Presentation of Neutralizing Antibodies in Single- or Pooled-Convalescent Immune Plasma from Donors to Prevent the Current SARS-CoV-2 Pandemic
Abstract
As there is no specific treatment yet, the fight against the COVID-19 pandemic is being carried out with great difficulty today. The use of immune plasma is seen as a promising option since there is expectation that it will reduce mortality, as in influenza pandemics experienced in 1918 and 2009. However, the safety and effectiveness of this treatment option against SARS-CoV-2 viruses are not known for certain. In addition, the optimal obtaining methods and protection time of neutralizing antibodies to be used to provide passive immunization are not fully known. Therefore, it would be very useful to investigate the most effective neutralizing antibody collection methods without disrupting the overall structure and effectiveness of the antibodies subject to the use of the convalescent immune plasma. For this purpose, we found it appropriate to prepare a broad review on the structure and properties of antibodies, as well as the principles and storage conditions of antibodies to be used in passive immunization.
Author Contributions
Academic Editor: Raul Isea, Fundación Instituto de Estudios Avanzados -IDEA, Venezuela.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Fatih Ozcelik, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
The antibodies produced by the plasma cells of the immune system and called immunoglobulin are molecules made of glycoprotein. Immunoglobulin isotypes include two light chains (LCs), each called kappa (κ) and lambda (λ), weighing 25 KDa, and two heavy chains (HCs), each weighing 50 KDa. Both light and heavy chains of the immunoglobulin molecule are divided into variable (in the heavy chain: VH and in the light chain: VL) and constant (CH and CL) domains. These domains in the fragment antigen-binding (Fab) in the antibody are called complementary-determining regions (CDRs). CDRs form the basis of the immunological specificity of the antibody 1,2. The three hypervariable regions (L1, L2 and L3; H1, H2 and H3) within each variable domain are responsible for the antigen-specific recognition function (Figure 1). Antibody-antigen interaction works with structural interaction motifs and amino acid residues. Antibody binding spaces are segmented at the motif level, not at the amino acid level. The vast majority of these are localized to CDRs 3. The part of the antibody that communicates with the immune system through complement and cell receptors is the Fc region. The Fc portion is located at the base of the Y-shaped antibody. The Fab parts, which interact with the antigen, are located at both upper ends of the Y. The main function of the humoral system is that plasma cells produce antibodies with these two regions. Humoral specificity represents the ability of antibodies to selectively interact with the antigen via epitope-paratope. The paratopic regions of the antibody, which recognize the epitope region of the antigen, are rich in important amino acid residues such as tyrosine and serine, and the antibody-antigen interaction in general is modular. Evolutionarily, it is advantageous to have short motifs instead of long motifs in these regions because short motifs reduce the potential escape area for antigens. However, these short motifs make it difficult for the immune system to distinguish the alien from the self. Of the CDRs regions, only the VH-CDR3 region is mandatory for antibody-antigen interaction. Other regions are thought to be necessary for fine tuning in antibody recognition 4, 5, 6, 7, 8. Specifically, the amino acid trend detected in CDRs in antibodies differs between antibody-antigen complexes. Antibodies recognize antigens by Fab regions. In this recognition, the paratope and the epitope regions are connected very precisely. Thanks to this binding, the marked antigen (bacteria, virus or other pathogens) is recognized and destroyed by the immune system including macrophages, or some of them are neutralized directly 9, 10, 11. This binding of the antibody breaks the potential of the pathogens that cause disease.
Figure 1.Antibody structure and functional Fc and Fab regions. C: Constant domain, V: Variable domain, H: Heavy chain, L: Light chain
Configuration of Antibodies and Disruptive Effects
Antibodies with globular protein structure are organized in a significant proportion with structures such as α-helix and β-sheet. In the rest, β-bends or coils can be observed. These structures are not random but relatively more regular structures. The side chains of the secondary structures which close to each other combine to form super secondary structures (motifs). When antibodies or proteins are denatured, random coils dominate the structure. The hydrophobic side chains of amino acids which make up these globular proteins, are embedded inside and hydrophilic groups are located on the surface of the protein. This structural organization creates the maximum number of hydrogen bonds, disulfide bonds and ionic interactions and ensures proper folding of proteins to perform their functions and increases their durability. Furthermore the chaperones play a major role in the formation of the three-dimensional structure of proteins during their synthesis 12, 13, 14, 15, 16, 17. Although the primary structure of proteins is formed with highly resistant peptide bonds, secondary, tertiary, and quaternary structures are composed of noncovalent weak bonds such as hydrogen, ionic and van der Waals forces, except disulfide. All these complex modular structures are sensitive to chemical, radioactive, heat, pressure, friction, oxidant and heavy metals (such as mercury, lead, zinc, manganese and cadmium). It has been reported that the stability and function of methionine and tryptophan residues of antibodies are sensitive to UV light, high temperature and reactive oxygen radicals 18, 19, 20, 21. It has also been found that freezing during storage of antibodies or other proteins can result in protein aggregation by increasing the acidification of the solution, affecting protein functions and disrupting the secondary and tertiary structure 22, 23, 24. However, it is believed that this effect will not be a problem for many polyclonal IgGs with a pI value (isoelectric point) of 4.7-7.0, except those with a pI value of 7.0. In general, antibodies should be stored in a sterile environment at ≤ 4 ° C 25. Moreover, storage at room temperature can often lead to disruption and immobility of antibodies due to microbial proliferation. Storage of lyophilized monoclonal antibodies at ≤ -20 ° C and the addition of sucrose (or trehalose) and / or polyols (glycerol and sorbitol) to the formulation retains their activity for a long time (3-5 years) 25,26. For any reason, proteins and antibodies that are damaged structurally or functionally are rapidly destroyed in the body. These endogenous proteins are marked by ubiquitin and shipped for destruction.
