Abstract
PEGylation is a well-established strategy for improving the target specificity, circulation time and stability of liposomes, thereby improving their stealth properties. This brief review provides an insight on the composition of PEGylated liposomes and the characteristics that dictate the functionality of PEGylated liposomes such as surface density, molecular weight, presence of linkers and acyl groups. Physicochemical techniques used to characterize the PEG liposomes and test their stability are also discussed along with their clinical implications. This review provides the readers with a broad range of understanding of various PEGylated lipids, techniques to access their stability in liposomal formulations and state-of -the-art development of PEGylated liposomal formulations.
Author Contributions
Academic Editor: Xiaoyong Lu, Department of Chemistry and Biochemistry, Ohio University, Athens, Ohio, 45701 USA.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Poornima Kalyanram, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Review of PEGylated Lipids
The lipid molecules comprise of a hydrophilic head group and hydrophobic tail region, in presence of an aqueous medium the lipids self-assemble to form liposomes. 1, 2, 3, 4, 5, 6 The presence of hydrophilic aqueous core and hydrophobic lipid bilayer region enables them to encapsulate both hydrophobic and hydrophilic molecules, making them ideal candidates for vaccines 7, 8, 9 and drug carriers10, 11 compared to metal 12, 13, polymer14, 15, 16, and dendrimer 17, 18, 19-based carries. The properties of liposomes can be tuned by inclusion of polymers 20, 21, 22, cholesterol23, 24, 25 or membrane proteins26, 27, choice of lipids and sizing methods to improve their biodegradability, biocompatibility and versatility7, 8, 28 for potential applications in encapsulation of drugs used in cancer therapy. Besides, liposomes can be customized according to size, charge and number of lamellae depending on the applications29, 30. The classification of liposomal drug delivery is broadly divided into conventional and long circulation liposomes.
Conventional and Long Circulating Liposomes
Conventional or the first-generation liposomes were developed commercially in the beginning of 1980s for the delivery of hydrophobic doxorubicin and amphotericin 31, 32. These liposomes comprised of a combination of cationic, anionic and zwitterionic phospholipids in conjunction with cholesterol. However, conventional liposomes were found to attract plasma proteins, also called as opsonin proteins, from the blood stream. The opsonins serve as identification markers for macrophages 33, 34. Opsonins attach themselves electrostatically to the surface of the lipids. The presence of opsonins on the liposomal surface makes them susceptible to macrophage attack and eventually, their removal through the reticuloendothelial system (RES). This phenomenon is also known as liposomal opsonization35, 36, 37. A comprehensive review on opsonization mechanisms of conventional liposomes is previously presented34. Therefore, various strategies have been exploited to induce stealth-ness in liposomes to prevent their opsonization and impart longer circulation in the bloodstream32, 38, 39. One of the commonly used techniques is introduction of steric stabilization by augmenting the liposomal surface with hydrophilic polymers as shown in Figure 135, 40, 41. Figure 1 represents the effects of steric stabilization on RES clearance; opsonins (shown in green) adhere only to conventional liposomes and not to the sterically stabilized liposomes (polymer chains shown in pink). This results in attack of conventional liposomes by the macrophage cells and subsequent clearance by RES. However, sterically stabilized liposomes have longer circulation times and successfully reach the tumor sites. Stealth nature of the liposomes is responsible for longer circulation times. This stealth nature is affected by the polymer’s hydrophilicity, spatial conformation, density and molecular weight 42, 43, 44.
Figure 1.Conventional vs long circulating liposomes and its effect on macrophage clearance
Some commonly reported polymers include PLA (poly lactic acid)42, polystyrene (PSt)43, 44, polyvinyl alcohol (PVA), polyacrylamide43, polysaccharides43, 45, 46, and Polyethylene glycol (PEG). However, PLA and PSt are highly anionic with zeta potentials ranging between −20 and −76 mV, due to the presence of carboxy groups, and these coatings are easily attacked by macrophages47, 48, 49, 50, 51, polysaccharide coatings on the other hand activate the immune system, due to the presence of hydroxyl groups, resulting in elimination52, 53. Liposomes coated with PVA and polyacrylamide copolymers have shown to suffer mechanical degradation and subsequent leaking of contents54.
Amongst the various polymers examined, PEG molecules being hydrophilic, biocompatible, non-immunogenic and uncharged36, 55, 56, 57, 58, 59 has demonstrated potential in steric stabilization. DoxilTM is the benchmark formulation demonstrating the stealth nature of PEG60, 61.
The stealth nature of PEG is attributed due to its hydrophilicity and uncharged surfaces. The inclusion of PEG increases the hydrophilicity and reduces the overall charge or the zeta potential on the surface of liposomes preventing opsonization62–64. This phenomenon has been demonstrated in previous studies, where coating of the PEG on anionic liposomes reduced the zeta potential to a near-neutral value, thereby increasing the circulation65, 66, 67.
PEG (Polyethylene Glycol) Linked Lipids
PEG (Poly-ethylene glycol) is non-ionic68, bio-compatible69, hydrophilic63, and easy to synthesize70 which makes it more favourable. PEGylated lipids consist of PEG chains, a linker, and a hydrophobic anchor 70. One of the ends of the PEG chain in PEGylated lipids is attached to the hydrophobic anchor through the linker.
Components in a PEGylated Lipid
The molecular weight of the PEG chains is usually in the range of 50kDa to 400 Da and can be functionalized by covalent conjugation with folate, biotin, amine, azide, carboxylic acid 71, 72. The functionalization of PEG lipids which is vital to increase its target specificity has been reported extensively 73 and is beyond the focus of this review.
The molecular weight and grafting density affect the conformation of PEG chains on the lipid bilayer surface. At lower grafting density, the PEG molecules assume a lesser interacting mushroom regime with conformed chains, and increasing grafting density, the PEG chains extend and branch out to interact with neighbouring molecules 74, 75. Brush regime is preferred to improve the stealth properties of a liposome as in this regime the interaction of the particle is less and diffuses faster through the tissues than the mushroom regime60. Moreover, opsonins bind predominantly through hydrophobic and electrostatic interactions. Non-ionic PEG chains in brush conformation render the surface of the liposome hydrophilic, reducing the interactions. Furthermore, some adsorbed opsonins tend to compress the brush like chains to mushroom like configuration. This change in energy impedes the attractive nature of opsonins from reaching the surface of the liposomes37, 76.
According to Alexander-deGennes theoretical model of polymer regimes, the transition from mushroom to brush regime depends on the distance (D) between the grafting sites and the length of the random PEG coils (Rf). Rf or Flory dimension is mathematically represented as aN3/5, where N is the degree of polymerization and a is the monomer size 77. The conditions for each of the configuration depicted in Figure 2 is as follows: (i) D > Rf, the PEG chains have very little interaction and follow interdigitated mushroom configuration (ii) Rf < D < 2Rf , a mushroom packing is observed (iii) D ≪ 2Rf, chains are closer and a brush like extended conformation is observed36, 40, 77. In all conformations, PEG chains form a fixed aqueous layer thickness (FALT) around the liposomes. This FALT value controls the regime type and circulation time of liposomes. It is reported that a combination of shorter and long PEG chain lengths help in lengthening the circulation time70.
