Abstract
Biogenic amines, which are responsible for the realization of many physiological conditions of our body, are compounds that can be produced by microorganisms especially in fermented foods with high protein content. They can have harmful effects on human health only when taken in high amounts with food. However, in individuals with impaired anti-toxic metabolism, which is responsible for detoxification, even lower amounts may cause toxic effects. The most common health effects are nausea, vomiting, severe headaches, hypotension, hypertension, tachycardia, various allergic reactions, abdominal pain and death in more severe cases. For these reasons, legislations on biogenic amines in foods have been established with some restrictions. Food producers have been asked to comply with these legislations. However, despite all precautions, biogenic amines in foods have not been completely removed. Further research is still needed to find effective solutions to prevent biogenic amine formation. In addition, consumers need to be made aware of this issue.
Author Contributions
Academic Editor: Mahipal Singh Sankhla, Galgotias University, Department of Forensic Science, India
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Fatih Ozcelik, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interest exists.
Citation:
Introduction
Fermented foods that are heat treated such as baking, pasteurization and sterilization, or produced by the ripening of foods because of the metabolic activities of microorganisms are consumed quite extensively todays. Fermented foods have been preferred due to their low pH, high salt concentration, long shelf life due to their low moisture content, unique flavors and colors. In addition, health-beneficial microorganisms found in fermented foods have led to an increase in preference. The digestibility of foods and the usefulness of amino acids increase because of the activities of these microorganisms. Some fermented foods may also exhibit probiotic properties. Yogurt, cheese and kefir made from milk; sausage, bacon and surströmming (fermented herring) made from meat products; pickles, sauerkraut (fermented cabbage) and miso (fermented soybeans) made from various vegetables and fruits; tarhana and bread made from cereal products; beer and wine are widely consumed foods that can be given as examples of these fermented foods. However, biogenic amines present in these foods or formed after fermentation may show toxic effects 1, 2, 3, 4, 5. Generally, even if they are toxic only when taken in large quantities, even lower amounts can cause serious health problems in people with impaired detoxification metabolism. Therefore, it is very important to know the physiological and pathological effects of biogenic amines that are naturally produced in our body.
The potential of biogenic amine formation of microorganisms in fermented foods can be quite different. This is related to decarboxylase enzyme activities of microorganisms. Therefore, detection of bacteria with decarboxylase enzyme activity is important to prevent the accumulation of biogenic amines in foods. Furthermore, these harmful compounds need to be known both for human health and for the quality of food. Today, in many countries, there are legislations on the content and amounts of biogenic amine in fermented products. These legislations and regulations are mostly aimed at preventing the accumulation of biogenic amine in food products. They are manufacturer oriented 1, 4, 5. However, these measures did not completely limit the formation of biogenic amine. Also increasing consumer awareness seems to be one of the most effective solutions in this regard.
Biogenic Amines
Biogenic amines are organic nitrogen compounds of small molecular weight. Biogenic amines are formed because of decarboxylation of amino acids during the conversion of carbohydrate units of lactic acid and organic compounds by microorganisms that enable the ripening of foods (fermentation). When taken in high amounts, they cause food intoxication. Toxic amounts of biogenic amines are produced during processing, preparation and storage of protein-rich plant and animal products. Natural biogenic amines in animal and human cells are formed by decarboxylation of amino acids or amination and transamination of aldehydes and ketones. They are especially molecules having aromatic and heterocyclic structures. Five of the natural biogenic amines (dopamine, norepinephrine, epinephrine, histamine and serotonin) play important roles in the body as neurotransmitters 6, 7, 8. However, they can cause harmful effects when taken with high amounts of food.
A suitable medium is required for decarboxylation reactions during the formation of biogenic amines. This medium is provided by the processes of preparation of foodstuffs, such as the presence of free amino acids, microbial contamination and heat treatment. Biogenic amines (such as adrenaline, histamine, tyramine, and serotonin) involved in normal physiological processes should be evaluated differently from toxic biological amines 6, 9, 10, 11. All catechol-derived biogenic amines are composed of tyrosine amino acid that is a common precursor. Biogenic amines can be detected very precisely by GS-MS or HPLC 12.