Depending on the characteristics and storage conditions of the antibody, the shelf life of the antibody can range from 3 days to one year at + 4 0C. However, some types of antibodies can be stored for much longer. To protect the activity, prevent the aggregation and extend the storage life, concentrated substances (~ 1 M) such as sucrose are added in a suitable temperature and pH environment 26, 27, 28. The stability of proteins is also related to the amount of aromatic amino acid residues that can absorb UV light in the environment. It was determined that the tryptophan residue of the IgG exposed to UV turned into glycine or glycine hydroperoxide 29,30. Therefore, the use of UV light exposed antibodies is not recommended. Antibodies can be affected by physical or mechanical trauma due to high frequency vibration, vortexing, shaking, defrosting, pump operation and filtration under pressure. It is also reported that such effects can disrupt the structure and function of antibodies and cause their aggregation 31, 32, 33, 34. The effect of exposure to various nonpolar surfaces and frictional interaction may impair the functions of proteins although the exact mechanisms are not fully understood 32,35,36. Therefore, it should be noted that antibody obtaining procedures should contain less procedures as much as possible without containing irradiation and mechanical stresses.
Neutralizing Antibody Types and Mechanism of Action
Immunoglobulins can be secreted by B cells (plasma cells) or can be found as transmembrane proteins on the non-polar surface of the B cells. Immunoglobulins have the ability to combine with the specific antigen (immunogen) that triggers their production 13, 14, 15, 16, 17. Antibodies can neutralize viruses by combining with neuraminidase (N) and hemagglutinin (H) (resembling sialic acid residues in red blood cells), which allows viruses to attach to respiratory epithelial cells. Then they can neutralize them 9,37. The recognition of antigens by B cells occurs with B cell receptors or immunoglobulin, while recognition by the T cell occurs only with the surface receptor. In the early period of exposure to the pathogen, IgM antibody is synthesized, while in the late period, the lower molecular weight IgG antibody is synthesized. The largest of the antibodies, IgM is a pentamer with ten antigen binding sites. Due to its’ larger size, it can show its effectiveness only in intravascular space. However, IgG antibody can easily circulate effectively between intravascular spaces and interstitial tissues. Secretarial IgA is located on the mucous surfaces where it is produced. This antibody forms the first line of defense against mucosal entering pathogens such as influenza, rhinovirus and human immunodeficiency virus (HIV). It is thought that IgA antibodies may also be effective against the novel corona virus (SARS-CoV-2), which enter through mucosal surfaces 38, 39, 40, 41, 42. IgM class macromolecules are potent agglutinator agents. They can quickly activate the complement system in the classical way. They are of great importance in the early phase of the primary immune response, as they are rapidly produced in a few days to provide protection when a foreign antigen enters the body. IgG, which has a longer life than IgM, is the dominant serum antibody. IgG also directly contributes to an immune response, including neutralization of toxins and viruses 43,44,45. Thanks to its specific antigen recognition function, this antibody provides protective immunity in blood and tissue against many types of microorganisms (bacteria, viruses and some fungi). In a study conducted by Castilla et al., cloning of a monoclonal antibody capable of neutralizing the corona virus, an infectious gastroenteritis agent, was achieved. These chimeric immunoglobulins were expressed in murine myeloma or COS cells, which are fibroblast-like cell lines derived from monkey kidney tissue. It was found that these secreted recombinant antibodies reduced the infectious virus more than 104 times. The researchers found that IgA can neutralize 50 times more TGEV than recombinant monomeric IgG containing the same antigenic binding site 40. These findings are in line with the knowledge that IgA in mucosal secretions provides the initial immunological barrier against many pathogens, especially viruses that invade the mucosal surfaces of the body 39,46,47. All these data suggest that neutralizing antibodies developed after corona virus infection may be effective in limiting the virus.
Collection of Convalescent Immune Plasma and Adverse Reactions
While animal viruses infect cells by binding to the cell type specific cell surface receptor, many antibodies that neutralize viruses directly inhibit viral binding to surface receptors. As with neutralization of toxins, high affinity IgA and IgG antibodies from these antibodies were found to be very important 9. In this context, antibody response developed by symptomatic or non-symptomatic people to combat the COVID-19 pandemic caused by the SARS-CoV-2 virus is considered as an option in treatment worldwide because no specific treatment and vaccine has been yet found in the fight against COVID-19 48,49. For this reason, it is recommended to use convalescent immune plasma, which is thought to contain high number of neutralizing antibodies. This approach is conceptually based on passive immunotherapy. However, the safety and efficacy of the convalescent immune plasma for COVID-19 therapy has not yet been confirmed. In addition, the duration of protection of antibodies with protective properties is uncertain. There are several methods of collecting the convalescent immune plasma from donors recovered from COVID-19 disease. The first is the provision of blood components by the conventional (classical) method. In fact, the principles used in the preparation of the fresh frozen plasma component today are the same as in this method. Similar methods were used in 1918 to obtain blood products in the Spanish flu pandemic. The other method is the convalescent immune plasma supplied using digital technology apheresis device. These two methods have either advantages and disadvantages 50, 51, 52, 53.
