Abstract
Objective
To compare Brief Adlerian Psychodynamic Psychotherapy (B-APP) plus venlafaxine versus venlafaxine plus usual care on pain and depressive symptomatology of depressed patients with cancer pain.
Methods
A total of 100 patients with pain and mood depression, according to DSM IV-TR, were randomized to receive treatment with B-APP plus venlafaxine (n=51) or venlafaxine plus usual care (n=49). The sample was evaluated at baseline and after 10 weeks with the Visual Analogue Scale (VAS); the Hospital Anxiety Depression Scale (HADS); the Montgomery Asberg Depression Rating Scale (MADRS); the Clinical Global Impressions (CGI); the Mini-Mental Adjustment to Cancer Scale (Mini-MAC); the European Organization for Research and Treatment of Cancer Quality-of Life Questionnaire (EORTC QLQ-C30) and the Dosage Record and Treatment Emergent Symptom scale (DOTES). Only at the endpoint was the Verona Service Satisfaction Scale (VSSS-54) also administered.
Results
A significant reduction in VAS and HADS scores was observed in both treatments, but a higher significance (p<0.01) was present only in subjects also treated with psychotherapy. A significant change was obtained in Mini-MAC scores (p<0.01) for Fighting Spirit, Fatalism, Anxious Preoccupation (p<0.01) and Avoidance items (p<0.05) only in patients treated with combined therapy. The combined group also showed more satisfaction with the treatment in their responses to the VSSS-54.
Conclusions
Brief Adlerian Psychodynamic Psychotherapy (B-APP) in combination with venlafaxine was superior to usual care and venlafaxine in improving depressive symptomatology and reducing pain.
Author Contributions
Academic Editor: Shuai Li, University of Cambridge. UK
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Andrea Bovero, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
In recent times, a growing number of epidemiological and clinical studies have shown the existence of complex relationships between cancer pain and mood disorders 1, 2. The treatment of anxiety and depression in patients with pain is therefore a must: basically, complete control of pain cannot be achieved without concomitant control of related emotional disturbances 3. Depressive mood reduces the pain threshold and increases, emotionally and cognitively, the pain perception, while chronic pain induces depression. Similar biological mechanisms underlie both pathologies: first of all, a neurotransmitter reduction, mainly concerning serotonin (5HT) and norepinephrine (NE); secondly, a cytokine imbalance, with an increase of pro-inflammatory agents and a modification of neurotrophic factors, can intervene in such mutual involvement 4.
Depression is the most prevalent psychological disorder among cancer patients and has a negative impact on quality of life and treatment adherence 5.
A meta-analysis of studies that assessed pain in patients with cancer estimated its prevalence at 53%. 6, 7.
Data suggest that the most effective treatment approaches to the treatment of depression and cancer pain include the combination of psychotherapeutic and pharmacological interventions 8, 9. Antidepressants (ADs) demonstrate important effects on mood but also on pain. Serotonin and norepinephrine reuptake inhibitors (SNRIs such as venlafaxine, duloxetine, milnacipran) showed greater efficacy on painful physical symptoms of depression 10, 11. Among SNRIs, venlafaxine shows peculiar pharmacodynamic and kinetic properties that suggest its use in oncological patients 12, 13.
Consistent evidence supports the efficacy of psychological interventions for alleviating pain related to depression when added to pharmacological treatment. Studies with varying results on the effects of Cognitive Behavioural therapy (CBT) for cancer-related emotional distress and pain have been published 14, 15, 16. Psychotherapies can act by modifying pain intensity and the pain affective component 17, 18. Conversely, only minimal data are available for psychological interventions other than Cognitive-Behavioural Therapy and, in particular, few studies have been carried out on psychodynamic psychotherapy 19, 20, 21, 22. Moreover there are not any clinical trials on Brief-Adlerian Psychodynamic Psychotherapy (B-APP) applied to oncological patients with pain.
The main object of the study was to evaluate the effects of adding a psychotherapeutic intervention, Brief Adlerian Psychodynamic Psychotherapy (B-APP) to venlafaxine (VLF) related to pain and depressive symptoms compared with usual care plus venlafaxine on depressed oncological patients with pain.
Materials and Methods
This is a randomized, controlled trial comparing B-APP plus venlafaxine versus VLF plus usual care. Randomization by a table of random number was centralized. Nine hundred and ninety five patients with cancer, of both sexes, from 18 to 75 years old, receiving ambulatory care at the Oncology Center of Citta’della Salute e della Scienza Hospital of Turin were screened for depression and pain.
Inclusion criteria were: a life expectancy ≥1 year, a Karnofsky Performance Status ≥70, a mood disorder according to DSM-IV-TR criteria, depression score (HADS-D ≥8) and concomitant relevant pain (VAS ≥5).