Dopamine
Dopamine, an important biogenic amine, responsible for the sense of motivation and reward, is synthesized by decarboxylation of 3,4-dihydroxyphenylalanine (Figure 1). Dopamine is commonly found in the corpus striatum, which plays an important role in the coordination of the body's motor movements 13, 14. This region is thought to be degenerate in Alzheimer's disease. Therefore, in order to compensate for the reduced dopamine, L-dopa is given which can cross the blood-brain barrier in treatment. However, the state of the decarboxylase enzymes that converts dopamine molecules and the peripheral catabolism of L-dopa should not be ignored. As is known, as soon as L-dopa is taken, it is rapidly catabolized in the intestine and peripheral tissues. Therefore, L-dopa is given together with carbidopa, a dopamine decarboxylase inhibitor that does not cross the blood-brain barrier, and selegiline, a monoamine oxidase (MAO) inhibitor 15, 16. However, hydrazine, a degradation product of carbidopa, is genotoxic and possibly carcinogenic. Dopamine is also used in shock treatment because it enlarges the renal arteries and activates β-adrenergic receptors in the heart and increases cardiac output.
Figure 1.Biogenic amine neurotransmitters synthesized from tyrosine amino acids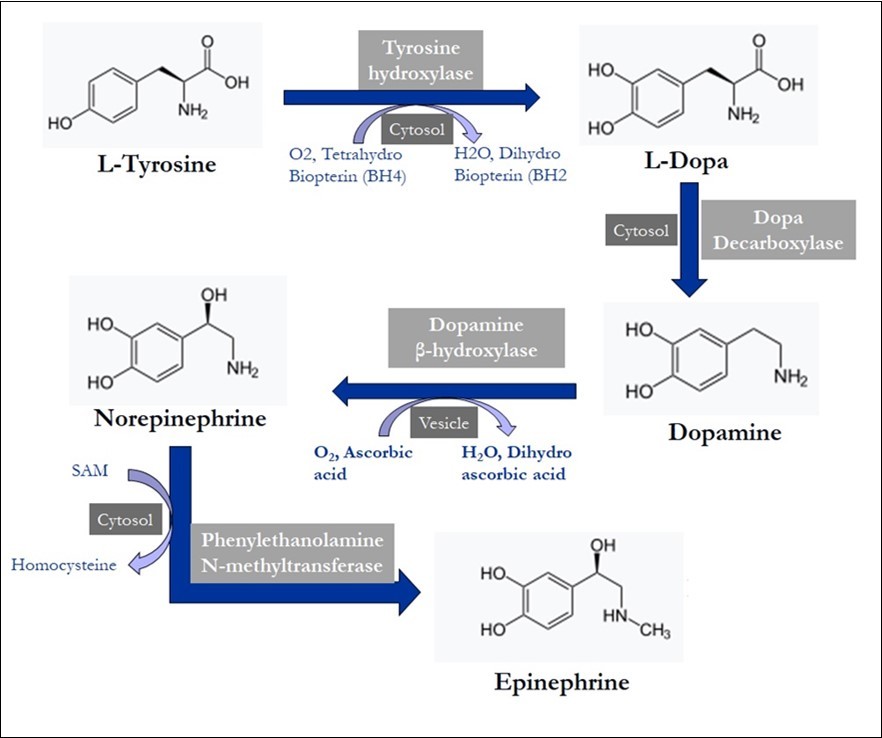
In a study investigating renal effects of biogenic amines; It has been found that dopamine and 5-hydroxytryptamine are converted to each other by the aromatic L-amino acid decarboxylase enzyme in the renal tubular cells 17. L-dopa and dopamine affect the transfer of Na+ outside the cell and K+ inside the cell by reversible inhibition of Na+-K+ ATPase. These vasoactive amines act on dopaminergic receptors in the renal and coronary vessels, increasing blood flow without altering blood pressure. Therefore, they accelerate glomerular filtration while increasing renal blood flow. It also inhibits aldosterone synthesis and Na-K ATPase pumps, resulting in natriuresis 18, 19. This makes it difficult to keep the water in the intravascular area. This also suggests that, biogenic amines taken through food can lead to blood pressure changes and renal effects, through intrarenal transformations. This will cause more serious health problems in patients with impaired anti-toxic metabolism, hypotensive/hypertensive patients, and those with heart or renal disease.
