On the Role of Cholecystokinin (CCK) in Fear and Anxiety: A Review and Research Proposal
Abstract
Cholecystokinin (CCK) is found in high concentrations in cortical and limbic structures including the amygdala of rodents, and evidence has been gathered supporting a role for CCK in the neurobiology of anxiety. A variety of animal models have been used to study a central state of fear or anxiety, state that appears to produce a complex pattern of behaviors highly correlated with each other. It is now well established that the amygdala in particular is a critical link in the pathway through which sensory stimuli come to acquire fear evoking properties. The purpose of the proposed experiments is to study the role of the putative neurotransmitter CCK in fear and anxiety in vivo by means of a methodology coupling electrochemical and electrophysiological measurements in various brain areas. Indeed, the association of in vivo differential pulse voltammetry (DPV) with in vivo extracellular single unit recording could be able to provide concomitant physiological and neurochemical indications and to relate them to behavioral events. To further study and support the initial observations pharmacological experiments will also be performed by means of CCK receptor agonists and antagonists. This may eventually lead to development of more effective pharmacological strategies for treating clinical anxiety disorders.
Author Contributions
Academic Editor: Sadia Batool, Preston University Kohat-Islambad, Pakistan.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Francesco Crespi
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
A variety of animal models have been used to study a central state of fear or anxiety. In some models fear is inferred when an animal freezes, thus interrupting an ongoing behavior such as pressing a bar or interacting socially with other animals. In other models fear is measured by changes in autonomic activity, such as heart rate, blood pressure, or respiration. Fear can also be measured by a change in simple reflexes or a change in facial expressions and mouth movements. Thus fear or anxiety appears to produce a complex pattern of behaviors that are highly correlated with each other.
A Behavioral Model to Study Fear and Anxiety
Classical conditioning paradigms are widely used to study emotional mechanisms of the brain 1, 2, 3, 4, 5. Fear conditioning is a form of Pavlovian classical conditioning. It is produced experimentally by arranging the occurrence of an insignificant or emotionally neutral sensory stimulus, the conditioning stimulus (CS), with an aversive stimulus, the unconditioned stimulus (US). The CS, by virtue of its relationship with the US, acquires aversive properties and comes to obtainresponses characteristically elicited by threatening stimuli. Thus, a tone that has been previously been paired with foot-shock elicits fear or defense responses. Because these responses are not elicited by the CS before the temporal pairing of the CS with the US, they can be referred to as learned or conditioned emotional responses. Indices of fear conditioning include the measurement of the activity of motor responses (i.e. induction of freezing behavior) and/or a variety of autonomic responses (changes in arterial pressure, heart rate, skin conductance, etc.). Fear conditioning, also known as defense conditioning, is often used as an experimental model of anxiety 6, 7.
Neural Systems Involved in Fear and Anxiety
Emotional responses to aversive stimuli are usually associated with limbic system components such as the hypothalamus, amygdala, and the cingulate cortex. Lesions of either the hypothalamus or the amygdala result in attenuation of blood pressure, heart rate, or behavioral immobility ("freezing") during the conditioned emotional response 5, 8, 9, 10. Cingulate cortex lesions result in attenuation of cardiovascular conditioned responses 1, 11.
It is now well established that the amygdala in particular is a critical link in the pathway through which sensory stimuli come to acquire fear evoking properties 5, 8, 12, 13, 14. Direct projections from the central nucleus of the amygdala to the lateral hypothalamus mediate the activation of the sympathetic autonomic nervous system that is seen during fear and anxiety 10. Projections to the dorsal motor nucleus of the vagus or parabrachial nucleus 15 are involved in several autonomic measures of fear and anxiety. Projections to the ventral tegmental area 16 may mediate stress-induced increases in dopamine metabolites in the prefrontal cortex. Release of norepinephrine or 5-HT onto motor neurons, via amygdaloid activation of the locus coeruleus or raphe' neurons, could lead to enhanced motor performances. The hippocampus is reciprocally connected with the amygdala 17 and interactions between the amygdala and hippocampus appear to play an important role in some cognitive functions 18, 19. Thus, the highly correlated set of behaviors seen during fear may result from activation of the amygdala, which then projects to specific target areas that are critical for specific symptoms of fear.
