Autism Spectrum Disorders and Gluten/Casein Free Diet Treatment: A Systematic Review (1990-2016)
Abstract
Background:
High number of patients suffering from autism spectrum disorders utilize dietary intervention methods, especially gluten-free/casein-free diet. In contrast with its’ extensive usage no sufficient and consistent data exist to support its’ efficiency and safety.
Objective:
The main aim of this systematic review is to provide a general look to the efficiency and safety of gluten-free/casein-free diet treatment for autism spectrum disorders.
Method:
Studies used in this systematic review are gathered from 3 online databases (PubMed, Embase and the Cochrane Library). Inclusion criteria are established for study selection. Articles published in a peer-reviewed article between 1990 and 2016 in English about our topic of interest and conducted with patients under the age of eighteen (18) are selected and further analyzed (“Level of Evidence” and “Grade of Recommendation” criteria are utilized).
Results:
Even though some studies with high values of “Level of Evidence” claim that gluten-free/casein-free diet is beneficial for patients suffering from autism spectrum disorders, studies with lower risk of bias demonstrate otherwise. Since studies such as case reports and cohort studies may contain bias associated with small sample size, absence of clear assessment methods, lack of randomization and short treatment period we suggest that there is no sufficient data to support gluten-free/casein-free diet treatment.
Conclusion:
Due to controversial outcomes from studies that have different “Level of Evidence” we speculate that efficiency of gluten-free/casein-free diet might be related to individual genetic differences. Therefore, studies conducted with large study groups could not provide statistically significant data to support this treatment option. Further studies should be conducted, especially in the field of genetics, in order to test our hypothesis.
Author Contributions
Academic Editor: Chung-Yi Chen, Fooyin University
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Mazlum Çopur, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Autism spectrum disorder (ASD) is a class of neurodevelopmental disorder that is characterized by impairment in social behaviors and non-verbal communicative skills and troubles in the perception and maintenance of social relationship according to the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5)1. Autism spectrum disorder includes autistic disorder, Asperger's disorder, childhood disintegrative disorder, and pervasive developmental disorder all of which were previously considered to be distinct diseases 1, 2. Although there is not a consensus on the definitive underlying cause of ASD, it has tight association with genetics, nutritional balances and pre- and perinatal disruptions. Studies have demonstrated that experiencing oxygen deprivation during birth process, being prematurely born and experiencing viral infections during pregnancy increase the risk of autism spectrum disorders. On average 0,6% of the population suffers from autism spectrum disorders which is independent from ethnic and cultural groups, however, prevalence of ASD is 3 times higher in males compared to females. One controversial explanation for the marked prevalence difference between genders is that prevalence of ASD is undervalued by physicians due to discrepancy in symptoms among genders. Patients suffering from ASD can have a wide range of intelligence test scores and even in rare instances they can have extra-ordinary talents is specific fields 2, 3
Diet treatment is a popular complementary treatment approach for ASD which includes gluten-free and casein-free diet (GFCF), increased omega 3 fatty acid intake and increased vitamin and mineral intake. GFCF diet is based on the opioid-excess theory that suggests gluteomorphine which is formed due to the metabolism of gluten and casomorphine that is formed due to the metabolism of casein can act as opiate-like chemicals and mimic their effect by binding to the specific receptors in central nervous system. By binding to those receptors casomorphine and gluteomorphine can enhance the activity of endogenous opioid system which is associated with the symptoms of ASD4
Omega 3 fatty acids are poly-unsaturated fatty acids some of which are essential fatty acids for human metabolism such as alpha-linolenic acid. Studies indicate that some of the fatty acids such as docosahexaenoic acid are necessary for proper development of the central nervous system and proven to have anti-inflammatory effects.Three out of four study conducted about the relationship between ASD and omega 3 fatty acids demonstrate that ASD patients have low levels of omega 3 fatty acids in blood compared to the controls. On the other hand, according to the systematic review conducted by Bent and Bertoglio there is not sufficient experimental evidence to support this treatment option even though some small studies claim otherwise 5
In the case of increased vitamin and mineral intake diet there are studies indicating the significance of well-balanced diet in the treatment of ASD 6, 7. In a study conducted by Dr. Bernard Rimland in 1978 75% of the ASD patients (12 out of 16) had showed improvement in autistic symptoms when they had been treated with B6, C and magnesium supplements. 6 Moreover, in another study conducted with 60 autistic children symptoms of autism diminished when they had been treated with vitamin B6 and magnesium supplements 7. Increased vitamin and mineral intake diet and increased omega 3 fatty acid intake diet will not be further discussed in the context of this systematic review.
According to a survey conducted in UK 29% of parents whose children suffer from ASD utilize GFCF diets that demonstrates the further need to conduct more comprehensive studies in this field 8 . In the context of this systematic review efficiency and safety of gluten-free/casein-free diet in the treatment of ASD will be discussed.
Materials and Method
Literature Search
We have searched our topic of interest via electronic databases (PubMed, Embase and the Cochrane Library) in August 2016 by utilizing Medical Subject Heading terms which are listed as follows: “autism”, “autism spectrum disorders”, “diet”, “gluten-free”, “casein-free”, “gluten”, “casein”, “GFCF” and their combinations. Afterwards, references of the selected articles and systematic reviews were searched by two authors in order to eliminate the risk of overlooking (Study selection process can be viewed from Figure 1).