Dopamine levels are associated with some urological diseases. For example, bupropion, a norepinephrine-dopamine reuptake inhibitor (NDRI), was found to reduce sperm motility in rats when taken at a dose of ≥30 mg/kg. However, it has been reported that methylphenidate, an NDRI, increases the rate of sperm with abnormal tail morphology in rats, increases the number of spermatogonia and sperm, and reduces the number of round spermatids. In addition, sibutramine has been reported to shorten the passage of sperm from the epididymis and reduce the total number of sperm in the epididymis, in rats 20. Chen G. et al. demonstrated that disruption of the Dopamine-2 receptor pathway in the basolateral amygdala in rats may contribute to the development of non-organic erectile dysfunction (ED) and that dopamine-2 receptor agonists may rehabilitate ED in rat models with non-organic ED 21.
Norepinephrine
Another biogenic amine, norepinephrine, is synthesized from dopamine by the dopamine β-hydroxylase enzyme found in the vesicles. From here, it is transported to the adrenergic nerve terminals. The most prominent class of neurons expressing norepinephrine are sympathetic ganglion cells 13, 14, 22. Norepinephrine is also an important transmitter of locus coeruleus, a brain stem core associated with sleep and alertness, attention and feeding behavior.As shown in Figure 2, norepinephrine is a neurotransmitter acting on smooth muscle in postganglionic sympathetic neurons, causing contraction or relaxation depending on the type of receptors. Dopamine is a neurotransmitter that modulates cholinergic transmission in autonomic ganglia. It also inhibits Na+ and H2O reabsorption in the kidneys and causes renal vasodilatation 23, 24, 25. Epinephrine and norepinephrine are neurohumoral agents that are released into the bloodstream by the adrenal medulla. The amount of epinephrine released is quite high compared to norepinephrine. All these biogenic amines are essential for the physiological functions of the body. However, when taken in high amounts, they cause serious problems.
Figure 2.Location of norepinephrine (NE), epinephrine (E) and acetylcholine (ACh) at preganglionic nerve and postganglionic nerve endings in sympathetic nervous system.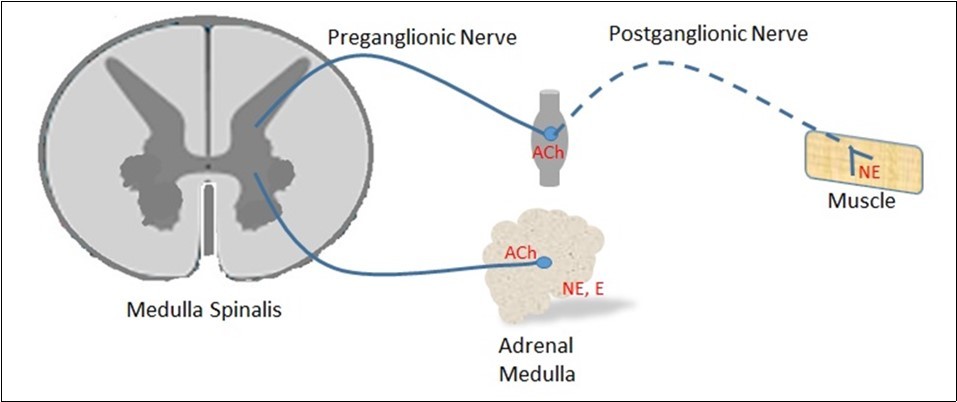
Excess norepinephrine in various tissues has been associated with some diseases. For example, In a study by Stein et al., It was reported that norepinephrine increased in the urine of patients with interstitial cystitis 26. Diederichs et al. have reported that intracavernous administration of norepinephrine (in a dose-dependent manner) reduced penile erection due to cavernous nerve. Sympathetic stimulation reduces or completely eliminates penile erection induced by acetylcholine or vasoactive intestinal polypeptide. It has been concluded that norepinephrine is an important neurotransmitter in the control of penile detumescence 27.