Exclusion criteria were: any mood disorders concomitant with pain, metastatic disease, some drugs interacting with venlafaxine such as tramadol, psychotic disorder, bipolar disorder, acute suicide risk and concurrent psychotherapy.
Of a total of 995 patients, 885 did not meet inclusion criteria, 10 did not meet SCID criteria for a depressive disorder or declined to participate. A total of 100 subjects were enrolled and randomly assigned to receive combined therapy (COM: VLF associated with B-APP) (n=51) or (VLF: venlafaxine plus usual care) (n=49).
Patients were evaluated by an expert psychiatrist of the Psycho-Oncology Unit (LA), Department of Neurosciences, University of Turin. The main baseline demographic and clinical characteristics are summarized in Table 1.
Table 1. Baseline Demographic and Clinical Characteristics| Subjects | N | 100 | ||
| Male | 45 | 45% | ||
| Sex | ||||
| Female | 55 | 55% | ||
| Mean | 54.63 | |||
| Age | Range | 23-77 | ||
| SD | 11.99 | |||
| Single | 12 | 12% | ||
| Married | 70 | 70% | ||
| Marital Status | ||||
| Divorced | 8 | 8% | ||
| Widowed | 10 | 10% | ||
| Employed | 48 | 48% | ||
| Work | Unemployed | 22 | 22% | |
| Retired | 30 | 30% | ||
| Colon-rectum | 19 | 19% | ||
| Breast | 15 | 15% | ||
| Lung | 15 | 15% | ||
| Prostate | 13 | 13% | ||
| Disease Site or Type | ||||
| Hepatic | 10 | 10% | ||
| Gastric | 10 | 10% | ||
| Dermatologic | 10 | 10% | ||
| Uterus-ovary | 8 | 8% | ||
| Local | 33 | 33% | ||
| Stage of Illness | ||||
| Loco-regional | 67 | 67% | ||
| Adjustment Disorder with Depression | 55 | 55% | ||
| Depressive Disorder not otherwise specified | 16 | 16% | ||
| Diagnosis | Major Depressive Disorder - single episode | 12 | 12% | |
| Major Depressive Disorder- recurrent episode | 5 | 5% | ||
| Dysthymia | 12 | 12% |
Besides patients had to meet at least one of the two following criteria in order to be eligible for the study: 1) the fixed-dose opioid had to be at least an equianalgesic daily dose of 60 mg of oral morphine and the patients were not expected to gain further therapeutic benefit by further increasing of the dose of the opioid therapy; 2) they were experiencing or had previously experienced intolerable side effects when increasing the dose of opioid therapy. In addition, in order to assess the antalgic add-on efficacy of venlafaxine, during the 10 weeks of this study, no change in the basic therapeutic regimen with analgesics was allowed. However, when pain breakthrough conditions were present, all analgesic adjunctive takings were recorded. The ten-week duration was decided in order to: a) verify the antidepressant response and the antalgic response to venlafaxine in the short-medium term; b) allow an adequate brief psychotherapy intervention, as stated in our previous study 22.
This study was carried out according to the “Declaration of Helsinki” (2002) and approved by the Citta’della Salute e della Scienza Hospital and University International Ethics Committee. The nature of the study and all procedures were fully explained to the patients and written informed consent to treatment was obtained from each patient.
Interventions
The drug, that is in insurance, was dispensed by the clinicians involved in the study. Venlafaxine was started at the dose of 37,5 mg/day and increased to 75 mg/day after a week. The rationale of the starting low dose is according to the need of titration in oncological patients with pain 4, 23. The choice of a fixed dose of 75 mg/day was related to a best balance between efficacy and tolerability in a group of frail patients with medium-high opioid dosages. Lorazepam 1-2 mg/day was allowed only during the first two weeks of treatment, in patients with anxiety and/or sleep disorders, and then was withdrawn.
The VLF group was subjected to a medical visit (LA) once a week for 10 weeks, in which physical parameters were evaluated and alteration of vital signs was recorded if present. The protocol included the discussion with oncologists about the effects and side effects of medication, and the provision of practical support.
The COM group carried out a B-APP with an expert psychotherapist (AB). B-APP is a time-limited psychodynamic psychotherapy (10 weekly individual sessions of 50 minutes), based on Alfred Adler’s theory of individual psychology 24. This technique has been described in a previous study 25 and has been used within a range of settings to treat various disorders 26, 27, 28 but this is the first time that it has been utilized with oncological patients. The B-APP theory refers to the following paradigms: the individual represents a psychosomatic unity integrated in a social context, the individual needs to build and regulate the image of self and bond patterns regulate human relationships, and they also constitute an unconscious ‘‘symbolic theme’’ connecting the elements of a lifestyle. Three process elements can be identified within B-APP: encouraging relationships, identifying the focus and determining areas of possible change within the focus. According to other psychodynamic treatments and to the recent literature 29 its objectives are: at least partial resolution of the focus problem; a decrease or non-increase of symptoms; a global increase of quality of life. The psychotherapist (AB) involved in this research was a licensed psychologist specifically trained in B-APP application at a certified school in Turin, Italy (SAIGA, Italian Adlerian Society Group and Analysis, certified by the Italian Ministry of University since 1994) and had personal analytic training and regular supervision with an Adlerian training supervisor (AF) to guarantee correct adherence to this psychological model.