Figure 1.Flow chart of the process of study selection for the systematic review (Software used to design this flow chart is available from: https://www.draw.io/)
Study Selection
Abstracts of each study have been examined by two authors independently in order to determine their eligibility according to the specific inclusion criteria. The articles that do not fit the inclusion criteria are eliminated and the others are further examined for the purpose of this systematic review. Inclusion criteria for article selection are as follows:
1. The study should be conducted with patients that are diagnosed with ASD and treated with gluten-free and/or casein free diet
2. The article should be published in a peer-reviewed journal between 1990 and 2016 in English
Participants of the studies should be under the age of 18.
Selected studies for the purpose of this systematic review can be seen from Table 1.
Table 1. Journals in which the selected studies were published and basic numerical data about the studies.| Article | Level of Evidence | Number of participants | Journal |
|---|---|---|---|
| Reichelt et al, 1990 11 | 4 | 15 | Applied Physiology, Nutrition and Metabolism |
| Knivsberg et al, 1990 12 | 4 | 15 | Brain Dysfunct |
| Sponheim , 1991 13 | 2 | 3 | Journal of Norwegian Medical Association |
| Lucarelli et al, 1995 14 | 4 | 30 | Panminerva Medica |
| Knivsberg et al, 1995 15 | 4 | Continium of Knivsberg et al, 1990 | Scandinavian Journal of Educational Research |
| Whiteley et al, 1999 16 | 4 | 22 | Autism |
| Knivsberg et al, 1999 17 | 6 | 1 | Nutritional Neuroscience |
| Cade et al, 2000 18 | 4 | 150 | Nutritional Neuroscience |
| Knivsberg et al, 2002 19 | 3 | 20 | Nutritional Neuroscience |
| Millward et al, 2004 20 | 1 | Systematic review | The Cochrane Database Of Systematic Reviews |
| Elder et al, 2006 21 | 2 | 15 | Journal of Autism and Developmental Disorders |
| Irvin, 2006 22 | 6 | 1 | Journal of Positive Behavior Interventions |
| Christison and Ivany , 2006 23 | 1 | Systematic review | Journal of Developmental and Behavioral Pediatrics |
| Mageshwari US, Minitha SJ, 2006 24 | 5 | 15 | Indian Journal of Nutrition |
| Patel and Curtis, 2007 25 | 5 | 10 | The Journal of Alternative and Complementary Medicine |
| Seung et al, 2007 26 | 2 | 13 | The Journal of Medical Speech-Language |
| Millward et al, 2008 27 | 1 | Systematic review | The Cochrane Database Of Systematic Reviews |
| Nazni et al, 2008 28 | 5 | 50 | Iranian Journal of Pediatrics |
| Hsu et al, 2009 29 | 6 | 1 | Chang Gung Medical Journal |
| Hyman et al, 2010 30 | 3 | 14 | The Brown University Child and Adolescent Behavior Letter |
| Mulloy et al, 2010 31 | 1 | Systematic review | Research in Autism Spectrum Disorders |
| Whiteley et al, 2010 32 | 2 | 72 | Nutritional Neuroscience |
| Mulloy et al, 2011 33 | 1 | Systematic review | Research in Autism Spectrum Disorders |
| Buie , 2013 34 | 1 | Systematic review | Clinical Therapeutics |
| Johnson et al, 2011 35 | 2 | 22 | The Journal of Developmental and Physical Disabilities |
| Pennesi, 2012 36 | 6 | 387 parents | Nutritional Neuroscience |
| Harris and Card, 2012 37 | 6 | 13 | Complementary Therapies in Medicine |
| Genuis et al, 2010 38 | 6 | 1 | Journal of Child Neurology |
| Hurwitz et al, 2013 39 | 1 | Systematic review | Journal of Early Intervention |
| Herbert and Buckley, 2013 40 | 6 | 1 | The Journal of Child Neurology |
| Mar ı ´- Bauset et al, 2014 41 | 1 | Systematic review | Journal of Child Neurology |
| Elder et al, 2015 42 | 1 | Systematic review | Nutrition and Dietary Supplements |
| Hyman et al, 2016 43 | 2 | 14 | Journal of Autism and Developmental Disorders |
Quality Assessment
After assessing the eligibility of studies to the inclusion criteria two authors have independently assessed the quality of each study according to the criteria established by Melnyk9 in the book entitled "Evidence-based practice in nursing and healthcare: a guide to best practice" (Brief explanation of the criteria used in this step can be seen from Table 2). quality assessment can be seen from Table 3).
Table 2. Quality Assessment Criteria for articles in this systematic review 9| Level of Evidence | Description |
|---|---|
| 1 | Evidence from a systematic review of all relevant randomized controlled trials (RCT's), or evidence-based clinical practice guidelines based on systematic reviews of RCT's |
| 2 | Evidence obtained from at least one well-designed Randomized Controlled Trial (RCT) |
| 3 | Evidence obtained from well-designed controlled trials without randomization, quasi-experimental |
| 4 | Evidence from well-designed case-control and cohort studies |
| 5 | Evidence from systematic reviews of descriptive and qualitative studies |
| 6 | Evidence from a single descriptive or qualitative study |
| 7 | Evidence from the opinion of authorities and/or reports of expert committees |
("Evidence-based practice in nursing and healthcare: a guide to best practice" by Bernadette M. Melnyk and Ellen Fineout-Overholt. 2005, page 10.)