The identity of the transmitters released into these target sites by amygdaloid neurons has been studied 20 and it is estimated that 25% of the neurons in central amygdala contains corticotropin-releasing factor, somatostatin and neurotensin. High density of benzodiazepine 21 and opiate receptors 22 are also found in the nuclei of the amygdala. Local infusion of the opiate agonist levorphanol into the central amygdala has anxiolytic effects in the social interaction test 23, 24. Local infusion of benzodiazepines into the amygdala has anxiolytic effects in the operant conflict test 25, 26, 27, 28.
Role of CCK in Fear and Anxiety
Cholecystokinin (CCK) is found in high concentrations in cortical and limbic structures of the temporal lobe, including the amygdala and the hippocampus of the rat 29, 30 and the monkey 31. Evidence has been gathered supporting a role for CCK in the neurobiology of anxiety 5, 32, 33, 34. CCK-induced excitation of rat hippocampus units is antagonized by benzodiazepine receptor agonists 35, suggesting that CCK might have anxiogenic properties 36, 37. Moreover, injections of CCK-8 into the central nucleus of the amygdala enhance arousal and fear in rats 38, 39. CCKB receptors antagonists show anxiolytic effects in several standard anxiety tests (the light/dark compartment test, the elevated plus-maze test, the social interaction test) 28, 32, 40, 41. Interestingly, delivery of electric shock to freely moving rats induces increase of CCK concentrations in limbic regions and in the prefrontal cortex 42. Administration of CCK-4 to healthy patients induces panic attacks 43 and produces respiratory changes in panic disorder and control patients 44.
Experimental Design and Methods
The purpose of the proposed experiments is to study the role of the putative neurotransmitter CCK in fear and anxiety. The development of very selective non-peptide antagonists for CCK subtypes (e.g. the CCKB antagonist G15013) will allow testing this hypothesis. The electrochemical methodology that joins in vivo electrophysiology with voltammetry using the same recording electrode will be applied 45, 46. This technique will be able to provide physiological and neurochemical information at the same time and to relate them to behavioral events. We will address the following questions:
Is the unit activity and the neurochemistry monitored in the amygdala modified by intravenous (iv) or intra-cerebroventricular (icv) injections of CCK and CCK antagonist G15013 in rats under urethane anaesthesia?
Is the unit activity and the neurochemistry modified in specific target areas of the amygdala (hippocampus, prefrontal cortex, or nucleus accumbens) by iv or icv injections of CCK and CCK antagonist G15013 in rats under urethane anaesthesia?
Does the selective CCK antagonist G15013 show anxiolytic effects during signaled (cued) fear conditioning tests? And if so are these effects correlated to changes in unit activity and neurochemistry in amygdala and/or in its projection areas?
Methods
Behavioral Test
A test well suited for the study of fear and anxiety will be applied 47. This test allows determining fear responses in a fast and reliable way.
Briefly, rats are placed individually in a rodent sound attenuating conditioning chamber (Coulbourn Instruments). The conditioned stimuli (CS) are an 800 Hz tone, amplified to 80 dB, and presented for 20 sec through a speaker located in the front panel of the chamber. The unconditioned stimulus (US) is a brief (0.5 mA, 500 msec) distributed delivery of direct current produced by a grid floor shocker.
On day 0, the animals are placed in the conditioning chamber for 20 min without stimulus presentation and are then returned to their home cages. On days 1 and 2 conditioning trials (consisting of 2 trials/day during which the US was presented during the last 500 msec of the 20 sec CS) are giving. Extinction trials (2 presentations of the CS alone) begin on day 3 and continue for 3 additional days. Freezing, used as the index of conditioned fear, is assessed by viewing the animals through a peephole in the sound-attenuating chamber and using stopwatches to measure freezing time. Freezing is defined as the absence of all movement except for respiratory-related movements 47. It is a very distinctive behavior that is easy to recognize and rarely occurs in non-fear situations 48, 49.
Micro-Electrodes for Concomitant Voltammetry and Electrophysiology
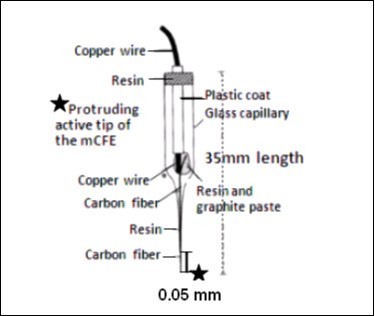
The micro-electrodes utilized here were prepared using a 12 µm-diameter carbon fiber (Carbone Lorraine, Lyon, France) and set as described earlier 45. They are cut under the microscope to leave 50-200 µm active area at the tip (Figure 1). This preparation gives the electrodes an impedance of 0.5-1.0 MOhms, allowing electrophysiological single cell activity discrimination 50.