Studies on H1N1 influenza pandemic, which started in Spain in 1918 and 2009, showed that blood transfusions applied at that time reduced mortality in influenza patients complicated by pneumonia 50,51,54. In addition, early transfusions have been reported to provide significant improvement in clinical signs and symptoms. However, significant side effects can be observed due to the use of convalescent immune plasma. Among these, allergic reactions, anaphylaxis, transfusion-related acute lung injury (TRALI) and hemolysis due to transfusion antibodies (especially against A and B blood group antigens) are the most feared side effects 55,56. However, in many studies, no serious side effects related to the convalescent immune plasma used for patients with H5N1 influenza and Severe acute respiratory syndrome (SARS) have been reported. It has been reported that mostly mild fever and chills are observed 57, 58, 59.
Whole blood, which is a mixture of different types of cells, colloids and crystalloids, is used by separating it into components according to need. Whole blood can be conventionally separated into blood components as erythrocyte concentrate, platelet concentrate, fresh frozen plasma and cryoprecipitate by the classical method (Figure 2). The same blood components can be obtained directly from the donor blood circulation using an apheresis device. In the apheresis method, besides these components, granulocyte and peripheral blood hematopoietic stem cell collection and therapeutic plasma exchange can be performed 55,60. Apheresis method is based on the difference of size / specific weights of blood cells. In this method, the centrifugation that distinguishes according to the specific weight and the filtration principle that distinguishes according to their sizes are used (Figure 3). Chromatography principle can be added to these principles depending on situation. In the principle of centrifugation, the device provides continuous blood flow from the patient to the centrifuge. An anticoagulant (usually citrate) is added before centrifugation. The centrifuge rotates at 2000–2500 rpm to separate the various components of anticoagulated blood by their density or specific weight. The separation takes place in a non-selective plasma layer with respect to the axis of rotation, the buffy layer of platelets, lymphocytes, monocytes, granulocytes and the most distant erythrocytes. The effectiveness of separation of various blood components depends on the dimensions of the centrifuge and the speed (revolutions per minute) and duration of the centrifuge, which creates the force of gravity. The filtration principle is also used in hemodialysis. Microprocessors in apheresis devices control the blood flow rate of the device, the amount of citrate anticoagulant mixed with the whole blood, the rotational speed of the centrifuge, the separation of the blood component and the rest of the components returned to the donor 52, 61, 62, 63, 64. Whole blood from is directed to the membrane with different sized pores (0.2-0.65 micron) at a given pressure and flow rate. Smaller structures from these pores can pass through the filter. It is an extremely comfortable and fast process.
Figure 2.Separation of blood components from the whole blood by classical-conventional method. Whole blood can be separated by two different methods depending on whether the first centrifugation speed is heavy and light spin. In the Buffy coat method, centrifugation starts with heavy spin.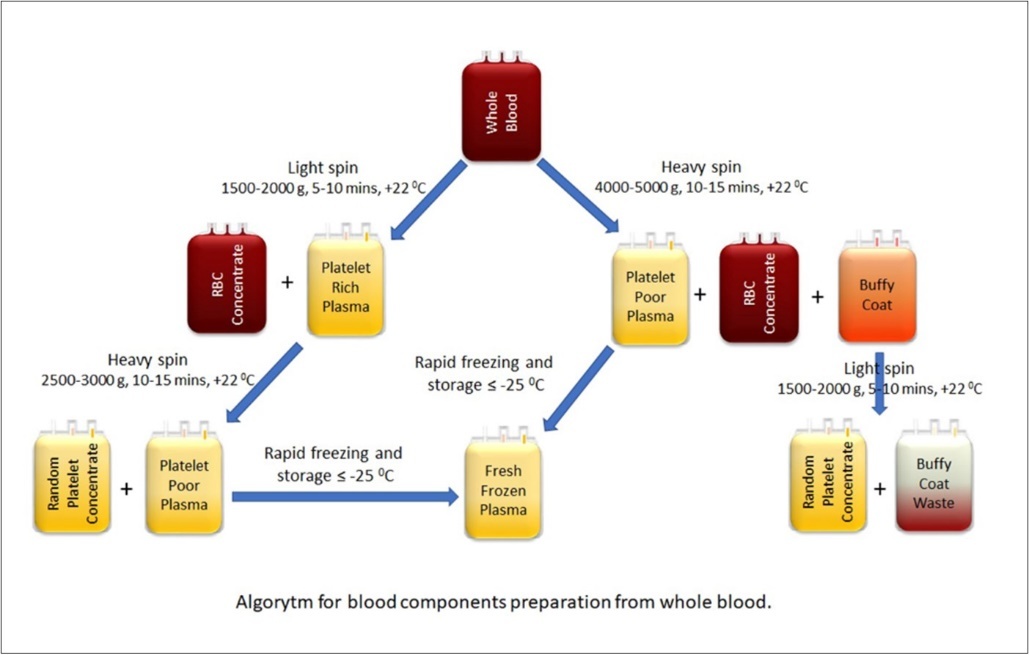