Table 3. Grade of Recommendation Criteria for quality assessment performed in this systematic review 10| Grade of Recommendation | Explanation |
| A | consistent level 1 studies |
| B | consistent level 2 or 3 studies or extrapolations from level 1 studies |
| C | level 4 studies or extrapolations from level 2 or 3 studies |
| D | level 5 evidence or troublingly inconsistent or inconclusive studies of any level |
(Available from: http://www.cebm.net/oxford-centre-evidence-based-medicine-levels-evidence-march-2009/) 10
In addition to “Level of Evidence” and “Grade of Recommendation” we also evaluate and assess each study individually depending on the criteria as listed above:
Presence of control group in the study
Methods utilized to assess the conditions of participants involved in studies (parental reports, direct observations, standardized questions, blood or urine tests etc.)
Homogeneity of the participants involved in each study (age, gender, ethnicity, other medical conditions etc.)
Size of the study group
Consistency of the data extracted in each study (absence of statistically significant deviations)
Table 4. Plot summary of the selected studies.| Article | Level of Evidence | Grade of Recommendation | Assessment | Results | Duration | Number of Participants |
|---|---|---|---|---|---|---|
| Millward et al, 2008 | 1 | A | Systematic review | No sufficient data to recommend GFCF diet treatment | - | - |
| Millward et al, 2004 | 1 | A | Systematic review | There is not enough empirical data to support GFCF diet in the treatment of ASD. | - | - |
| Christison and Ivany , 2006 | 1 | B | Systematic review | Inadequate evidence to support GFCF diet in the treatment of ASD | - | - |
| Mulloy et al, 2010 | 1 | A | Systematic review | No sufficient data to recommend GFCF diet treatment | - | - |
| Mulloy et al, 2011 | 1 | A | Systematic review | Significant improvements in some ASD subgroups whereas no significant improvement in others | - | - |
| Buie , 2013 | 1 | A | Systematic review | Although there are small subgroups in which there is a correlation between gluten and ASD, there is not sufficent information to confirm this theory | - | - |
| Hurwitz et al, 2013 | 1 | A | Systematic review | No significant improvement and high family investment | - | - |
| Mar ı ´- Bauset et al, 2014 | 1 | A | Systematic review | No sufficient data to recommend GFCF diet treatment | - | - |
| Elder et al, 2015 | 1 | A | Systematic review | No sufficient data to recommend GFCF diet treatment | - | - |
| Sponheim , 1991 | 2 | B | Visual Analogue Scale and Real Life Rating Scale | No significant improvement | 6 months | 7 |
| Elder et al, 2006 | 2 | B | Urinary peptide levels and standardized assessment questions | No significant improvement | 6 weeks | 15 |
| Seung et al, 2007 | 2 | B | Analysis of video recordings and verbal and non-verbal communication assessment | No significant improvement | 6 weeks | 13 |
| Whiteley et al, 2010 | 2 | B | Urinary peptide levels and standardized assessment questions | Significant improvements in some ASD subgroups whereas no significant improvement in others | 2 years | 72 |
| Hyman et al, 2016 | 2 | B | Behaviour assessment, autism symptoms | No statistically significant differences were noted between treatment groups. | 4-6 weeks | 14 |
| Hyman et al, 2010 | 2 | C | Direct observations and video recording | No significant improvement in autistic symptoms | 12 weeks | 14 |
| Knivsberg et al, 2002 | 3 | C | Behaviour assessment, autism symptoms (additional statistical tests) | Significant improvement in the diet group even though small improvements are observed also in the control group | 1 year | 20 |
| Johnson al, 2011 | 3 | C | Child Behavior Checklist and direct behavioral assessment | No statistically significant differences were noted between treatment groups. | 3 months | 22 |
| Article | Level of Evidence | Grade of Recommendation | Assessment | Results | Duration | Number of Participants |
| Reichelt et al, 1990 | 4 | C | Peptiduria test and retrorespective comparison | Improvement in patients treated with GF diet over 50%. | 3 months | 15 |
| Knivsberg et al, 1990 | 4 | C | Urinary peptide levels and standardized assessment questions | High levels of improvement in the GFCF diet group compared to the control group | 1 year | 15 |
| Lucarelli et al, 1995 | 4 | C | immunological signs of food allergy, levels of IgA antigen specific antibodies | Improvement in behaviour and decrease in spesific IgA suggesting relationship between casein and autism. | 2 months | 36 |
| Knivsberg et al, 1995 | 4 | C | Urinary peptide levels and standardized assessment questions | Significant improvement in behavior and social skills | 1 year | Continium of Knivsberg et al, 1990 |
| Whiteley et al, 1999 | 4 | C | Urinary peptide levels and standardized assessment questions | Significant improvement in behavior and social skills | 5 months | 22 |
| Cade et al, 2000 | 4 | C | Behavioral assessment and urinary peptide levels | Decrease in autism symptoms | 3 months | 150 |
| Mageshwari US, Minitha SJ, 2006 | 5 | D | Direct observation | Improvements in autistic symptoms | 3 months | 15 |
| Patel and Curtis, 2007 | 5 | D | Behavioral assessment and urinary heavy metal levels | Behavioral improvement and lower levels of heavy metal in urine | 3-6 months | 10 |
| Nazni et al, 2008 | 5 | D | Day-to-day direct observation | GFCF diet is beneficial for treating behavioral symptoms of ASD | 2 months | 50 |
| Knivsberg et al, 1999 | 6 | D | Standardized assessment questions | Improvement in social skills | 2 year | 1 |
| Irvin, 2006 | 6 | D | Direct observation | No significant behavioral improvement | 4 days | 1 |
| Pennesi, 2012 | 6 | D | Parent reports and standardized assessment questions | Significant improvement in behaviour and social skills | - | 387 parents |
| Harris and Card, 2012 | 6 | D | Parental reports and standardized assessment questions | No statistically significant association between ASD and GFCF diet | - | |
| 13 | ||||||
| Herbert and Buckley, 2013 | 6 | D | Childhood Autism Rating Scale, direct observation | Improvement in language, auditory sensitivity and autistic symptoms | 14 months | 1 |
| Hsu et al, 2009 | 6 | D | Direct observation | Improvement in social interactions, eye contact and autistic symptoms | 2.5 months | 1 |
| Genuis , 2010 | 6 | D | Parent reports, direct observations and standardized assessment questions | Significant improvement in symptoms and resolution of gastrointestinal problems | 6 months | 1 |
Results
Thirty-three articles varying from case reports to systematic reviews are analyzed in terms of the duration of treatment, methods utilized to assess the conditions of the participants, number of participants, publication year and trustworthiness of the studies. Afterwards, studies are categorized depending on their “Level of Evidence” and results of each category will be discussed separately (Table 4).
