Abstract
Background
The morbidity and mortality associated with the multi drug resistant Gram negative bacterial infections pose a significant and growing challenge to clinical practitioners. Klebsiella pneumoniae, which is a substantial nosocomial pathogen and confer high levels of resistance to broad-spectrum antibiotics including carbapenems. The aim of this study is to determine the resistance profiles of Klebsiella pneumoniae strains isolated from various clinical specimens.
Material and Method
In this study, the antibiotic susceptibility profiles of 2452 Klebsiella pneumoniae strains isolated from various clinical specimens between July 2015 and November 2019 were considered retrospectively. Blood culture vials were incubated in Bac T / Alert 3D automated system, other samples were cultured on blood and Eosin Methylene-blue (EMB) medium. The identification and antibiotic susceptibility tests of the isolated bacteria were performed by VITEC 2 Compact ID-AST cards (BioMérieux, Marcy l’Etoile, Fransa) and the results were evaluated according to the standards of the Europian Committee on Antimicrobial Susceptibility testing (EUCAST). The first sample of each patient was included in the study. The strains with inappropriate results were re-studied with gradient agar diffusion test (BioMérieux, Marcy l’Etoile, Fransa).
Results
The study included a total of 2452 Klebsiella pneumoniae strains and the distribution of samples was as follows: 894 (36, 5 %) blood, 790 (32, 2%) bronchial lavage, 211 (8,6 %) wound, 148 (6 %) drainage, 148 (6 %) peritoneal fluid, 95 (3, 9 %) tracheal aspirates, 64 (2,6%) catheter, 62 (2,5 %) urine, 40 (1,6%) other samples (pleural fluid, cerebrospinal fluid, throat ). In this study, antibiotic susceptibility test results indicated that the resistance to tigecycline was lowest (0, 3%). High colistin resistance ratio (33, 5 %) in carbapenem-resistant Klebsiella pneumoniae was considered to be of concern.
Conclusion
In this study, high rates of resistance to carbapenems were noteworthy. Determination of hospital antimicrobial resistance rates will be useful in developing antibiotic use policies of each hospital, in the treatment of causative agents, in selecting antibiotics according to antimicrobial susceptibility.
Author Contributions
Academic Editor: Jun Wan, Department of Medical and Molecular Genetics, Indiana University School of Medicine, United States.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Selin UGRAKLI, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
The worldwide increase in the occurrence of antimicrobial resistance (AMR) and prevalence of infections with multidrug-resistant (MDR) Gram negative Enterobacteriaceae has become a global health problem. Antimicrobial resistance could be accelerated by the misuse of antibiotics in animals and humans, leading to restricted treatment options, poor clinical outcomes and increasing hospital costs 1, 2. The majority of infections caused by K. pneumoniae are healthcare-associated and can emerge rapidly between patients via the hands of hospital staff, leading to nosocomial outbreaks 3. K. pneumoniae is encountered as an opportunistic pathogen in humans and other mammals, colonizing the gastrointestinal tract, skin and nasopharynx 4. Colonization of Klebsiella pneumoniae has been reported to be associated with long-term hospitalization, previous operation history, intravenous and urinary catheter administration and overuse of antibiotics 5. This appreciable efficiency of colonization, enhanced by acquired resistance to antibiotics, enables K. pneumoniae to persist and spread rapidly in health care settings 6. Carbapenemases are major enzyme groups responsible for antibiotic resistance in Enterobacteriaceae. Carbapenamase genes are often located on plasmids that can be exchanged between Gram negative bacteria 3. Treatment options for infections with carbapenemase-producing Klebsiella pneumoniae strains are limited and associated with high mortality 7. In many countries large percentages of antibiotics used in animals belong to what is categorized as critically important antimicrobials by the World Human Organisation (WHO) 8. Widespread use of antibiotics in animals as metaphylaxis or growth promoter may contrubite the existence of super bugs and the dramatic emerge of resistance gene over the world 9.
Therefore, surveillance programs are needed to establish antibiotic resistance policies, to determine treatment schemes for infections with multi drug resistance bacteria and to observe emerging new resistance phenotypes.
The aim of this study was determine the antibiotic susceptibility of Klebsiella pneumoniae isolated in our hospital and put in order empirical treatment schemes based on these data.
