Abstract
The samples of flatfishes were collected along the Mon State from July to December 2017 including Mawlamyine, Kyaikkhami, Setse, Zee-Phyu-Thaung Belugyun island, Paung and Thaton. The total 21 species of flatfishes were recorded in Mon State. Moreover, the relative abundance, diversity and similarity were conducted using Simpson Index, Shannon index and Jacquard’s index etc. According to the present study, the Cynoglossidae was the most dominant family and the most dominant species was Cynoglossusarelfor the present study. The relative abundance of Mawlamyine and November was relatively the highest. Furthermore, Mawlamyine was the highest diversity and Thaton was the lowest diversity. The JI value between Mawlamyine and Paung is the highest while it is the lowest between Setse and Thaton. Likewise, the JI value between August and September and the value between September and November are the same as well as the highest while it is the lowest for the comparison between July and August. The present study also expects to become a basic informational providing data for further observing (in Academic) on Order Pleuronectiformes (flatfishes) along the Mon State.
Author Contributions
Academic Editor: Cesar Amaral, DNA Diagnostic Laboratory, University of the State of Rio de Janeiro, Brazil.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 ThetHtwe Aung, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Pleuronectiformes were first named in 1758 by Linnaeus; pleuromeaning "on side" and nectomeaning "swim". Flatfishes are easy to recognize since this is the only group of fishes that is not bilaterally symmetrical. The ventral side of the body is eyeless and white, while the dorsal is dark and has both eyes. They swim by the undulation of the body, and usually remain close to the bottom on the continental shelf.
Flatfishes are one of the important components in several demersal communities around the world. Flatfishes are also important predators in many demersal communities. They may serve as an important link between demersal production and human consumption9.
The flatfishes are one of the most important economic groups of marine fishes in Myanmar. From the commercial point of view, all species are locally known as “Khway-Shar”. It yields 2277.35 tones and by percentage, it had 4.41 % of the Myanmar marine fish landings in Yangon, the business city of Myanmar for 200510.
The present study area, called Mon state is an administrative division of Myanmar. It is sandwiched between Kayin State in the east, the Andaman Sea in the west, Bago Region in the north and Tanintharyi Region in the south. The land area is 12, 155 km2. The state of capital is Mawlamyine. Bordering Bago Division in the south of Sittaung river mouth, Kayin state in the east, Thailand and Taninthayi division in the south and Andaman Sea and Gulf of Mottama in the west, Mon state is situated between Latitudes 14° 52′ and 17° 32′ N and Longitudes 96° 51′ and 98° 13′ E.
Numerous studies have been done on the distribution, abundance and fisheries biology of flatfish in many different places around the world3,4,5. However, with the exception of a few taxonomic studies, there was no study about that in Myanmar. Therefore, the objectives of this study is to understand distribution, abundance and diversity of flatfishes along the Mon State, Myanmar.
Materials and Methods
Sampling Sites
The Specimens Identified in this Study were Collected from the Following Seven Places.
1. Mawlamyine (Lat. 16° 29′ N, Long. 97° 37′ E),
2. Kyaikkhami (Lat. 16° 03′ N, Long. 97° 33′ E),
3. Setse (Lat. 15° 56′ N, Long. 97° 37′ E),
4. Zee-Phyu-Thaung (Lat. 15° 15′ N, Long. 98° o′ E),
5. Belugyun island (Lat. 16° 12' N and 16° 32' N and Long. 79° 52' and 79° 35' E),
6. Paung (Lat. 16° 37' N and 97° 34'E) and
7. Thaton (Lat. 17° 00' N and 97° 22'E) (Figure 1)
Figure 1.Map showing the specimens collection sites of the flatfishes (Order Pleuronectiformes) from Mon State
Samples Collecting and Observation
From July to December 2017, the samples of flatfish species were randomly collected on monthly basis, at seven stations along the Mon State, not only from fishing boats but also from the fishery markets. The fresh samples were put in the bags and containers. For environmental parameters, the water samples in each station were measured their temperature (ᵒC), salinity (‰) and pH with thermometer, refractometer and portable pH meter. The samples were brought back to the laboratory, rinsed and identified using the illustration handbooks produced by the F.A.O species identification sheets6 and other available references. 1, 2, 7 After identification, each species was counted and the total length and weight of fishes were measured. All data were then digitized and saved for later analysis. (Table 1).