Level of Evidence = 1
This category includes 9 different systematic reviews listed as follows: Millward et al, 2004, Christison and Ivany, 2006, Millward et al, 2008, Mulloy et al, 2010, Mulloy et al, 2011, Buie 2013, Hurwitz et al, 2013, Marı´-Bauset et al, 2014 and Elder et al 2015 (Table 4).
Systematic review conducted by Christison and Ivany in 2006 concludes that there is not sufficient data to support GFCF diet in ASD treatment by analyzing six uncontrolled clinical trials and a single-blind randomized controlled trial. We believe that the review conducted by Christison and Ivany is inadequate to reach a valid conclusion due to the absence of control group in most of the studies 23.
Hurwitz et al, 2013 reached the conclusion that elimination of gluten and casein from diet of an ASD patient would not cause significant improvement in ASD symptoms by analyzing 5 study (same conclusion with Christison and Ivany, 2006) 39. Moreover, Hurwitz et al, 2013 states that 3 out of 5 study demonstrate that GFCF diet is not beneficial for ASD patients, two of which were reliable studies since they were blinded studies comparing participants’ behavior and found consistent outcomes. On the other hand, other 2 studies claim that GFCF diet has positive impacts on ASD patients. However, these two studies do not reveal reliable results because of not having placebo control (high risk of bias)39.
Millward et al, 2004 states that the results about cognitive skills, linguistic ability and motor ability are unclear, whereas, there is an important beneficial effect in autistic traits associated with GFCF diet. Millward et al, 2004 touches upon the fact that there are wide confidence intervals in the outcomes since their review is a small-scale study20. On the other hand, Millward et al, 2008 concludes that there is not statistically significant difference in any of the traits between the control group and treatment group 27. Similarly, Marı´-Bauset et al, 2014, Mulloy et al, 2010 and Mulloy et al, 2011 argue that there is not sufficient data to support opioid-excess theory and, thus GFCF diet in treatment of ASD 31, 33, 41.There is a consensus on studies’ about this topic being weak and limited . Moreover, all of these studies indicate that GFCF diet can be resorted by medical professionals if the patient experiences dramatic behavioral improvement associated with GFCF diet or if the patient has a confirmed allergy towards gluten and/or casein 20, 27, 31, 33, 41.
Even though Buie 2013 reaches the same conclusion as all other systematic reviews, Buie 2013 emphasizes the possibility that inconsistent and insufficient data regarding to GFCF diet might be related to genetic differences and possible gene expression differences between individuals suffering from ASD 34.The emphasis of Buie 2013 about the individualistic differences and individual-based treatment strategies should not be overlooked.
Level of Evidence = 2
This category includes six studies all of which are well-conducted randomized controlled trials aim to reach out a conclusion about the efficiency of GFCF diet in ASD treatment (Table 4).
Sponheim, 1991 has both conducted double-blind gluten/placebo studies (3 patients) and open study (4 patients), thus become a fundamental study in this category. Sponheim, 1991 argues that GF diet is not beneficial for ASD patients, also in some instances it becomes a negative factor that causes further social isolation of patient 13. Elder et al, 2006 conducted randomized, double-blind study with 15 patient (age 2-16) and argues that there is no connection between GFCF diet and ASD symptoms despite the presence of improvement in some patients (statistically insignificant). One must consider the fact that results of Elder et al, 2006 are preliminary and based on a short-term treatment, thus not complete 21.
Hyman et al, 2016 is a double-blind, placebo-controlled study conducted with 14 children (age 3-5) which demonstrates no statistically significant benefit associated with GFCF diet treatment for patients suffering from ASD (Similar results were established by Hyman et al, 2010 in a randomized, double-blind study) 30, 43. Depending of the size of study group (72 patients) and longer treatment period (24 months) Whiteley et al, 2010 is an attention-grabbing study. It promotes the same conclusion reached by other studies in this group, though, Whiteley et al, 2010 does not provide conclusive evidence due to lack of placebo control 32. Even though these studies utilize distinct assessment methods to evaluate patiens’ conditions they reveal supportive outcomes 21, 26, 30, 43.
