Abstract
Idiopathic granulomatous mastitis was initially scripted in 1972 by Kessler and Wolloch and is cogitated as an infrequent, benign, chronic inflammatory disorder. It commonly implicates females of reproductive age group, especially women who have breast fed in the preceding five to six years. Idiopathic granulomatous mastitis is commonly discerned in Asia and Mediterranean region although true incidence is undetermined. Diverse ethnicities delineate a diverse disease prevalence. Elimination of adjunctive causes of granulomatous inflammation makes idiopathic granulomatous mastitis a diagnosis of exclusion. As per the natural history, idiopathic granulomatous mastitis is designated as a self limiting disorder 1.
Author Contributions
Academic Editor: El-Sabbagh AH, professor of plastic surgery, faculty of medicine, mansoura university, Egypt.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Anubha Bajaj
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Disease Characteristics
Essentially an uncommon condition of obscure origin, granulomatous mastitis simulates an abscess or carcinoma breast on radiography. As radiographic features are inconclusive, definitive diagnosis is usually obtained with a core tissue biopsy and histological analysis. Repetitive tissue sampling is required to exclude a metamorphosis into carcinoma breast. Associated aetiological factors such as various infections, duct ectasia or a distinctive autoimmune mechanism may not be discovered.
Granulomatous mastitis frequently emerges in the third or fourth decade of life with a demonstrable age range betwixt 11 years to 83 years. Reoccurrence is observed in a significant percentage of instances and varies betwixt 5% to 50%. An infectious aetiology of granulomatous mastitis requires elimination as corticosteroids are contraindicated in infectious aetiologies 2, 3.
Disease Pathogenesis
Mechanism of inflammation in idiopathic granulomatous mastitis is comprised of an injurious deterioration of ductal epithelium followed by metamorphosis and luminal secretions within the lobular connective tissue. Localized inflammation within the connective tissue accompanied by migration of macrophages and lymphocytes within the inflamed region evokes a focal, granulomatous inflammatory response. However, pre-emptive factor triggering the inflammation remains elusive.
Trigger factors indicated are constituted by pregnancy, lactation, autoimmune disorders, hyperprolactinaemia and tobacco consumption. Enhanced severity and duration of disease can be cogitated in inflammation arising within the puerperal period 2, 3.
Of obscure aetiology, factors such as hormonal imbalance, autoimmunity, undetermined microbiological agents, smoking contraceptive pills, lactation, hyperprolactinaemia, infection with Corynebacterium and antitrypsin deficiency are implicated in the genesis of granulomatous mastitis. Certain aetiological factors may be undiscernible with the employment of current diagnostic modalities 3.
Clinical Elucidation
Majority of subjects (90%) depict a unilateral lesion appearing as a palpable mass or an abscess situated in the upper segment of breast. The tender, erythematous, inflamed mass detectable within the breast parenchyma enlarges gradually and can be associated with pain and lymphadenopathy. Emergence of a hard, irregular nodule in the breast parenchyma can recapitulate carcinoma breast. Nipple and cutaneous retraction as well as nipple discharge can be discerned. Enunciated mass in the breast parenchyma or nodule can depict a sequential breast abscess which can drain spontaneously. Comprehensive breast parenchyma may be incriminated barring the nipple. Multiple fistulae or sinus tracts can be exemplified in advanced disease 3, 4.
Histological Elucidation
On gross examination, a firm to hard, mildly nodular mass is delineated.
Core needle biopsy of idiopathic granulomatous mastitis enunciates multiple aggregates of non- caseating epitheloid cell granulomas within and encompassing breast lobules, constituted of epitheloid histiocytes, lymphocytes, neutrophils and multinucleated giant cells. Granulomatous inflammation is predominantly lobulo-centric. The inflammation is preponderantly composed of lymphocytes, plasma cells, epitheloid histiocytes, multinucleated giant cells and neutrophils. Neutrophils can configure micro-abscesses and encompass vacant micro-cystic cavities, morphological features which are in common with cystic neutrophilic granulomatous mastitis. Non specific lobulitis along with a lymphoid and plasma cell infiltrate accompanies the granulomatous inflammation. Necrosis is usually absent. Neutrophilic micro-abscesses can be accompanied by fistula formation 4, 5.