Figure 2.Regimes in a PEG coated liposome a) Interdigitated Mushroom; b) Mushroom Regime; c) Brush Regime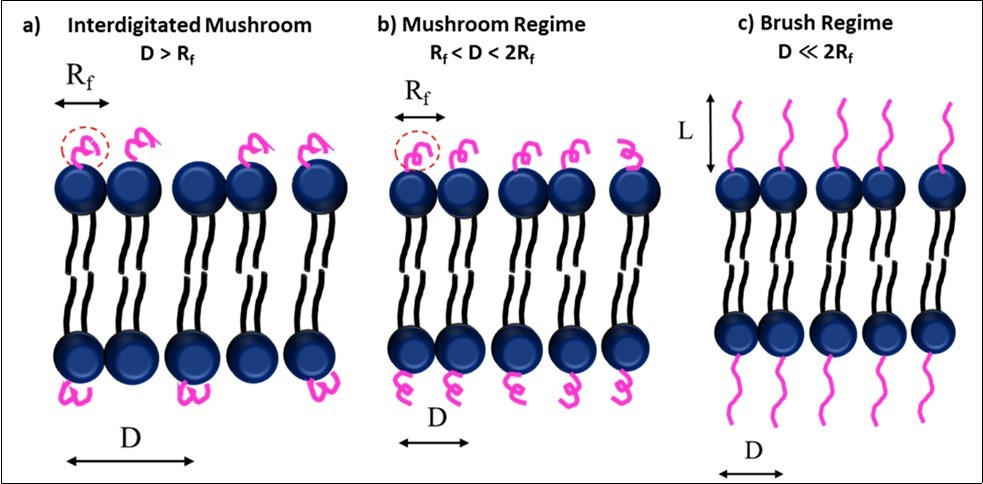
The PEG chains are further attached to the hydrophobic acyl anchor through linker moieties. Common linker moieties include phosphate ester, ether, disulfide, carboxyl ester, amide, and peptide linkages78, 79, 80, 81, 82. The linker moieties affect the surface charge and binding properties of the liposomes. The conventional linker has been the phosphate linker, based on its presence or absence the PEGylated lipids are categorized as PEG-phospholipids and PEG-non-phospholipids 70. The third component of PEG lipids is the hydrophobic anchor comprising of acyl groups that associate into the lipid bilayer with PEG chains branching into the aqueous region. The acyl groups determine the lamellar or micellar morphology of the lipid assemblies20 and the extent of inclusion of PEGylated lipid in the liposome 83, 84. In case of PEG-phospholipids, the acyl group is usually a fatty acid chain such as distearoyl, dipalmitoyl or dimyristoyl that is covalently bonded to the polar phospholipid head group70. These heads groups are then attached to a linear chain methoxy-PEG (mPEG) as shown in Figure 3a. It is found that the length of the mPEG chains which is directly related to the molecular weight influences the FALT values, thereby determining the circulation time. Shorter m-PEG chains (350-750 Da) have shown relatively limited effects to avoid macrophage clearance due to their inherent mushroom configuration. Increasing the mPEG chain length has shown to increase the circulation time85, 86, 87, 88, 89, 90. Additionally, mPEG phospholipids having a mixture of PEG chain lengths in the same molecule (1,2-distearoyl-sn-glycero-3-phosphoethanolamine-PEG2) elongates the circulation time and has increased tumor contact time because of its differing hydrophilic and hydrophobic properties88. It is also been found that concentration of mPEG-phospholipids in a liposome influence the rate of release of drugs. The higher molecular weight mPEG-phospholipids transform a diffusion-controlled drug release to an interfacial-controlled drug release91.
Figure 3.Structure of Phospholipid PEG a) DSPE-PEG2000 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-methoxy(polyethylene glycol); Non-Phospholipid PEG b) C8 PEG2000 Ceramide N-octanoyl-sphingosine-1-{succinylmethoxy(polyethylene glycol)2000}; c) DMG-PEG 2000 1,2-dimyristoyl-rac-glycero-3-methoxypolyethylene glycol-2000; d) Chol-PEG600 Cholesterol-(polyethylene glycol-600)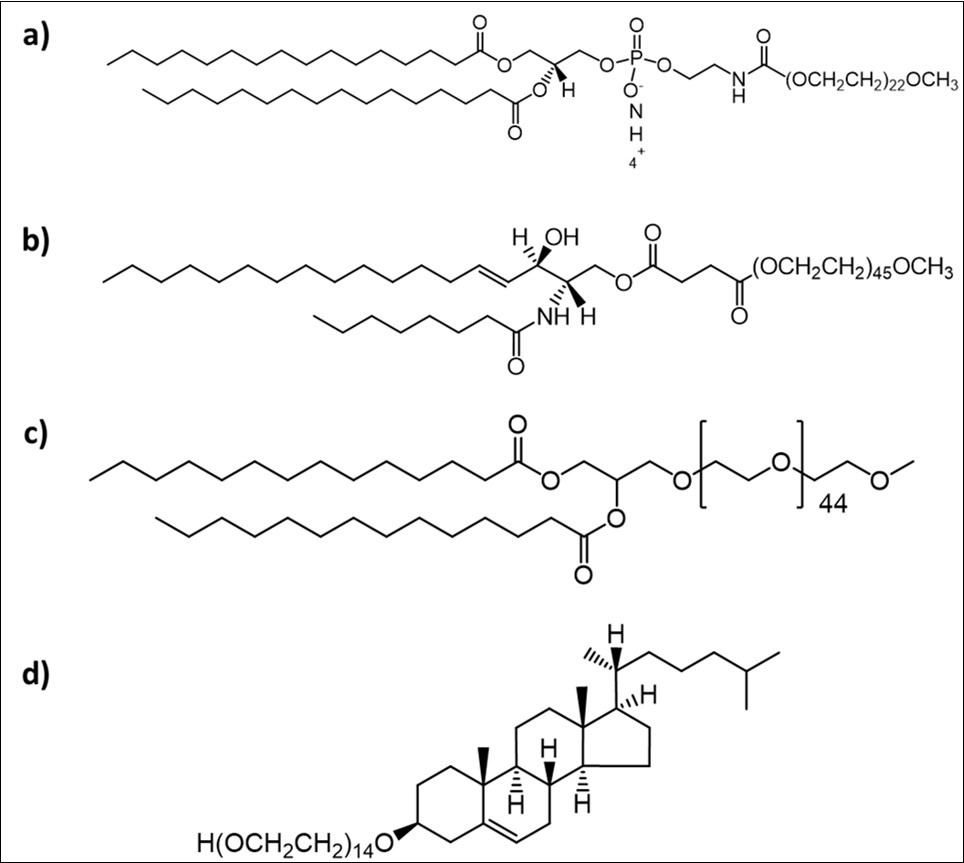
Conventional phosphate linkers are anionic in nature that can activate the complement system, which is responsible for innate immunity, leading to hypersensitivity reactions, however neutralizing these anionic charges, with cationic groups or methylation has shown to thwart this activation 92. The costs associated with production of phospholipids is not justified due to proneness to enzymatic degradation by lipases and phospholipases 93, 94, 95, 96, 97 that results in their rapid clearance. Consequently, PEG chains conjugated to glycerolipids, sphingolipids and cholesterol shown in Figure 3b-d have been explored as alternatives to PEG-phospholipids. A study investigating the different lengths of PEGylated ceramides on the circulation times of liposomes concluded that longer acyl chained ceramides (C24) compared to shorter chain ceramides (C8), had stronger anchoring properties that resulted in longer circulation time and higher drug release rates98, 99.
Cholesterol linked PEG or Cholesteryl-PEG (Chol-PEG) includes cholesterol as the hydrophobic anchor. Due to the lipophilicity, compatibility with other lipids and stabilizing properties, cholesterol is a favourable choice for anchoring PEG chains through ester bonds5, 93, 100. Chol-PEG is found to regulate the membrane fluidity, which helps in adding stability to the bilayer and prevents the leakage of drugs 70. Additionally, the drug loading efficiency of Chol-PEG is found to be dependent on the percentage of Chol-PEG. Lower Chol-PEG ratios in the formulation could encapsulate more amount of drugs101, 102, 103. Studies have reported the synthesis of pH cleavable PEG chains by linking Cholesterol through succinate and carbamate linkers104, 105. Despite its advantages, Chol-PEG demonstrated shorter circulation time than PEG phospholipids. This is because cholesterol anchors deep into the hydrophobic zone of the lipids, counteracting the advantages offered by PEG chains. The addition of extra linkers is proposed to overcome this limitation, however complicates the synthesis process.25, 70.
Furthermore, it is necessary to cleave the PEG chains after the circulation time is achieved to increase the absorption of drugs25, 105. This is accomplished by attaching linker moieties that either cleave the PEG chains upon the reaching the target site or when exposed to a suitable stimulus such as change in pH, temperature, or in the presence of an enzyme in the cellular microenvironment106, 107. Some prominent moieties that are used as cleavers include vinyl ether bond, hydrazine bond, disulphide, peptide and ester bonds108. Vinyl ether bonds are non-reactive in near neutral and basic conditions that makes them labile in acidic conditions especially under pH < 5 109. Vinyl ether linked PEG to dioleoyl phosphatidyl ethanolamine (DOPE) liposomes have demonstrated efficacious release of therapeutic contents post the cleavage of PEG moiety under varying pH conditions 109. It is also observed these PEG conjugated vinyl ether bonds are cleavable by reactive singlet oxygen generated encapsulated by photosensitizers 105. Peptide bonds, are cleaved only in the presence of tumor specific enzymes such as matrix metalloproteinases (MMPs)110, 111, 112. In-vitro studies have shown that peptide conjugated PEG lipids have longer circulations times and prefer to accumulate on tumor sites due to the enhanced permeability and retention effect (EPR), which arise from lack of draining in the tumor tissue110. For intracellular delivery specific, PEGylated liposomes conjugated with disulfide bonds are desirable 113, 114, 115 because the disulfide linkage in nanocarriers gets cleaved in the presence of glutathione, an antioxidant overexpressed in the cytoplasm of cancerous cells. Furthermore, many studies indicate that antibody delivery by cleavable PEG linked by disulfide bond can potentially be used in cancer diagnosis and treatment113, 116, 117.