Response to pharmacological treatment and adverse events were also reviewed.
Study Measures
Patients were assessed at baseline visit (T0), where all their demographic and clinical data were recorded and after 10 weeks (T1) at the end of the study with several rating scales. Rating scales were performed by psychologists (VI; AM) blind to the patient’s kind of therapy.
All raters were adequately trained in the use of the rating scale to ensure good internal consistency and inter-rater reliability. The day before baseline visit (T0), the Structured Clinical Interview (SCID) for DSM-IV-TR criteria was conducted 30.
The Hospital Anxiety and Depression Scale (HADS) 31: a 14-item self-report scale that provides scores on depression and anxiety, recently proposed by our group as a useful screening tool in oncological patients 32. The Montgomery Asberg Depression Rating Scale (MADRS): 33 a hetero-evaluated scale designed to be sensitive to changes in the severity of depression. The Mini-Mental Adjustment to Cancer (Mini-MAC) 34: a self–report scale that measures coping styles with cancer, such as Helplessness/Hopelessness, Anxious Preoccupation, Fighting Spirit, Avoidance and Fatalism. The Visual Analogue Scale (VAS) 35 assesses the current intensity of pain. The European Organization for Research and Treatment of Cancer – Quality of Life Questionnaire Core 30 (EORTC QLQ-C30) 36: a cancer-specific, self-administered, structured questionnaire designed for use in clinical trials. It is composed by five functional subscales (Physical, Role, Emotional, Cognitive and Social), three symptom subscales (Fatigue, Pain and Nausea), a global health item, a quality of life item and 6 items describing relevant cancer-oriented symptoms (dyspnea, insomnia, appetite, constipation, diarrhoea, financial difficulties).The Clinical Global Impression (CGI) 37: a well-known assessment tool administered by a clinician to evaluate the severity of emotional illness and to assess the changes in illness degree compared to the previous evaluation. The Dosage Record and Treatment Emergent Symptom Scale (DOTES) 37: evaluates venlafaxine side effects, assessing the severity of adverse effects of clinical treatments, their possible relation with pharmacological treatment and any measure adopted to manage these adverse effects. The Verona Service Satisfaction Scale (VSSS-54) 38: a self-report, multidimensional scale administered in order to assess patients’ satisfaction and overall perception of the clinical outcome in relation to symptomatology reduction and interpersonal improvements. In this study it was submitted only 10 weeks after the first assessment, when all the rating scales were submitted again. The scale is composed of eight areas of patient satisfaction: overall satisfaction, professional skills and behavior, symptoms, comprehension, family relationships, social relationships, working capacity and self-care.
Statistical Analysis
SPSS version 21.0 (IBM Corp. Released 2012. IBM SPSS Statistics for Windows, Version 21.0. Armonk, NY, USA) was used for analysis and an intent-to-treat approach, i.e. including all enrolled patients taking at least one dose of medication and with at least one follow-up assessment, was used for the primary end-point analysis. Cardinal variables were described by using the mean and standard deviation. The Paired-Samples t-Test was used to evaluate significant differences within each group between assessments at baseline (T0) and after 10 weeks (T1). Significant differences were calculated between COM and VLF groups by the Independent-Samples t-Test. As for ordinal variables, significant differences within each group between T0 and T1 were calculated using the Wilcoxon Signed Ranks Test; whereas the Mann Whitney U Test was used to evaluate significant differences between the COM and VLF groups.
Results
Of 995 patients consecutively evaluated in Citta’della Salute e della Scienza Hospital, 106 patients fulfilled the study selection criteria. One hundred depressed patients with cancer pain agreed to participate in this study, the others refused for various reasons (2 insufficient motivation, 3 burden of cancer, 1 other reasons). Sample demographic, psychiatric and oncological characteristics are described in Table 1. All the patients were treated with a fixed daily dose of venlafaxine 75 mg extended release (XR). All patients of the COM group attended the individual sessions of psychotherapy.
Ninety-four patients (94%) completed the study, five dropped out because of drug-related side effects (tachycardia, agitation, hypertension, headache) and one died at home because of the progression of oncological pathology.
The statistical analysis shows homogeneous demographic and clinical characteristics while the comparison between groups at T0 highlighted no significant difference on all rating scales.
Depression and Anxiety Symptoms
Mean self-evaluated HADS anxiety and depression scores decreased significantly in both treatment regimens during the 10 weeks of the study (Figure 1). In both groups a significant difference of depression and anxiety symptoms between T0 and T1 was observed. The Independent Samples t-Test nevertheless showed a more significant improvement in the COM group than in the VLF group.