In vivo Differential Pulse Voltammetry (DPV) Recordings
For in vitro and in vivo DPV measurements the carbon fiber electrode is set as described earlier 45, 51. Briefly, it is electrically treated firstly with a voltage from zero to 3 Volts, 70 Hz, 10 s, then with continuous potentials (+1.5 Volts, 5 s and −0.9 Volts, 5 s), so as to permit the measurement of three oxidation signals related to ascorbic acid, dopamine and serotonin metabolites, respectively, as well as that of a further oxidation peak when the differential pulse voltammetric (DPV) recordings were made in the same solution with the addition of the amino acids or neuropeptides such as CCK and when the DPV scan rate used was 10mV/s from -250 to +950mV at a step size of 50mV (see Figure 2). The DPV measurements of oxidation potentials and of oxidation currents are done automatically every 5min by the Autolab polarograph (potentiostat/galvanostat Ecochemie, The Netherlands) linked to an IBM pc computer equipped with a general purpose electrochemical system software (GPES) package.
Figure 2.Traces obtained in vivo in the amygdala of a single animal by means of the same micro-electrode: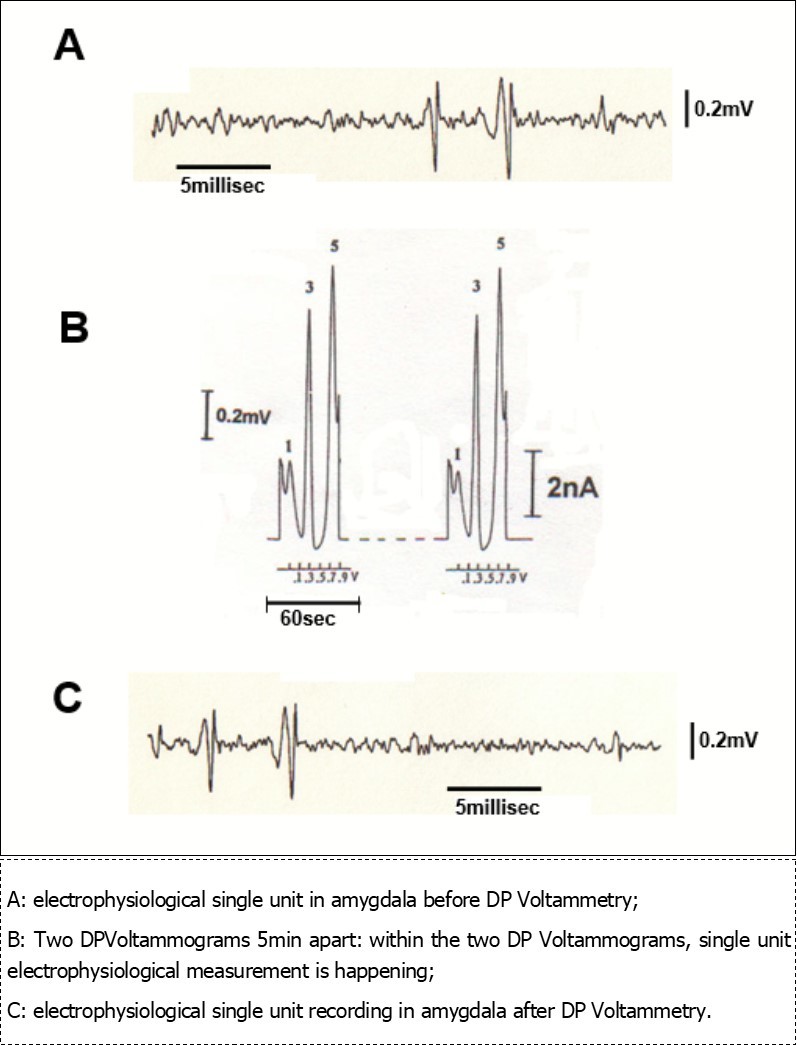
In Vivo Extracellular Single Unit Recording
The unit wave signals are fed into an amplifier (Fintronics). The signals are also led to an Axon Instruments system for acquisition and subsequently off-line analysis. Single unit discrimination is accomplished through a window discriminator (FHC Instruments) connected to an oscilloscope (Textronics)
For freely moving animal preparations a chronic stage will be used. The animals are placed in a stereotaxic frame under pentobarbital anaesthesia (40mg/kg). An opening is drilled through the skull above the CNS structure of interest (e.g. amygdala, hippocampus, etc.). The microsensor is then located into the area selected as described earlier 51 and fixed into a head stage mounted with dental acrylic on the skull of the animal. After a recovery of 5-7 days the animals will be ready for behavioral tests.