Experimental Techniques for the Stability Assessment of PEGylated Liposomes
Thermal and physical stability are the important factors to consider in liposome-based vaccine and drug carrier design systems. A variety of analytical and optical methods are being used to characterize the physicochemical properties and assess the stability of PEGylated liposomes for in vivo studies and clinical trials as summarized in Figure 4. Studies from our laboratory have used a combination of techniques such as dynamic light scattering (DLS), differential scanning calorimetry (DSC), Nuclear magnetic resonance (NMR), Fourier-transform infrared spectroscopy (FTIR), and cryogenic electron microscopy (EM) to elucidate the structure and properties of the liposomes. These studies provide insights on their steric and thermal stability of the liposomes. However, techniques such as NMR, FTIR and EM are very sample specific and require an elaborate and specific set up. On the other hand, DSC and DLS are relatively simple techniques which still provide desired information about the sample stability. A brief review on DLS and DSC studies is as follows.
Figure 4.Assessment of physicochemical properties of PEGylated Liposome
DLS provides hydrodynamic diameter, polydispersity index, and zeta potential of the liposomal solution as shown in Figure 4. A monochromatic light reflected in the liposomal solution gets scattered due to the Brownian motion of the liposomes in the solution4, 5, 118, 119. The motion is related to the diffusion coefficient of the liposomes. Temperature and viscosity of the liposomal solution also influences the size measurement. The diffusion coefficient (Dτ) is related to the hydrodynamic radius (RH) is given by the stokes-Einstein equation120:
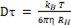 ….(1)
….(1)
Where kB is Boltzmann coefficient (1.380×10−23 kg.m2. s−2.K−1), T is an absolute temperature, and η is the viscosity of the aqueous medium. In addition to the hydrodynamic radius measurements, this technique also generates the poly dispersity index (PDI) of the liposomes which determines the size uniformity of the liposomes.
Another important parameter that determines the circulation ability of the PEG liposomes is the FALT, which is derived from the zeta potential or surface charge of the liposomes 121, 122, 123. The Zeta potential is measured using the Laser Doppler electrophoresis technique. When PEGylated liposomes are dispersed in water and when voltage is applied, a layer of ions from the solution strongly bind to the liposome surface forming a stern layer. This charged layer induces loose adhesion of ions of opposite charge called the diffuse layer. The two layers combined is called the electrical double layer. The electrical potential measured at the surface of the electrical double layer is called the zeta potential.
When an electric field is applied to a liposomal suspension, the liposomes move towards the oppositely charged electrodes of zeta potential measurement cell. The ratio between the velocity of motion of the particles and the electric field is called the electrophoretic mobility (µe) which is related to zeta potential (z) by the Henry equation,
 ..(2)
..(2)
Where ε and η are the dielectric constant and the absolute zero-shear viscosity of the aqueous medium. f(k.α) is known as “the Henry function”, where α is the radius of the particle and k is known as the Debye-Huckel parameter, which represent the thickness of the electrical double layer119, 124.
The zeta potential can be calculated from the electrophoretic velocity of the particles using Helmholtz-Smoluchowski equation
μe = VpE = ζεη ...(3)
where Vpis the electrophoretic velocity, ζ is the zeta potential, ε is the permittivity, and η is the viscosity of the medium.
The FALT measurement is calculated from the slope of the graph of the zeta potential vs Debye-Huckel parameter
The influence of PEG chains on the orientation of the lipids in the liposome and other morphological characteristics can be obtained by cryo-transmission electron microscopy (cryo-TEM), phase contrast microscopy (PCM), atomic force microscopy (AFM) and fluorescence microscopy (FM). Sample preparation in the case of EM and AFM and presence of other dyes in FM can generate spatial and resolution artefacts 125, 126. The use of X-Ray to obtain information on the structural properties is constrained because of the difficulty arising in crystallizing the samples resulting in lack of long-range order127, 128.
Our previous works have extensively used Differential scanning calorimetry (DSC) to gain an insight on the thermal stability and stressed induced in the lipid bilayer due to annealing and presence of peptides, and porphyrin, non-porphyrin amphiphilic fluorophores2, 3, 4, 5. DSC measures change in c heat capacity (Cp)as a function of temperature. PEGylated lipid molecules have characteristic pretransition and melting temperature that is accompanied during their thermal transition from gel to fluid crystalline transition which is endothermic in nature. The nature of the thermal transition is affected in the presence of molecules in the lipid bilayer. Change in enthalpy of transition is measured from the area under the melting transition and mathematically represented by the following equation 2, 3, 4, 5, 6:
Calorimetric Enthalpy
The change in shape of the melting peak of the lipids imply the presence of surface bound or encapsulated therapeutic molecules. Additional information on the cooperativity of these molecules can be obtained by computing thermodynamic parameters of the lipid-molecule system. 130, 131.
Benefits of PEGylated Liposomes in the Clinics
Based on previous discussions on the composition, properties, and advantages offered by PEGylated lipid molecules, it is clear that PEGylated formulations provide shielding effects from macrophages, offer longer circulation time, and allow preferential accumulation to sites of interests (such as tumors). Due to the substantial research in stealth technology, several PEGylated liposomes have been tested for clinical trials and approved by the FDA for treatment of patients. PEGylated lipids are typically used in combination with regular phospholipids and cholesterol in various formulations. The first approved PEGylated liposomal formulation was Doxil®. Doxil®, is an intravenous formulation developed by Sequus Pharmaceuticals, USA in 1995 for the delivery of DOX (doxorubicin) to treat ovarian and breast cancer. This formulation has 5 mol% of phospholipid DSPE-PEG 2000, 39 mol% cholesterol and 59 mol % hydrogenated soy phosphatidylcholine (HSPC) 125, 126, 127. Subsequently, Onivyde™, which has another anticancer agent, irinotecan, was approved by the FDA for the treatment of metastatic adenocarcinoma. Onivyde™ contains distearoyl-sn-glycero-phosphoethanolamine (DSPC), cholesterol and distearoylphosphatidyl ethanolamine (DSPR-PEG2000) in the ratio of 3:2:0.015128, 129.
Several other formulations which contain PEGlyated phospholipids and Chol-PEG are currently in different phases of clinical trials. The active ingredient and composition of the formulations has been tabulated below in Table 1130.
Table 1. FDA approved Formulations involving PEGylated Liposomes| Formulation | Active Ingredient | Composition | Stage | Application | Company |
| Doxil132–134 | Doxorubicin | HSPC:Cholesterol:PEG 2000-DSPE(56:39:5 molar ratio | Approved | Ovarian/Breast Cancer | Sequus Pharmaceuticals (1995) |
| Onivyde135, 136 | Irinotecan | DSPC:MPEG-2000: DSPE (3:2:0.015 molar ratio | Approved | Metastatic adenocarcinoma of the pancreas | Merrimack Pharmaceuticals Inc. (2015) |
| Thermodox142 | Doxorubicin | DPPC, Myristoyl stearyl phosphatidylcholineand DSPE-N-(amino(polyethylene glycol)-2000) | Phase 3 | Hepatocellular carcinoma | CelsionCorporation |
| Lipoplatin143, 144 | Cisplatin | DPPG, soy phosphatidyl choline,mPEG-distearoyl phosphatidylethanolaminelipid conjugate and cholesterol | Phase 3 | Non-small cell Lung Cancer | RegulonInc. |
| S-CKD602145, 146 | Potent topoisomerase I inhibitor | Phospholipids covalently bound to mPEG | Phase 2 | Cancer | Alza Corporation |
| 2B3-101141 | Doxorubicin | Glutathione PEGylated liposomes | Phase 1 | Solid Tumors | 2-BBB therapeutic |
| MCC-465147 | Doxorubicin | DPPC, cholesterol and maleimidated palmitoylphosphatidyl ethanolamine; immunoliposomestagged with PEG and the F(ab0)2 fragment ofhuman monoclonal antibody GAH | Phase 1 | Metastatic Stomach Cancer | Mitsubishi TanabePharma Corporation |
Thus, the stealth nature of PEGylated liposomes makes them suitable for encapsulation of variety of therapeutics. A very interesting recent application of PEGylated lipids is to use it in combination with photopolymerizable lipids (for example, DC8,9PC (1,2 bis (tricosa-10, 12-diynoyl)-sn-glycero-3-phosphocholine)). However, these studies are relatively recent, and their potential is still not fully explored. Therefore, we chose to summarize these observations to abreast and update readership in the field of lipids (see section below).
DC8,9PC is a polymeric lipid having a tubular morphology. Previously, DC8,9PC has been successively used in combination with DPPC for light-triggered delivery of a photosensitizer (HPPH, currently in clinical trials. This patented technology 132 is being developed by Nano-RedTMfor various therapeutic interventions 133. In our recent studies, we have described utilization of the PEGylated phospholipid DSPE-PEG2000 to render tubule-forming photopolymerizable lipid, DC8,9PC to a stable vesicular morphology 131. Therefore. the presence of hydrophilic PEG chains can induce formation of lamellar structure, thus resulting in eliminating the need for the presence of DPPC and therefore resulting in binary lipid stealth nanosystem. The DC8,9PC formulations could incorporate up to 20 mol% of and DSPE-PEG-2000 and encapsulated HPPH at very high efficiency 131. To our knowledge, inclusion of such high concentration of PEG lipids in a lipid formulation has not been shown previously. Similarly, another group also investigated the encapsulation of dexamethasone, a potent rheumatoid arthritis drug134 in this formulation. Both these studies have demonstrated the application of DC8,9PC to achieve longer circulation, stealth nature, and preferential accumulation of these formulations in the respective sites of interest in animals.