Epinephrine
Epinephrine is present in the brain at lower levels than other catecholamines. It is also called adrenaline. Phenylethanolamine-N methyl transferase, which synthesizes epinephrine, is present only in neurons that secrete epinephrine. The function of epinephrine-containing neurons in the central nervous system is not fully understood. There are two main enzymes involved in the catabolism of catecholamines, mitochondrial monoamine oxidase (MAO) and cytoplasmic catechol O methyl transferase (COMT) in neurons and glial cells. Inhibitors of these enzymes are used as antidepressants in psychiatry clinics 22, 28, 29. Especially, it is produced by the adrenal medulla, which is a postsynaptic ganglion organ, reaching the peripheral tissues through circulation. It is produced by the adrenal medulla, which is a postsynaptic ganglion, and reaches to peripheral tissues through circulation.
Epinephrine has many vital effects and has place in treatments. For example, it is still used successfully in the treatment of emergency priapism cases 30. In addition, intravesical administration of epinephrine has been inhibited hemorrhage and inflammation in bladder in a rat model that has cyclophosphamide-induced hemorrhagic cystitis 31.
Histamine
Histamine is a biogenic amine synthesized from histidine by histidine decarboxylase. Histamine methyltransferase and MAO are involved in its catabolism. Histamine and histidine decarboxylase are found in neurons in the hypothalamus and histamine receptors are intensely found in neurons in almost all regions of the brain and spinal cord. Histamine mediates arousal and attention in the central nervous system, as does acetylcholine and norepinephrine 32, 33. In peripheral tissues, histamine is released from mast cells in response to tissue damage and allergic reactions. Histamine is also effective in modulating bladder contraction. Stromberga et al. reported that the stimulation of H1 histaminic receptors caused contractions in both the lamina propria and detrusor tissue of the urinary bladder. Activation of H2 histaminic receptors is only prevents H1 receptor-mediated contractions in the lamina propria, not in detrusor tissue. Based on these results, H1 and H2 receptors may be potential targets in the future, in the treatment of overactive bladder and other bladder contraction disorders 34.
Serotonin
Serotonin (5-hydroxytryptamine) is synthesized from tryptophan, an essential amino acid (Figure 3). Tryptophan is taken into the neurons by a membrane transporter and is initially hydroxylated by the enzyme tryptophan-5-hydroxylase. This stage is the control step of serotonin synthesis. Then, this hydroxylated form is decarboxylated and the synthesis of serotonin is completed. Serotonin is found intensely in neurons in the pons and brain stem. It has an important role in sleeping and staying awake. Its effects in the synaptic range are terminated by withdrawing from the intersynaptic junction, like other biogenic amines. Many antipsychotic drugs used in the treatment of depression and anxiety are thought to have serotonergic effects 22, 35, 36. Mitochondrial MAO has an important role in its catabolism. The NE and 5-HT monoaminergic systems play an important role in controlling a wide range of behavioral and physiological processes, including sleep, cognition, and mood. Homeostasis of central NE and 5-HT neurons is achieved by signal controls of the NE and 5-HT autoreceptors. In addition to its neuropsychiatric effects, serotonin has many associations with other systems and diseases. For example, Siddiqui et al. have reported that blockade of 5-Hydroxytryptamine 1A (5HT1A) receptors and, to a greater extent, 5-Hydroxytryptamine 1B (5HT1B) receptors significantly inhibits bladder cancer cell growth and may be used in the treatment of bladder cancer 37.