Table 1. Area-wise environmental parameters from July to December 2017| Study areas | Parameters | July | August | September | October | November | December | |
| 1 | Mawlamyine | TemperatureᵒC | 30.0 | 29.5 | 29.0 | 28.0 | 27.0 | 26.5 |
| Salinity ‰ | 25.5 | 25.0 | 23.5 | 26.5 | 23.5 | 25.5 | ||
| pH | 7.2 | 7.2 | 7.4 | 7.4 | 7.4 | 7.6 | ||
| 2 | Kyaikkhami | TemperatureᵒC | 29.5 | 27.5 | 28.5 | 28.0 | 27.0 | 26.5 |
| Salinity ‰ | 28.0 | 24.5 | 28.0 | 26.5 | 27.0 | 25.5 | ||
| pH | 7.4 | 7.2 | 7.4 | 7.6 | 7.6 | 7.8 | ||
| 3 | Setse | TemperatureᵒC | 29.5 | 28.0 | 28.0 | 27.5 | 27.0 | 26.5 |
| Salinity ‰ | 30.5 | 26.0 | 26.5 | 27.0 | 27.5 | 27.0 | ||
| pH | 7.6 | 7.2 | 7.4 | 7.6 | 7.8 | 7.6 | ||
| 4 | Zee-Phyu-Thaung | TemperatureᵒC | 30.0 | 28.5 | 28.0 | 27.5 | 27.5 | 26.0 |
| Salinity ‰ | 30.5 | 27.5 | 29.5 | 30.0 | 30.5 | 29.5 | ||
| pH | 7.8 | 7.6 | 7.8 | 7.8 | 8.0 | 7.8 | ||
| 5 | Belugyun island | TemperatureᵒC | 29.0 | 27.5 | 28.0 | 28.5 | 27.5 | 26.0 |
| Salinity ‰ | 26.5 | 25.0 | 28.0 | 27.0 | 28.0 | 25.0 | ||
| pH | 7.4 | 7.4 | 7.6 | 7.8 | 7.8 | 8.0 | ||
| 6 | Paung | TemperatureᵒC | 30.0 | 27.5 | 28.5 | 27.5 | 26.5 | 26.0 |
| Salinity ‰ | 26.0 | 25.0 | 26.0 | 27.0 | 27.5 | 26.0 | ||
| pH | 7.4 | 7.4 | 7.4 | 7.6 | 7.8 | 7.8 | ||
| 7 | Thaton | TemperatureᵒC | 30.0 | 28.5 | 28.0 | 28.0 | 27.5 | 26.5 |
| Salinity ‰ | 25.5 | 25.0 | 25.5 | 25.0 | 26.5 | 25.5 | ||
| pH | 7.4 | 7.2 | 7.2 | 7.4 | 7.2 | 7.6 |
Statistical Analysis
The relative abundance (percentage of catch) of fish across different sites was calculated. RA of individual species was calculated by the following formula.
Number of samples of particular species x 100 Total number of sample
The species richness indices were calculated using Simpson Index (D).
Simpson Index (D) =
In the Simpson index, D is richness, p is the proportion (n/N) of individuals of one particular species found (n) divided by the total number of individuals found (N), Σ is still the sum of the calculations, and s is the number of species.
Evenness was calculated by the following formula.
J′ = H/lnS
In the formula, J′ is evenness, S= the number of species, H is richness.
The species diversity indices were calculated using Shannon index.
Shannon index (H) =
In the Shannon index, H is diversity, p is the proportion (n/N) of individuals of one particular species found (n) divided by the total number of individuals found (N), ln is the natural log, Σ is the sum of the calculations, and s is the number of species.
Similarity of the species in all sampling station was calculated using Jacquard’s index:
Sj = j / x+y+j
Where Sj is the similarity between any two zones X and Y, j the number of species common to both the zones X and Y, x the total number of species in zone X and y total number of species in zone Y. The similarity in species composition has shown as a dendrogram, obtain from the JI coefficients of similarity using the average linkage method.
Results and Discussion
A total of 5 families, including 21 species of flatfishes were recorded in Mon State. The Psettodidae which included 1 genus and 1 species, the Paralichthyidae, which included 1 genus and 2 species, the Bothidae which included 1 genus and 1 species, the Soleidae which included 4 genera and 7 species, the Cynoglossidae which included 2 genera and 10 species were found along the Mon state in this study.