Conclusions
Stealth nature and long circulation properties of PEGylated liposomes has made them an attractive choice for drug delivery applications. However, certain studies have indicated certain unexpected immunological response to PEGylated liposomes. One such response is the accelerated blood clearance (ABC), the periodic administration of PEGylated liposomes results in production of antibodies against the carrier148–150. This has reduced the effectiveness of the course of the treatment and caused hypersensitive immune reactions. Therefore, more research is required to understand the intrinsic mechanisms responsible for these responses. This would support in designing PEGylated liposomes that can be used throughout a course of the treatment.
References
- 1.Matos C, Moutinho C, Lobão P. (2012) Liposomes as a Model for the Biological Membrane: Studies on Daunorubicin Bilayer Interaction. , The Journal of Membrane Biology 245(2), 69-75.
- 2.Gupta A, Mandal D, Ahmadibeni Y, Parang K, Bothun G. (2011) Hydrophobicity drives the cellular uptake of short cationic peptide ligands. https://doi.org/10.1007/s00249-011-0685-4. , European Biophysics Journal 40(6), 727-736.
- 3.Ye G, Gupta A, DeLuca R, Parang K, G D Bothun. (2010) Bilayer disruption and liposome restructuring by a homologous series of small Arg-rich synthetic peptides. https://doi.org/10.1016/j.colsurfb.2009.10.016. Colloids and Surfaces B: Biointerfaces 76(1), 76-81.
- 4.Kalyanram P, Ma H, Marshall S, Goudreau C, Cartaya A. (2020) . Interaction of Amphiphilic Coumarin with DPPC/DPPS Lipid Bilayer: Effects of Concentration and Alkyl Tail Length. https://doi.org/10.1039/D0CP00696C. Physical Chemistry Chemical Physics .
- 5.Kalyanram P, Gupta A, Stadler I. (2019) Interaction of Riboflavin-5-Phosphate With Liposome Bilayers. , https://doi.org/10.4018/jnn.2018010103. Journal of Nanotoxicology and Nanomedicine 3(1), 49-59.
- 6.Gupta A, Gupta R, Kurwardkar S. (2014) Liposome-encapsulated antimicrobial peptides: Potential infectious diseases therapy. https://doi.org/10.4018/978-1-4666-6363-3.ch014. In Handbook of Research on Diverse Applications of Nanotechnology in Biomedicine, Chemistry, and Engineering IGI Global 301-332.
- 7.Alavi M, Karimi N, Safaei M. Advanced Pharmaceutical Bulletin.Tabriz University of Medical (2017) Application of various types of liposomes in drug delivery systems. Sciences. https://doi.org/10.15171/apb.2017.002
- 8.R A Schwendener. (2014) 1). Liposomes as vaccine delivery systems: A review of the recent advances. https://doi.org/10.1177/2051013614541440. Therapeutic Advances in Vaccines.SAGEPublications Ltd .
- 9.Henriksen-Lacey M, K S, Andersen P, Perrie Y, Christensen D. (2011) Liposomal vaccine delivery systems. https://doi.org/10.1517/17425247.2011.558081. Expert Opinion on Drug Delivery.Taylor&Francis.
- 10.J O Eloy, Claro de Souza, Petrilli M, Barcellos R, J P A et al. (2014) Liposomes as carriers of hydrophilic small molecule drugs: Strategies to enhance encapsulation and delivery. https://doi.org/10.1016/J.COLSURFB.2014.09.029. Colloids and Surfaces B: Biointerfaces 123, 345-363.
- 11.V P Torchilin. (2005) Recent advances with liposomes as pharmaceutical carriers. https://doi.org/10.1038/nrd1632. Nature Reviews Drug Discovery 4(2), 145-160.
- 12.Ma P, Xiao H, Li C, Dai Y, Cheng Z. (2015) Inorganic nanocarriers for platinum drug delivery. https://doi.org/10.1016/J.MATTOD.2015.05.017. Materials Today 18(10), 554-564.
- 13.S J, Rivera-Gil P, Montenegro J-M, W J Parak, Smedt S C De. (2011) Cellular toxicity of inorganic nanoparticles: Common aspects and guidelines for improved nanotoxicity evaluation. https://doi.org/10.1016/J.NANTOD.2011.08.001. Nano Today. 6(5), 446-465.
- 14.Xiao B, M K Han, Viennois E, Wang L, Zhang M. (2015) Hyaluronic acid-functionalized polymeric nanoparticles for colon cancer-targeted combination chemotherapy. , https://doi.org/10.1039/c5nr04831a. Nanoscale 7(42), 17745-55.
- 15.Chopra M, Jain R, A K Dewangan, Varkey S, Mazumder S. (2016) Design of Curcumin Loaded Polymeric Nanoparticles-Optimization, Formulation and Characterization. , Journal of Nanoscience and Nanotechnology 16(9), 9432-9442.
- 16.Nagavarma B V N, Yadav H K S, Ayaz A, L S Vasudha, H G Shivakumar. (2012) Different techniques for preparation of polymeric nanoparticles- A review. , Asian Journal of Pharmaceutical and Clinical Research 5(3), 16-23.
- 17.Cheng Y, Li M, Xu T. (2008) Potential of poly(amidoamine) dendrimers as drug carriers of camptothecin based on encapsulation studies. https://doi.org/10.1016/j.ejmech.2007.09.030. , European Journal of Medicinal Chemistry 43(8), 1791-1795.
- 18.M T, Nakanishi Y, D J Kroll, A P Griset, M A Carnahan. (2006) Dendrimer-encapsulated camptothecins: Increased solubility, cellular uptake, and cellular retention affords enhanced anticancer activity in vitro. https://doi.org/10.1158/0008-5472.CAN-06-2066. , Cancer Research 66(24), 11913-11921.
- 19.Xie J, Gao Y, Zhao R, P J Sinko, Gu S. (2015) Ex vivo and in vivo capture and deactivation of circulating tumor cells by dual-antibody- coated nanomaterials. https://doi.org/10.1016/j.jconrel.2015.04.036. , Journal of Controlled Release 209, 159-169.
- 20.Belsito S, Bartucci R, Sportelli L. (2001) Lipid chain length effect on the phase behaviour of PCs/PEG: 2000-PEs mixtures. A spin label electron spin resonance and spectrophotometric study. https://doi.org/10.1016/S0301-4622(01)00201-0. , Biophysical Chemistry 93(1), 11-22.
- 21.Takeuchi H, Kojima H, Yamamoto H, Kawashima Y. (2000) Polymer coating of liposomes with a modified polyvinyl alcohol and their systemic circulation and RES uptake in rats. https://doi.org/10.1016/S0168-3659(00)00260-1. , Journal of Controlled Release 68(2), 195-205.
- 22.Dzieciuch M, Rissanen S, Szydłowska N, Bunker A, Kumorek M. (2015) Pegylated liposomes as carriers of hydrophobic porphyrins. , https://doi.org/10.1021/acs.jpcb.5b01351. Journal of Physical Chemistry B 119(22), 6646-6657.
- 23.Khajeh A, Modarress H. (2014) The influence of cholesterol on interactions and dynamics of ibuprofen in a lipid bilayer. , https://doi.org/10.1016/j.bbamem.2014.05.029. Biochimica et Biophysica Acta - Biomembranes 1838(10), 2431-2438.
- 24.Vabbilisetty P, X L Sun. (2014) Liposome surface functionalization based on different anchoring lipids via Staudinger ligation. https://doi.org/10.1039/c3ob41721b. Organic and Biomolecular Chemistry 12(8), 1237-1244.
- 25.Xu H, K Q Wang, Y H Deng, D W Chen. (2010) Effects of cleavable PEG-cholesterol derivatives on the accelerated blood clearance of PEGylated liposomes. , https://doi.org/10.1016/j.biomaterials.2010.02.049. Biomaterials 31(17), 4757-4763.
- 26.Kenworthy A. (2002) Peering inside lipid rafts and caveolae. , Trends in Biochemical Sciences 27(9), 10-1016.
- 27.W H Binder, Barragan V, F M Menger. (2003) . Domains and Rafts in Lipid Membranes. https://doi.org/10.1002/anie.200300586. Angewandte Chemie International Edition 42(47), 5802-5827.
- 28.S K Murthy. (2007) Nanoparticles in modern medicine: state of the art and future challenges. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/17722542 , International journal of nanomedicine 2(2), 129-41.