Figure 1.HADS mean scores at T0 and T1 assessments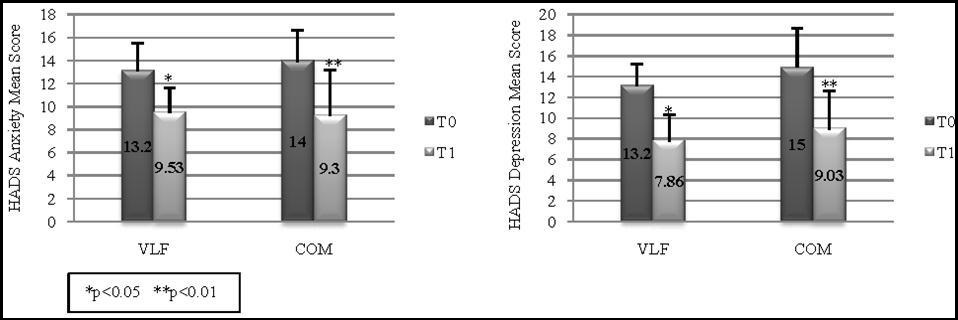
Figure 2 shows the hetero-evaluated MADRS for two groups. The Independent Samples t-Test emphasized a greater change in the COM group than in the VLF group for the Pessimistic Thoughts subitem: at T0 the data showed no significant difference, whereas at T1 a significant difference (p<0.01) between the two groups was observed.
Figure 2.MADRS mean scores pain
VAS demonstrated a significant difference between assessments in perception of the intensity of pain (Figure 3). These data are confirmed by the Independence Samples t-Test which highlighted a higher significant improvement in the COM group (p<0.01) than in the VLF group (p<0.05).
Coping Styles
The Mini-MAC showed important changes in switching coping strategies. Between T0 and T1 in the usual care group only a trend of a non-significative reduction was found, while the subjects treated with combined therapy showed significant changes in Fatalism, Fighting Spirit, Anxious Preoccupation and Avoidance (Figure 4). In addition, the Independent Samples t-Test highlighted a significant difference (p<0.05) between groups in improvement on the Avoidance subscale.
Quality of Life
Quality of Life, measured by the EORTC-QLQ-C30, also demonstrated an improvement for both groups in treatment in all functional and symptom scores. The differences between treatments were in favor of the COM group with regard to cognitive functioning, emotional functioning and global QoL (Table 2).
Table 2. EORTC QLQ-C30 Health-related quality of life scores| T0 | T1 | p-value | ||||
| VLF | COM | VLF | COM | VLF | COM | |
| FUNCTIONAL SCORES | ||||||
| Cognitive functioning | 43.33±20.69 | 63.75±18.96 | 31.25±14.21 | 45.83±13.67 | p<0.05 | p<0.01 |
| Emotional functioning | 67.29±18.18 | 78.96±17.25 | 47.71±17.48 | 58.33±16.36 | p<0.05 | p<0.01 |
| Physical functioning | 66.67±21.10 | 51.87±17.72 | 56.58±22.04 | 41.80±18.94 | p>0.05 | p>0.05 |
| Role functioning | 58.58±23.18 | 38.04±15.66 | 55.41±27.74 | 32.58±8.80 | p>0.05 | p>0.05 |
| Social functioning | 45.25±23.72 | 57.92±15.57 | 38.42±23.07 | 48.33±14.40 | p>0.05 | p>0.05 |
| SYMPTOM SCORES | ||||||
| Nausea and vomiting | 54.08±19.37 | 38.18±17.88 | 31.67±11.24 | 34.17±11.34 | p<0.01 | p>0.05 |
| Pain | 58.73±20.78 | 58.73±22.15 | 47.92±14.71 | 40.75±113.21 | p<0.05 | p<0.05 |
| Appetite loss | 51.67±27.80 | 58.33±22.10 | 32.50±13.37 | 39.17±15.65 | p<0.01 | p<0.01 |
| Constipation | 57.29±23.22 | 35.83±21.46 | 38.95±15.89 | 33.33±16.52 | p<0.01 | p>0.05 |
| Diarrhea | 28.33±10.85 | 31.67±13.02 | 26.00±9.95 | 29.17±14.80 | p>0.05 | p>0.05 |
| Dyspnoea | 31.50±16.09 | 59.67±25.29 | 29.17±11.53 | 40.80±13.94 | p>0.05 | p<0.01 |
| Fatigue | 68.79±14.10 | 66.57±13.63 | 54.08±19.37 | 45.83±13.67 | p<0.01 | p<0.01 |
| Insomnia | 53.33±18.26 | 74.17±17.96 | 37.33±15.91 | 47.50±16.54 | p<0.01 | p<0.01 |
| Financial Difficulties | 32.50±18.74 | 50.00±18.57 | 29.17±14.80 | 45.00±13.77 | p>0.05 | p>0.05 |
| QUALITY OF LIFE SCORES | ||||||
| Global physical condition | 55.04±15.36 | 50.91±15.72 | 67.88±13.61 | 64.11±11.65 | p<0.01 | p<0.01 |
| Global quality | 51.45±17.74 | 57.47±20.70 | 64.09±15.28 | 70.78±12.54 | p<0.05 | p<0.01 |
Patient Satisfaction
At the end of the treatments, the total VSSS score, which quantifies the patients’ satisfaction, was 22.58±2.38 for the usual care group and 28.91±2.61 for the combined group. Patients treated with combined therapy referred a statistically higher satisfaction in the subscales concerning prevention, symptoms, comprehension of their problems, interpersonal relationships and in relation to working capacity and self-care. The Independent Samples t-Test highlighted significant differences between the two groups in the subscales concerning prevention, interpersonal relationships, working capacity and self-care (p<0.01) in the COM group.