Material and Methods
A total of 2452 K. pneumoniae strains isolated from various clinical specimens sent to our laboratory from different clinics of Meram Medical Faculty Hospital between July 2015 - November 2019 were included in the study. Except for blood cultures, other submitted clinical samples were incubated on 5% sheep blood agar and Eosin Methylene Blue agar (EMB) at 37 ° C for 18-24 hours. Blood culture vials were incubated in an automated system (BacT / Alert 3D, BioMérieux, France) for 5 days. After the positive signal was obtained, Gram staining was inoculated on appropriate media as indicated above. The isolated strains were identified using VITEK 2 Compact (BioMerieux Inc., France) system. Antibiotic susceptibility tests were performed with the VITEK 2 (Bio Merieux Inc., France) automated system and evaluated according to the recommendations of the European Committee on Antimicrobial Susceptibility Testing guidelines 10. The broth microdilution method (BMD) proposed by EUCAST for detecting colistin sensitivity in Klebsiella pneumoniae could not be applied because it was a retrospective study. Antibiotic susceptibility was re-studied by E-test (BioMérieux,Marcy l’Etoile, Fransa) in incompatible strains.
Results
The isolated strains included in the study were mostly isolated from blood; this was followed by bronchial lavage, wound, drainage and other clinical specimens. The distribution of isolated K. pneumoniae according to clinical samples is shown in Table 1.
Table 1. Distribution of isolated Klebsiella pneumoniae strains according to clinical samples| Sample type | Number | Persentage (%) |
|---|---|---|
| Blood | 894 | 36,5 |
| Bronchial lavage fluid | 790 | 32,2 |
| Wound | 211 | 8,6 |
| Drainage | 148 | 6 |
| Peritoneal fluid | 148 | 6 |
| Tracheal aspirates | 95 | 3,9 |
| Catheter | 64 | 2,6 |
| Urine | 62 | 2,5 |
| Other * | 40 | 1,6 |
| Total | 2452 | 100 |
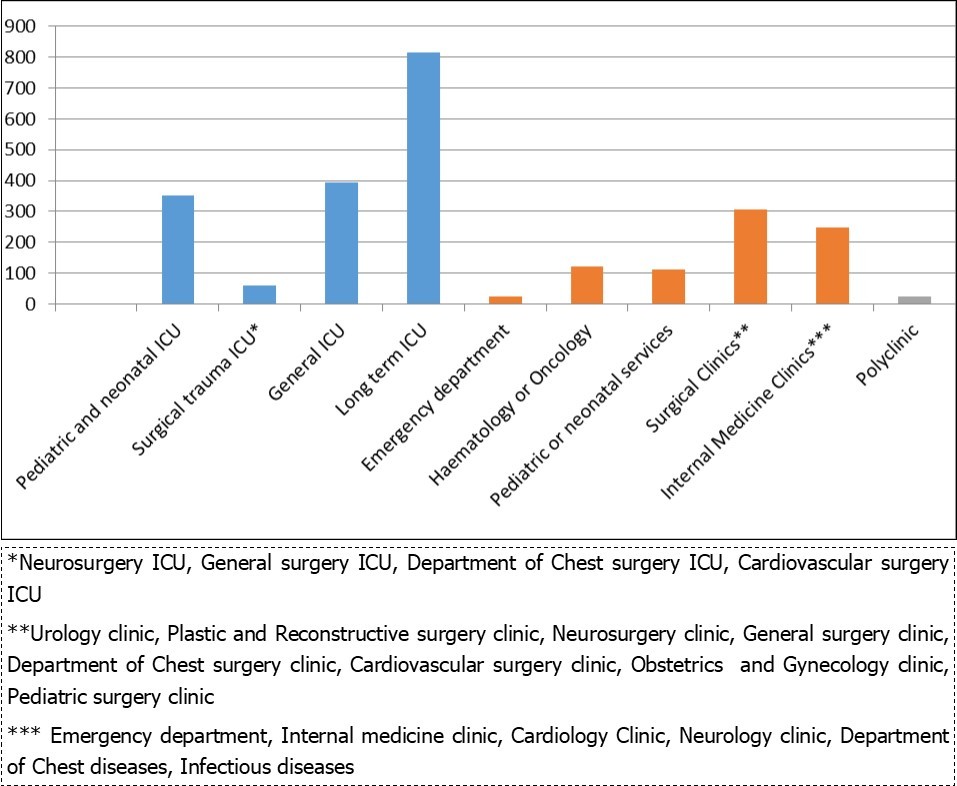
Of the isolates, 1616 (65,9 %) were obtained from intensive care units, 813 (33,2 %) from service and 23 (0,9 %) from polyclinic patients. The disribution of isolated strains according to clinics is given in Table 2 in detail. The antibiotic susceptibility test results of K. pneumoniae strains indicated that the most effective antibiotics were tigecycline, colistin and amicasin respectively. The detected high rates of meropenem resistance (63,6 %) were found to be concern. Resistance ratios and numbers of the isolates are given in Table 3.