Figure 3.Centrifugation and filtration principle in collecting immune plasma component with apheresis device.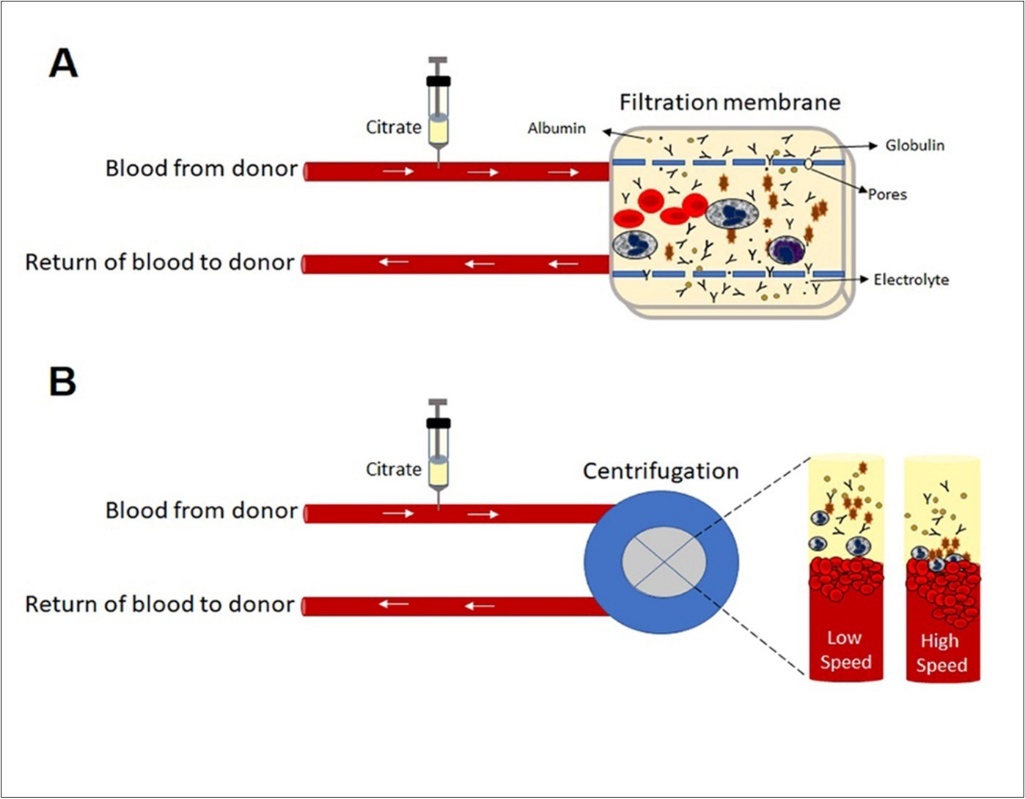
Apheresis gives the advantage of being able to collect a higher amount of plasma (ranging up to 200-600 ml) from a single person, while preventing unnecessary red cell loss from the donor. However, a maximum of 150-200 ml of convalescent immune plasma can be obtained from 1 unit of whole blood (approximately 450 ml) collected randomly by classical conventional method. However, a maximum of 150-200 ml of convalescent immune plasma can be obtained from 1 unit of whole blood (approximately 450 ml) collected randomly by classical conventional method. However, as mentioned above, we think that the application of IV citrate during filtration in apheresis method, the small porous structure of the filter membrane, the risk of contact with the active surface, physical and mechanical effects due to filtration under the pump and pressure may partially impair the structure and functions of the antibodies and cause aggregation. In addition, applied irradiation (gamma rays or UVC sterilization) or chemical pathogen inactivation procedures to kill viruses and bacteria in plasmas obtained by classical or apheresis method and freezing-thawing procedures may result in a higher amount of neutralizing antibody loss 67,68. By the way, it is reported that the Coronaviruses can remain stable at -25 0C.
Current researchers show that people with SARS-CoV-2 infection develop neutralizing antibodies against to the virus. This neutralizing formation varies depending on viruses and individual differences in the immune system. While some people respond strongly to the same pathogen, some people may have very low levels of neutralizing antibodies in their blood 48, 69, 70, 71, 72, 73, 74. Also, it is not clear whether the antibodies against SARS-CoV-2 provide immunity against the subsequent infection. To clarify this issue, Grzelak et al conducted a study in which people who had COVID-19 infection investigated the presence of seroconversion and neutralizing antibodies. In this study, they investigated antibodies against nucleoprotein (N) or trimeric Spike (S) virus proteins by ELISA method. Antibodies against the virus were detected 4-15 days after the onset of the disease. They found seropositivity in 29% of people with several symptoms (within 15 days after symptoms) and in 3% of healthy donors selected from the region with the COVID-19 outbreak 48. These findings were evidence that neutralizing antibodies were produced at a level that could be measured against the virus. Generally, it is believed that the neutralization of viruses by antibodies occurs when antibodies bind to the proteins of the virus that attach to cell receptors. Thus, antibodies prevent the virus from adhering to epithelial cells. Again, mucosal secretion antibodies of type IgA have been found to prevent virus attachment to the cell by a similar mechanism 46, 75, 76, 77, 78. In a study conducted to take advantage of the neutralization properties of antibodies, transient expression genes of flavivirus were transplanted into vertebrate cells to obtain antibodies that protect against virus infection 76. These and similar studies show that neutralizing antibodies are important option in resistance to the virus.