- 29.Akbarzadeh A, Rezaei-Sadabady R, Davaran S, S W Joo, Zarghami N. (2013) Liposome: Classification, preparation, and applications. https://doi.org/10.1186/1556-276X-8-102. , Nanoscale Research Letters 8(1), 102.
- 30.Maherani B, Arab-Tehrany E, Mozafari R, Gaiani M, C et al. (2011) Liposomes: A Review of Manufacturing Techniques and Targeting Strategies. https://doi.org/10.2174/157341311795542453. Current Nanoscience 7(3), 436-452.
- 31.Bozzuto G, Molinari A. (2015) 2). Liposomes as nanomedical devices. https://doi.org/10.2147/IJN.S68861. International Journal of Nanomedicine.DoveMedicalPressLtd .
- 32.Sercombe L, Veerati T, Moheimani F, S Y Wu, A K Sood. (2015) Advances and challenges of liposome assisted drug delivery. https://doi.org/10.3389/fphar.2015.00286. Frontiers in Pharmacology. Frontiers. , Media S.A
- 33.S M Moghimi, A C Hunter. (2001) Recognition by macrophages and liver cells of opsonized phospholipid vesicles and phospholipid headgroups. https://doi.org/10.1023/A: 1011054123304. Pharmaceutical Research.
- 34.Yan X, G L Scherphof, Kamps J A A M. (2005) Liposome opsonization. https://doi.org/10.1081/LPR-64971. , Journal of Liposome Research
- 35.Pozzi D, Colapicchioni V, Caracciolo G, Piovesana S, A L Capriotti. (2014) Effect of polyethyleneglycol (PEG) chain length on the bio-nano- interactions between PEGylated lipid nanoparticles and biological fluids: From nanostructure to uptake in cancer cells. , https://doi.org/10.1039/c3nr05559k. Nanoscale 6(5), 2782-2792.
- 36.Vonarbourg A, Passirani C, Saulnier P, J P Benoit. (2006) Parameters influencing the stealthiness of colloidal drug delivery systems. , https://doi.org/10.1016/j.biomaterials.2006.03.039. Biomaterials 27(24), 4356-4373.
- 37.D E Owens, N A Peppas. (2006) 3). Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. https://doi.org/10.1016/j.ijpharm.2005.10.010. International Journal of Pharmaceutics .
- 38.Marqués-Gallego P, Kroon A I P M de. (2014) Ligation Strategies for Targeting Liposomal Nanocarriers. https://doi.org/10.1155/2014/129458. BioMed Research International .
- 39.P, Biswas S, V P Torchilin. (2013) Current trends in the use of liposomes for tumor targeting. , https://doi.org/10.2217/nnm.13.118. Nanomedicine 8(9), 1509-1528.
- 40.Marsh D, Bartucci R, Sportelli L. (2003) 2). Lipid membranes with grafted polymers: Physicochemical aspects. https://doi.org/10.1016/S0005-2736(03)00197-4. Biochimica et Biophysica Acta-Biomembranes.Elsevier.
- 41.Galindo-rodriguez S, Alle E, Fessi H, Doelker E. (2004) . Physicochemical Parameters Associated with Nanoparticle Formation in the Salting-out , Nanoprecipitation Methods. https://doi.org/10.1023/B:PHAM.0000036917.75634.be. Pharmaceutical Research 21(8), 1428-1439.
- 42.Lemarchand C, Gref R, Couvreur P. (2004) 1). Polysaccharide- decorated nanoparticles. https://doi.org/10.1016/j.ejpb.2004.02.016. , European Journal of Pharmaceutics and Biopharmaceutics.Elsevier
- 43.Gref R, Domb A, Quellec P, reviews T B-… drug delivery. (1995) The controlled intravenous delivery of drugs using PEG-coated sterically stabilized nanospheres. ncbi.nlm.nih.gov. Retrieved from
- 44.Vonarbourg A, Passirani C, Saulnier P, J P Benoit. (2006) 1). Parameters influencing the stealthiness of colloidal drug delivery systems.
- 45.Vittaz M, Bazile D, Spenlehauer G, Verrecchia T, Veillard M. (1996) Effect of PEO surface density on long-circulating PLA-PEO nanoparticles which are very low complement activators. https://doi.org/10.1016/0142-9612(95)00322-3. , Biomaterials 17(16), 1575-1581.
- 46.Montdargent B, Maillet F, Carreno Paule, Jozefowicz M, Kazatchkinet M et al. (1993) Regulation by sulphonate groups of complement activation induced by hydroxymethyl groups on polystyrene surfaces. https://doi.org/10.1016/0142-9612(93)90024-V. , Biomaterials 14(3), 203-208.
- 47.S E Dunn, Brindley A, S, M C Davies, Illum L. (1994) Polystyrene-Poly (Ethylene Glycol) (PS-PEG2000) Particles as Model Systems for Site Specific Drug Delivery. 2. The Effect of PEG Surface Density on the in Vitro Cell Interaction and in Vivo Biodistribution. https://doi.org/10.1023/A: 1018939521589. Pharmaceutical Research: An Official , Journal of the American Association of Pharmaceutical Scientists 11(7), 1016-1022.
- 48.Passirani C. (2005) J.B. delivery and targeting of proteins, &, undefined. (n.d.). Complement activation by injectable colloidal drug carriers. , New York
- 49.Wilhelm C, Billotey C, Roger J, J N Pons, J C Bacri. (2003) Intracellular uptake of anionic superparamagnetic nanoparticles as a function of their surface coating. https://doi.org/10.1016/S0142-9612(02)00440-4. , Biomaterials 24(6), 1001-1011.
- 50.K D Lee, Hong K, Papahadjopoulos D. (1992) Recognition of liposomes by cells: In vitro binding and endocytosis mediated by specific lipid headgroups and surface charge density. https://doi.org/10.1016/0005-2736(92)90086-2. , BBA - Biomembranes 1103(2), 185-197.
- 51.Roser M, Fischer D, Kissel T. (1998) Surface-modified biodegradable albumin nano- and microspheres. II: Effect of surface charges on in vitro phagocytosis and biodistribution in rats. https://doi.org/10.1016/S0939-6411(98)00038-1. , European Journal of Pharmaceutics and Biopharmaceutics 46(3), 255-263.
- 52.J K Gbadamosi, A C Hunter, S M Moghimi. (2002) PEGylation of microspheres generates a heterogeneous population of particles with differential surface characteristics and biological performance. https://doi.org/10.1016/S0014-5793(02)03710-9. , FEBS Letters 532(3), 338-344.
- 53.Mosqueira V C F, Legrand P, Gref R, Heurtault B, Appel M. (1999) Interactions between a macrophage cell line (J774A1) and surface-modified Poly(D,L-lactide) nanocapsules bearing poly(ethylene glycol). https://doi.org/10.3109/10611869909085493. , Journal of Drug Targeting 7(1), 65-78.
- 54.Montdargent B, Maillet F, Carreno M, M J Biomaterials. (1993) undefined. (n.d.). Regulation by sulphone groups of complement activation induced by hydroxymethyl groups on polystyrene surfaces. 14(3), 203-208.
- 56.Stolnik S, Illum L, S. (1995) Long circulating microparticulate drug carriers. https://doi.org/10.1016/0169-409X(95)00025-3. Advanced Drug Delivery Reviews.
- 57.Maruyama K, Yuda T, Okamoto A. (1992) undefined. (n.d.). Prolonged circulation time in vivo of large unilamellar liposomes composed of distearoyl phosphatidylcholine and cholesterol containing amphipathic poly (ethylene.
- 58.Illum L, Jacobsen L, Müller R, Mak E, Biomaterials S D.Surface characteristics and the interaction of colloidal particles with mouse peritoneal macrophages.
- 59.Österberg E, Bergström K, Holmberg K, T P Schuman, J A Riggs. (1995) Protein‐rejecting ability of surface‐bound dextran in end‐on and side‐on configurations: Comparison to PEG. https://doi.org/10.1002/jbm.820290610. , Journal of Biomedical Materials Research 29(6), 741-747.
- 60.C K Anders, Adamo B, Karginova O, A M Deal, Rawal S. (2013) . Pharmacokinetics and Efficacy of PEGylated Liposomal Doxorubicin in an Intracranial Model of Breast Cancer. https://doi.org/10.1371/journal.pone.0061359. PLoS ONE 8(5), 61359.
- 61.Gabizon A, Shmeeda H, Barenholz Y. (2003) Pharmacokinetics of pegylated liposomal doxorubicin: Review of animal and human studies. https://doi.org/10.2165/00003088-200342050-00002. Clinical Pharmacokinetics.
- 62.Vlerken L E Van, T K Vyas, M. (2007) Poly(ethylene glycol)-modified nanocarriers for tumor-targeted and intracellular delivery. https://doi.org/10.1007/s11095-007-9284-6. , Pharmaceutical Research 24(8), 1405-1414.