Table 2. The isolates of Klebsiella pneumoniae strain distribution to clinics Table 3. The distribution of resistance rates of isolated Klebsiella pneumoniae| Antibiotics | Number of resistant strains | Ratio of resistant strains (%) |
|---|---|---|
| Amoxicillin clavulonic acid | 2264 | 92,3 |
| Amikacin | 1120 | 45,7 |
| Ceftazidime | 2122 | 86,5 |
| Ciprofloxacin | 1862 | 75,9 |
| Ceftriaxone | 2220 | 90,5 |
| Colistin | 822 | 33,5 |
| Cefuroxime | 2252 | 91,8 |
| Cefuroxime axetil | 2452 | 100 |
| Cefazolin | 2293 | 93,5 |
| Ertapenem | 1841 | 75 |
| Cefepime | 241 | 87 |
| Cefoxitin | 1744 | 71,1 |
| Gentamicin | 1290 | 52,6 |
| Meropenem | 1559 | 63,6 |
| Trimethoprim- sulfamethoxazole | 1900 | 77,5 |
| Tigecycline | 9 | 0,4 |
| Piperacillin- tazobactam | 2133 | 87 |
Discussion
The resistance rates that we found in our study have increased over the years, especially for critical broad-spectrum antibiotics. It may be due to the fact that the isolates included in the study were mostly obtained from intensive care patients (65,9 %) in order to detect high antibiotic resistance rates. Resistant nosocomial infections are the most common places are intensive care units (ICU). Empirically, missuse / overuse of broad-spectrum antibiotics leads to the selection of resistant strains. Prolonged hospitalization, invasive procedures, immune suppression of patients facilitate colonization of resistance organisms in these patients. Therefore, multidrug-resistant infections are more common in this patient group and treatment options are very limited 11. The Turkish national AMR surveillance system was established in 2011. The national reference laboratory collects data on AMR at the Public Health Institution of Turkey of the Ministry of Health. Antibiotic susceptibility testing results from blood and CSF culture isolates are collected into a standard database from participating laboratories. The data sents to Central Asian and Eastern European Surveillance of Antimicrobial Resistance (CAESAR). The CEASAR 2017 annual report Turkey data for K. pneumoniae antibiotic resistance ratio are as follows; 77 % amoxicillin-clavulanic acid, 59 % piperacillin-tazobactam, 68 % third-generation cephalosporins, 71 % ceftazidime, ertapenem, 30 % carpapenems, 48% aminoglicozid, 22% amikacin, 55 % fluoroquinolones and multidrug resistance bacteria percentage is 35 % 12.
Resistance to carbapenems (imipenem / meropenem) in the 2018 CAESAR report was comparable to that in previous years for K. pneumoniae. High levels of resistance of K. pneumoniae to third-generation cephalosporins (cefotaxime / ceftriaxone and ceftazidime) and fluoroquinolones (ciprofloxacin / levofloxacin / ofloxacin) were observed 13. In our study, ceftriaxone resistance was determined above country data. This may be due to the fact that it is a commonly used agent in empirical treatment of infections with both Gram positive and Gram negative bacteria in our hospital.
Carbapenems are broad-spectrum antibiotics containing the carbapenem ring additional to beta-lactam ring. The carbapenem groups of these antibiotics are supposed to protect the beta-lactam ring from destructive enzymes produced by bacteria. These antibiotics break down the bacterial cell wall at the penicillin-binding protein via a beta-lactam ring 14, 15.
