Abstract
The objective of this study was to confirm the nutritional risk factors for asthma and obesity in adolescents using a cross-sectional survey. We included male and female adolescents aged 10 to 19 years of age in this study. The indicators of nutritional status used in this study were body mass index, the height-for-age index, and arm and waist circumferences. We used a food frequency questionnaire to estimate the average consumption of sodium. Physical activity was assessed using the International Physical Activity Questionnaire, and asthma characteristics were evaluated using the International Study of Asthma and Allergies in Childhood (ISAAC). Factors associated with asthma in bivariate and multivariate analyses were assessed using the Poisson regression analysis. The significance level was set at 5%, and the analyses were performed using the SPSS statistical package. We evaluated 1362 students with an average age of 15.65 ± 1.24 years. There was a positive risk for asthma between females (PR = 1.41) and asthma severity in adolescents with a high sodium intake (PR = 2.30). Within overweight adolescents, the prevalence for asthma risk washigher among females (PR = 1.66) and the high sodium intake group (PR = 1.98). An increased risk for asthma severity correlated with high sodium intake (PR = 3.07). There was a higher risk for asthma and its severity in females with high sodium intake. The same pattern was observed for adolescents with excessive weight.
Author Contributions
Academic Editor: Justin Friedlander, Fox Chase/Einstein Urologic Institute
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2016 Franceliane Jobim Benedetti, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Asthma and obesity are diseases that have reached epidemic proportions, especially in westernized countries. Theories have been proposed to explain the association between obesity and asthma. Theoretical assumptions included he direct effects of obesity on functional, genetic, hormonal, and immunological mechanisms and inflammatory responses1, 2, 3. Moreover, increased weight has been associated with lifestyle and eating habits4, 5.
Significant changes in dietary and nutritional standards have been proposed worldwide. These changes are characterizedas part of aprocess callednutritional transition. There is an increasing consumption of processed foods and a more sedentary lifestyle provided by technological advances, especially among children and adolescents who remain in front of televisions, videogames and computer devices for many hours6, 7. The low level of physical activity in adolescents with asthma may be associated with obesity because these verity of the disease can contribute to a sedentary lifestyle; however, obesity and a large amount of time spent watching TV can increase the risk of respiratory symptoms8, 9.
In addition to the relationship with respiratory health and sedentary behavior, the habit of watching television promotes a change in eating routine and increases the risk of obesity in children and adolescents10. These habits are also associated with a higher consumption of salty snacks11 that are rich in sodium and increase the risk of bronchial hyperresponsiveness and wheezing in children and adolescents12.
Despite the promising discoveries and hypotheses available in the current literature, finding son the prevalence of asthma, the effects of a sedentary lifestyle and the role of sodium are scarce. Therefore, this study assessed the nutritional risk factors for asthma and obesity in adolescents.
Methodology
This study consisted of across-sectional observational design and was completed with a group of adolescents from 16 public high schools located in the urban area of the city of Santa Maria-RS/Brazil from May to November 2012.
We included male and female adolescents aged 10 and 19 years who were regularly enrolled in the schools where data collection occurred. All subjects agreed to participate, and their parents signed an informed consent form. We excluded three adolescents with cerebral palsy and two adolescents with Down syndrome.
Sampling was performed by randomly polling classes. The sample size estimation to show statistically significant differences with a RR= 2.0 for the prevalence of obesity (15%) among adolescents with and without asthma (21%), a significance level of 0.05 and a power level of 80% was calculated as 395 individuals.
The research project used to develop this study was approved by the Ethics Committee in research of the Rio Grande do Sul Federal University (UFRGS) under the protocol n. 20009. All participants received the results of their assessment, and the school received the overall results of the student analyses.
The data referring to sample characterizations, anthropometric evaluations and asthma characteristics were collected by the authors and previously trained students from the undergraduate program in nutrition. The questionnaires were applied in the classroom for all of the present students and in the absence of the teacher.
Anthropometric measurements were assessed individually and in duplicate using standard techniques and calibrated equipment in a private room. We assumed a maximum difference value of 1.0cm or 100g between measurements. For the analysis, we used the average value of each measure 13.
The body mass weight in kg was obtained while the adolescents were barefoot and wearing minimal clothing using platform scales (Plenna, São Paulo, Brazil) with a maximum load of 150kg and a range of 100g. Height was measured while the subjects were standing barefoot using an extensible stadiometer (Sanny, São Paulo, Brazil) fixed on a flat wall without a baseboard 13.
The anthropometric indicators used included the body mass index for age (BMI-Z) and the height-for-age index (H/A-Z). These parameters were analyzed with Anthro Plus version 1.04, and the results were obtained using the z-score and classified according to WHO guidelines 13. Adolescents with a BMI-Z greater than +1 were classified as overweight.