Multinucleated giant cells are detected in an estimated three fourths (78.5%) instances. Plasma cells are discernible in around half (53.9%) of the subjects and usually appear at the margins of cystic vacuoles with centric accumulation of neutrophils within the granulomas.
Special stains such as Gomori-Methanamine Silver (GMS) and Periodic acid Schiff’s (PAS) stain for fungal spores or mycelia are usually non reactive. Acid fast bacilli or Mycobacterial species are usually not detected on Zeihl Neelsen stain. Gram’s stain is mandated for discovering gram positive bacilli. Fungal and mycobacterial cultures usually do not reveal any results. Axillary and internal mammary lymphadenopathy is observed 4, 5.
Fine needle aspiration can demonstrate a cellular exudate indicative of an abscess with an intense inflammatory infiltrate preponderantly composed of neutrophils. Ductal epithelial cells may be absent. Cytological examination delineates abundant and accumulated epitheloid histiocytes, neutrophils, lymphocytes with a variable degree of granuloma formation and an absence of necrosis 5.
Figure, Figure 2, Figure 3, Figure 4, Figure 5, Figure 6, Figure 7, Figure 8, Figure 9, Figure 10.
Figure 1.Granulomatous mastitis with the configuration of epitheloid cell granuloma and a lymphoid and plasma cell infiltrate9.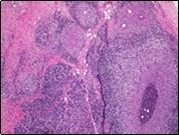
Figure 2.Granulomatous mastitis with articulated epitheloid cell granuloma, lymphocytic, plasma cell and neutrophilic infiltration10.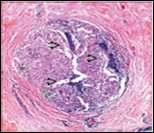
Figure 3.Granulomatous mastitis with scattered langhans and foreign body giant cells, epitheloid cell granuloma and lymphoid ingress11.
Figure 4.Granulomatous mastitis with epitheloid cell granuloma, langhans and foreign body giant cells with lymphocytic rimming12.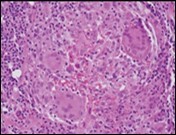
Figure 5.Granulomatous mastitis with disseminated epitheloid cell granuloma, langhans giant cells, lymphocytic and plasma cell dispersal13.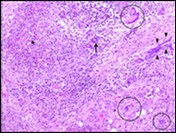
Figure 6.Granulomatous mastitis with numerous epitheloid cell granuloma, foreign body giant cells and a peripheral rimming with lymphocytes14.
Figure 7.Granulomatous mastitis with scattered epitheloid cells, neutrophilic exudate and aggregates of epithelod cells15.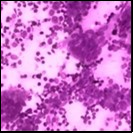
Figure 8.Granulomatous mastitis with disseminated epitheloid cells admixed with neutrophils, lymphocytes and plasma cells 16.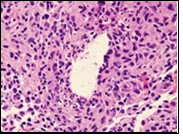
Figure 9.Granulomatous mastitis with circumscribed epitheloid cell granuloma, foreign body giant cells and a perimeter of lymphocytes 17.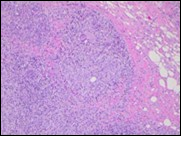
Figure 10.Granulomatous mastitis with a ductal circumscription of epitheloid cells, plasma cells, lymphocytes and neutrophils 18.
Differential Diagnosis
Idiopathic granulomatous mastitis requires a segregation from diseases such as peri-ductal mastitis or carcinoma breast.
An infective aetiology such as fungal or mycobacterial infection, autoimmune conditions delineated as sarcoidosis with distribution of non-caseating granulomas, devoid of a lobular distribution and Wegener’s granulomatosis with polyangiitis demonstrating necrotizing vasculitis require distinction.