It is known that in patients with immune system depression or weakness, COVID-19 disease progresses seriously, and the neutralizing antibodies against SARS-CoV-2 virus cannot sufficiently be created. It is believed that the use of neutralizing antibodies originated from those who have previously had this disease would be beneficial in patients with COVID-19. Plasma donors can be considered as candidates at least 14 days after all symptoms have improved and the COVID-19 PCR test has become negative. Donor candidates may become donors after compulsory tests and standard procedures established by regional committee. It is especially emphasized that male donors should be primarily selected to prevent the risk of Transfusion Related Acute Lung Injury (TRALI) 48,52. However, we think that the effectiveness of neutralizing antibodies in immune plasmas to be obtained from donors will vary depending on the methods of collection, storage conditions and whether they are exposed to radiation or chemical agents that disrupt the antibody structure. Apheresis devices allows the collection more often and amount of plasma by preventing the collection of unnecessary components; however, during this procedure plasma undergoes a heavy filtering and certain filtering process. Furthermore, a freezing procedure is required in order to stock the immune plasma for longer time periods. In addition, pathogen inactivation processes (radioactive irradiation: gamma irradiation and biochemical treatment) to prevent transmission of infections can disrupt the conformational structure of antibodies in the immune plasma. In this way, the neutralizing antibodies that are denatured or whose load is altered should not be expected to play an effective role in virus inactivation. According to our experience and feedback, the expected benefit could not obtain if plasma collected by apheresis method. However, the results have not been compared with the classical conventional methods yet. Our research on this topic will be presented later. This situation contrasts with the results of blood transfusions used during the Spanish virus pandemic in 1918. The main reason might be the utilization of different collection methods and the area of expertise. Because the immune plasmas used in Spanish virus outbreak were randomly collected convalescent immune plasma by classical methods and were not subjected to pathogen inactivation or freezing. In the other word, neutralizing antibodies were used where in vivo conditions maintained significantly.
Due to a significant number of COVID-19 donors failed to produce sufficient neutralizing antibodies may be the answer to why immune plasma use has failed 48,52. We propose that such a problem can be solved by pooling of convalescent immune plasma from at least 3 or 4 donors. In this way, the possibility of the success of neutralizing antibodies in the immune plasma will be increased 3 to 4 times. Since such an approach may cause volume loading in a patient with heart failure and volume overload, the condition of the patient must be evaluated before the administration and the amount should be adjusted case by case. Another suggestion is to give pooled convalescent immune plasmas without freezing and exposure to pathogen inactivation (irradiation or biochemical treatment) in order to protect neutralizing antibodies. However, we also know that such an application will be a subject to a lot of criticism by clinicians. The method we recommend is like the use of pooled platelet suspension applied today. In addition, the risk of transfusion reaction due to transfusion of pooled convalescent immune plasma formed from 3 or 4 donors is not higher than the pooled platelet suspension. Fever or mild allergic reactions that are mostly related to protein can be eliminated by using acetaminophen or antihistamine agents depending on the patients’ condition 79,80. Again, the extent to which the pathogen inactivation procedure is required in patients with high viral load and the danger of not performing pathogen inactivation is controversial. The benefit-harm situation should be considered here. Another situation to be investigated is the effectiveness of pooled convalescent immune plasma use. Unfortunately, studies comparing the classical conventional and apheresis method of convalescent immune plasma are quite limited.
Conclusion
In the fight against the COVID-19 pandemic, obtaining the collected neutralizing antibodies with the method that will suffer the least is important for the success of the treatment. In addition, studies in which apheresis and classical methods are compared to single donor and pooled options are needed to determine the most appropriate collection method.
References
- 1.Inbar D, Hochman J, Givol D. (1972) . Localization of Antibody-Combining Sites within the Variable Portions of Heavy and Light Chains.Proceedings of the National Academy of Sciences 69(9), 2659-2662.
- 2.Xu J L, Davis M M. (2000) . Diversity in the CDR3 Region of VH Is Sufficient for Most Antibody Specificities.Immunity 13(1), 37-45.
- 3.Kunik V, Peters B, Ofran Y. (2012) . Structural Consensus among Antibodies Defines the Antigen Binding Site. Baker B, ed.PLoSComputational Biology 8(2), 1-12.
- 4.Robin G, Sato Y, Desplancq D, Rochel N, Weiss E et al. (2014) . , Restricted Diversity of Antigen Binding Residues of Antibodies Revealed by Computational Alanine Scanning of 227 Antibody–Antigen Complexes.Journal of Molecular Biology 426(22), 3729-3743.
- 5.Gainza P, Sverrisson F, Monti F, Rodolà E, Bronstein M et al. (2019) Deciphering interaction fingerprints from protein molecular surfaces.bioRxiv. 1-44.