For arm circumference (AC) measurements, we used an inextensible measuring tape (Secca, ON, USA) that was positioned perpendicular to the long axisof the arm at the midpoint (distance between the acromion and theolecranon). The measurement value was classified based on Frisancho 14.
Waist circumference (WC) was measured usinga Secca® (ON, USA) in elastic measuring tape positioned at the minimum circumference between the iliac crest and the last rib. An excess of abdominal fat was defined as a WC higher than the 80th percentile for age and sex 15.
We used a food frequency questionnaire for foods with high sodium content (FFQSo) to estimate the average daily intake of sodium. The survey inquires about 15 types of food, including the frequency and portion size consumed. The final result was calculated as the average consumption of sodium in grams/day 16. Intake values above the Tolerable Upper Intake Level (UL) for age 17 were considered inadequate. The cut-off for the UL was based on the high sodium intake of Brazilians18, 19
The typical physical activity of adolescents was assessed using the short and validated version of the Brazilian adolescents of the International Physical Activity Questionnaire (IPAQ), which classifies populations into three categories: insufficiently active, sufficiently active and very active 20. The survey included questions regarding the practice of sports and the daily hours of exposure to television, videogames and computers.
Asthma characteristics were evaluated by the International study of asthma and allergies in childhood (ISAAC) self-administered questionnaire, which was validated after its translation into Brazilian Portuguese 21.
Asthma was assessed by the overall ISAAC score, as recommended by Wandalsen et al. 22. To identify adolescents with asthma, we used acut-off score ≥6 on the ISAAC questionnaire 21. More severe asthma cases were defined by wheezing in the last 12 months and one or more positive responses to the following questions: inability to speak at least two complete words during an attack of wheezing, more than 12 crises in the last year, and greater than one night per week of nocturnal awakenings23, 24.
Statistical Analyses
The results are expressed as the mean and standard deviation. To assess the distribution of variables, the Kolmogorov-Smirnov test was used. To analyze the association between qualitative variables, the chi-square test was applied. Gross and adjusted estimation of prevalence ratios were calculate dusing a Poisson regression analysis with a confidence interval of 95% (95% CI) and a robust adjustment of variance in bivariate and multivariate analyses, respectively. Covariates with a value of p <0.20 in the bivariate analysis were used in the Poisson multiple regression analysis (multivariate analysis).
The cut-off level for significance was 5%, and the analyses were performed using SPSS (Statistical Package for the Social Sciences) software version 18.0.
Results
We evaluated 1362 students from 57 classes across 16 high schools in the city of Santa Maria/RS. The average age of the adolescents was 15.65 ± 1.24 years, and the prevalence of signs and symptoms related to asthma among all adolescents were stratified by BMI-Z (Table 1). There were no significant differences in the signs and symptoms of asthma between adolescents with and without obesity.
Table 1. The prevalence of signs and symptoms related to asthma in overweight and non-overweight adolescents according to the ISAAC questionnaire.| Questionnaire | Adolescents | |||
|---|---|---|---|---|
| Total * (n=1362) | Non-overweight * (n=995) | Overweight * (n=367) | P | |
| Wheezing or whistling at any time | 588 (43,2) | 424(51,1) | 164(12,0) | 0,493 |
| Wheezing or whistling in the chest | 282 (20,7) | 221(15,5) | 71(5,2) | 0,452 |
| Attacks of wheezing | 38 (2,8) | 25(1,8) | 71(5,2) | 0,306 |
| Sleep been disturbed due to wheezing | 179 (13,1) | 136(10,0) | 43(3,2) | 0,344 |
| Wheezing to limit your speech | 46 (3,4) | 33(2,4) | 13(1,0) | 0,848 |
| Ever asthma | 219 (16,1) | 149(10,9) | 70(5,1) | 0,069 |
| Wheezy during or after exercise | 217 (16) | 162(12,0) | 55(4,1) | 0,564 |
| Dry cough at night | 491 (36,2) | 360(26,5) | 131(9,7) | 0,882 |
| Score and cut-off point (≥6) | 279(20,5) | 207(15,2) | 72(5,3) | 0,796 |
| Severe asthma | 78(5,7) | 56(4,1) | 22(1,6) | 0,631 |
Table 2 shows the physical activity and nutritional characteristics of the study participants. There is greater prevalence of obesity in females. There were statistically significant differences between overweight adolescents and adolescents with asthma, particularly in females. A total of 57.1% of the overweight adolescents with severe asthma consumed a high amount of sodium on a daily basis.