Previous experiments using such microsensors for concomitant in vivo electrophysiological and voltammetric measurements demonstrated that there is no reciprocal influence upon the respective signals recorded in vivo. And that that no damage is done to the single unit recordings by the current used for voltammetry 46. Here, Figure 2 shows single cell discrimination and voltammetry recording in a Central Amygdala preparation. DP Voltammograms lasting 60sec each are carried out every 5 min. In the interval time of 5 min electrophysiological measurements are performed. It appears evident that electrophysiological monitoring of unit activity is not altered after voltammetry recording and this during 1 hour of control analysis. This is in accord with the above mentioned studies that have been performed in other brain areas, therefore further supporting this methodological approach. DPVoltammograms show three signals at approximately +50mV; +300mV and +650mV, respectively. They correspond to the oxidation of endogenous cathechol (peak 1), indole (peak 3) and peptidergic (peak 5) compounds, respectively. In particular peak 5 recorded in amygdala may corresponds to the oxidation of endogenous CCK 51, 52.
Pharmacology
In the experiments performed under urethane anaesthesia, CCK and CCK antagonist will be administered iv (in the lateral tail vein) or icv (in a lateral ventricle) when the drugs do not readily pass the blood-brain barrier. In freely behaving animals experiments a 23 gauge guide canula will be implanted in a lateral ventricle and secured to skull screws with dental cement. Drugs will be then injected before the behavioral test through a 30 gauge canula extending out of the previously implanted guide canula by 1mm.
Specific Experiments
Effects of CCK and CCKs Antagonists on Amygdala in Rats Under Anaesthesia
As a first step towards the understanding of the effects of CCK and CCKs antagonist on the amygdala, the rates and changes in the electrophysiology and receptor release in animals restrained under urethane anaesthesia will be determined. Although high concentrations of CCK are found in amygdala (see INTRODUCTION), no information is known about the response of single cells to CCK or CCK antagonist injections in the experimental conditions used here i.e. monitoring neurotransmitter release through voltammetry and record at the same time unit activity with electrophysiology techniques. Changes in number of spikes of single cells will be correlated to changes in CCK release. Other neurotransmitter systems will be observed as well. Dopamine (DA) levels for example could be affected by CCK or CCK antagonist injections due to a possible coexistence of DA and CCK in amygdala. Indeed, the influence of CCK on this aminergic system has been observed 33, 54. In rodents, it has been shown that memory enhancement produced by subcutaneous (sc) administration of CCK-8 involves the amygdala 38, 55, 56. Fekete and Coll. Injected CCK-8 into the central amygdala and found enhanced fear in rats 38. Are these effects associated to an increase in single cell activity in amygdala or only a general increase of CCK concentrations will be detected? This preliminary phase will allow answering some of these questions.
Effects of CCK and CCKB Antagonists on Target Areas of the Amygdala in Rats Under Anaesthesia
A highly correlated set of behaviors seen during fear could be a result of the activation of the amygdala that project to target areas critical for various symptoms of fear and anxiety. During the years, number of investigations has focused on the modulation of DA release by CCK 57, 58, 59, 60. Using single cell 61 or electrochemical 62 techniques, some studies had demonstrated involvement of D-2 DA receptors in the interaction DA-CCK in the nucleus accumbens. No study, however, has ever analyzed electrophysiology activity and neurochemistry outcome together in the same experiment. The experiments proposed here will concentrate in areas of projections of the amygdala: nucleus accumbens, prefrontal cortex, and hippocampus; cell firing and receptor release in animals under urethane anaesthesia will be analyzed. Recordings will be made in these projection areas during CCK or CCK-B antagonist G15013 injections in the lateral ventricle. Some experiments could involve stimulation of the amygdala and recording in the target areas to study directly the effects of CCK drugs on these output pathways.