Figure 3.Synthesis of 5-hydroxytryptamine from tryptophan amino acid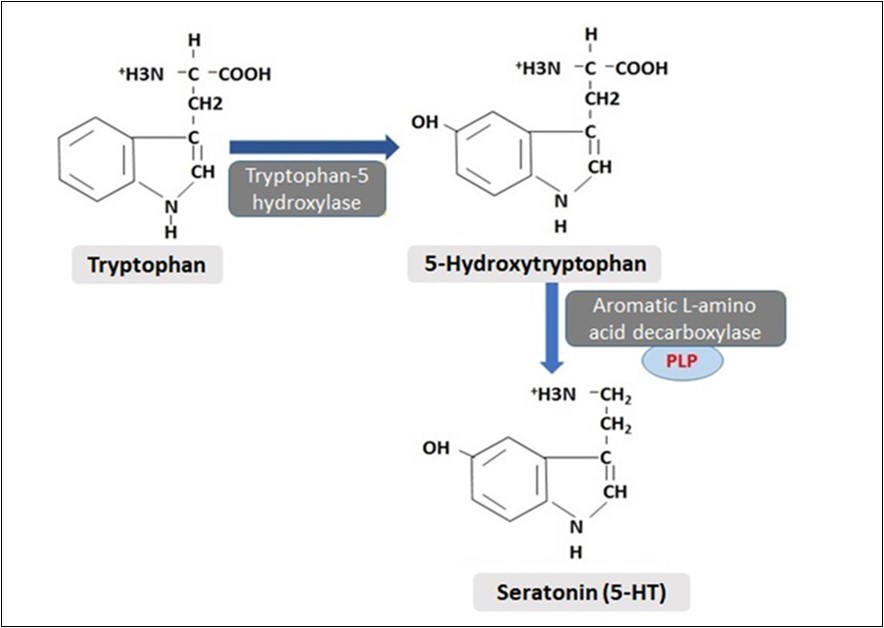
Discussion
Although biogenic amines are present in low concentrations in fresh foods, they can lead to higher effects than expected, especially when they taken with protein-rich animal foods. Biogenic amines in processed fermented foods, biogenic amines formed by microbial decarboxylation in foods, biogenic amines formed by deamination of aldehydes and ketones can also increase blood levels to high levels 38, 39. As in high protein foods such as fermentation or heat-treated fish and fish products, meat and meat products, milk and milk products; Low protein foods such as wine, beer, vegetables, fruits may also contain high levels of biogenic amine 38, 40.
In addition to natural biogenic amines, there are many different types of biogenic amines that have risk of oral intake. The most important of these are cadaverine, putrescine, spermine, spermidine, agmatine, phenylethylamine, tyramine, tryptamine, histamine, cadaverine, agmatine, spermine and spermidine 41, 42. Each of these biogenic amines, even when taken in high amounts alone, have potential toxic risks. Moreover, the toxic effects are usually due to the effect of several biogenic amines rather than a single biogenic amine. The most toxic of these biogenic amines and the most important in terms of food safety are histamine and tyramine 43. They are mostly produced by fermented foods and intensive microbial activity. When histamine is taken at high concentrations, it causes adrenaline and noradrenaline release, stimulation of smooth muscles, stimulation of sensory and motor neurons, hypotension, and allergic reactions. Tyramine causes hypertension, tachycardia, hypercapnia, hyperglycemia and headache.
Some biogenic amines provide aroma and taste to foodstuffs. This is known as esterification. Some can react with nitrites to convert to carcinogenic substances (such as nitrosamine). However, they do not pose a serious health hazard unless biogenic amines are taken at a high rate and the individual's monoamine oxidase and other natural antitoxic catabolism (such as cytochrome P450 activity) deteriorates. In addition, many biogenic amines are neutralized while it was yet in the intestine. Because they are inactivated by the intestinal flora. In the first stage, liver metabolizes histamine and many biogenic amines taken by food in the first stage. However, harmful effects may occur when exposed to high levels of biogenic amines. These effects include headache, dizziness, nausea, vomiting, diarrhea, intestinal distension and hypertension. The simplest way to protect from biogenic amines taken with food is to consume food fresh without spoiling. Especially histamine, cadaverine, putrescine, tyramine and spermidine concentrations are used as indicators of freshness in foods. Therefore, it should be ensured that processed fermented products and foods with long storage life are not consumed intensively 8, 44, 45, 46. In addition peoples who have hypertension, lung and heart disease, vitamin deficiency (Vitamin B12, folate deficiency), inflammatory bowel disease (Crohn's disease and ulcerative colitis), immune system insufficiency (Because they contain less amino oxidase enzyme in the intestine) are, must avoid foods that have high biogenic amines levels because they are more sensitive to biogenic amines 7, 47.