Table 2. The effect of asthma on the nutritional status and physical activity level of adolescents.| Variables | Adolescents | Asthma | Severe asthma | ||||
|---|---|---|---|---|---|---|---|
| Total (n=1362) | Overweight * (n=367) | Total (n=279) | Overweight * (n=72) | Total (n=78) | Overweight * (n=22) | ||
| Sex | Male | 574(42.1) | 155(42.2) | 85(30.5)** | 22(30.6)** | 23(29.5)** | 7(31.8) |
| Female | 788(57.9) | 212(57.8) | 194(69.5) | 50(69.4) | 55(70.5) | 15(68.2) | |
| H/A-Z | Appropriate | 1340(98.4) | 335(96.7)** | 273(97.3) | 72(100) | 74(94.9)** | 22(100) |
| Short | 22(1.6) | 12(3.3) | 6(2.2) | 0 | 4(5.1) | 0 | |
| WC | Without excess | 862 (63.3) | 224(61) | 182(65.2) | 44(61.1) | 49(62.8) | 12(54.5) |
| With excess | 500(36.7) | 143(39) | 97(34.8) | 28(38.9) | 29(37.2) | 10(45.5) | |
| AC | Eutrophic | 792 (82.2) | 212(82.8) | 163(81.5) | 41(77.4) | 50(87.7) | 14(87.5) |
| Obesity | 172(17.8) | 44(17.2) | 37(18.5) | 12(22.6) | 7(12.3) | 2(12.5) | |
| IPAC | Insuff. active | 806 (67.4) | 210(67.3) | 172(65.6) | 47(69.1) | 46(63.9) | 14(67.1) |
| Suff. active | 270 (22.6) | 72(23.1) | 61(23.3) | 14(20.6) | 20(27.8) | 4(23.3) | |
| Very active | 120 (10) | 30(9.6) | 29(24.2) | 7(10.3) | 6(8.3) | 2 (9.6) | |
| Tv/video | <4 hours | 363(30.3) | 84(26.8) | 73(27.9) | 17(25) | 21(29.2) | 3(15) |
| >4 hours | 834(69.7) | 229(73.2) | 189(72.1) | 51(75) | 51(70.8) | 17(85) | |
| Sports | No | 545(45.7) | 134(43.1) | 127(48.5) | 26(38.2) | 34(47.2) | 7(35) |
| Yes | 648(54.3) | 177(56.9) | 135(51.5) | 42(61.8) | 38(52.8) | 13(65) | |
| Sodium/day | Adequate | 716(74.3) | 191(75.5) | 124(70.1) | 25(54.3)** | 28(57.1)** | 6(42.9)** |
| High | 248(25.7) | 78(24.5) | 53(29.9) | 21(45.7) | 21(42.9) | 8(57.1) | |
Based on the height-for-age index, we determined that out of 22 adolescents with short stature, 12 were classified as overweight. Among asthmatic students, there was no individual that was overweight and had a short stature.
The bivariate and multivariate analyses adjusted for the age of all adolescents, showed a significant risk for asthma in females and severe asthma in adolescents with high sodium intake (Table 3).
Table 3. The risk factors for asthma and asthma severity among adolescents identified with bivariate and multivariate analyses| Variables | Asthma | Severe asthma | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total * (n=1362) | Total ** (n=1362) | Total * (n=1362) | Total ** (n=1362) | |||||||||
| PR | CI 95% | P | PR | CI 95% | P | PR | CI 95% | P | PR | CI 95% | P | |
| BMI-ZNon-overweight Overweight | Ref.0.94 | --0.74-1.20 | -0.623 | - | -- | - | Ref.1.07 | --0.66-1.72 | -0.793 | - | -- | - |
| SexMaleFemale | Ref.1.66 | --1.32-2.09 | -<0.01† | Ref.1.41 | --1.06-1.87 | -0.017† | Ref.1.75 | --1.09-2.80 | -0.021† | Ref.1.57 | --0.86-2.84 | -0.155 |
| WCWithout excess With excess | Ref.0.92 | --0.74-1.15 | -0.473 | - | -- | - | Ref.1.01 | --0.65-1.58 | -0.938 | - | -- | - |
| ACEutrophicObesity | Ref.1.04 | --0.76-1.42 | -0.823 | - | -- | - | Ref.0.64 | --0.29-1.38 | -0.258 | - | -- | - |
| TV/video<4 hours>4 hours | Ref.1.12 | --0.88-1.43 | -0.341 | - | -- | - | Ref.1.05 | --0.64-1.73 | -0.832 | - | -- | - |
| IPACInsuff. activeSuff. activeVery active | Ref.0.930.88 | --0.63-1.370.63-1.24 | -0.7340.477 | - | -- | - | Ref.1.481.14 | --0.61-3.590.49-2.61 | -0.3850.755 | - | -- | - |
| SportsYesNo | Ref.1.12 | --0.90-1.38 | -0.298 | - | -- | - | Ref.1.06 | --0.68-1.66 | -0.784 | - | -- | - |
| Sodium/day AdequateHigh | Ref.1.24 | --0.93-1.65 | -0.146 | Ref.1.29 | --0.96-1.75 | -0.083 | Ref.2.16 | --1.24-3.74 | -0.006 | Ref.2.30 | --1.37-4.17 | -0.006† |
As demonstrated in Figure 1 and Table 4, overweight adolescents that were female and had a high sodium intake showed a significant risk of asthma in the bivariate analysis. However, in the multivariate analysis, only high sodium intake was associated with asthma. The multivariate analysis also showed that severe asthma was more prevalent in adolescents with a high sodium intake.