Level of Evidence = 3
Despite the fact that two studies in this category, Knivsberg et al, 2002 and Johnson et al, 2011, are designed differently we believe that they are equal in terms of trustworthiness and “Level of Evidence” (Table 4).
Positive correlation between GFCF diets and ASD treatment has been demonstrated for the first time in this systematic review by Knivsberg et al, 2002. Even though the study group is not large (20 participants) use of two-tailed Mann–Whitney U-test, p-test and mean value analysis reduces the risk of bias. The comparison between control group and diet group indicates significant improvement in the diet group even though small improvements are observed also in the control group.These statistically insignificant improvements in the control group might be related to the fluctuations in autism symptoms as argued by the authors 19.On the other hand, Johnson et al, 2011, is a non-randomized, single blinded study which also rejects the relation between gluten and casein free diet and autistic behaviors 35.
Level of Evidence = 4
Lucarelli et al, 1995 is a double blind study which examines the effect of gluten-free and casein-free diet on patients individually. Lucarelli et al, 1995 concludes that significant improvement is associated with GF and CF diets. Additionally, Lucarelli et al, 1995 indicates that casein challenge is related to the worsening of autistic symptoms which becomes a supportive evidence to the interest of this study 14.
Similar results are established by Knivsberg et al, 1990 and Knivsberg et al, 1995 both of which utilize urinary peptide level analysis and standard assessment questionaries 12, 15. Also, Cade et al, 2000 demonstrates that schizophrenia or autism is associated with exorphins formed due to incomplete digestion of gluten and casein in the intestine by immunoglobulin analysis as claimed by opioid-excess theory 18.
On the other hand, controversial findings are established by Whiteley et al, 1999 which argues that GFCF is associated with behavioral and social improvements whereas no significant decrease in specific urinary compounds are observed expected by opioid-excess theory. Therefore, outcomes of this study point out the possibility that possible success of eliminating gluten and casein from diet might be related to a different reason, rather than opioid-excess theory 16.
Level of Evidence = 5
Patel and Curtis, 2007 is a qualitative study performed with a small study group comprised of 10 participants. This study indicates significant improvement in social interaction, concentration, writing, language, and behavior in all of the participants and decrease in urinary heavy metal levels (lead) in every participant 25. Nazni et al, 2008 investigates the efficiency of gluten-free diet and casein-free diet separately and concludes that they are both beneficial for autistic patients (Higher improvements are observed in gluten-free diet compared to casein-free diet). Other study in this category conducted by Mageshwari and Minitha, 2006 provide supportive evidence to the conclusion reached by Patel and Curtis, 2007 and Nazni et al, 2008 and demonstrates improvements in behavioral symptoms of autism 24, 28.
Level of Evidence = 6
This category consists of five case reports and two studies based on parental reports. Even though Irvin, 2006 does not suggest any association between ASD and gluten-free diet, Knivsberg et al, 1999, Hsu et al, 2009 and Genius, 2010 claim otherwise 17, 22, 29, 38 Moreover, Herbert and Buckley, 2013 shows improvements in autistic symptoms, language skills and auditory sensitivity. 40 These studies will not be further discussed since they are case reports which may include high levels of bias associated with small study groups and unreliable assessment methods.
Adverse Effects
After completing the discussion about the efficiency of GFCF diet for patients suffering from ASD one should consider adverse effects associated with the implementation of GFCF diet. Despite the fact that GFCF diet is a relatively new treatment method for ASD numerous studies exist about its’ safety, as well as its’ efficiency. Although 2 studies, Cornish et al, 2002 and Johnson et al, 2011, report no significant side-effects associated with GFCF diet in any kind of macro or micronutrient, most of the studies report lower bone density and calcium intake 35, 44, 45, 46, 47, 48. In addition to lower calcium intake and bone density Neumayer et al, 2012 also reports lower vitamin D and lactose intake from gut 49.
Another aspect of gluten free diet, effects on microbiome in human gut, is investigated in Bonder et al, 2016. According to their 4 weeks of study gluten-free diet results in moderate but significant changes in human gut microbiome, especially for 8 bacteria species. For instance, remarkable decrease in Veillonellaceae, known for lactate fermentation, and Ruminococcus bromii, known for starch degradation is reported 50 Nevertheless, there is an obvious need to conduct more comprehensive research about the safety of GFCF diet since none of the previous studies were conducted with large study groups.
Conclusion
The main purpose behind categorizing studies in this systematic review depending on their “Level of Evidence” is to examine and assess the outcomes of each study as well as its’ trustworthiness. By categorizing studies depending on their “Level of Evidence” we emphasize the contrast between outcomes of studies with high trustworthiness and low trustworthiness (high risk of bias). In general studies conducted with a small study group (have high values of “Level of Evidence”) illustrate significant improvements in autistic symptoms when participants are treated with GFCF diets, whereas, studies conducted with a large study group or systematic reviews argue that association between ASD and GFCF diet is statistically insignificant (Figure 2). Even though some of the participant in those studies with high trustworthiness have showed improvement in some autistic symptoms, due to the size of study group they become statistically insignificant. Thus, we speculate that the efficiency of GFCF diet and validity of opioid-excess theory might be related to individual genetic differences. Therefore, rather than rejecting opioid-excess theory and use of GFCF diet as a treatment option for ASD patients we claim that GFCF diet should remain as a treatment option for ASD and physicians should consider its’ implementation depending on the patient. Further studies, especially in the field of genetics, is needed in order to prove our hypothesis about the relation between genetic characteristics and the efficiency of GFCF diet in ASD patients.