Aromatic amines were first identified as carcinogens in the paint industry. Especially, 4-aminobiphenyl and 2-naphthylamine are well-known human bladder carcinogens. These heterocyclic aromatic amines are protein pyrolysis products which are also found in over-fried foods and tobacco smoke 48. Tobacco use and exposure to occupational aromatic amines are two identified environmental risk factors for bladder cancer, and the prevention of exposure to them has contributed significantly to the reduction of mortality, especially among men 49. The International agency for research on cancer (IARC) has reported that in most countries, in cancers related with cigarette which is aromatic amine supply, the attributed risk had reached up to 50% in men and 25% in women 50. Moreover, it has been reported that one of the most well-defined risk factors in renal cell carcinoma is cigarette, the source of aromatic amine 51. In addition, animal studies have shown that α-difluoromethylornithine, a polyamine biosynthesis inhibitor, can inhibit skin tumor formation 52.
Sıddıqui et al. found that 5-hydroxytryptamine causes proliferation in cells and 5-hydroxytryptamine antagonists cause inhibition of proliferation in various tumor cells such as prostate carcinoma, lung carcinoma and colonic carcinoma 53. In a study which investigating the effects of serotonin on human prostate carcinoma by Abdul et al reported that serotonin, a biogenic amine, increases the growth of prostate tumors and therefore may be a target for treatment 54.
Conclusion
As a result, because of biogenic amines are a predisposing factor for cancer as well as causing many toxic effects, the amounts of biogenic amines in animal and vegetable foods are important. Therefore, there is still a need for researches, which will find effective solutions to prevent biogenic amine formation. In addition, both producers and consumers should be made aware of this issue.
Disclosure
There is no authors’ potential conflict of interest in this paper. The authors received no financial support for this study.
References
- 1.Spano G, Russo P, Lonvaud-Funel A, Lucas P.Alexandre H,et al.(2010). Biogenic amines in fermented foods.Eur. , J. Clin. Nutr.Eur J Clin Nutr 64, 95-100.
- 2.Linares D M, Martín M, Ladero V, Alvarez M A, Fernández M. (2011) . Biogenic Amines in Dairy Products.Crit Rev Food Sci. Nutr.51(7),691–703 .
- 3.Mulaw G, Sisay Tessema T, Muleta D, Tesfaye A. (2019) In Vitro Evaluation of Probiotic Properties of Lactic Acid Bacteria Isolated from Some Traditionally Fermented Ethiopian Food Products.Int. , J. Microbiol.2019,1–11
- 4.Shalaby A R. (1996) Significance of biogenic amines to food safety and human health.Food Res. Int.29(7),675–690
- 5.Bodmer S, Imark C, Kneubühl M. (1999) Biogenic amines in foods: Histamine and food processing.Inflamm. Res.48(6),296–300
- 6.Purves D, Augustine G J, Fitzpatrick D. (2001) . The Biogenic Amines. inNeuroscience(2ndedition. Purves D. et al.) Sunderland (MA):Sinauer Associates,Inc .
- 7.Bardócz S. (1995) Polyamines in food and their consequences for food quality and human health.Trends Food Sci. Technol.6(10),341–346
- 8.Rice S L, Eitenmiller R R, Koehler P E. (1976) Biologically Active Amines in Food: A Review.J. Milk Food Technol.39(5),353–358.doi.org/10.4315/0022-2747-39.5.353.
- 9.Kalač P. (2014) Health effects and occurrence of dietary polyamines: A review for the period 2005-mid 2013.Food Chem.161. 27-39.
- 10.Silla Santos MH. (1996) Biogenic amines: their importance in foods.Int. , J Food Microbiol.29(2-3),213–231
- 11.Ruiz-Capillas C, Herrero A M. (2019) . Impact of Biogenic Amines on Food Quality and Safety.Foods.8(2). pii: E62 .
- 12.Önal A. (2007) A review: Current analytical methods for the determination of biogenic amines in foods.Food Chemistry. 103(4), 1475-1486.
- 13.Doeun D, Davaatseren M, Chung M S. (2017) . Biogenic amines in foods.Food Sci. Biotechnol.26(6),1463–1474
- 14.Nagatsu T, Levitt M, Udenfriend S. (1964) Tyrosine Hydroxylase. The initial step in norepinephrine biosynthesis. , J Biol Chem 239, 2910-2917.