Despite a varied differential diagnosis, granulomatous mastitis is predominantly an idiopathic disorder2, 3. Possible association of Corynebacterium infection with granulomatous mastitis can be observed and is designated as cystic neutrophilic granulomatous mastitis. Aforesaid condition typically displays lipo- granulomas comprised of clear, vacuolated spaces with a peripheral rimming of neutrophils along with a circumscription of granulomatous inflammation 2, 3.
A distinctive histological attribute of granulomatous inflammation admixed with acute inflammation and concurrent cystic spaces indicates a probable accumulation with exceptional, gram positive bacilli within the clear, vacuolated spaces. Microbiological analysis for isolation of Corynebacterium, particularly Corynebacterium kroppenstedtii is necessitated in concurrence with aforesaid morphological aspects to arrive at a cogent diagnosis. Corynebacterium is a gram positive bacillus which appears as a constituent of skin microbiota. Thus, a possible source can be on account of bacterial colonization, infection or contamination. Corynebacterium kroppenstedtii can be isolated in around two fifths (40%) instances of definitive granulomatous lobar mastitis 6, 7.
Granulomatous mastitis necessitates a demarcation from Squamous Metaplasia of Lacteriferous ducts (SMOLD), a condition which demonstrates epithelium lined lacteriferous ducts with concurrent squamous metaplasia, essentially occurs in the nipple and delineates a singular sinus tract at the edge of the aerola. Multinucleated giant cells can be associated with keratinous debris. The condition can be incited by tobacco intake 2, 3.
Investigative Assay
Ultrasonography depicts cutaneous thickening, cutaneous retraction and a persistent, poorly circumscribed, hypoechoic mass of varying diameter. The mass is initially subdermal and multiple, deep-seated masses may be cogitated, occasionally extraneous to the breast tissue. Lymphadenopathy may or may not be detectable. Ultrasonography is beneficial in discerning infectious conditions such as effusions, inflammation of mammary parenchyma and fatty tissues, abscess formation along with the emergence of a fistulous tract 6, 7.
Magnetic resonance imaging (MRI) depicts a heterogeneous admixture of fibrous and glandular tissue along with several, tiny, fluid-filled cavities rimmed with inflammatory cells and granulomas within the breast tissue, a few of which progress to fistula formation 6, 7.
Magnetic resonance imaging can depict a gradually evolving mass or a non-specific, heterogeneous enhancement or a clustered augmentation with ring formation within the breast tissue. Adjunctive findings on magnetic resonance imaging are defined as cutaneous thickening and retraction along with retraction of the nipple. Mammography delineates an enhanced echogenicity, focal asymmetry of the nodule disassociated with nodular distortion and micro-calcifications along with infrequent, well-defined mammary nodules. Doppler studies exemplify an amplification of nodular vascularization. Levels of certain cytokines such as interleukin 33(IL-33) are enhanced in idiopathic granulomatous mastitis with a sensitivity of 93.7% and specificity of 96%, values which are beneficial in segregating granulomatous mastitis from carcinoma breast 2, 3.
Nevertheless, aforesaid non – specific imaging features can be recapitulated by carcinoma breast. Thus, a cogent, non invasive diagnostic modality to differentiate granulomatous mastitis from carcinoma breast remains to be established.
Core needle biopsy is a sensitive technique (94.5%) to detect granulomatous mastitis whereas a fine needle aspiration is efficacious in around 39% instances. Also, fine needle aspiration may be inadequate in demarcating idiopathic granulomatous mastitis from adjunctive granulomatous diseases of the breast 7, 8.