Efficacy and Safety Balance
First of all the Mann-Whitney U Test showed no differences at baseline between the two groups. The CGI was used to give a global judgment on the severity of psychological illness. The Wilcoxon Signed Ranks Test emphasized a significant improvement of severity of illness in both groups (p<0.05 for the VLF group; p<0.01 for the COM group).
The Mann-Whitney U Test highlighted a significant difference between the two groups (p<0.05), with the COM group making a larger improvement at T1. Side effects of the treatment were evaluated with the Dosage Record and Treatment Emergent Symptom scale (DOTES). Fifty-four patients completed the study, five dropped out (4 in VLF and 1 in COM) because of side effects: agitation (2 patients), tachycardia, hypertension, headache (1 patient for each side effect) and one died at home because of the progression of oncological pathology. Finally, direct confirmation of the good balance between the efficacy and safety of venlafaxine in our oncological patients is given by the CGI efficacy index, which graphically evaluates the therapeutic response (in ordinate) and the adverse events (in abscissa): 80% of patients treated with venlafaxine alone and 93% of those treated with combined therapy are located on the left side of the graph, showing a good balance between the efficacy and tolerability of the psychopharmacological treatment.
Discussion
This study represents the first randomized, controlled clinical trial which assesses the effectiveness of B-APP plus venlafaxine compared with venlafaxine related to pain and depressive symptoms among Italian patients with non-metastatic cancer who met depression diagnostic criteria according to DSM-IV-TR. In addition, this study represents the first one in which B-APP for cancer patients has been used.
The first important outcome measure of the study was the reduction of pain perception. At the end of the study, cancer pain perception in the sample decreased and the intensity of pain is <5 at VAS but the COM group showed a more significant improvement (p<0.01) than the VLF group (p<0.05). Our study showed that the efficacy of pharmacotherapy on depressed oncological patients with pain, should be positively reinforced by brief psychotherapy, according to previous data 39, 40, 41. Psychological intervention, together with pharmacological treatment, modified patients’ interpretations, expectations and meaning of oncological pain, reducing pain perception and improving their quality of life 42 more than pharmacological treatment alone. Besides an essential component needed for efficacy of a psychotherapy for pain reduction seems to include enhancement of self-efficacy or confidence in one’s ability to manage pain 43, 44.
Mood improvement was demonstrated by a significant reduction, from baseline to the 10 weeks endpoint, in the self-rated Hospital Anxiety and Depression Scale (HADS) in both groups, although it was more significant in the COM group (p<0.01) than the VLF group (p<0.05). According with previous studies, we found that cancer patients with higher level of depression and pain showed a worst quality of lifeand also that the combined intervention could preserve a better sense of well-being and could support participants appraising positively their stressful and threatening disease9, 45, 46. B-APP associated with venlafaxine could allow patients to explore their feelings, needs and to interpret their experience with a supportive and facilitating therapist. In fact, the theraphist focuses on the integration and interpretation of the material brought by the patient, in order to facilitate him/her insight into its origin and bring about emotional and cognitive changes.
Furthermore these data could support the relation between emotional disorders and physical symptoms, because patients with these disorders were more likely to report moderate to mild degree of global suffering 47. In fact, chronic pain, is often related to depression and depression in itself should induce a reduction of the pain threshold, with an amplification of painful symptomatology and a worsening quality of life 48. These findings support the importance of screening for, monitoring and treating both pain and depressive symptoms concurrently in patients with cancer.
The hetero-evaluated Montgomery Asberg Depression Rating Scale (MADRS) total scale confirmed a significant statistical reduction in both groups, again more significant in the COM group (p<0.01) than the VLF group (p<0.05). So venlafaxine shows efficacy not only with somatic but also with emotional and cognitive symptoms, as confirmed by MADRS scores, as shown in Figure 2, according to Hotopf et al. (2002) 49. Our data demonstrated that the association of psychotherapy with antidepressants increases the effectiveness of venlafaxine on several depressive clusters, as also observed by De Maat’s study (2008) 50. In fact the statistical analysis emphasized a greater reduction of Pessimistic Thoughts subitem in the COM group than in the VLF group, suggesting that the supportive role of psychotherapy strengthened the therapeutic action of the antidepressant.