In table 2, Psettodidae was only found in Zee-Phyu-Thaung, Bothidae was only found in Setse and Paralichthyidae was found in Zee-Phyu-Thaung and Setse. Cynoglossidae was found in every station abundantly but Soleidae was found in Mawlamyine, Paung and Thaton. Therefore, it can be inferred Psettodidae, Bothidae and Paralichthyidae can survive in higher salinity than Cynoglossidae and Soleidae. Of seven stations, the most different kind of species were collected in Mawlamyine and the lowest were collected in Thaton.
Table 2. Showing species list of fishes caught by different types of fishing gear and their habitats in sampling areas.| Sr. No | Scientific Name | Sampling areas | Type of gear | ||||||||||
| Mawlamyine | Kyaikkhami | Setse | Zee-Phyu-Thaung | Belugyun island | Paung | Thaton | Bag net | Gill net | Cast net | Trawl net | Drift net | ||
| 1. | Psettodes erumei | - | - | - | + | - | - | - | - | - | - | + | + |
| 2. | Pseudorhombus arsius | - | + | - | + | - | - | - | - | - | + | + | |
| 3. | P. dupliciocellatus | - | - | - | + | - | - | - | - | - | - | + | + |
| 4. | Engyprosopongrandisquama | - | - | + | - | - | - | - | - | - | - | + | + |
| 5. | Heteromycteris oculus | - | + | - | - | - | - | - | - | - | - | + | + |
| 6. | Synapture albomaculata | + | - | - | - | - | - | - | - | - | - | + | - |
| 7. | Zebraiszebra | - | - | + | - | - | - | - | + | + | + | + | + |
| 8. | Z.synapturoides | - | - | + | - | - | - | - | + | + | + | + | + |
| 9. | Euryglossa harmandi | + | - | - | - | - | + | - | + | - | + | - | - |
| 10. | E. orientalis | + | - | - | - | + | + | + | + | - | + | - | - |
| 11. | E. panoides | + | - | - | - | + | + | - | + | - | - | ||
| 12 | Paraplagusi bilineata | + | + | + | _ | + | _ | _ | + | + | + | + | + |
| 13. | P. ablochii | - | - | + | - | - | - | - | + | + | + | + | + |
| 14. | Cynoglossus lida | + | - | - | - | - | + | - | + | + | + | - | - |
| 15. | C. Bengalensis | + | - | - | - | - | + | + | + | + | + | - | - |
| 16. | C. semifasciatus | + | - | - | - | - | + | + | + | + | + | - | - |
| 17. | C.cynoglossus | + | - | - | - | + | + | - | + | + | + | - | - |
| 18. | C. carpenteri | - | + | + | + | - | - | - | - | - | - | + | + |
| 19. | C. lingua | + | + | + | + | - | + | - | - | - | - | + | + |
| 20 | C.macrolepidotus | + | + | + | + | - | - | - | - | - | - | + | + |
| 21. | C.arel | + | + | + | + | + | + | + | - | - | - | + | + |
| Total | 12 | 7 | 9 | 7 | 4 | 9 | 4 | ||||||
In addition, flatfishes are caught mostly using bottom trawl nets by fishermen in all stations as the flatfishes are close to the bottom and swim by undulation of the body. Besides bag nets, gill nets, cast nets and drift nets are used for catching flatfishes in some stations. Soleidae appears to be caught mostly by bag nets and cast nets and Psettodidae, Paralichthyidae and Bothidae appear to be caught mostly by bottom trawl nets in Mon State.
Due to sampling limits in this study, potential bias may be an issue, especially as large variations in catch were observed among vessels. However, since our samples were taken randomly from different catches deposited in the market, it was believed such bias was minimized. it cannot be excluded that the possibility that rare flatfish species were under-represented in the study (in terms of their number and abundance) because many of these species are small in size and deposited as trash fish and so could not be examined completely.
Of the total sample number 378 of 21 species in seven stations, samples were obtained the total number of 128 in Mawlamyine, 79 in Kyaikkhami, 29 in Setse, 53 in Zee-Phyu-Thaung, 19 in Belugyun, 48 in Paung and 19 in Thaton (table 3).