Anxiolytic Effects of CCK B Antagonists During Fear Conditioning Test
The aim of this experiment is to study the effect of CCK and CCK antagonists on a fear conditioning test. Animals will be implanted with a carbon fiber electrode as already described 45, 51. After a recovery of 5-7 days unit discharge rates and transmitter release will be measured before, during, and after the conditioning test. After few presentations (2 trials a day for only 2 days) of the tone (the CS) the animals will be conditioned. On the 3rd day of the test they exhibit freezing behavior at the sound of the tone (see METHODS). Thus, simultaneous electrochemical (voltammetric) and electrophysiological recordings will be performed in the amygdala, or in target areas like the nucleus accumbens or the prefrontal cortex. During the state of anxiety of the animal detection of changes in the unit activity rates and/or in the release of endogenous chemicals are expected. Are CCK levels increasing in the amygdala during the CS presentation and the freezing of the animal? Are DA levels also changed in prefrontal cortex or in the nucleus accumbens as a consequence of the state of fear/anxiety? An increase of CCK concentration in limbic regions after inescapable foot-shock has been reported 34, 42, 63. Therefore it was concluded that this increase was due to a state of stress in the animal. It is possible, however, that only pain mechanisms were involved in their experiment, given the role of CCK in analgesia 34. The present experimental behavioural approach should be able to dissociate pain mechanisms from the fear /anxiety state. By recording before conditioning during foot-shock presentation and after conditioning when the animal freezes to the CS (the tone) and no foot-shock is presented, it would be possible to determine what system is involved: if CCK levels increase during conditioning but not in the presence of the foot-shock, it can be proposed that CCK is selectively involved in fear and anxiety responses.
In a second phase of this study another group of animals will be implanted also with a canula aimed at the lateral ventricle to inject icv CCKB antagonist G15013. In such way it will be feasible to monitor putative changes in the behavior, single cell activity (electrophysiology) and chemical level (voltammetry). In particular, if the selective CCKB antagonist will implement anxiolytic effects, the animal behavior will show no or reduced freezing response to the CS, as well as no or not significant electrophysiological and chemical changes compared to control animals.
Conclusions
There is a strong evidence for a crucial role of the amygdala in fear and anxiety. An increasing number of studies are emerging to establish CCK as an important modulator system for anxiety 60, 64, 65.
A better understanding of brain systems that inhibit the amygdala, as well as the role of its high levels of peptides, could be attained with the proposed approach and may eventually lead to development of more effective pharmacological strategies for treating clinical anxiety disorders.
References
- 1.S L Buchanan, D A Powell. (1982) Cingulate cortex: its role in Pavlovian conditioning.Comp. , Physiol. Psychol 96, 755-774.
- 2.D H Cohen. (1980) Functional neuroanatomy of a conditioned response. In:Neural mechanisms of goal-directed behavior and learning,ed. by R.F. Thompson, L.H. Hicks, and V.B. Shvyrkov. New York:Academic 283-302.
- 3.Davis M. (1992) The role of the amygdala in fear-potentiated startle: implications for animal models of anxiety.Trend. in Pharmacological Sciences 13(1), 35-41.
- 4.Kapp B S, Pascoe J P, Bixle M A. (1984) The amygdala: a neuroanatomical systems approach to its contributions to aversive conditioning. In:The neuropsychology of memory,ed. by N. Butters and L. R. Squire , New York: Guilford 473-488.
- 5.J E LeDoux. (2011) . Emotion In Comprehensive Physiology,R.Terjung.(Ed.), doi-10.1002/cphy.cp010510 .
- 6.Marks I, Tobena A. (1990) Learning and unlearning fear: a clinical and evolutionary perspective.Neurosci. , andBiobehavRev 14, 365-384.
- 7.Lewis M D. (2005) Bridging emotion theory and neurobiology through dynamic systems modeling.Behavioral and. Brain Sciences,28 2, 169-194.
- 8.B S Kapp, R C Frysinger, Gallagher M, Haselton J. (1979) Amygdala central nucleus lesions: effects on heart rate conditioning in the rabbit.Physiol.Behav. 23, 1109-1117.
- 9.O A Smith, C A Astley, J L DeVito, J M Stein, R E Walsh. (1980) Functional analysis of hypothalamic control of the cardiovascular responses accompanying emotional behavior.Federation. Proc 29, 2487-2494.
- 10.J E LeDoux, Iwata J, Cicchetti P, D J Reis. (1988) Different projections of the central amygdaloid nucleus mediate autonomic and behavioral correlates of conditioned fear.J.Neurosci. 8, 2517-2529.