- 16.Thanvi B R. (2004) Long term motor complications of levodopa: clinical features, mechanisms, and management strategies.Postgrad. , Med 80, 452-458.
- 17.Itskovitz H D, Chen Y H, Stier C T. (1988) Reciprocal renal effects of dopamine and 5-hydroxytryptamine formed within the rat kidney.Clin. , Sci.75,503–507
- 18.Aperia A, Bertorello A, Seri I. (1987) Dopamine causes inhibition of Na+-K+-ATPase activity in rat proximal convoluted tubule segments.Am. , J. Physiol. Physiol.252,F39–F45
- 19.Satoh T, Ominato M, Katz A I. (1995) Different Mechanisms of Renal Na-K-ATPase Regulation by Dopamine. in the Proximal and Distal Nephron.Hypertens. Res.18,S137–S140 .
- 20.Beeder L A, Samplaski M K. (2019) Effect of antidepressant medications on semen parameters and male fertility.Int. , J 27(1), 39-46.
- 21.Chen G, Chen J, Yang B, Yu W, Chen Y.Dai Y.(2019) Dopamine D2 receptors in the basolateral amygdala modulate erectile function in a rat model of nonorganic erectile dysfunction.Andrologia.51(1):e13160.
- 22.Goridis C, Rohrer H. (2002) Specification of catecholaminergic and serotonergic neurons.Nat. , Rev. Neurosci.3,531–541
- 24.Rome A, Bell C. (1983) Catecholamines in the sympathetic nervous system of the domestic fowl.J. , Auton. Nerv 8(4), 331-342.
- 26.Stein P C, Torri A, Parsons C L. (1999) . Elevated urinary norepinephrine in interstitial cystitis.Urology53,1140–1143
- 27.Diederichs W, Stief C G, Lue T F, Tanagho E A. (1990) . Norepinephrine Involvement in Penile Detumescence.J. Urol.143,1264–1266 .
- 29.Feighner J P. (1999) Mechanism of action of antidepressant medications.J. Clin. Psychiatry60 Suppl 4,4–11; discussion 12-3.
- 30.Molina L, Bejany D, Lynne C M, Politano V A. (1989) Diluted Epinephrine Solution for the Treatment of Priapism.J. , Urol,141: 1127-1128.
- 31.Chow Y C, Yang S, Huang C J, Tzen C Y.Huang PL,et al.(2006) Epinephrine promotes hemostasis in rats with cyclophosphamide-induced hemorrhagic cystitis.Urology67(3),636-641.
- 32.Watanabe T, Taguchi Y, Maeyama K, Wada H. (1991) Formation of Histamine: Histidine Decarboxylase. inHistamine and histamine antagonists(eds. Arrang, J.-M. & Uvnas, B.) 145-163.
- 33.Krusong K, Ercan-Sencicek A G, Xu M, Ohtsu H.Anderson GM,et al.(2011) High levels of histidine decarboxylase in the striatum of mice andrats.Neurosci. 495(2), 110-114.
- 34.Stromberga Z, Chess-Williams R, Moro C. (2019) Histamine modulation of urinary bladder urothelium, lamina propria and detrusor contractile activity via H1 and H2 receptors.Sci. Rep.9(1),3899
- 35.Yohn C N, Gergues M M, Samuels B A. (2017) The role of 5-HT receptors in depression.Mol. Brain10(1),28
- 36.Launay J M, Schneider B, Loric S, Da Prada M, Kellermann O. (2006) Serotonin transport and serotonin transporter-mediated antidepressant recognition are controlled by 5-HT 2B receptor signaling in serotonergic neuronal cells.FASEB J.20(11),1843–1854.
- 37.Siddiqui E J, Shabbir M A, Mikhailidis D P, Mumtaz F H, Thompson C S. (2006) The effect of serotonin and serotonin antagonists on bladder cancer cell proliferation.BJU Int.97(3). 634-639.
- 38.Ten Brink B1, Damink C, Joosten H M. (1990) Huis in 't Veld JH. Occurrence and formation of biologically active amines in foods.Int. , J Food 11(1), 73-84.