- 63.Veronese F, today G P-D discovery. (2005) . undefined. (n.d.). PEGylation, successful approach to drug delivery. Elsevier
- 64.Knop K, Hoogenboom R, Fischer D, U S Schubert. (2010) 23). Poly(ethylene glycol) in drug delivery: Pros and cons as well as potential alternatives. https://doi.org/10.1002/anie.200902672. Angewandte Chemie - International Edition.
- 65.J S Suk, Xu Q, Kim N, Hanes J, L M Ensign. (2016) 1). PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. https://doi.org/10.1016/j.addr.2015.09.012. Advanced Drug Delivery Reviews.ElsevierB.V.
- 66.M N Khalid, Simard P, Hoarau D, Dragomir A, J C Leroux. (2006) Long circulating poly(ethylene glycol)-decorated lipid nanocapsules deliver docetaxel to solid tumors. https://doi.org/10.1007/s11095-006-9662-5. , Pharmaceutical Research 23(4), 752-758.
- 67.G P Zara, Cavalli R, Bargoni A, Fundaro A, Vighetto D. (2002) Intravenous administration to rabbits of non-stealth and stealth doxorubicin-loaded solid lipid nanoparticles at increasing concentrations of stealth agent: Pharmacokinetics and distribution of doxorubicin in brain and other tissues. https://doi.org/10.1080/10611860290031868. , Journal of Drug Targeting 10(4), 327-335.
- 68.Zarrintaj P, M R Saeb, S H Jafari, Mozafari M. (2019) Application of compatibilized polymer blends in biomedical fields. https://doi.org/10.1016/B978-0-12-816006-0.00018-9. In Compatibilization of Polymer Blends: Micro and Nano Scale Phase Morphologies, Interphase Characterization, and Properties 511-537.
- 69.M P Sikka, V K Midha. (2019) The role of biopolymers and biodegradable polymeric dressings in managing chronic wounds. https://doi.org/10.1016/b978-0-08-102192-7.00016-3. In Advanced Textiles for Wound Care 463-488.
- 70.O K Nag, Awasthi V. (2013) 25). Surface engineering of liposomes for stealth behavior. https://doi.org/10.3390/pharmaceutics5040542. Pharmaceutics. Multidisciplinary Digital Publishing Institute (MDPI) .
- 71.Rahme K, Dagher N. (2019) 1). Chemistry routes for copolymer synthesis containing peg for targeting, imaging, and drug delivery purposes. https://doi.org/10.3390/pharmaceutics11070327. Pharmaceutics. MDPI AG
- 72.Ulbrich K, Holá K, Šubr V, Bakandritsos A, Tuček J. (2016) . Targeted Drug Delivery with Polymers and Magnetic Nanoparticles: Covalent and Noncovalent Approaches, Release Control, and Clinical Studies. https://doi.org/10.1021/acs.chemrev.5b00589. Chemical Reviews 116(9), 5338-5431.
- 73.M K Riaz, M A Riaz, Zhang X, Lin C, K H Wong. (2018) 9). Surface functionalization and targeting strategies of liposomes in solid tumor therapy: A review. https://doi.org/10.3390/ijms19010195. , International Journal of Molecular Sciences. MDPI AG
- 74.Garbuzenko O, Barenholz Y, Priev A. (2005) Effect of grafted PEG on liposome size and on compressibility and packing of lipid bilayer. https://doi.org/10.1016/j.chemphyslip.2005.02.003. Chemistry and Physics of Lipids 135(2), 117-129.
- 75.Bartucci R, Pantusa M, Marsh D, Sportelli L. (2002) Interaction of human serum albumin with membranes containing polymer-grafted lipids: Spin-label ESR studies in the mushroom and brush regimes. , Biochimica et Biophysica Acta - Biomembranes 1564(1), 237-242.
- 76.J S Suk, Xu Q, Kim N, Hanes J, L M Ensign. (2016) 1). PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. https://doi.org/10.1016/j.addr.2015.09.012. Advanced Drug Delivery Reviews.ElsevierB.V.
- 77.A K Kenworthy, Hristova K, Needham D, T J McIntosh. (1995) Range and magnitude of the steric pressure between bilayers containing phospholipids with covalently attached poly(ethylene glycol). https://doi.org/10.1016/S0006-3495(95)80369-3. , Biophysical Journal 68(5), 1921-1936.
- 78.O K Nag, V R Yadav, Hedrick A, Awasthi V. (2013) Post-modification of preformed liposomes with novel non-phospholipid poly(ethylene glycol)-conjugated hexadecylcarbamoylmethyl hexadecanoic acid for enhanced circulation persistence in vivo. https://doi.org/10.1016/j.ijpharm.2013.02.026. , International Journal of Pharmaceutics 446(1), 119-129.
- 79.Karathanasis E, A L, Bhavane R, R V Bellamkonda, A V. (2005) Preparation of in vivo cleavable agglomerated liposomes suitable for modulated pulmonary drug delivery. https://doi.org/10.1016/j.jconrel.2004.11.009. , Journal of Controlled Release 103(1), 159-175.
- 80.Heyes J, Hall K, Tailor V, Lenz R, MacLachlan I. (2006) Synthesis and characterization of novel poly(ethylene glycol)-lipid conjugates suitable for use in drug delivery. , https://doi.org/10.1016/j.jconrel.2006.02.012. Journal of Controlled Release 112(2), 280-290.
- 81.Ambegia E, Ansell S, Cullis P, Heyes J, Palmer L. (2005) Stabilized plasmid-lipid particles containing PEG-diacylglycerols exhibit extended circulation lifetimes and tumor selective gene expression. , https://doi.org/10.1016/j.bbamem.2005.02.001. Biochimica et Biophysica Acta - Biomembranes 1669(2), 155-163.
- 82.Wang R, Xiao R, Zeng Z, Xu L, Wang J. (2012) Application of poly(ethylene glycol)- distearoylphosphatidylethanolamine (PEG-DSPE) block copolymers and their derivatives as nanomaterials in drug delivery. https://doi.org/10.2147/IJN.S34489. International Journal of Nanomedicine.Int JNanomedicine .
- 83.Hu Q, C R Shew, M B Bally, T D Madden. (2001) Programmable fusogenic vesicles for intracellular delivery of antisense oligodeoxynucleotides: Enhanced cellular uptake and biological effects. , Biochimica et Biophysica Acta - Biomembranes 1514(1), 1-13.
- 84.Shimada K, Matsuo S, Sadzuka Y, Miyagishima A, Nozawa Y. (2000) Determination of incorporated amounts of poly(ethylene glycol)-derivatized lipids in liposomes for the physicochemical characterization of stealth liposomes. , International Journal of Pharmaceutics,203(1–2): 255-263.
- 85.Zeisig R, Shimada K, Hirota S, Arndt D. (1996) Effect of sterical stabilization on macrophage uptake in vitro and on thickness of the fixed aqueous layer of liposomes made from alkylphosphocholines. https://doi.org/10.1016/S0005-2736(96)00167-8. Biochimica et Biophysica Acta (BBA) -. , Biomembranes 1285(2), 237-245.
- 86.Sadzuka Y, Nakade A, Tsuruda T, Sonobe T. (2003) Study on the characterization of mixed polyethyleneglycol modified liposomes containing doxorubicin. https://doi.org/10.1016/S0168-3659(03)00248-7. , Journal of Controlled Release 91(3), 271-280.
- 87.Sadzuka Y, Nakade A, Hirama R, Miyagishima A, Nozawa Y. (2002) Effects of mixed polyethyleneglycol modification on fixed aqueous layer thickness and antitumor activity of doxorubicin containing liposome. , International Journal of Pharmaceutics,238(1–2): 171-180.
- 88.Sugiyama I, Sadzuka Y. (2013) Change in the Character of Liposomes as a Drug Carrier by Modifying Various Polyethyleneglycol-Lipids. , Biological and Pharmaceutical Bulletin 36(6), 900-906.
- 89.Sugiyama I, Sadzuka Y. (2011) Correlation of Fixed Aqueous Layer Thickness Around PEG-modified Liposomes with. In Vivo Efficacy of Antitumor Agent-containing Liposomes. https://doi.org/10.2174/157016311798109344. Current Drug Discovery Technologies 8(4), 357-366.
- 90.T M Allen, Hansen C, Martin F, Redemann C, Yau-Young A. (1991) Liposomes containing synthetic lipid derivatives of poly(ethylene glycol) show prolonged circulation half-lives in vivo. https://doi.org/10.1016/0005-2736(91)90246-5. , BBA - Biomembranes 1066(1), 29-36.
- 91.Er Y, T J Barnes, Fornasiero D, C A Prestidge. (2009) The encapsulation and release of guanosine from PEGylated liposomes. https://doi.org/10.1080/08982100802673940. , Journal of Liposome Research 19(1), 29-36.
- 92.R E Gibson. (2011) Molecular Imaging, Principles and Practices. , Molecular Imaging and Biology 13(3), 10-1007.