Carbapenem resistance in Enterobacteriaceae occurs through many mechanisms. The most common one is inactivation of carbapenems via production of carbapenemases. The other resistance mechanisms include porin selectivity and increased efflux pumps 15. Three main carbapenemases have been reported, which are Klebsiella pneumoniae carbapenemase (KPC), New Delhi metallo-lactamase (NDM), and Oxacillin-hydrolysing carbapenemase (OXA). First OXA-48 positive K.pneumoniae was reported from Turkey. Then nasocomial outbreaks with strains carrying OXA-48 from the country have been reported 16. Turkey is an endemic region for OXA-48 producers. According to CAESAR 2018 annual data; although in most countries carbapenem resistance proportions in K.pneumoniae are low (especially in northern Europe), Georgia, Italy, Turkey and Ukraine reported ratios between 25 % and 50 %, Greece and Belarus notified propotions exceeding 50 %. These ratios for the related report are shown on the geographical map (Figure 1) 13. Identifying reservoir regions for these resistance genes and sending data from these regions to regular surveillance may hamper the worldwide distribution of resistance 3, 12, 13, 17.
Figure 1.Carbapenem-resistant K.pneumoniae in the European Region (EARS-Net and CAESAR) , 2017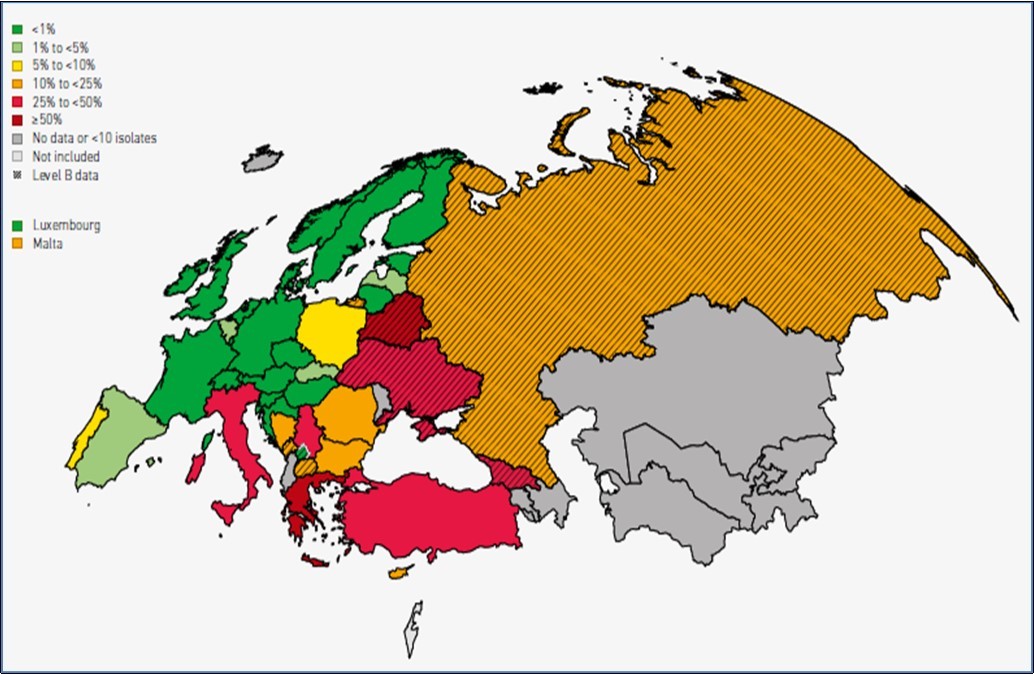
In our study, the increase in resistance to carbapenems from broad-spectrum antibiotics over the years and the high rates detected were considered as worrying. 99.1% of the strains in our study were the samples of hospitalized patients and the majority of them were in the intensive care units (65, 9 %). The high number of carbapenem-resistant K. pneumoniae and their high ratios of resistance are of concern and likely represent the dissemination of resistant clones in the health care setting. In our data, patients with nosocomial infections were overrepresented. This case mixture should be taken into account when interpreting the data.
Colistin resistance poses an important public health risk because it further limits treatment alternatives in patients with infections caused by health care associated, including carbapenem resistance K. pneumoniae. Colistin resistance may occur during treatment, depending on use, or may be through plasmid-mediated gene transfer. In our study, we found colistin resistance to 33.5 %. EUCAST recommends broth microdilution as the only valid method for colistin. However, there is no available data on colistin resistance rates due to the lack of necessary infrastructure of the participating laboratories for surveillance 3. Therefore, we could not compare the resistance rates.
Aminoglycosides were found to be more effective than most antibiotics in our study. It can be considered as an alternative agent in combination therapy. The resistance of tigecycline was very low (0, 4 %). The resistance rates of tigecycline are consistent with the literature data Although it is accepted as a good treatment option for multi-drug resistant K. pneumoniae, the attainable drug concentrations at several anatomic sites are suboptimal. Thus use of this agent is limited in severe infections 18. It is known that regularly setting antibiotic resistance profiles in hospitals and arranging treatment protocols are important especially in the planning of empirical treatment. Considering the increased resistance of meropenem in the regulation of empirical treatment protocols of our hospital, it is recommended to use colistin and tigecycline in limited cases.