Table 3. The sample number of flatfish species for each station of Mon State from July to December 2017| Family | Scientific name | No. of specimens | ||||||||
| Sampling areas | Mawlamyine | Kyaikkhami | Setse | Zee-Phyu-Thaung | Belugyun island | Paung | Thaton | Total | ||
| Psettodidae | Psettodes erumei | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | |
| Paralichthyidae | Pseudorhombus arsius | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 2 | |
| P. dupliciocellatus | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | ||
| Bothidae | Engyprosopon grandisquama | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | |
| Soleidae | Synapture albomaculata | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | |
| Heteromycteris oculus | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | ||
| Zebrais zebra | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 2 | ||
| Z. synapturoides | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | ||
| Euryglossa harmandi | 7 | 0 | 0 | 0 | 0 | 3 | 0 | 10 | ||
| E. orientalis | 6 | 0 | 0 | 0 | 2 | 5 | 2 | 15 | ||
| E. panoides | 1 | 0 | 0 | 0 | 1 | 0 | 1 | 3 | ||
| Paraplagusi bilineata | 1 | 1 | 3 | 0 | 1 | 0 | 0 | 6 | ||
| P. ablochii | 0 | 0 | 4 | 0 | 0 | 0 | 0 | 4 | ||
| Cynoglossus lida | 3 | 0 | 0 | 0 | 0 | 2 | 0 | 5 | ||
| C. Bengalensis | 37 | 0 | 0 | 0 | 0 | 18 | 15 | 75 | ||
| Cynoglossidae | C. semifasciatus | 20 | 0 | 0 | 0 | 0 | 10 | 0 | 31 | |
| C. cynoglossus | 32 | 0 | 0 | 0 | 6 | 10 | 0 | 48 | ||
| C. carpenteri | 0 | 2 | 1 | 1 | 0 | 0 | 0 | 4 | ||
| C. lingua | 1 | 1 | 1 | 4 | 0 | 0 | 1 | 8 | ||
| C. macrolepidotus | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 4 | ||
| C. arel | 17 | 71 | 15 | 45 | 9 | 0 | 0 | 154 | ||
| Total | 128 | 79 | 29 | 53 | 19 | 48 | 19 | 378 | ||
The relative abundance of number of species of Mawlamyine is relatively the highest (34%) of all stations. The relative abundance of number of species of Kyaikkhami (21%) is the higher as compared to Zee-Phyu-Thaung. In the same way, the relative abundance of number of species of Paung (12%) is higher than Setse (7%) and the relative abundance of number of species of Belugyun and Thaton are the same with 5% respectively (figure 2A).
Of the total sample number 378 of 21 species, samples were obtained the total number of 36 in July, 41 in August, 75 in September, 86 in October, 88 in November and 52 in December (table 4).
In the figure 2B, the relative abundance 23% in November was the highest month, followed by October 22%, September 19%, December 13% and August 11% respectively. The relative abundance in July is the lowest (9%).
Figure 2.Comparison relative abundance percentage of sample number of flatfish species A) for each station and B) for each month from July to December 2017.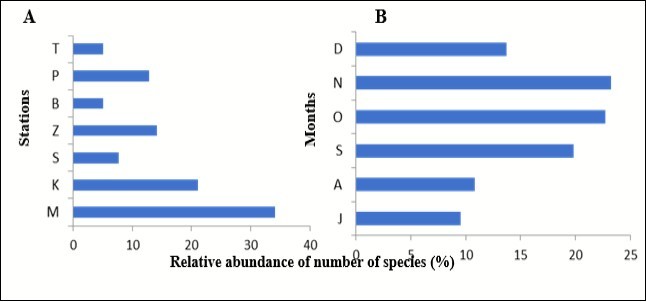
| Family | Scientific name | No. of specimens | Total | ||||||
| Month | July | August | September | October | November | December | |||
| Psettodidae | Psettodes erumei | 0 | 1 | 0 | 0 | 0 | 0 | 1 | |
| Paralichthyidae | Pseudorhombus arsius | 0 | 0 | 0 | 1 | 0 | 1 | 2 | |
| P. dupliciocellatus | 0 | 0 | 0 | 0 | 0 | 1 | 1 | ||