- 93.Bastiat G, Oligere P, Karlsson G, Edwards K, Lafleur M. (2007) Development of non-phospholipid liposomes containing a high cholesterol concentration. , https://doi.org/10.1021/la700824m. Langmuir 23(14), 7695-7699.
- 94.P A Carlson, M H Gelb, Yager P. (1997) Zero-order interfacial enzymatic degradation of phospholipid tubules. , Biophysical Journal 73(1), 230-238.
- 95.Yoshikawa N, Fumoto S, Yoshikawa K, Hu D, Okami K. (2020) Interaction of lipoplex with albumin enhances gene expression in hepatitis mice. , https://doi.org/10.3390/pharmaceutics12040341. Pharmaceutics 12(4), 341.
- 96.Bonté F, R L Juliano. (1986) Interactions of liposomes with serum proteins. https://doi.org/10.1016/0009-3084(86)90079-4. Chemistry and Physics of Lipids,40(2–4): 359-372.
- 97.Moghimi Moein, Hamad S, Andresen I, L T, Jørgensen K et al. (2006) Methylation of the phosphate oxygen moiety of phospholipid‐methoxy(polyethylene glycol) conjugate prevents PEGylated liposome‐mediated complement activation and anaphylatoxin production. , The FASEB Journal 20(14), 2591-2593.
- 98.Shimada K, Matsuo S, Sadzuka Y, Miyagishima A, Nozawa Y. (2000) Determination of incorporated amounts of poly(ethylene glycol)-derivatized lipids in liposomes for the physicochemical characterization of stealth liposomes. , International Journal of Pharmaceutics,203(1–2): 255-263.
- 99.M S Webb, Saxon D, Wong F M P, H J Lim, Wang Z. (1998) Comparison of different hydrophobic anchors conjugated to poly(ethylene glycol): Effects on the pharmacokinetics of liposomal vincristine. , Biochimica et Biophysica Acta - Biomembranes 1372(2), 272-282.
- 100.Beugin S, Edwards K, Karlsson G, Ollivon M, Lesieur S. (1998) New sterically stabilized vesicles based on nonionic surfactant, choleserol, and poly(ethylene glycol)-cholesterol conjugates. https://doi.org/10.1016/S0006-3495(98)78026-9. , Biophysical Journal 74(6), 3198-3210.
- 101.Kuang Y, Liu J, Liu Z, Zhuo R. (2012) Cholesterol-based anionic long-circulating cisplatin liposomes with reduced renal toxicity. , https://doi.org/10.1016/j.biomaterials.2011.10.081. Biomaterials 33(5), 1596-1606.
- 102.Addeo R, Faiola V, Guarrasi R, Montella L, Vincenzi B. (2008) Liposomal pegylated doxorubicin plus vinorelbine combination as first-line chemotherapy for metastatic breast cancer in elderly women ≥65 years of age. https://doi.org/10.1007/s00280-007-0605-6. Cancer Chemotherapy and Pharmacology. 62(2), 285-292.
- 103.Deniz A, Sade A, Severcan F, Keskin D, Tezcaner A. (2010) Celecoxib-loaded liposomes: Effect of cholesterol on encapsulation and in vitro release characteristics. , https://doi.org/10.1042/BSR20090104. Bioscience Reports 30(5), 365-373.
- 104.J A Boomer, M, H D Inerowicz, R H Haynes, V S Patri. (2009) Cytoplasmic delivery of liposomal contents mediated by an acid-labile cholesterol-vinyl ether-PEG conjugate. , https://doi.org/10.1021/bc800239b. Bioconjugate Chemistry 20(1), 47-59.
- 105.Komeda C, Ikeda A, Kikuchi J, Ishida-Kitagawa N, Tatebe H. (2013) A photo-triggerable drug carrier based on cleavage of PEG lipids by photosensitiser-generated reactive singlet oxygen. , https://doi.org/10.1039/c2ob27199k. Organic & Biomolecular Chemistry 11(16), 2567.
- 106.Cai X, Dong C, Dong H, Wang G, G M Pauletti. (2012) Effective gene delivery using stimulus-responsive catiomer designed with redox-sensitive disulfide and acid-labile imine linkers. , https://doi.org/10.1021/bm2017355. Biomacromolecules 13(4), 1024-1034.
- 107.R M Sawant, J P Hurley, Salmaso S, Kale A, Tolcheva E. (2006) SMART” drug delivery systems: Double-targeted pH-responsive pharmaceutical nanocarriers. https://doi.org/10.1021/bc060080h.Bioconjugate Chemistry. 17(4), 943-949.
- 108.Fang Y, Xue J, Gao S, Lu A, Yang D. (2017) Cleavable PEGylation: a strategy for overcoming the “PEG dilemma” in efficient drug delivery. https://doi.org/10.1080/10717544.2017.1388451. Drug Delivery 24(2), 22-32.
- 109.Shin J, Shum P, D H Thompson. (2003) Acid-triggered release via dePEGylation of DOPE liposomes containing acid-labile vinyl ether PEG-lipids. https://doi.org/10.1016/S0168-3659(03)00232-3. , In Journal of Controlled Release 91, 187-200.
- 110.Hatakeyama H, Akita H, Ito E, Hayashi Y, Oishi M. (2011) Systemic delivery of siRNA to tumors using a lipid nanoparticle containing a tumor-specific cleavable PEG-lipid. , https://doi.org/10.1016/j.biomaterials.2011.02.045. Biomaterials 32(18), 4306-4316.
- 111.Hatakeyama H, Ito E, Akita H, Oishi M, Nagasaki Y. (2009) A pH-sensitive fusogenic peptide facilitates endosomal escape and greatly enhances the gene silencing of siRNA-containing nanoparticles in vitro and in vivo. https://doi.org/10.1016/j.jconrel.2009.06.008. , Journal of Controlled Release 139(2), 127-132.
- 112.Hatakeyama H, Akita H, Kogure K, Oishi M, Nagasaki Y. (2007) Development of a novel systemic gene delivery system for cancer therapy with a tumor-specific cleavable PEG-lipid. https://doi.org/10.1038/sj.gt.3302843. Gene Therapy 14(1), 68-77.
- 113.W J, Welker E, Narayan M, H A Scheraga. (2000) Disulfide bonds and protein folding. , https://doi.org/10.1021/bi992922o. Biochemistry 39(15), 4207-4216.
- 114.Shaunak S, Godwin A, J W Choi, Balan S, Pedone E. (2006) Site-specific PEGylation of native disulfide bonds in therapeutic proteins. , https://doi.org/10.1038/nchembio786. Nature Chemical Biology 2(6), 312-313.
- 115.Z Y Guo, X Y Jia, Y M Feng. (2004) Replacement of the interchain disulfide bridge-forming amino acids A7 and B7 by glutamate impairs the structure and activity of insulin. , https://doi.org/10.1515/BC.2004.151. Biological Chemistry 385(12), 1171-1175.
- 116.Wu Y, Chen W, Meng F, Wang Z, Cheng R. (2012) Core-crosslinked pH-sensitive degradable micelles: A promising approach to resolve the extracellular stability versus intracellular drug release dilemma. https://doi.org/10.1016/j.jconrel.2012.07.011. , In Journal of Controlled Release 164, 338-345.
- 117.Dong H, Tang M, Li Y, Li Y, Qian D. (2015) 1). Disulfide-bridged cleavable PEGylation in polymeric nanomedicine for controlled therapeutic delivery. https://doi.org/10.2217/nnm.15.38. Nanomedicine. Future Medicine Ltd .
- 118.Gupta A, Gupta R, Kurwadkar S. (2014) Liposome encapsulated antimicrobial peptides: potential infectious diseases therapy:. Handbook of Research on Diverse Applications of Nanotechnology in Biomedicine, Chemistry and Engineering. IGI GlobalPublisher .
- 119.Gupta A. (2011) Investigation Of Interaction Of Cationic Amphiphiles With Model Membranes And Their Application In Drug Delivery.UniversityofRhodeIsland.
- 120.Stetefeld J, S A McKenna, T R Patel. (2016) Dynamic light scattering: a practical guide and applications in biomedical sciences. https://doi.org/10.1007/s12551-016-0218-6. Biophysical reviews. 8(4), 409-427.
- 121.Kronberg B, Dahlman A, Carlfors J, Karlsson J, Artursson P. (1990) Preparation and evaluation of sterically stabilized liposomes: Colloidal stability, serum stability, macrophage uptake, and toxicity. https://doi.org/10.1002/jps.2600790803. , Journal of Pharmaceutical Sciences 79(8), 667-671.
- 122.Papahadjopoulos D, T M Allen, Gabizon A, Mayhew E, Matthay K. (1991) Sterically stabilized liposomes: Improvements in pharmacokinetics and antitumor therapeutic efficacy. https://doi.org/10.1073/pnas.88.24.11460. Proceedings of the National Academy of Sciences of the United States of America 88(24), 11460-11464.