A further interesting finding of this study is that psychological treatment was associated with significant changes in measures of coping strategies 51. Greater improvements were found in some subscales of the Mini-MAC in patients treated with venlafaxine combined with B-APP: particularly an increase of Fighting Spirit and a decrease of Fatalism, Anxious Preoccupation and Avoidance coping styles. In this context, brief psychotherapy is used to improve adaptive coping styles and to maintain adherence with the therapeutic program of oncological patients with pain: when depression and pain are comorbid, the patients’ adherence with treatments was reduced 52.
In this study, patients’ quality of life, evaluated with EORTC-QLQ-C30, improved over time in both groups, together with a significant reduction in scores for pain and depression symptoms. These improvements were observed both with physical symptoms and functional scales. The COM group, particularly, showed a higher significant response in emotional and cognitive functioning, global physical condition and global quality of life. These results support the notion that combined treatment compared with usual care improves significantly patient’s quality of life 9.
These findings suggest that patients with an higher level of a sense of well-being, were more capable of confronting the reality of the situation and this implied that also most of the participants attempted to develop effective coping strategies in order to maintain their better psychosocial well-being 53. Analyzing the VSSS-54, the subjects were satisfied with the treatment they had received as long as the intervention was effective. In particular, it was observed that brief psychotherapy, when associated with venlafaxine, showed greater improvements in prevention (p<0.01), social relations (p<0.01), working capacity (p<0.01) and self-care (p<0.01). In fact combined therapy, allows to confront the impact of pain on patients’social and existential concerns and to listen the needs of the patients, may be considered very relevant in determining their quality of life 54.
Conclusions
This study provides data supporting the effectiveness of B-APP used in combination with venlafaxine in reducing cancer pain and depression.
This study presents several limitations: the small number of patients recruited, may reflect the difficulties in recruiting patients with strict criteria, the use of an open-label design was caused by deontological aspects in patients with persistent pain. The use of restricted and limited Statistical tools could be another limitation 55, 56, 57. The use of a non-manualised type of psychotherapy (B-APP) could be a limitation, even though some studies have demonstrated that non-manualised treatments are as effective as manualised therapies. The final limitation is that our analysis was restricted to the short-term effects of time-limited treatments. Further controlled trials on the synergistic activity of psychotherapies and pharmacotherapies in oncological patients are needed.
Conflict of Interests
This is a spontaneous study without pharmaceutical grant sponsorisation. The authors of this manuscript have no financial or other relationships that might lead to a conflict of interests.
References
- 1.L S Porter, F J Keefe. (2011) Psychosocial issues in cancer pain. , Current Pain Headache Reports 15, 263-270.
- 2.Teunissen S C C M, A De Graeff, Voest E E. (2007) Are anxiety and depressed mood related to physical symptom burden? A study in hospitalized advanced cancer patients. , Palliat Med 21(4), 341-346.
- 3.Reich M. (2008) Depression and cancer: recent data on clinical issues, research challenges and treatment approaches. , Curr Opin in Oncol 20(4), 353-359.
- 4.Torta R G V, Munari J. (2010) Symptom Cluster: depression and pain. Surgical Oncology. 19(3), 155-157.
- 5.Li M, Fitgerald P, Rodin G. (2012) Evidence-based treatment of depression in patients with cancer. , J Clin Oncol 30(8), 1187-1196.
- 6.Mitchell A, Chan M, Bhatti H, Halton M, Grassi L. (2011) Prevalence of depression, anxiety, and adjustment disorder in oncological, hematological, and palliative care settings: a meta-analysis of 94 interview based studies. , Lancet Oncol 12, 160-174.
- 7.VandenBeukenvanEverdingen M, Deriijke K. (2012) Chronic pain in cancer survivors: a growing issue. , J Pain Palliat Care Pharmacother 26, 385-387.
- 8.I D Glik. (2004) Adding psychotherapy to pharmacotherapy data, benefits and guidelines for integration. , Am J Psychother 58(2), 186-208.
- 9.Rodriguez Vega BR, Palao A, Torres G, Benito G, Pérez E. (2011) Combined therapy versus usual care for the treatment of depression in oncologic patients: a randomized controlled trial. , Psycho-Oncology 20, 943-952.
- 10.R M Hirschfeld, S A Montgomery, Aguglia E, P L Delgado, Gastpar M. (2002) Partial response and nonresponse to antidepressant therapy: current approaches and treatment options. , J Clin Psychiatry 63(9), 826-837.