- 6.Chuang G-Y, Zhou J, Acharya P. (2019) Structural Survey of Broadly Neutralizing Antibodies Targeting the HIV-1 Env Trimer Delineates Epitope Categories and Characteristics of Recognition.Structure. 27(1), 196-206.
- 7.Jespersen M C, Mahajan S, Peters B, Nielsen M, Marcatili P. (2019) . Antibody Specific B-Cell Epitope Predictions: Leveraging Information From Antibody-Antigen Protein Complexes.Frontiers in Immunology.10. doi: 10.3389/fimmu.2019.00298 .
- 8.Akbar R, Robert P A, Pavlović M. (2019) A compact vocabulary of paratope-epitope interactions enables predictability of antibody-antigen binding.bioRxiv.
- 9.Janeway C, Travers P, Walport M, Shlomchik M. (2001) . The Immune System in Health and Disease. In:Immunobiology. 5th ed. New York:Garland Science; 425-596.
- 10.Maverakis E, Kim K, Shimoda M. (2015) Glycans in the immune system and The Altered Glycan Theory of Autoimmunity: A critical review.Journal of Autoimmunity. 57, 1-13.
- 11.Sela-Culang I, Kunik V, Ofran Y. (2013) . The Structural Basis of Antibody-Antigen Recognition.Frontiers in Immunology.4. doi: 10.3389/fimmu.2013.00302 .
- 12.Gilliland G L, Luo J, Vafa O, Almagro J C. (2012) Leveraging SBDD in protein therapeutic development: Antibody engineering.Methods in Molecular Biology. 841, 321-349.
- 13.Padlan E A. (1977) Structural basis for the specificity of antibody–antigen reactions and structural mechanisms for the diversification of antigen-binding specificities.Quarterly Reviews of Biophysics. 10(1), 35-65.
- 14.Amzel L M, Poljak R J. (1979) Three-Dimensional Structure of Immunoglobulins.Annual Review of Biochemistry. 48(1), 961-997.
- 15.Davies D R, Metzger H. (1983) Structural Basis of Antibody Function.Annual Review ofImmunology. 1(1), 87-115.
- 16.Wilson I A, Stanfield R L. (1994) Antibody-antigen interactions: new structures and new conformational changes.Current Opinion in Structural Biology. 4(6), 857-867.
- 17.Chiu M L, Goulet D R, Teplyakov A, Gilliland G L. (2019) Antibody Structure and Function: The Basis for Engineering Therapeutics.Antibodies. 8(4), 2-80.
- 18.Chumsae C, Gaza-Bulseco G, Sun J, Liu H. (2007) Comparison of methionine oxidation in thermal stability and chemically stressed samples of a fully human monoclonal antibody.Journal of Chromatography B.850(1-2):. 285-294.
- 19.Wei Z, Feng J, Lin H-Y. (2007) Identification of a Single Tryptophan Residue as Critical for Binding Activity in a Humanized Monoclonal Antibody against Respiratory Syncytial Virus.Analytical Chemistry. 79(7), 2797-2805.
- 20.Lam X M, Yang J Y, Cleland J L. (1997) Antioxidants for Prevention of Methionine Oxidation. in Recombinant Monoclonal Antibody HER2.Journal of Pharmaceutical Sciences 86(11), 1250-1255.
- 21.Davies K J, Delsignore M E, Lin S W. (1987) Protein damage and degradation by oxygen radicals. II. Modification of amino acids.The Journal of biological chemistry. 262(20), 9902-9907.
- 22.Murphy B M, Swarts S, Mueller B M, Geer P van der, Manning M C et al. (2013) Protein instability following transport or storage on dry ice.Nature Methods. 10(4), 278-279.
- 23.Szenczi Á, Kardos J, Medgyesi G A, Závodszky P. (2006) The effect of solvent environment on the conformation and stability of human polyclonal IgG in solution.Biologicals. 34(1), 5-14.
- 24.Park J, Nagapudi K, Vergara C, Ramachander R, Laurence J S et al. (2013) . Effect of pH and Excipients on Structure, Dynamics, and Long-Term Stability of a Model IgG1 Monoclonal Antibody upon Freeze-Drying.Pharmaceutical Research 30(4), 968-984.
- 25.Sydow J F, Lipsmeier F, Larraillet V. (2014) . Structure-Based Prediction of Asparagine and Aspartate Degradation Sites in Antibody Variable Regions. Dübel S, ed.PLoSONE 9(6), 1-13.
- 26.Chang L, Shepherd D, Sun J, Tang X, Pikal M J. (2005) Effect of sorbitol and residual moisture on the stability of lyophilized antibodies: Implications for the mechanism of protein stabilization in the solid state.Journal of Pharmaceutical Sciences. 94(7), 1445-1455.
- 27.Argentieri M C, Pilla D, Vanzati A. (2013) Antibodies are forever: a study using 12-26-year-old expired antibodies.Histopathology. 63(6), 869-876.
- 28.Laskowski T J, Hazen A L, Collazo R S, Haviland D. (2020) Rigor and Reproducibility of Cytometry Practices for Immuno‐Oncology: A multifaceted challenge.Cytometry Part A. 97(2), 116-125.