| Bothidae | Engyprosopon grandisquama | 1 | 0 | 0 | 0 | 0 | 0 | 1 | |
| Soleidae | Synapture albomaculata | 0 | 0 | 1 | 0 | 1 | 0 | 2 | |
| Heteromycteris oculus | 0 | 0 | 0 | 0 | 0 | 1 | 1 | ||
| Zebrais zebra | 2 | 0 | 0 | 0 | 0 | 0 | 2 | ||
| Z. synapturoides | 0 | 0 | 1 | 0 | 0 | 0 | 1 | ||
| Euryglossa harmandi | 0 | 1 | 0 | 3 | 4 | 2 | 10 | ||
| E. orientalis | 0 | 6 | 4 | 0 | 4 | 1 | 15 | ||
| E. panoides | 0 | 0 | 1 | 0 | 2 | 0 | 3 | ||
| Paraplagusi bilineata | 0 | 3 | 2 | 0 | 1 | 0 | 6 | ||
| P. ablochii | 2 | 0 | 0 | 2 | 0 | 0 | 4 | ||
| Cynoglossus lida | 0 | 4 | 0 | 1 | 0 | 0 | 5 | ||
| C. Bengalensis | 2 | 5 | 18 | 10 | 28 | 12 | 75 | ||
| Cynoglossidae | C. semifasciatus | 4 | 1 | 5 | 10 | 5 | 6 | 31 | |
| C. cynoglossus | 12 | 8 | 0 | 6 | 17 | 5 | 48 | ||
| C. carpenteri | 0 | 0 | 2 | 0 | 2 | 0 | 4 | ||
| C. lingua | 0 | 2 | 1 | 3 | 0 | 2 | 8 | ||
| C. macrolepidotus | 1 | 0 | 2 | 0 | 0 | 1 | 4 | ||
| C. arel | 12 | 10 | 38 | 50 | 24 | 20 | 154 | ||
| Total | 36 | 41 | 75 | 86 | 88 | 52 | 378 | ||
In figure 3A, Mawlamyine was the highest diversity whereas, Thaton was the lowest diversity. Although Paung and Belugyun have the same value of diversity, the richness of Paung is much higher than Belugyun’s richness. It could be possible that there was an individual dominance in Paung station. Therefore, various species are not found there but the number of individual is high.
The similarity in species composition among the stations was analyzed using the Jaccard index for calculating the extent of similarity between pairs of data sets. Cluster analyses identified two assemblages (with a similarity of 14%), which corresponded to the group of Mawlamyine, Paung and the group of Kyaikkhami, Setse, Zee-Phyu-Thaung, Belugyun and Thaton (figure 4A).
Figure 3.A) the differences among station and B) months of Diversity, evenness and richnessamong the six months from July to December 2017.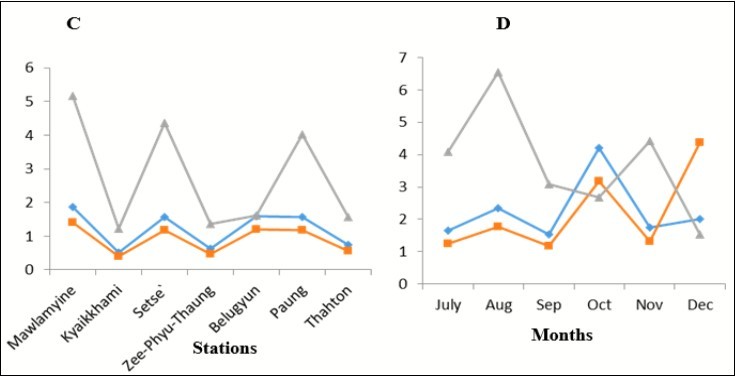
In figure 3B, October was the highest value of diversity and August was the highest value of richness. Likewise, September was the lowest value of diversity and December was the lowest value of richness. On the other hand, August and November were high value of richness which the values of diversity are low. So, these months can be the result of an individual dominance. In another way, in spite of being high diversity in October and December, their richness is low because there will be various kinds of species in these months but the number of samples are scare.
The similarity in species composition among the months was analyzed using the Jaccard index for calculating the extent of similarity between pairs of data sets. The JI value between August and September and the value between September and November are the same as well as the highest while it is the lowest for the comparison between July and August. Cluster analyses identified two assemblages (with a similarity of 53%), which corresponded to the group of September, November, August and the group of July, December, October (figure 4B).
Figure 4.A) Dendrogram of similarity among the seven stations; B) among the six months from July to December 2017.