Early detection of carbapenem-producing isolate is one of the most important measures to prevent hospital-acquired outbreaks. The establishment of institutional, regional, national and global surveillance programs should be aimed to prevent the spread of antibiotic resistance.
Conclusion
Antimicrobial resistance rates and distributions could vary between countries, regions and clinics. Wars, travels, health tourism, climate change, overuse of antibiotics in livestock facilitate the distribution of antibiotic resistance in the world and the emerge of new resistance phenotypes. Antibiotic resistance is a dynamic process that requires close monitoring and urgent measures should be taken. The one health approach should be kept in mind when countries take precautions.
Acknowledgements
The study was not supported by a financial institution.
References
- 1.Chiotos K, Hayes M, Gerber J, Tamma P D. (2016) Carbapenem-resistant Enterobacteriaceae infections in children. Current infectious disease reports. 18(1), 2.
- 2.Zając M, Sztromwasser P, Bortolaia V, Leekitcharoenphon P, Cavaco L M.(2019)."Occurrence and characterization of mcr-1-positive Escherichia coli isolated from food-producing animals in Poland, 2011-2016." Frontiers in microbiology 10:. 1753.
- 3.European Centre (2019) for Disease Prevention and Control. Surveillance of antimicrobial resistance. in Europe 2018 , Stockholm: ECDC; .
- 4.Bagley S T. (1985) Habitat association of Klebsiella species. , Infection Control & Hospital Epidemiology 6(2), 52-58.
- 5.Temiz H, Özbek E, VURAL D G, Özekinci T. (2015) Klebsiella İzolatlarının Antimikrobiyal Direnç Oranlarının Değerlendirilmesi. Türk Mikrobiyol Cem Derg. 45, 68-74.
- 6.Jarvis W R, Munn V P, Highsmith A K, Culver D H, HughesJM. (1985) The epidemiology of nosocomial infections caused byKlebsiella pneumoniae. , Infection Control & Hospital Epidemiology 6(2), 68-74.
- 7.Reyes J, Aguilar A C, Caicedo A. (2019) Carbapenem-ResistantKlebsiella pneumoniae: Microbiology Key Points for Clinical Practice. , International Journal of General Medicine 12, 437.
- 8. (2017) World Health Organization. Global priority list of antibiotic-resistant bacteria to guide research, discovery, and development of new antibiotics.
- 9.T R Walsh. (2018) A one-health approach to antimicrobial resistance. , Nature microbiology 3(8), 854.
- 11.Dursun A, Özsoylu S, Kılıç H, Kılıç A U, Akyıldız B N. (2018) Antibiotic Susceptibilities of Pseudomonas aeruginosa,Klebsiella pneumoniaeand Acinetobacter baumannii Strains Isolated from Patients in the Pediatric Intensive CareUnit Turkish. , Journal of Intensive Care 16(3), 109-114.
- 14.Nordmann P, Naas T, Poirel L. (2011) Global spread of carbapenemase-producing Enterobacteriaceae. Emerging infectious diseases. 17(10), 1791.
- 15.H Z Smith, Kendall B. (2019) Carbapenem Resistant Enterobacteriacea (CRE). In StatPearls[Internet].StatPearlsPublishing.
Cited by (2)
- 1.Toledano-Tableros José Eduardo, Gayosso-Vázquez Catalina, Jarillo-Quijada Ma Dolores, Fernández-Vázquez José Luis, Morfin-Otero Rayo, et al, 2021, Dissemination of blaNDM–1 Gene Among Several Klebsiella pneumoniae Sequence Types in Mexico Associated With Horizontal Transfer Mediated by IncF-Like Plasmids, Frontiers in Microbiology, 12(), 10.3389/fmicb.2021.611274
- 2.Huang Wen-Chun, Wong Min-Yi, Wang Ssu-Han, Hashimoto Masayuki, Lin Meng-He, et al, 2021, The Ferric Citrate Uptake System Encoded in a Novel blaCTX–M–3- and blaTEM–1-Harboring Conjugative Plasmid Contributes to the Virulence of Escherichia coli, Frontiers in Microbiology, 12(), 10.3389/fmicb.2021.667782