Therapeutic Options
A consensus for optimal therapeutic modality for managing idiopathic granulomatous mastitis remains elusive. Granulomatous mastitis can be treated with various modalities such as systemic therapy composed of oral and topical corticosteroids, immune-suppressants or surgical excision. Therapeutic intervention is mandated for symptomatic palliation. Untreated granulomatous mastitis can spontaneously resolve in roughly one to two years. Granulomatous mastitis can be treated medically with antibiotics, systemic steroids as with prednisone or mycophenolate, non steroidal anti inflammatory drugs(NSAIDs), immunosuppressive agents such as methotrexate or azathioprine 7, 8.
Surgical extermination of granulomatous inflammation followed by placement of a surgical drain within the wound with concomitant administration of steroids are a preferred approach. Also, antibiotics following a wide surgical eradication of the nodule with a broad margin of the healthy tissue or a mastectomy with subsequent immune suppression can be adopted. Alternatively, a surgical excision with a wide perimeter of uninvolved tissue with concurrent antibiotic administration can be adopted. Localized excision with a broad perimeter of normal tissue is employed in around three fifths (59%) of subjects and usually the wound heals within a minimal period. Drainage of associated breast abscess is also employed. Localized surgical eradication of the nodule can be accompanied by complications such as infection of the surgical site, wound disruption, disease reoccurrence and delayed wound healing 7, 8.
Median healing time is cogitated at around 84 days whereas median time for emergence of reoccurrence is at an estimated 280 days. Incriminated elderly populations depicting associated comorbid conditions usually demonstrate a delayed healing. Delayed healing is enunciated in an estimated 10% to 50% instances. Surgical intervention depicts wound complications in nearly half (52%) the subjects and relapse of granulomatous mastitis following surgical excision is observed in approximately 13.3% individuals 7, 8.
Therapy with steroids can be contemplated as the preferential treatment for idiopathic granulomatous mastitis. Adoption of steroids for treating granulomatous mastitis is efficacious and granulomas can be resolved in the absence of a surgical option. Primary therapy with steroids is beneficial although the treatment may extend up to six months. Essentially a conservative form of management, administration of corticosteroids delineates a superior response in an estimated three fourths(77.8%) subjects. Steroids can wither the lesion when administered pre-operatively or post- operatively in persistent masses.
Immune-suppressive agents such as methotrexate or azathioprine can be adopted in treating reoccurrences of inflammation or in subjects demonstrating deleterious outcomes of prednisone 7, 8. Granulomatous mastitis depicts elevated proportion of relapse in an estimated half (50%) the instances on account of adoption of suboptimal therapeutic modalities.
Average period of recovery of granulomas usually exceeds >one year. Chronic and reoccurring forms of disease can emerge due to adoption of various therapeutic measures 7, 8.
References
- 1.Kessler E. (1972) Granulomatous mastitis: a lesion clinically simulating carcinoma”. , Am J Clin Pathol 58(6), 642-646.
- 2.Chirappapha P, Thwaweepworadej P. (2018) Idiopathic granulomatous mastitis: a retrospective cohort between study between 44 patients with different treatment modalities”. , Ann Med Surg 36, 162-167.
- 3.Halim N A, Uthman I. (2018) Idiopathic granulomatous mastitis presenting as a breast pseudo-tumour: case reports with review of literature” Case Reports in Rheumatology. 1-5.
- 4.Altintoprak F, Kivilcim T. (2014) . , Aetiology of idiopathic granulomatous mastitis” World J Clin Cases 2(12), 852-858.
- 5.Uysal E, Soran A. (2018) Granulomatous Mastitis Study G : Factors related to recurrence of idiopathic granulomatous mastitis: what do we learn from a multicentre study”. , ANZ J Surg 88(6), 635-639.
- 6.Akahane K, Tsunoda N. (2013) Therapeutic strategy for granulomatous lobular mastitis: a clinicopathological study of 12 patients”. , Nagoya J Med Sci,75(3-4): 193-200.
- 7.Yabanoglu H.Colakoglu T et al (2015). ”A comparative study of conservative versus surgical treatment protocols for 77 patients with idiopathic granulomatous mastitis”. , Breast J 21(4), 363-369.