- 123.Zeisig R, Shimada K, Hirota S, Arndt D. (1996) Effect of sterical stabilization on macrophage uptake in vitro and on thickness of the fixed aqueous layer of liposomes made from alkylphosphocholines. https://doi.org/10.1016/S0005-2736(96)00167-8. , Biochimica et Biophysica Acta - Biomembranes 1285(2), 237-245.
- 124.Bhattacharjee S. (2016) DLS and zeta potential – What they are and what they are not? https://doi.org/10.1016/J.JCONREL.2016.06.017. , Journal of Controlled Release 235, 337-351.
- 125.Galdiero S, Falanga A, Cantisani M, Vitiello M, Morelli G. (2013) Peptide-Lipid Interactions: Experiments and Applications. , International Journal of Molecular Sciences 14(9), 18758-18789.
- 126.A C, Ribeiro D, Nunes C, Reis S. (2016) Biophysics in cancer: The relevance of drug-membrane interaction studies. , https://doi.org/10.1016/J.BBAMEM.2016.06.025. Biochimica et Biophysica Acta (BBA) - Biomembranes 1858(9), 2231-2244.
- 127.Szyk A, Wu Z, Tucker K, Yang D, Lu W. (2006) Crystal structures of human alpha-defensins. HNP4, HD5, and HD6. https://doi.org/10.1110/ps.062336606. Protein science a publication of the Protein Society 15(12), 2749-60.
- 128.I L Karle, M A Perozzo, V K Mishra, Balaram P. (1998) Crystal structure of the channel-forming polypeptide antiamoebin in a membrane-mimetic environment. Proceedings of the National Academy of Sciences of the United States of America Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/9576911 95(10), 5501-4.
- 129.Matthew Ryan Preiss, Gupta Anju, G D Bothun. (2013) Liposome-Nanoparticle Assemblies, Bionanotechnology: Biological self-assembly and its applications. , Norwich, UK
- 130.Joanne P, Galanth C, Goasdoué N, Nicolas P, Sagan S. (2009) Lipid reorganization induced by membrane-active peptides probed using differential scanning calorimetry. , https://doi.org/10.1016/j.bbamem.2009.05.001. Biochimica et Biophysica Acta (BBA) - Biomembranes 1788(9), 1772-1781.
- 131.Kiricsi M, E J Prenner, Jelokhani-Niaraki M, Lewis R N A H, R S Hodges. (2002) The effects of ring-size analogs of the antimicrobial peptide gramicidin S on phospholipid bilayer model membranes and on the growth of Acholeplasma laidlawii B. , European Journal of Biochemistry 269(23), 5911-5920.
- 132.Batist G. (2007) Cardiac safety of liposomal anthracyclines. https://doi.org/10.1007/s12012-007-0014-4. Cardiovasc Toxicol , In Cardiovascular Toxicology 7, 72-74.
- 133.Gabizon A, Catane R, Uziely B, Kaufman B, Safra T. (1994) Prolonged Circulation Time and Enhanced Accumulation. in Malignant Exudates of Doxorubicin Encapsulated in Polyethylene-glycol Coated Liposomes 1. CANCER Retrieved from https://cancerres.aacrjournals.org/content/54/4/987.short 154.
- 134.Gabizon A, Shmeeda H, Barenholz Y. (2003) Pharmacokinetics of pegylated liposomal doxorubicin: Review of animal and human studies. https://doi.org/10.2165/00003088-200342050-00002. , Clinical Pharmacokinetics 42(5), 419-436.
- 135.K H, Drummond, C D, Kripotin D.US20160030341A1 - Liposomes Useful for Drug Delivery - Google Patents. Retrieved from https://patents.google.com/patent/US20160030341A1/en.
- 136.D C, C O Noble, Guo Z, Hong K, J W Park. (2006) Development of a highly active nanoliposomal irinotecan using a novel intraliposomal stabilization strategy. https://doi.org/10.1158/0008-5472.CAN-05-4007. , Cancer Research 66(6), 3271-3277.
- 137.Bulbake U, Doppalapudi S, Kommineni N, Khan W. (2017) Liposomal Formulations in Clinical Use: An Updated Review. , https://doi.org/10.3390/pharmaceutics9020012. Pharmaceutics 9(4), 12.
- 138.Viard M, Reichard H, B A Shapiro, F A Durrani, A J Marko. (2018) Design and biological activity of novel stealth polymeric lipid nanoparticles for enhanced delivery of hydrophobic photodynamic therapy drugs. , https://doi.org/10.1016/j.nano.2018.07.006. Nanomedicine: Nanotechnology, Biology, and Medicine 14(7), 2295-2305.
- 139.A J. (2018) Photoactivatable lipid-based nanoparticles as vehicles... - Google Scholar. U.S. Patent and Trademark Office. Retrieved from https://scholar.google.com/scholar?oi=gsb40&q=Photoactivatable lipid-based nanoparticles as vehicles for dual agent delivery&lookup=0&hl=en.
- 140.NanoRED. (2020) Revolutionizing Cancer Care. (n.d.). Retrieved from http://www.nanoredbiotech.com/
- 141.P J Gaillard, Appeldoorn C C M, Dorland R, J Van Kregten, Manca F. (2014) Pharmacokinetics, brain delivery, and efficacy in brain tumor-bearing mice of glutathione pegylated liposomal doxorubicin (2B3-101). https://doi.org/10.1371/journal.pone.0082331. PLoS ONE, 9(1)
- 142.Chen J, C Q He, A H Lin, Gu W, Z P Chen. (2014) Thermosensitive liposomes with higher phase transition temperature for targeted drug delivery to tumor. https://doi.org/10.1016/j.ijpharm.2014.09.009. , International Journal of Pharmaceutics 475(1), 408-415.
- 143.Terwogt Meerum, M J, Groenewegen G, Pluim D, Maliepaard M et al. (2002) Phase I and pharmacokinetic study of SPI-77, a liposomal encapsulated dosage form of cisplatin. https://doi.org/10.1007/s002800100371. Cancer Chemotherapy and Pharmacology 49(3), 201-210.
- 144.Boulikas T. (2009) Clinical overview on LipoplatinTM: A successful liposomal formulation of cisplatin. https://doi.org/10.1517/13543780903114168. Expert Opinion on Investigational Drugs.
- 145.W C Zamboni, J L Eiseman, Strychor S, P M Rice, Joseph E. (2011) Tumor disposition of pegylated liposomal CKD-602 and the reticuloendothelial system in preclinical tumor models.
- 146.W C Zamboni, L J Maruca, Strychor S, B A Zamboni, Ramalingam S. (2011) Bidirectional pharmacodynamic interaction between pegylated liposomal CKD-602 (S-CKD602) and monocytes in patients with refractory solid tumors.
- 147.Danhier F. (2016) 28). To exploit the tumor microenvironment: Since the EPR effect fails in the clinic, what is the future of nanomedicine? https://doi.org/10.1016/j.jconrel.2016.11.015. Journal of Controlled Release.
- 148.Mohamed M, Lila Abu, S A, Shimizu T, Alaaeldin E et al. (2019) 31). PEGylated liposomes: immunological responses. https://doi.org/10.1080/14686996.2019.1627174. Science and Technology of Advanced Materials.TaylorandFrancisLtd .
- 149.Khan A, Szebeni J. (2003) undefined. (n.d.). Complement activation following first exposure to pegylated liposomal doxorubicin (Doxil®): Possible role in hypersensitivity reactions. mayoclinic.pure.elsevier.com.
- 150.Szebeni J, Bedocs P, Rozsnyay Z, Weiszhár Z, Urbanics R. (2012) Liposome-induced complement activation and related cardiopulmonary distress in pigs: Factors promoting reactogenicity of Doxil and AmBisome. , https://doi.org/10.1016/j.nano.2011.06.003. Nanomedicine: Nanotechnology, Biology, and Medicine 8(2), 176-184.
Cited by (3)
- 1.Kalyanram Poornima, Puri Anu, Gupta Anju, 2022, Thermotropic effects of PEGylated lipids on the stability of HPPH-encapsulated lipid nanoparticles (LNP), Journal of Thermal Analysis and Calorimetry, 147(11), 6337, 10.1007/s10973-021-10929-6
- 2.Dopierała Katarzyna, Weiss Marek, Krajewska Martyna, Błońska Justyna, 2023, Towards understanding the binding affinity of lipid drug carriers to serum albumin, Chemistry and Physics of Lipids, 250(), 105271, 10.1016/j.chemphyslip.2022.105271
- 3.Gyanani Vijay, Goswami Roshan, 2023, Key Design Features of Lipid Nanoparticles and Electrostatic Charge-Based Lipid Nanoparticle Targeting, Pharmaceutics, 15(4), 1184, 10.3390/pharmaceutics15041184