Figure 1.The factorsassociated with asthmaand asthma severityamong overweightwere identified with abivariate analysis. Waist circumference (WC), Arm circumference (AC), International Physical Activity Questionnaire (IPAQ), 95% confidence interval (CI95%), prevalence ratio (PR). Poisson regression: *bivariate analysis, ** multiple regression,†p<0,05.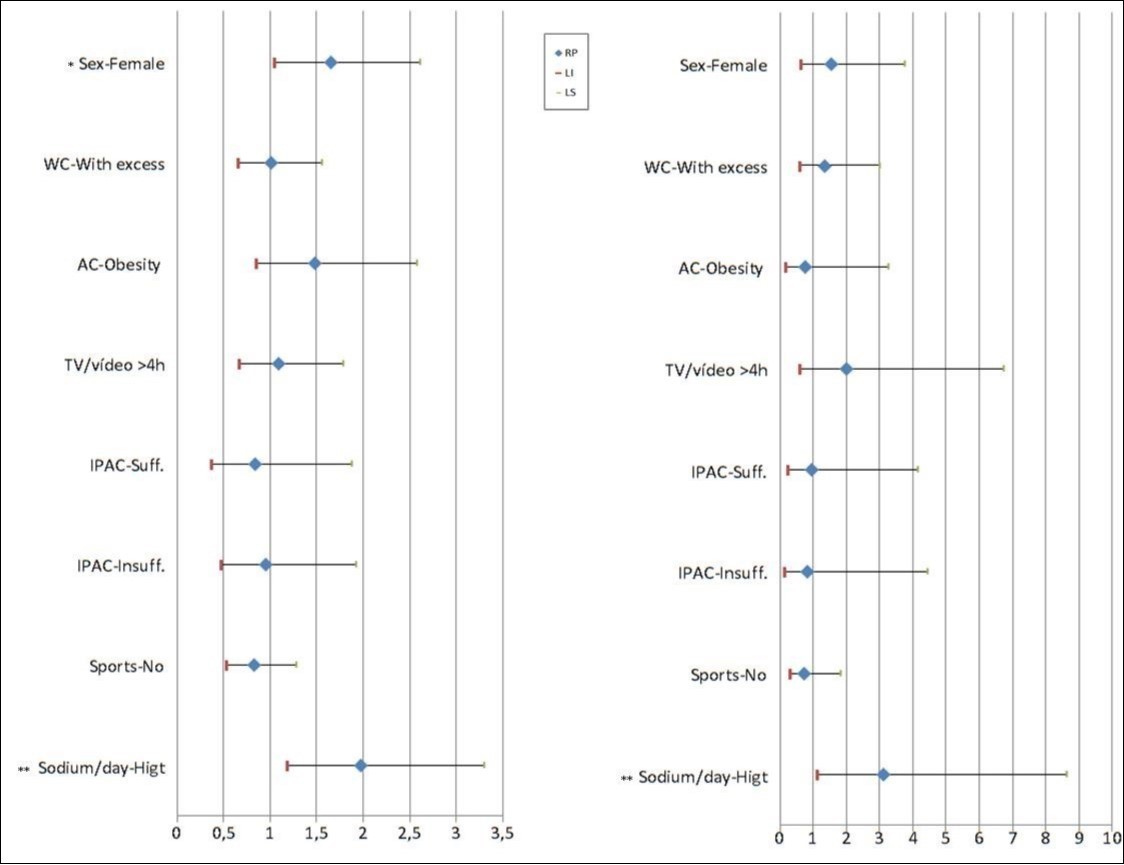
| Variables | Asthma | Severe asthma | ||||
|---|---|---|---|---|---|---|
| Overweight * (n=367) | Overweight * (n=367) | |||||
| PR | CI95% | P | PR | CI95% | P | |
| SexMaleFemale | Ref.1.03 | --0.59-1.78 | -0.911 | - | -- | - |
| ACEutrophicObesity | Ref.1.71 | -- 0.98-2.97 | -0.058 | - | -- | - |
| TV/video<4 hours>4 hours | - | -- | - | Ref.3.84 | --0.49-2.95 | -0.196 |
| Sodium/day AdequateHigh | Ref.2.48 | --1.46-4.19 | -0.001† | Ref.3.07 | -1.03-9.13 | -0.043† |
Discussion
In this study, female sex and high sodium intake increased the risk for asthma. There was an additional risk for asthma in females and those with high sodium intake among overweight adolescents. There were no differences in the signs and symptoms of asthma between overweight and non-overweight adolescents; however, females with asthma showed a higher prevalence of obesity.