Figure 2.Percentile of studies at each “Level of Evidence” that support gluten-free and/or casein-free diet in the treatment of autism spectrum disorders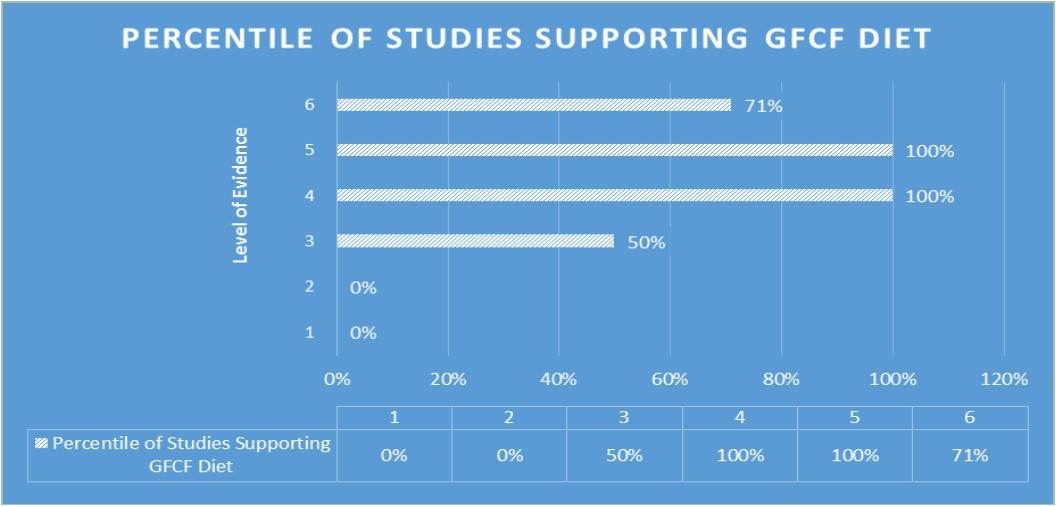
One other factor that needs to be evaluated when assessing the efficiency of GFCF diet on patients suffering from autism spectrum disorders is the sensitivity of individuals participating in the studies to gluten and/or casein. Neither of the studies analyses whether the individuals have any kind of sensitivity or allergy towards casein except one (Lucarelli et al, 1995), whereas, only a limited number of studies assess individuals in that manner. Lucarelli et al, 1995 hypothesized a relation between food allergy, mainly against casein and lactalbumin, and infantile autism, though, their hypothesis has not been confirmed by other studies 14. Genuis et al, 2010 illustrates connection between coeliac disease and autism through a case report involving 5-year-old boy and improvement in symptoms of both autism and gastrointestinal disorders 38. In addition, Whiteley et al, 1999 states that no test had been performed to test the sensitivity of individuals to gluten but there had been indicators of allergy and sensitivity towards gluten mostly based on parental reports 16. Similar observations are made by et al, 2012 36. To conclude, in the studies conducted so far in the literature gluten and/or casein sensitivity of patients are either not assessed or gluten/casein sensitive individuals constitute statistically insignificant portion of the study groups. Thus, there is not adequate data to separately analyze effects of GFCF diet on gluten and/or casein sensitive patients.
Furthermore, adverse effects related to the implementation of GFCF diet such as weight gain, obesity and deficiencies in vitamin B, iron, minerals and vitamin D should not be overlooked and necessary precautions should be taken.
Author Contributions
M.Ç.: Design, Literature Search, Study Selection, Study Assessment, Article Writing; S.Ç.: Literature Search, Study Selection, Study Assessment, Article Writing.
*No conflict of interest is reported.
References
- 1. (2013) American Psychiatric Association, American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5. Washington,DC:American Psychiatric Association.
- 2.Dombrowski S. (2014) Autism spectrum disorder. Salem Press Encyclopedia Of Health [serial online]. Available from: Research Starters. , Ipswich, MA. AccessedSeptember13,2016
- 4.Hunter L, O’Hare A, Herron W, Fisher L, Jones G. (2003) Opioid peptides and dipeptidyl peptidase in autism.Developmental Medicine And Child Neurology [serial online]; 45(2), 121-128.Available from:MEDLINE. , Ipswich, MA. AccessedSeptember13,2016
- 5.Bent S, Bertoglio K, Hendren R. (2009) Omega-3 fatty acids for autistic spectrum disorder: a systematic review. Available from: CINAHL Plus with Full Text , Ipswich, MA. AccessedSeptember13,2016, Journal Of Autism & Developmental Disorders [serial online] 39(8), 1145-1154.
- 6.Rimland B, Callaway E, Dreyfus P. (1978) The effects of high doses of vitamin B6 on autistic children: a double-blind crossover study. , American Journal of Psychiatry 135, 472-75.
- 7.Martineau J, Barthelemy C, Garreau B, Lelord G. (1985) Vitamin B6, magnesium, and combined B6-Mg: therapeutic effects in childhood autism. , Biol. Psychiatry 20, 467-78.