- 29.Haywood J, Mozziconacci O, Allegre K M, Kerwin B A, Schöneich C. (2013) . Light-Induced Conversion of Trp to Gly and Gly Hydroperoxide in IgG1.Molecular Pharmaceutics 10(3), 1146-1150.
- 30.Du C, Barnett G, Borwankar A. (2018) Protection of therapeutic antibodies from visible light induced degradation: Use safe light in manufacturing and storage.European. , Journal of Pharmaceutics and Biopharmaceutics 127, 37-43.
- 31.Kiese S, Papppenberger A, Friess W, Mahler H-C Shaken. (2008) . Not Stirred: Mechanical Stress Testing of an IgG1 Antibody.Journal of Pharmaceutical Sciences 97(10), 4347-4366.
- 32.Wang W. (1999) Instability, stabilization, and formulation of liquid protein pharmaceuticals.International. , Journal of Pharmaceutics 185(2), 129-188.
- 33.Telikepalli S N, Kumru O S, Kalonia C. (2014) . Structural Characterization of IgG1 mAb Aggregates and Particles Generated Under Various Stress Conditions.Journal of Pharmaceutical Sciences 103(3), 796-809.
- 34.Randolph T W, Schiltz E, Sederstrom D. (2015) . Do Not Drop: Mechanical Shock in Vials Causes Cavitation, Protein Aggregation, and Particle Formation.Journal of Pharmaceutical Sciences 104(2), 602-611.
- 35.Maa Y-F, Hsu C C. (2000) Effect of high shear on proteins.Biotechnology and Bioengineering. 51(4), 458-465.
- 36.Colombié S, Gaunand A, Lindet B. (2001) Lysozyme inactivation under mechanical stirring: Effect of physical and molecular interfaces.Enzyme and Microbial Technology.28(9-10). 820-826.
- 37.Byrd-Leotis L, Cummings R D, Steinhauer D A. (2017) The Interplay between the Host Receptor and Influenza Virus Hemagglutinin and. , Neuraminidase.International Journal of Molecular Sciences 18(7), 1-22.
- 38.Schroeder H W, Cavacini L. (2010) Structure and function of immunoglobulins.Journal of Allergy and Clinical Immunology. 125(2), 41-52.
- 40.Castilla J, Sola I, Enjuanes L. (1997) Interference of coronavirus infection by expression of immunoglobulin G (IgG) or IgA virus-neutralizing antibodies.Journal of virology. 71(7), 5251-5258.
- 41.Kulkarni V, Ruprecht R M. (2017) Mucosal IgA responses: Damaged in established HIV infection-yet, effective weapon against HIV transmission.Frontiers in. Immunology.8.doi: 10.3389/fimmu.2017.01581 .
- 42.Béné M C, M de Carvalho Bittencourt, Eveillard M, Y Le Bris. (2020) . Good IgA Bad IgG in SARS-CoV-2 Infection?Clinical Infectious Diseases.1(4): 1-1.
- 43.Boes M. (2000) Role of natural and immune IgM antibodies in immune responses.Molecular Immunology. 37(18), 1141-1149.
- 44.Cavacini L A, Kuhrt D, Duval M, Mayer K, Posner M R. (2003) . Binding and Neutralization Activity of Human IgG1 and IgG3 from Serum of HIV-Infected Individuals.AIDS Research and Human Retroviruses 19(9), 785-792.
- 45.Scharf O, Golding H, King L R. (2001) . Immunoglobulin G3 from Polyclonal Human Immunodeficiency Virus (HIV) Immune Globulin Is More Potent than Other Subclasses in Neutralizing HIV Type 1.Journal of Virology 75(14), 6558-6565.
- 46.Mazanec M B, Coudret C L, Fletcher D R. (1995) Intracellular neutralization of influenza virus by immunoglobulin A anti-hemagglutinin monoclonal antibodies.Journal of virology. 69(2), 1339-1343.
- 47.Staats H F, Jackson R J, Marinaro M, Takahashi I, Kiyono H et al. (1994) Mucosal immunity to infection with implications for vaccine development.Current Opinion in Immunology. 6(4), 572-583.
- 48.Grzelak L, Temmam S, Planchais C. (2020) SARS-CoV-2 serological analysis of COVID-19 hospitalized patients, pauci-symptomatic individuals and blood donors.medRxiv. 1-18.
- 49.Cunningham A C, Goh H P, Koh D. (2020) Treatment of COVID-19: old tricks for new challenges.Critical Care. 24(91), 1-2.
- 50.Luke T C, Kilbane E M, Jackson J L, Hoffman S L. (2006) Meta-Analysis: Convalescent Blood Products for Spanish Influenza Pneumonia: A Future H5N1. , Treatment?Annals of Internal Medicine 145(8), 599-609.
- 51.Marano G, Vaglio S, Pupella S. (2016) Convalescent plasma: New evidence for an old therapeutic tool?Blood Transfusion. 14(2), 152-157.
- 52.Seghatchian J, Lanza F. (2020) Convalescent plasma, an apheresis research project targeting and motivating the fully recovered COVID 19 patients: A rousing message of clinical benefit to both donors and recipients alike.Transfusion and Apheresis Science. 1-4.
- 53.Phelan A L. (2020) COVID-19 immunity passports and vaccination certificates: scientific, equitable, and legal challenges.The Lancet. 395(10237), 1595-1598.