Asthma and obesity are among the most prevalent diseases in children. Both pathologies have increased in parallel over the recent decades, particularly in western countries. Therefore, the possibility of the association and risk between asthma and obesity has garnered a great deal of attention. Many studies used across-sectional design to show an association between asthma and obesity in the adult population, especially in women. This association in children and adolescents remains contradictory 3, 21.
Recently, cross-sectional studies and studies with a prospective design 25, 26, 27, 28, 31, 32, 33 reported an association between asthma and obesity in children and adolescents, especially among women 31, 33. Notably, the 95th percentile for BMI was used as the cut-off for nutritional status 25, 26, 27. These results are consistent with the hypothesis described in the meta-analyses completed by Beuther and Sutherland 36 and Chen et al. 37, which concludes that the incidence of asthma is weight-dependent. In contrast, other authors 29, 30, 34, 35 did not observe this association in the pediatric population. However, these authors used the 85th percentile for BMI as the cut-off point. This cut-off value corresponds to a z-score +1, which was the cut-off value used in the present study.
Our findings are in accordance with other studies 38, 39 that showed a higher prevalence of asthma or severe asthma in females (OR= 1.48 to 2.44). In the current study, the prevalence rate for asthma in all female participants and in all overweight adolescents was 1.66. This meta-analysis also showed that overweight/obese males are more susceptible to asthma (RR: 2.47 for boys, 1.25 for girls); however, a weight-dependence for the incidence of asthma was only significant in girls 37.
There is a significant debate regarding the effect of gender on asthma. Previous studies showed that boys have a higher incidence of asthma in childhood, but girls show the higher incidence during adolescence. Potential mechanisms to explain this phenomenon include pulmonary mechanics, sleep disorders and leptin, and the influence of hormones, such as estrogen 37, 40.
Notably, the majority of studies used BMI as the sole indicator of nutritional status. New findings indicate that body composition, including WC, in the pediatric population may increase the risk of asthma 3; however, we did not observe this characteristic in the present study. Interestingly, there was a tendency toward a positive risk for asthma based on the AC within overweight adolescents. This indicator reflects body composition, and it can be used as a practical and feasible measurement during clinical examination, especially when weight and height data are not available 41.
Possible explanations for the relationship between excess adipose tissue and asthma include genetic factors, mechanical changes, airway hyperresponsiveness, alteration of hormonal substances, such as cytokines and chemokines, and changes in lifestyle, including physical activity and nutrition 42.
Another indicator of nutritional status is the H/A index. A total of 2% of adolescents with asthma and 5% of adolescents with severe asthma also had a short stature; however, no adolescent with a small stature was overweight. These results are consistent with the study by Baum et al. 43 that reported children with allergy-related diseases, including asthma, are more likely to have a low height for their age. Asthmatics show a characteristic pattern of growth, i.e., decreased growth rate, especially patients with severe asthma 44. Studies suggest that the use of inhaled corticosteroids at the recommended doses does not adversely affect the final growth of children and adolescents 45, 46.
The nutritional status of adolescents is likely related to asthma, and many theories have been proposed to elucidate the mechanism of this association. Dietary measures should be considered in analyses because the energetic control and quality of diet are considered possible inflammatory stimuli. Few studies have investigated the expenditure and consumption of energy, which are determinants of energy balance and body weight. In children and adolescents with asthma, we found that the resting energy expenditure is similar between children with and without asthma; however, the estimated energy intake exceeds the resting energy expenditure in children with asthma 47.
These data reinforce that the increase in the prevalence of asthma, especially in westernized countries, is associated with environmental factors, such as dietary intake and sedentary lifestyle. Indeed, these elements likely play a fundamental rolein the development and progression of asthma 48.