Wang et al.8 had examined the seasonal dynamics of flatfish assemblage in the coastal waters off northeastern Taiwan. In their study, the Bothidae was the most dominant family, accounting for 81% of the total catch, while the most dominant species was the Pseudorhombuspentophthalmus. Species richness and diversity were high between the end of fall and the end of spring, but low in summer. In contrast, the Cynoglossidae was the most dominant family and the most dominant species was Cynoglossusarelfor the present study. It can be possible due to the geographical differences.
Table 5 shows the number of samples in each station bimonthly. Of the 375 total number of samples, Mawlamyine (128 number of samples) was the highest number station and then the highest number of samples were within September and October. In addition to, the samples could be collected 79 numbers in Kyaikkhami, 29 numbers in Setse, 53 numbers in Zee-Phyu-Thaung, 19 numbers in Belugyun, 48 numbers in Paung and 19 numbers in Thaton. In almost all station, the highest sample numbers were in September and October.
Table 5. Comparison of the total number of the sample number of flatfish species in each station by bi-monthly.| Station No | Station name | Duration | Total Sampled Fish | ||
| July- Aug | Sep- Oct | Nov- Dec | |||
| 1. | Mawlamyine | 28 | 52 | 48 | 128 |
| 2. | Kyaikkhami | 18 | 38 | 23 | 79 |
| 3. | Setse | 5 | 11 | 13 | 29 |
| 4. | Zee-Phyu-Thaung | 17 | 29 | 7 | 53 |
| 5. | Belugyun island | 2 | 5 | 12 | 19 |
| 6. | Paung | 4 | 20 | 24 | 48 |
| 7. | Thaton | 1 | 5 | 13 | 19 |
Conclusion
Mon State is more adjacent to the brackish water rather than the marine areas. Therefore, it can be concluded that flatfishes are brackish water or marine species. In the present study, Cynoglossidae was the most abundant among order- Pleuronectiformes. As a result, Cynoglossidae was also one of the important exported fishes in Mon State. Moreover, Mawlamyine was the highest diversity and the most abundant as it is one major city of Myanmar and most of the fishes all around the Mon State are exported to there.
Acknowledgements
I’m deeply grateful to my dearest parents, U Kyin Aung and Daw Myint Myint San for their kind moral and financial support to reach the goal of this work.
References
- 1.K L Lapierre. (2007) Taxonomic Revision of the Genera Achiroides, Brachirus, Dexillus. and Paradicula (Pleuronectiformes: Soleidae). MSc. Thesis. Ottawa-Carleton Institute of Biology. 197pp .
- 2.Win Hla, Twin Swe, Pe Myint, Maung Myint. (2008) . Commericial fishes of Myanmar. Myanmar Fishery Products Processors and Exporters Association 1-248.
- 3.Amezcua F, Nash R D M. (2001) Distribution of the order Pleuronectiformes in relation to the sediment type in the North Irish Sea,”. , Journal of Sea Research 45, 293-301.
- 4.M R Gibson. (1998) Recent trends in abundance, recruitment, and fishing mortality for winter flounder in Narragansett Bay and Rhode Island coastal waters,” RI Division of Fish and Wildlife. Report to the Rhode Island Marine Fisheries Council. , USA
- 5.Minami T. (1981) The early life history of a flounder Limanda yokohamae,”. , Nippon Suisan Gakkaishi 47, 1411-1419.
- 6.Bruin G H P De, B C Russell, Bogusch A. (1995) FAO species identification field guide for fishery purposes. The marine fishery resources of Sri Lanka. , Rome, FAO 400, p..
- 7.Froese R, Pauly D. (2018) FishBase. World Wide Web electronic publication. http://www.Fishbase.org
- 8.Wang Shyh-bin, Chen Wen-Kwen, Shoou- Jeng Joung. (2013) Seasonal Dynamics of flatfish assemblage in coastal waters off northeastern Taiwan: with implications for shift in species dominance of the ecosystem. , Journal of Marine Science and Technology 21, 23-30.
Cited by (1)
- 1.Tripathy Prasanta Kumar, Seth Jaya Kishor, Dixit Prasanna Kumar, 2022, Geometric morphometric analysis of flatfishes of Gopalpur-on-Sea, Odisha coast, India, Proceedings of the Indian National Science Academy, 88(1), 90, 10.1007/s43538-021-00061-2