- 8.Winburn E, Charlton J, McConachie H, McColl E, Parr J et al. (2014) Parents’ and Child Health Professionals’ Attitudes Towards Dietary Interventions for Children with Autism Spectrum Disorders. , Journal Of Autism & Developmental Disorders 44(4), 747-757.
- 9.B M Melnyk, Fineout-Overholt E. (2005) Evidence-based practice in nursing & healthcare: a guide to best practice. , Philadelphia: Lippincott Williams,Wilkins
- 10. (2009) Oxford Centre for Evidence-based Medicine - Levels of Evidence (March2009) - CEBM. (n.d.). Retrieved from http://www.cebm.net/oxford-centre-evidence-based-medicine-levels-evidence-march-2009/
- 11.K L Reichelt, Ekrem J, Scott H. (1990) Gluten, milk proteins and autism: dietary intervention effects on behavior and peptide secretion. , J Appl Nutr 42, 1-11.
- 12.A M Knivsberg, Wiig K, Lind G, Nødland M, K L Reichelt. (1990) Dietary intervention in autistic syndromes. , Brain Dysfunct 3, 315-317.
- 13.Sponheim E. (1991) Gluten free diet in infantile autism. , Journal of Norwegian Medical Association 111, 704-707.
- 14.Lucarelli S, Frediani T, A M Zingoni, Ferruzzi F, Giardini O et al. (1995) Food allergy and infantile autism. , Panminerva Med 37, 137-141.
- 15.A M Knivsberg, K L Reichelt, Nødland M, Høien T. (1995) Autistic syndromes and diet: a follow-up study. , Scan. J. Educ. Res 39, 223-236.
- 16.Whitley P, Rodgers J, Savery D, Shattock P. (1999) A gluten-free diet as an intervention for autism and associated spectrum disorders: Preliminary findings. , Autism 3, 45-66.
- 17.Knivsberg A, Reichelt K, Nodland M. (1999) Dietary intervention for a seven year old girl with autistic behavior. , Nutritional Neuroscience 2, 435-439.
- 18.Cade R, Privette M, Fregly M, Rowland N, Z J Sun et al. (2000) Autism and schizophrenia: intestinal disorders. , Nutritional. Neuroscience 3, 57-72.
- 19.Knivsberg A, Reichelt K, Høien T, Nødland M. (2002) A Randomized Controlled Study of Dietary Intervention in Autistic Syndromes. Nutritional Neuroscience [serial online], 5(4), 251-261.Available from:Academic Search Complete. , Ipswich, MA. AccessedSeptember10,2016
- 20.Millward C, Ferriter M, Calver S, Connell-Jones G. (2004) Gluten- and casein-free diets for autistic spectrum disorder. The Cochrane Database Of Systematic Reviews [serial online], (2), CD003498. Available from: MEDLINE , Ipswich, MA. AccessedSeptember10,2016 .
- 21.Harrison-Elder J, Shankar M, Shuster J, Theriaque D, Burns S et al. (2006) The Gluten-Free, Casein-Free Diet In Autism:. , Results of A Preliminary Double Blind Clinical Trial. Journal Of Autism & DevelopmentalDisorders [serial online]. Available from: Academic Search Complete,Ipswich, MA. AccessedSeptember10,2016; 36(3), 413-420.
- 22.Irvin D. (2006) Using analog assessment procedures for determining the effects of a gluten-free and casein-free diet on rate of problem behaviors for an adolescent with autism. Behavioral Interventions [serial online]. Available from: Academic Search Complete. , Ipswich, MA, AccessedSeptember10,2016; 21(4), 281-286.
- 23.Christison G, Ivany K. (2006) Elimination diets in autism spectrum disorders: any wheat amidst the chaff?. Journal Of Developmental And Behavioral Pediatrics: JDBP [serial online]. Available from: MEDLINE, Ipswich, MA. Accessed September10,2016; 27(2 Suppl): 162-171.
- 24.U S Mageshwari, S J Minitha. (2006) Impact of dietary exclusion of casein and gluten on selected autistic children. , Ind J Nutr Diet 43, 183-191.
- 25.Patel K, Curtis L. (2007) A comprehensive approach to treating autism and attention-deficit hyperactivity disorder: A pre-pilot study. , Journal of Alternative and Complementary Medicine 13, 1091-1097.
- 26.Seung H, Rogalski Y, Shankar M, Elder J. (2007) The gluten- and casein-free diet and autism: Communication outcomes from a preliminary double-blind clinical trial. , Journal of Medical Speech-Language Pathology 15, 337-345.
- 27.Millward C, Ferriter M, Calver S, Connell-Jones G. (2008) Gluten- and casein-free diets for autistic spectrum disorder. The Cochrane Database of Systematic Reviews [serial online]. 2, 003498.
- 28.Peerkhan N, Edward-Gnanaraj W, Veerappan N. (2008) Impact of Casein and Gluten Free Dietary Intervention on Selected Autistic Children. , Iranian Journal Of Pediatrics [serial online] 3, 244.
- 29.Chia-Lin H, Delmar C Y L, Chia-Lin C, Chin-Man W, Alice M K W. (2009) The Effects of A Gluten and Casein-free Diet in Children with Autism: A Case Report. , Chang Gung Medical Journal [serial online] 04, 459.
- 30.Hyman S, Stewart P, Smith T. (2010) The gluten- and casein-free diet in children with autism. The Brown University Child and Adolescent Behavior Letter [serial online], 3. Available from: Academic OneFile , Ipswich, MA. AccessedSeptember10,2016 .