There was no statistically significant change in the risk of asthma based on the level of physical activities, sedentary activities and sports practice; however, the risk for severe asthma was higher among students classified as insufficiently active, and the risk doubled for overweight students that spent more than 4 hours a day doing sedentary activities. A previous study in children found no association between regular sport activity and asthma symptoms, but children who spent five or more hours per day watching television were more likely to have wheezing and asthma 48. Cabral et al. 49 studied children with asthma and found a higher prevalence of exercise-induced bronchospasm in more serious asthma cases; however, the intensity of the bronchoconstriction response to exercise was not related to asthma severity. Rodrigues et al. 50 observed that obese students showed more hyperresponsiveness to exercise. The authors did not consider this a reason to limit physical activity but rather suggested that this limitation be combined with other factors related to adipose tissue.
Notably, over 70% of the overweight adolescents evaluated in this study watched television or played videogames for more than four hoursper day. The exposure to television is associated with an increase in the consumption of salty snacks, and these two behaviors are implicated in both obesity and the development of asthma 11, 5.
The consumption of fast foods that are high-calorie and rich in sodium and saturated/trans-fat is associated with the symptoms of wheezing and bronchial hyperresponsiveness in children; thus, diet plays an important role in the pathogenesis of asthma 5, 48.
The risk of asthma and increased asthma severity in all participants with high sodium intake was approximately two-fold higher. For overweight children, this risk was three-fold higher. Corroborating these findings, previous studies showed that the addition of salt to food led to a greater than two-fold increased risk for wheezing and asthma 48. The consumption of snacks (three times/week) was associated with asthma symptoms at a risk of 4.8 5.
The influence of sodium on asthma is not yet fully understood. One potential explanation is that sodium exacerbates asthma via the hypersensitization of bronchial smooth muscle. This muscle is permeable to sodium, and an increase in sodium consumption leads to muscle hyperpolarization. Another hypothesis is that the asthma is associated with increased ouabain, an inhibitor of the sodium/potassium pump that causes sodium intracellular accumulation. However, the relationship between the response capacity of the airways (asthma) and urinary sodium excretion (an indicator of intake) in response to increased sodium intake is unclear 12. Based on the currently available evidence, we cannot conclude that reducing salt in the diet will help treat or manage asthma symptoms; however, sodium intake among asthmatics should abide by the recommendations available for the age-group of the patient 17.
During adolescence, many changes occur that influence the development of particular lifestyles, including nutrition and physical activity. We are the first to report a Brazilian study that specifically investigated the prevalence of asthma based on the sodium intake of adolescents; however, one limitation of the study is that the effects of asthma protective nutrients as potential confounders were not assessed. Another limitation is the lack of uniform criteria for the definition of asthma across studies. The ISAAC questionnaire is widely used for this purpose because of its specificity; however, the symptoms obtained by this survey are subject to biases because the information is self-reported.
Conclusions
This study found no differences between adolescents with asthma and obesity; however, the prevalence of asthma was higher in overweight females. There was a higher risk for females and those with a high sodium intake for asthma and severe asthma, respectively. Among overweight adolescents, there was an additional risk for females and those with a high sodium intake. Asthma and obesity are multifactorial diseases with environmental factors that are significant predictors of quality of life. Thus, we suggest further research is needed to clarify the relationship between asthma and obesity and the influence of sodiumon this association. Adolescents need to learn about these risk factors because school is an ideal location for health promotion and risk prevention.
References
- 1.Asher M I, Keil U, Anderson H R, Beasley R, Crane J. (1995) . International Study of Asthma and Allergies in Childhood (ISAAC): rationale and methods. The European respiratory journal 8(3), 483-91.
- 3.Musaad S M, Patterson T, Ericksen M, Lindsey M, Dietrich K.Comparison of anthropometric measures of obesity in childhood allergic asthma: central obesity is most relevant. The Journal of allergy and clinical immunology. 123(6), 1321-7.
- 4.Romieu I, Mannino D M, Redd S C, McGeehin M A.Dietary intake, physical activity, body mass index, and childhood asthma. in the Third National Health And Nutrition Survey (NHANES III). Pediatric pulmonology 38(1), 31-42.
- 5.Arvaniti F, Priftis K N, Papadimitriou A, Yiallouros P, Kapsokefalou M.Salty-snack eating, television or video-game viewing, and asthma symptoms among 10- to 12-year-old children: the PANACEA study. , Journal of the American Dietetic Association 111(2), 251-7.
- 6.Kumanyika S K.Minisymposium on obesity: overview and some strategic considerations. Annual review of public health. 2001, 293-308.
- 7.Monteiro C A, Mondini L, de Souza AL, Popkin B M. (1995) The nutrition transition in Brazil. European journal of clinical nutrition. 49(2), 105-13.
- 8.Lang D M, Butz A M, Duggan A K, Serwint J R. (2004) Physical activity in urban school-aged children with asthma. Pediatrics.