- 11.Esin E, Yalcin S. (2014) Neurophatic cancer pain: what were dealing with? How to manage it? Onco Targets Ther. 7, 599-612.
- 12.Eardley W, Toth C. (2010) An open label, non-randomized comparison of venlafaxine and gabapentin as monotherapy or adjuvant therapy in the management of neuropathic pain in patients with pheripheral neuropathy. , J Pain Res 3, 33-49.
- 13.Amr Mohamed, Y, Ayman Abd Al-Maksoud, Y. (2010) Evaluation of efficacy of the perioperative administration of venlafaxine or gabapentin on acute and postmastectomy pain. , Clin J Pain 26, 381-385.
- 14.Tatrow K, G H Montgomery. (2006) Cognitive Behavioral Therapy Techniques for Distress and pain in breast cancer patients: a meta-analysis. , J Behav Med 29(1), 17-27.
- 15.Sheinfeld Gorin S, Krebs P, Badr H, Janke E A, Jim H S. (2012) Meta-analysis of psychosocial interventions to reduce pain in patients with cancer. , J Clin Oncol 30, 539-547.
- 16.Syrjala K L, Jensen M P, Mendoza E, Yi J C, Fisher H M. (2014) Psychosocial and behavioral approaches to cancer pain management. , J Clin Oncol 32, 1703-1711.
- 17.E A Mundy, Du Hamel KN, Montgomery G H. (2003) The efficacy of behavioural interventions for cancer treatment related side effects. , Semin Clin Neuropsychiatry 8(4), 253-275.
- 18.Rodin G, Lloyd N, Katz M, Green E, J A Mackay. (2007) Supportive Care Guidelines Group of Cancer Care Ontario Program in Evidence Based Care. The treatment of depression in cancer patients: a systematic review. Support Care in Cancer. 15(2), 123-136.
- 19.M E Beutel, Weibflog G, Leuteritz K, Haselbacher A, Ruckes C. (2014) Efficacy of short-term psychodynamic psychotherapy (STPP) with depressed breast cancer patients: results of a randomized controlled multicenter trial. , Ann Oncol 25, 378-384.
- 20.Ludwig G, Krenz S, Zdrojewski C, Bot M, Rousselle I. (2014) Psychodynamic interventions in cancer care II: psychometric results of a randomized control trial. , Psycho-Oncology 23, 65-74.
- 21.Zwerenz R, Beutel M, Imruck I B, Wiltink J, Haselbacher A. (2012) Efficacy of psychodynamic short –term psychotherapy for depressed cancer patients: study protocol for a randomized controlled trial. doi: 10.1186/1471-2407-12-578.BMC. , Cancer 12, 57.
- 22.Bovero A, Ferrero A, Torta R. (2006) A new approach on oncological pain in depressed patients: data from a clinical study using Brief-Adlerian Psychodynamic Psychotherapy. , Psycho-Oncology. 15 (Suppl 1, 182-183.
- 23.Torta R, Leombruni P, Borio R, Castelli L. (2011) Duloxetine for the treatment of mood disorder in cancer patients: a 12- week case-control clinical trial. , Hum Psychopharmacol 26, 291-299.
- 25.Fassino S, Amianto F, Ferrero A. (2008) Brief-Adlerian Psychodynamic Psychotherapy: theoretical issues and process indicators. , Panminerva Med 50(2), 165-175.
- 26.Ferrero A, Simonelli B, Simona Fassina S, Cairo E, Abbate-Daga G. (2016) Psychopathological Functioning Levels (PFLs) and their possible relevance in psychiatric treatments: a qualitative research project. doi: 10.1186/s12888-016-0940-4. BMC Psychiatry;6: 253
- 27.Ferrero A, Pierò A, Fassina S, Massola T, Lanteri A. (2007) A 12-month comparison of brief psychodynamic psychotherapy and pharmacotherapy treatment in subjects with generalized anxiety disorders in a community setting. , Eur Psychiatry J 22(8), 530-539.
- 28.Ferrero A. (2012) The Model of Sequential Brief-Adlerian Psychodynamic Psychotherapy (SB-APP): Specific Features in the Treatment of Borderline Personality Disorder. Res Psychother. 15(1), 32-45.
- 29.M J Hilsenroth, E J Peters, S J Ackerman. (2004) The development of therapeutic alliance during psychological assessment: patient and therapist perspectives across treatment. , J Pers Assess 83(3), 332-344.
- 30.M B First, R L Spitzer, Gibbson M, Janet W. (2002) Structured Clinical Interview for DSM-IV-TR Disorders, Clinician Version (SCID-CV).Washington, DC:AmericanPsychiatryPress.
- 31.A S Zigmond, R P Snaith. (1983) The Hospital Anxiety and Depression Scale. Acta Psychiatr Scand. 67(6), 361-370.