- 11.Sehlmeyer C, Schöning S, Zwitserlood P, Pfleiderer B. (2009) Human Fear Conditioning and Extinction.In Neuroimaging: A Systematic ReviewPloSonehttps://doi.org/10.1371/journal.pone.0005865.
- 12.D C Blanchard, R J Blanchard. (1972) Innate and conditioned responses to threat in rats with amygdaloid lesions.Comp. Physiol. Psychol.81,281-290
- 13.J E LeDoux, Sakaguchi J, Iwata J, D J Reis. (1986) Destruction of intrinsic neurons in the lateral hypothalamus disrupts the associative conditioning of autonomic but not behavioral emotional responses in the rat.Brain Res.38,161-166.
- 14.Davis M, J M Hitchock, J B Rosen. (1987) Anxiety and the amygdala: pharmacological and anatomical analysis of the fear-potentiated startle paradigm. In:The psychology of learning and motivation. ed. by G.H. Bower. San Diego: 263-305.
- 15.D A Hopkins, Holstege G. (1978) Amygdaloid projections to the mesencephalon, pons and medulla oblungata in the cat.Exper. Brain Res.32,529-547.
- 16.R M Beckstead, V B Domesick, Nauta W J H. (1979) Efferent connections of the substantia nigra and ventral tegmental area in the rat.Brain Res. 175, 191-217.
- 17.J L Price, D J Amaral. (1981) An autoradiographic study of the projections of the central nucleus of the monkey amygdala. , J.Neurosci 1, 1242-1259.
- 18.J P Aggleton. (1993) The contribution of the amygdala to normal and abnormal emotional states.Trend in. , Neurosciences 16, 328-333.
- 19.Tovote P, Fadok J, Lüthi A. (2015) Neuronal circuits for fear and anxiety.Nature Reviews. Neuroscience16 317-331.
- 20.Gray T S. (1989) In:Neuropeptides and stressed, by Y, Tache JE. , Morley MR,BrownSpringer-Verlag,1: 92-106.
- 21.D L Niehoff, M J Kuhar. (1983) Benzodiazepine receptors: localization in rat amygdala.J.Neurosci. 3, 2021-2097.
- 22.Goodman R R, Snyder S H, Kuhar M J, Young W S. (1980) Differentiation of delta and mu opiate receptor localization by light microscopic autoradiography.Proc. , Nat. Acad. Sci. U. S.A 77, 2167-2174.
- 23.S E File, Rodgers R J. (1979) Partial anxiolytic actions of morphine sulphate following microinjection into the central nucleus of the amygdala in rats.Pharmacol.Biochem.Behav. 11, 313-318.
- 24.G W Pasternak, Y X Pan. (2013) Mu Opioids and Their Receptors:. , Evolution of a Concept.Pharmacological Reviews 65(4), 1257-1317.
- 25.Nagy J, Zambo K, Decsi L. (1979) Anti-anxiety action of diazepam after intra-amygdaloid application in the rat.Neuropharmacol. 18, 573-576.
- 26.E N Petersen, Scheel-Kruger J. (1982) The GABAergic anticonflict effect of intra-amygdaloid benzodiazepines demonstrated by a new water lick conflict paradigm. In:Behavioral models and analysis of drug action. ed. by M.Y. Spiegelstein and A. Levy.Amsterdam:Elsevier
- 27.Shibata K, Y Gomita Kataoka, Y, Ueki S. (1982) Localization of the site of the anticonflict action of the benzodiazepines in the amygdaloid nucleus of the rat.Brain Res. 234, 442-446.
- 28.Gross J J. (2013) Handbook of Emotion Regulation. , Second Edition, J. Gross Ed,TheGuilfordPress,NewYork,N.Y
- 29.Vanderhaeghen J, J C Signeau, Gept W. (1975) New peptide in vertebrate CNS reacting with antigastrin antibodies.Nature 257. 604-605.
- 30.M C Beinfeld, D K Meyer, R L Eskay, R T Jensen, M J Brownstein. (1981) The distribution of cholecystokinin immunoreactivity in the central nervous system of the rat as determined by radioimmunoassay.Brain Res. 212, 51-57.
- 31.M F Kritzer, R B Innis, P S Goldman-Rakic. (1988) Regional distribution of CCK receptors in the macaque medial temporal lobe determined by in vitro receptor autoradiography.Comp. , Neural 276, 219-230.