- 31.Mulloy A, Lang R, O’Reilly M, Sigafoos J, Lancioni G et al. (2010) Review: Gluten-free and casein-free diets in the treatment of autism spectrum disorders: A systematic review. Research In Autism Spectrum Disorders [serial online] 4, 328-339.
- 32.Whiteley P, Shattock P, Carr K, Hooper M, Todd L. (2010) How could a gluten-and casein-free diet ameliorate symptoms associated with autism spectrum conditions?. Autism Insights [serial online], 2010, 39. Available from: Academic OneFile. , Ipswich, MA. AccessedSeptember10,2016
- 33.Mulloy A, Lang R, O’Reilly M, Sigafoos J, Lancioni G et al. (2011) Addendum to “gluten-free and casein-free diets in treatment of autism spectrum disorders: a systematic review”. Res Autism Spectrum Disorders. 5(1), 86-88.
- 34.Buie T. (2013) The Relationship of Autism and Gluten. Clinical Therapeutics [serial online], (5), 578. Available from: Academic OneFile. , Ipswich, MA. AccessedSeptember10,2016
- 35.Johnson C, Handen B, Zimmer M, Sacco K, Turner K. (2011) Effects of Gluten Free / Casein Free Diet in Young Children with Autism: A Pilot Study. Journal Of Developmental And Physical Disabilities [serial online]. Available from: Academic OneFile,Ipswich,MA. , AccessedSeptember10,2016; 3, 213.
- 36.Pennesi C, Klein L. (2012) Effectiveness of the gluten-free, casein-free diet for children diagnosed with autism spectrum disorder: Based on parental report. , Nutritional Neuroscience [serial online] 15(2), 85-91.
- 37.Harris C, Card B. (2012) A pilot study to evaluate nutritional influences on gastrointestinal symptoms and behavior patterns in children with Autism Spectrum Disorder. Complementary Therapies. In Medicine [serial online]. Available from: MEDLINE , Ipswich, MA, AccessedSeptember10,2016; 20(6), 437-440.
- 38.Genuis S, Bouchard T. (2010) Celiac Disease Presenting as Autism. , Journal Of Child Neurology [serial online] 25(1), 114-119.
- 39.Hurwitz S. (2013) The Gluten-Free, Casein-Free Diet and Autism: Limited Return on Family Investment. , Journal Of Early Intervention [serial online] 35(1), 3-19.
- 40.Herbert M, Buckley J. (2013) Autism and Dietary Therapy: Case Report and Review of the Literature. Journal Of Child Neurology [serial online]. Available from: Publisher Provided Full Text Searching File,Ipswich. , MA. AccessedSeptember10,2016; 28(8), 975.
- 41.Marí-Bauset S, Zazpe I, Mari-Sanchis A, Llopis-González A, Morales-Suárez-Varela M. (2014) Evidence of the gluten-free and casein-free diet in autism spectrum disorders: a systematic review. , Journal Of Child Neurology [serial online]. December 29(12), 1718-1727.
- 42.Elder J, Kreider C, Schaefer N, M de Laosa. (2005) A review of gluten- and casein-free diets for treatment of autism: 2005-2015. Nutrition And Dietary Supplements [serial online],2015,87.
- 43.Hyman S, Stewart P, Smith T. (2016) The gluten-free/casein-free diet: A double-blind challenge trial in children with autism. Journal Of Autism AndDevelopmental Disorders [serial online]. Available from: Academic OneFile. , Ipswich, MA. Accessed September10,2016;1:
- 44.Cornish E. (2002) Gluten and casein free diets in autism: a study of the effects on food choice and nutrition. , J. Hum. Nutr. Diet 15, 261-269.
- 45.Konstantynowicz J, T V Nguyen, Kaczmarski M. (2007) Fractures during growth: Potential role of a milk-free diet. , Osteoporos Int 18, 1601-1607.
- 46.M L Hediger, L J England, C A Molloy, K F Yu, Manning-Courtney P et al. (2008) Reduced bone cortical thickness in boys with autism or autism spectrum disorder.doi: 10.1007/s10803-007-0453-6.J. Autism Dev. Disord;38: 848–856
- 47.Monti P M, N P Barnett, Colby S M, C J Gwaltney, Spirito A et al. (2007) Motivational interviewing versus feedback only in emergency care for young adult problem drinking. , Addiction 102, 1234-1243.
- 48.K E Black, J A Murray, C S David. (2002) HLA-DQ determines the response to exogenous wheat proteins: a model of gluten sensitivity in transgenic knockout mice. , J. Immunol 169, 5595-5600.
Cited by (7)
This article has been cited by 7 scholarly works according to:
Citing Articles:
Z. Taha, Khalid A. Abdalhai - (2021) Semantic Scholar
Open Access Macedonian Journal of Medical Sciences (2021) OpenAlex
Katarzyna Tarnowska, E. Gruczyńska-Sękowska, D. Kowalska, E. Majewska, M. Kozłowska et al. - Critical reviews in food science and nutrition (2021) Semantic Scholar
Critical Reviews in Food Science and Nutrition (2021) OpenAlex
(2019) Crossref
Vandenhoeck & Ruprecht eBooks (2019) OpenAlex
International Journal Of Nutrition (2017) OpenAlex