- 40.Flick G J, Granata L A. (2004) Biogenic amines in foods. inToxins. in Food(eds. Dabrowski, W. M. & Sikorski, Z. E.),CRCPress,121–154
- 41.Halász A, Baráth Á, Simon-Sarkadi L, Holzapfel W. (1994) Biogenic amines and their production by microorganisms in food.Trends Food Sci. Technol.5, 42–49
- 43.Food European. (2011) Scientific Opinion on risk based control of biogenic amine formation in fermented foods.EFSA. J.9 2393.
- 44.Stratton J E, Hutkins R W, Taylor S L. (1991) Biogenic Amines in Cheese and other Fermented Foods: A Review.J Food Prot.54(6),460-470.
- 45.Shalaby A R. (1996) Significance of biogenic amines to food safety and human health.Food Res. Int.29(7),675–690
- 46.Taylor S L, Eitenmiller R R. (1986) Histamine Food Poisoning: Toxicology and Clinical Aspects.CRC Crit Rev Toxicol.17(2),91–128.
- 47.MBR Rodriguez, S Carneiro C da, S Feijó MB da, CAC Júnior, Mano S B. (2014) Bioactive Amines: Aspects of Quality and Safety in Food.Food Nutr. 5(2), 138-146.
- 49.Pelucchi C, Bosetti C, Negri E, Malvezzi M, La Vecchia C. (2006) Mechanisms of Disease: the epidemiology of bladder cancer.Nat. Clin. Pract. Urol.3(6),327–340
- 50.IARC. (2004) Monographs on the Evaluation of Carcinogenic Risks to Humans. Tobacco Smoke and Involuntary Smoking. IARC Working Group on the Evaluation of Carcinogenic Risks to Humans,Lyon 83.
- 51.McLaughlin J K, Lipworth L, Tarone R E, Blot W J. (2006) Renal cancer. In:. Schottenfeld D, Fraumeni JF Jr, editors.Cancer Epidemiology and Prevention. Third Edition.New York, NY:Oxford University 1087-1100.
- 52.Takigawa M, Simsiman R C, Boutwell R K, Verma A K. (1983) Inhibition of mouse skin tumor promotion and of promoter-stimulated epidermal polyamine biosynthesis by α-difluoromethylornithine. Cancer Res. 43(8), 3732-3738.
Cited by (7)
- 1.Wang Lingyun, Xin Shuqi, Zhang Chufeng, Ran Xueguang, Tang Hao, et al, 2021, Development of a novel chromophore reaction-based fluorescent probe for biogenic amines detection, Journal of Materials Chemistry B, 9(45), 9383, 10.1039/D1TB01791H
- 2.Omidiran Adebukola Tolulope, Jenfa Mary Damilola, 2023, , , (), 539, 10.1016/B978-0-323-98341-9.00027-X
- 3.Unabia Romnick B., Reazo Renzo Luis D., Rivera Rolen Brian P., Lapening Melbagrace A., Omping Jahor L., et al, 2024, Dopamine-Functionalized Gold Nanoparticles for Colorimetric Detection of Histamine, ACS Omega, (), 10.1021/acsomega.3c10123
- 4.Bekhit Alaa El-Din A., Holman Benjamin W.B., Giteru Stephen G., Hopkins David L., 2021, Total volatile basic nitrogen (TVB-N) and its role in meat spoilage: A review, Trends in Food Science & Technology, 109(), 280, 10.1016/j.tifs.2021.01.006
- 5.Gurwitz Bracha, Ray Sidhartha D, 2024, , , (), 649, 10.1016/B978-0-12-824315-2.00211-6
- 6.Wójcik Wojciech, Łukasiewicz Monika, Puppel Kamila, 2021, Biogenic amines: formation, action and toxicity – a review, Journal of the Science of Food and Agriculture, 101(7), 2634, 10.1002/jsfa.10928
- 7.Abré Marina Ghislaine, Kouakou-Kouamé Clémentine Amenan, N’guessan Florent Kouadio, Teyssier Corinne, Montet Didier, 2023, Occurrence of biogenic amines and their correlation with bacterial communities in the Ivorian traditional fermented fish adjuevan during the storage, Folia Microbiologica, 68(2), 257, 10.1007/s12223-022-01010-2