- 9.Tsai H J, Tsai A C, Nriagu J, Ghosh D, Gong M.Associations of BMI, TV-watching time, and physical activity on respiratory symptoms and asthma in 5th grade schoolchildren in Taipei, Taiwan. The Journal of asthma : official journal of the Association for the Care of Asthma. 44(5), 397-401.
- 10.Bar-on M E.The effects of television on child health: implications and recommendations. Archives of disease in childhood. 83(4), 289-92.
- 11.Wiecha J L, Peterson K E, Ludwig D S, Kim J.Sobol A et al. When children eat what they watch: impact of television viewing on dietary intake in youth. Archives of pediatrics & adolescent medicine 160(4), 436-42.
- 12.Hirota S A, Janssen L J.Sodium and asthma: something borrowed, something new? American journal of physiology Lung cellular and molecular physiology. 2007-293.
- 14.Frisancho A R.Triceps skin fold and upper arm muscle size norms for assessment of nutritional status. The American journal of clinical nutrition. 27(10), 1052-8.
- 15.Taylor R W, Jones I E, Williams S M, Goulding A.Evaluation of waist circumference, waist-to-hip ratio, and the conicity index as screening tools for high trunk fat mass, as measured by dual-energy X-ray absorptiometry, in children aged 3-19 y. The American journal of clinical nutrition. 72(2), 490-5.
- 16.Ferreira-Sae M C, Gallani M C, Nadruz W, Rodrigues R C, Franchini K G.Reliability and validity of a semi-quantitative FFQ for sodium intake in low-income and low-literacy Brazilian hypertensive subjects. Public health nutrition. 12(11), 2168-73.
- 17.Food, Board Nutrition. (2000) Dietary reference intakes for water, potassium, sodium, chloride, and sulfate. Washington (DC): National Academy Press;.
- 18.Sarno F, Claro R M, Levy R B, Bandoni D H, SRG Ferreira.Estimativa de consumo de sódio pela população brasileira, 2002-2003. Revista de Saúde Pública. 2009, 219-25.
- 19.GVd Veiga, RSd Costa, Araújo M C, AdM Souza, Bezerra I N.Inadequação do consumo de nutrientes entre adolescentes brasileiros. Revista de Saúde Pública. 2013, 212-21.
- 20.Guedes D P, Lopes C C, JERP Guedes.Reprodutibilidade e validade do Questionário Internacional de Atividade Física em adolescentes. Revista Brasileira de Medicina do Esporte. 2005, 151-8.
- 21.Sole D, Vanna A T, Yamada E, Rizzo M C, Naspitz C K. (1999) International Study of Asthma and Allergies in Childhood (ISAAC) written questionnaire: validation of the asthma component among Brazilian children. , Journal of investigational allergology & clinical immunology 8(6), 376-82.
- 22.Wandalsen N F, Gonzalez C, Wandalsen G F, Solé D.Avaliação de critérios para o diagnóstico de asma através de um questionário epidemiológico. , Jornal Brasileiro de Pneumologia 2009, 199-205.
- 23.Mallol J, Crane J, E von Mutius, Odhiambo J, Keil U.The International Study of Asthma and Allergies in Childhood (ISAAC) Phase Three: a global synthesis. Allergologia et immunopathologia. 41(2), 73-85.
- 24.SdM Simões, SSd Cunha, Barreto M L, ÁA Cruz.Distribuição da gravidade da asma na infância. , Jornal de Pediatria 2010, 417-23.
- 25.Visness C M, London S J, Zeldin D C.Association of Childhood Obesity with Atopic and Non-Atopic Asthma: Results from the National Health and Nutrition Examination Survey 1999-2006. , The Journal of Asthma 47(7), 822-9.
- 26.Okabe Y, Itazawa T, Adachi Y, Yoshida K, Ohya Y. (2011) Association of overweight with asthma symptoms in Japanese school children. Pediatrics International. 53-2.
- 27.Chu Y-T, Chen W-Y, Wang T-N, Tseng H-I, Wu J-R.Extreme BMI predicts higher asthma prevalence and is associated with lung function impairment in school-aged children. Pediatric pulmonology. 44(5), 472-9.
- 28.Cibella F, Cuttitta G, La Grutta S, Melis M R, Bucchieri S.A cross-sectional study assessing the relationship between BMI, asthma, atopy, and eNO among schoolchildren. Annals of allergy, asthma & immunology : official publication of the American College of Allergy, Asthma. , Immunology 107(4), 330-6.
- 29.Leung T F, Kong A P, Chan I H, Choi K C, Ho C S.Association between obesity and atopy in Chinese schoolchildren. International archives of allergy and immunology. 149(2), 133-40.