- 54.NPAS Johnson, Mueller J. (2002) Updating the Accounts: Global Mortality of the 1918-1920 "Spanish". , Influenza Pandemic.Bulletin of the History of Medicine 76(1), 105-115.
- 55.Basu D, Kulkarni R. (2014) Overview of blood components and their preparation.Indian. , Journal of Anaesthesia 58(5), 529-537.
- 56.O’Shaughnessy D F, Atterbury C, Bolton Maggs P. (2004) Guidelines for the use of fresh-frozen plasma, cryoprecipitate and cryosupernatant.British. , Journal of Haematology 126(1), 11-28.
- 57.Yeh K-M, Chiueh T-S, Siu L K. (2005) Experience of using convalescent plasma for severe acute respiratory syndrome among healthcare workers in a Taiwan hospital.Journal of Antimicrobial Chemotherapy. 56(5), 919-922.
- 58.Cheng Y, Wong R, YOY Soo. (2005) Use of convalescent plasma therapy in SARS patients in Hong Kong.European. , Journal of Clinical Microbiology & Infectious Diseases 24(1), 44-46.
- 59.Hellstern P, Muntean W, Schramm W, Seifried E, Solheim B G. (2002) Practical guidelines for the clinical use of plasma.Thrombosis Research.;. 107, 53-57.
- 61.McLeod B C. (2009) Therapeutic apheresis: history, clinical application, and lingering uncertainties.Transfusion. 50(7), 1413-1426.
- 62.Wehrli G. (2011) Therapeutic apheresis instrumentation.Journal of Clinical Apheresis. 26(5), 286-290.
- 63.Expert WHO. (2007) Committee on biological standardization.Recommendations on the Production, Control and Regulation of Human Plasma for Fractionation. Fifty-Sixth Report. http://www.ncbi.nlm.nih.gov/pubmed/18314862 , Geneva;
- 64.Runkel S, Haubelt H, Hitzler W, Hellstern P. (2005) The quality of plasma collected by automated apheresis and of recovered plasma from leukodepleted whole blood.Transfusion. 45(3), 427-432.
- 65. (2017) . EDQM.European Committee (Partial Agreement) on Blood Transfusion (CD-P-TS ). Guide to the Preparation, Use and Quality Assurance of Blood Components, 19th Edition. F-67081 , Strasbourg, France; .
- 66.Mundt J M, Rouse L, Bossche J Van den, Goodrich R P. (2014) . Chemical and Biological Mechanisms of Pathogen Reduction Technologies.Photochemistry and Photobiology 90(5), 957-964.
- 67.Lamarre A, Talbot P J. (1989) Effect of pH and temperature on the infectivity of human coronavirus 229E.Canadian Journal of Microbiology. 35(10), 972-974.
- 68.Geller C, Varbanov M, Duval R. (2012) Human Coronaviruses: Insights into Environmental Resistance and Its Influence on the Development of New Antiseptic Strategies.Viruses. 4(11), 3044-3068.
- 69.Wölfel R, Corman V M, Guggemos W. (2020) Virological assessment of hospitalized patients with COVID-2019.Nature. 581(7809), 465-469.
- 70.Wu F, Wang A, Liu M. (2020) Neutralizing antibody responses to SARS-CoV-2 in a COVID-19 recovered patient cohort and their implications.medRxiv.
- 72.Poh C M, Carissimo G, Wang B. (2020) Potent neutralizing antibodies in the sera of convalescent COVID-19 patients are directed against conserved linear epitopes on the SARS-CoV-2 spike protein.bioRxiv. 1-11.
- 73.NMA Okba, Müller M A, Li W. (2019) Patients.Emerging Infectious Diseases. Severe Acute Respiratory Syndrome Coronavirus 2−Specific Antibody Responses in Coronavirus Disease 26(7), 10-3201.
- 74.Zhao J, Yuan Q, Wang H. (2020) Antibody responses to SARS-CoV-2 in patients of novel coronavirus disease 2019.Clinical Infectious Diseases. 1-22.
- 75.Duan L, Bagasra O, Laughlin M A, Oakes J W, Pomerantz R J. (1994) Potent inhibition of human immunodeficiency virus type 1 replication by an intracellular anti-Rev single-chain antibody.Proceedings of the National Academy of Sciences. 91(11), 5075-5079.
- 76.Jiang W, Venugopal K, Gould E A. (1995) Intracellular interference of tick-borne flavivirus infection by using a single-chain antibody fragment delivered by recombinant Sindbis virus.Journal of virology. 69(2), 1044-1049.
- 77.Mazanec M B, Kaetzel C S, Lamm M E, Fletcher D, Nedrud J G. (1992) Intracellular neutralization of virus by immunoglobulin A antibodies.Proceedings of the National Academy of Sciences. 89(15), 6901-6905.
- 78.Mazanec M B, Nedrud J G, Kaetzel C S, Lamm M E. (1993) A three-tiered view of the role of IgA. in mucosal defense.Immunology Today 14(9), 430-435.
Cited by (4)
This article has been cited by 4 scholarly works according to:
Citing Articles:
Xin Qian, Eunyee Koh, F. Du, Sungchul Kim, Joel Chan - CHI Extended Abstracts (2020) Semantic Scholar
