Avsar D, Çevik C, Türeli C. (1996) İskenderun Körfezi için yeni bir tür olan (Rhopilemanomadica)’nın biyometrisi ve Yumurtalık Koyundaki bulunurluğu. , XIII. Ulusal Biyoloji Kongresi, 17-20 Eylül1996 İstanbul, Düzenleyen Kuruluş: İ.Ü, Fen Fakültesi, Biyoloji Bölümü
- 11.B S Galil, Zenetos A. (2002) A sea change. Exotics in the Eastern Mediterranean. In: Leppäkoski E. Gollasch S. Olenin S (eds). Invasive aquatic species of Europe: distriution, impacts, and management. , Dordrecht 325-336.
- 12.Öztürk B, İşinibilir M. (2010) An alien jellyfishRhopilema nomadicaand its impacts to the Eastern Mediterranean part of Turkey. , J. Black Sea/Mediterranean Environment 16(2), 149-156.
- 13.Gülşahin N, A N Tarkan. (2011) The first confirmed record of the alien jellyfishRhopilema nomadicaGalil,1990 from the southern Aegean coast of Turkey. S95-S97, http://dx.doi.org/10.3391/ai.2011.6.S1.022 , Aquatic Invasions 6(1).
- 14.Sakınan S. (2011) Recent occurrence of Indo-Pacific jellyfishRhopilema nomadicain North-Eastern Levantine Sea. First National Workshop on Jellyfish and other Gelatinous Species in Turkish Marine Waters, Published by Turkish Marine Research Foundation , Istanbul, Turkey 35, 73-77.
- 15.Siokou-Frangou I, Sarantakos K, D C Epaminondas. (2006) . First record of the scyphomedusaRhopilemanomadicaGalil 1990 (Cnidaria: Scyphozoa: Rhizostomeae) in Greece. Aquatic Invasions 1, 194-195.
- 16.Deidun A, Arrigo S, Piraino S. (2011) The westernmost record ofRhopilemanomadica(Galil. in the Mediterranean – off the Maltese Islands. Aquatic Invasions, http://dx.doi.org/10.3391/ai.2011.6.S1.023 6(1), 99-103.
- 17.Yahia M N D, Yahia O K D, Gueroun S K M, Aissi M, Deidun A. (2013) . The invasive tropical scyphozoanRhopilema nomadicaGalil,1990 reaches the Tunisian coast of the Mediterranean Sea, Bio Invasions Records 2(4), 319-323.
- 18.Balistreri1 P, Spiga A, Deidun A, S K Gueroun, Yahia M N D. (2017) Further spread of the venomous jellyfishRhopilemanomadicaGalil, Spannier & Ferguson. , (Rhizostomeae, Rhizostomatidae) in the western Mediterranean, Bio Invasions Records 6(1), 19-24.
- 19.El-Regal Abu, MA, Temraz T A. (2016) Blooming of the nomad jelly fishRhopilema nomadicaalong the Egyptian Mediterranean coasts. , Rapp. Comm. int. Mer Médit 41, 490.
- 20.Crocetta F, Agius D, Balistreri P, Bariche M, Y K Bayhan. (2015) . , New Mediterranean Biodiversity Records (October,2015), Mediterranean Marine Science 16, 472-488.
- 21.ICES. (2016) Report of the Working Group on. Introductions and Transfers of Marine Organisms (WGITMO). Olbia: ICES CM 2016/SSGEPI: 10, 201.
- 22.F P Wilkerson, R C Dugdale. (1983) Possible connection between sewage effluent, nitrogen levels and jellyfish blooms. In:. Jellyfish blooms in the Mediterranean Proceedings of the 11 Workshop on Jellyfish in the Mediterranean Sea. Map Technical Reports Series no. 47 .
- 23.Bingel F, Avsar D, A C Gucu. (1991) Occurrence of jellyfish in Mersin Bay. 1n: Jellyfish blooms in the Mediterranean. Proceedings of the 1 Workshop on Jellyfish in the Mediterranean Sea. Map Technical Reports Series no. 47 .
- 24.Goy J, Morand P, Etienne M. (1989) Long term fluctuations ofPelagianoctiluca(Cnidaria. Scyphomedusa) in the western Mediterranean Sea. Prediction by climatic variables. , Deep Sea Res 36(2), 269-280.
Cited by (28)
This article has been cited by 28 scholarly works according to:
Citing Articles:
Environmental Microbiome (2025) OpenAlex
Environmental Microbiome (2025) Crossref
Research Square (Research Square) (2024) OpenAlex
Anastasiia Iakovleva, Arseniy R. Morov, Dror L. Angel, T. Guy-Haim - Scientific Reports (2024) Semantic Scholar
Scientific Reports (2024) OpenAlex
Scientific Reports (2024) Crossref
Noga Barak, Vera Brekhman, Dikla Aharonovich, Tamar Lotan, Daniel Sher - bioRxiv (2024) Semantic Scholar
bioRxiv (Cold Spring Harbor Laboratory) (2024) OpenAlex
J. Douek, Giovanni Giallongo, Zoya Harbuzov, B. Galil, Baruch Rinkevich - Journal of Marine Science and Engineering (2024) Semantic Scholar
Journal of Marine Science and Engineering (2024) OpenAlex
Journal of Marine Science and Engineering (2024) Crossref
Marine Ecology Progress Series (2024) Crossref
Sabry A. El-Naggar, Wesam M. Salama, N. El-Desouki, Ghada Tabl, Rasha Abo Jobier et al. - Journal of Bioscience and Applied Research (2023) Semantic Scholar
Journal of Bioscience and Applied Research/Journal of Bioscience and Applied Research (2023) OpenAlex
H. Dror, D. Angel - Marine Ecology Progress Series (2022) Semantic Scholar
Marine Ecology Progress Series (2022) OpenAlex
W. Salama, S. El-Naggar, Hadeer Attia, Ezar H Hamed - Egyptian Journal Of Zoology (2022) Semantic Scholar
Egyptian Journal Of Zoology (2022) OpenAlex
D. Edelist, Ø. Knutsen, I. Ellingsen, Sanna Majaneva, N. Aberle et al. - Frontiers in Marine Science (2022) Semantic Scholar
Frontiers in Marine Science (2022) OpenAlex
Frontiers in Marine Science (2022) Crossref
Giovanni Giallongo, J. Douek, Zoya Harbuzov, B. Galil, B. Rinkevich - Biological Invasions (2021) Semantic Scholar
Biological Invasions (2021) OpenAlex
Biological Invasions (2021) Crossref
J. Douek, Zoya Harbuzov, B. Galil, B. Rinkevich - Molecular Biology Reports (2020) Semantic Scholar
Molecular Biology Reports (2020) OpenAlex
D. Edelist, T. Guy‐Haim, Z. Kuplik, Noa Zuckerman, P. Nemoy et al. - Journal of Plankton Research (2020) Semantic Scholar
Journal of Plankton Research (2020) OpenAlex
Journal of Plankton Research (2020) Crossref
T. Guy‐Haim, Maxim Rubin‐Blum, E. Rahav, Natalia Belkin, J. Silverman et al. - (2020) Semantic Scholar
T. Guy‐Haim, Maxim Rubin‐Blum, E. Rahav, Natalia Belkin, J. Silverman et al. - Biogeosciences (2020) Semantic Scholar
Biogeosciences (2020) OpenAlex
Biogeosciences (2020) Crossref