- 30.MdPC Bertolace, Toledo E, PPdO Jorge, Liberatore Junior RDR.Association between obesity and asthma among teenagers. , Sao Paulo Medical Journal 2008, 285-7.
- 31.Kattan M, Kumar R, Bloomberg G R, Mitchell H E.Calatroni A et al. Asthma control, adiposity, and adipokines among inner-city adolescents. The Journal of allergy and clinical immunology. 125(3), 584-92.
- 32.Ginde A A, Santillan A A, Clark S, Camargo C A.Body mass index and acute asthma severity among children presenting to the emergency department. Pediatric allergy and immunology : official publication of the European Society of Pediatric Allergy and Immunology. 21(3), 480-8.
- 33.Lang J E, Hossain J, Dixon A E, Shade D, Wise R A et al.Does age impact the obese asthma phenotype? Longitudinal asthma control, airway function, and airflow perception among mild persistent asthmatics. , Chest 140(6), 1524-33.
- 34.Hom J, Morley E J, Sasso P, Sinert R.Body mass index and pediatric asthma outcomes. Pediatric emergency care. 25(9), 569-71.
- 35.Peters J I, McKinney J M, Smith B, Wood P, Forkner E.Impact of obesity in asthma: evidence from a large prospective disease management study. , Annals of allergy, asthma & immunology : official publication of the American College of Allergy, Asthma, & Immunology 106(1), 30-5.
- 36.Overweight.obesity, and incident asthma: a meta-analysis of prospective epidemiologic studies. American journal of respiratory and critical care medicine. 175(7), 661-6.
- 37.Chen Y C, Dong G H, Lin K C, Lee Y L.Gender difference of childhood overweight and obesity in predicting the risk of incident asthma: a systematic review and meta-analysis. Obesity reviews : an official journal of the International Association for the Study of Obesity. 14(3), 222-31.
- 38.Moreau D, Kalaboka S, Choquet M, Annesi-Maesano I Asthma.obesity, and eating behaviors according to the diagnostic and statistical manual of mental disorders IV in a large population-based sample of adolescents. The American journal of clinical nutrition. 89(5), 1292-8.
- 39.He Q Q, Wong T W, Du L, Jiang Z Q, Qiu H.. Respiratory health in overweight and obese Chinese children. Pediatric pulmonology 44(10), 997-1002.
- 40.Hancox R J, Milne B J, Poulton R, Taylor D R, Greene J M.Sex differences in the relation between body mass index and asthma and atopy in a birth cohort. American journal of respiratory and critical care medicine. 171(5), 440-5.
- 41.Zemel B S, Riley E M, Stallings V A. (1997) Evaluation of methodology for nutritional assessment in children: anthropometry, body composition, and energy expenditure. Annual review of nutrition. 1997, 211-35.
- 42.Papoutsakis C, Priftis K N, Drakouli M, Prifti S, Konstantaki E.Childhood overweight/obesity and asthma: is there a link? A systematic review of recent epidemiologic evidence. , Journal of the Academy of Nutrition and Dietetics 113(1), 77-105.
- 43.Baum W F, Schneyer U, Lantzsch A M, Kloditz E.Delay of growth and development in children with bronchial asthma, atopic dermatitis and allergic rhinitis. Experimental and clinical endocrinology & diabetes : official journal, German Society of Endocrinology [and] German Diabetes Association 110(2), 53-9.
- 44.Sant´Anna C A, Solé D, Naspitz C K.Short stature in children with respiratory allergy. Pedriatric Allergy and Immunology. 7(4), 187-92.
- 45.Arend E E, Fischer G B, Mocelin H, Medeiros L.Corticóide inalatório: efeitos no crescimento e na supressão adrenal. , Jornal Brasileiro de Pneumologia 2005, 341-9.
- 46.Bensch G W, Greos L S, Gawchik S, Kpamegan E, Newman K B.Linear growth and bone maturation are unaffected by 1 year of therapy with inhaled flunisolide hydrofluoroalkane in prepubescent children with mild persistent asthma: a randomized, double-blind, placebo-controlled trial. Annals of allergy, asthma & immunology : official publication of the American College of Allergy, Asthma. , Immunology 107(4), 323-9.
- 47.Benedetti F J, Mocelin H T, Bosa V L, de Mello ED, Fischer G B.Energy expenditure and estimated caloric intake in asthmatic adolescents with excess body weight. Nutrition. , Burbank, Los Angeles, County, Calif) 26(10), 952-7.
- 48.Corbo G M, Forastiere F, M De Sario, Brunetti L, Bonci E.Wheeze and asthma in children: associations with body mass index, sports, television viewing, and diet. , Epidemiology 19(5), 747-55.