- 32.Castelli L, Binaschi L, Caldera P, Mussa A, Torta R. (2010) Fast screening of depression in cancer patients: the effectiveness of the HADS. , Eur J Cancer Care 20(4), 528-533.
- 33.S A Montgomery, Asberg M. (1979) A new depression scale designed to be sensitive to change. , Br J Psychiatry 134, 328-389.
- 36.B K Aaronson, Ahmedazai S, Bullinger M.for EORTC Study Group on Quality of Life(1991)The EORTC core quality of life questionnaire:interim results of an international field study.In D Osoba(Ed.),Effect of cancer on quality of life.CRC Press Inc:Boca. , Raton
- 37.Guy W. (1976) Clinical Global Impression (C.G.I.) In. ECDEU, Assessment Manual for Psychopharmacology US Dept Health, Education and Welfare publication (ADM): National Institute of Mental Health , Rockville 218-222.
- 38.Ruggeri M, Dell’Agnola R. (1993) The development and use of the Verona Expectation for Care Scale (VECS) and the Verona Service Satisfaction Scale (VSSS) for measuring expectations and satisfaction with community-based psychiatric services in patients, relatives and professionals. , Psychological Medicine 23(2), 511-523.
- 39.Bovero A, Torta R, Varetto A. (2005) Depressione e dolore cronico. Valutazione del trattamento integrato psicoterapeutico-psicofarmacologico nel dolore oncologico versus il trattamento psicofarmacologico. Proceedings of XII Congresso Nazionale della Società Italiana di Cure Palliative, Avril19-22,Firenze .
- 40.Rodin G, Katz M, Lloyd N, Green E, J A Mackay. (2007) Treatment of depression in cancer patients. Current Oncology. 14(5), 180-188.
- 41.Institute ofMedicine: relieving pain in America: a blueprint for transforming prevention, care, education, and research.Http://www.iom.edu/reports/2011/Relieving painin America a blueprint for transforming prevention care education research. aspx.
- 42.Arntz A, Claassens I. (2004) The meaning of pain influences its experienced intensity.Pain;109(1-2): 20. 25.
- 43.Johannsen M, Farver I, Beck N, Zachariae R. (2013) The efficacy of psychosocial intervention for pain in breast cancer and survivors: a systematic review and meta-analysis. Breast Canc Res Treat. 138, 675-690.
- 44.Marie N, Luckett T, P M Davidson, Lovell M, Lai S. (2013) Optimal patients education for cancer pain: a systematic review and theory- based-based meta-analysis. , Support Care Cancer 21, 3529-3537.
- 45.Black B, Herr K, Fine P, Sanders S, Tang X. (2011) The relationship among pain, nonpain symptoms and quality of life measures in older adults with cancer receiving hospice care. , Pain Medicine 12(6), 880-889.
- 46.L F Brown, Kroenke K, D E Theobald. (2010) The association of depression and anxiety with health-related quality of life in cancer patients with depression and/or pain. , Psycho-Oncology 19(7), 734-741.
- 47.Ieraci V, Bovero A, Pennazio F, Torta R. (2014) Pain, depression and coping styles: assessment and evaluation in cancer pain population. doi: 10.4172/2167-0846.S2-004. J Pain Relief;S2: 004
- 48.H L Wang, Kroenke K, Wu J, Tu W, Theobald D. (2013) Predictors of cancer related pain improvement over time. , Psychosom Med 74, 642-647.
- 49.Hotopf M, Chidgey J, Addington-Hall J, K L Ly. (2002) Depression in advanced disease: a systematic review Part 1. Prevalence and case finding. , Palliat Med 16(2), 81-97.
- 50.S De Maat, Dekker J, Schoevers R, G van Aalst, C Gijsbers-van Wijk. (2008) Short psychodynamic supportive psychotherapy, antidepressants, and their combination in the treatment of major depression: a mega-analysis based on three randomized clinical trials. Depress and Anxiety. 25(7), 565-574.
- 51.Reuter K, Classen C C, J A Roscoe, G R Morrow, Kirshner J. (2006) Association of coping style, pain, age and depression with fatigue in women with primary breast cancer. , Psycho-Oncology 15(9), 772-779.
- 52.Krenz S, Godel C, Stagno C, Stiefel F, Ludwig G. (2014) Psychodynamic interventions in cancer care II: a qualitative analysis of the therapists’ reports. , Psycho-Oncology 23, 75-80.
- 53.Bovero A, Leombruni P, Miniotti M, Torta R. (2012) Religiosity, pain and depression in advanced cancer patients. , WCPRR 8(1), 51-59.
- 54.M S Walker, D M Zona, E B Fisher. (2006) Depressive symptoms after lung cancer surgery: their relation to coping style and social support. , Psycho-Oncology 15, 684-693.
- 55.Kar P, Li S, Narasimhan H. (2016) . Online Optimization Methods for the Quantification Problem. The 22nd ACM SIGKDD 1625-1634.