- 32.Harro J, Vasar E, Bradwejn J. (1993) CCK in animal and human research on anxiety.Trends inPharmacol. , Sciences 14, 244-249.
- 33.J N Crawley. (1991) . , Cholecystokinin-dopamine interactions.Trend in Pharmacological Science 12, 232-236.
- 34.P V Jetty, D S Charney, A W Goddard. (2001) . , Neurobiology of Generalized Anxiety Disorder.Psychiatric Clinics of North America 24(1), 75-97.
- 35.Bradwejn J, deMontigny C. (1984) Benzodiazepines antagonize cholecystokinin-induced activation of rat hippocampal neurones.Nature 312. 363-364.
- 36.C F Netto, F S Guimaraes. (2004) . , Anxiogenic Effect of Cholecystokinin in the Dorsal Periaqueductal Gray.Neuropsychopharmacology 29, 101-107.
- 37.T A Lovick. (2008) Pro-nociceptive action of cholecystokinin in the periaqueductal grey: A role in neuropathic and anxiety-induced hyperalgesic states,Neuroscience&BiobehavioralReviews,32(4):. 852-862.
- 38.Fekete M, Lengyel A, Hegedus B, Perike B, Zarandy M et al. (1984) Further analysis of the effects of cholecystokinin octapeptides on avoidance behaviour in rats. , Eur. J. Pharmacol 98, 79-91.
- 39.M E Bowers, D C Choi, K J Ressler. (2012) Neuropeptide regulation of fear and anxiety: Implications of cholecystokinin, endogenous opioids, and neuropeptide Y.Physiology. , Behavior 107(5), 699-710.
- 40.C A Hendrie, J C Neill. (1992) Ethological analysis of the role of CCK. in anxiety. In:Multiple cholecystokinin receptors in the CNS ed. by C.T. Dourish, S.J. Cooper, S.D. Iversen and L.L. Iversen. Oxford:OxfordUniversityPress 132-142.
- 41.Griebel G, Perrault G, D J Sanger. (1997) CCK receptor antagonists in animal models of anxiety: Comparison between exploration tests, conflict procedures and a model based on defensive behaviours.Behavioural Pharmacology,8(67):. 549-560.
- 42.R A Siegel, E M Duker, Fuchs E, Pahnke U, Wuttke W. (1984) Responsiveness of mesolimbic, mesocortical and hippocampal cholecystokinin and substance P neuronal systems to stress, in the male rat.Neurochem. , Int 6, 783-789.
- 43.Bradwejn J, Koszycki D, Meterissian G. (1990) Cholecystokinin-Tetrapeptide Induces Panic Attacks in Patients with Panic Disorder.Can. , J. Psych 35, 83-85.
- 44.Bradwejn J, Koszycki D, Bourin M. (1991) Enhanced sensitivity to cholecystokinin tetrapeptide in panic disorder: clinical and behavioral findings.Arch. , Gen. Psych 48, 603-607.
- 45.Crespi F. (1990) In vivo voltammetry with micro-biosensors for analysis of neurotransmitter release and metabolism.J.Neurosci. , Methods 34, 53-65.
- 46.Crespi F. (2002) In vivo voltammetry and concomitant electrophysiology at a single biosensor to analyse ischaemia, depression and drug dependence.Journal of Neuroscience Methods. 119, 173-184.
- 47.R G Phillips, J E LeDoux. (1992) Differential contribution of amygdala and hippocampus to cued and contextual fear conditioning.Behav.Neurosci. 106, 274-285.
- 48.M S Fanselow, F J Helmstetter. (1988) Conditional analgesia, defensive freezing, and benzodiazepines.Behav.Neurosci. 2, 233-243.
- 49.S F Maier. (1992) Role of fear in mediating shuttle escape learning deficit produced by inescapable shock.J. Exp. Psychol. Anim.Behav. Proc.16,137-149
- 50.Bordi F, J E LeDoux. (1992) Sensory tuning beyond the sensory system: an initial analysis of auditory response properties of neurons in the lateral amygdaloid nucleus and overlying areas of the striatum.J.Neurosci. 12, 2493-2503.
- 51.Crespi F. (2013) Invasive or Non-Invasive Techniques and Sensors for Real Time In Vivo Sensing in the Brain" chapter in the book,Laboratory and Clinical Research: "Microelectrodes: Techniques, Structures for Biosensing and Potential Applications",NovaSciencePublishers,Inc.
- 52.Crespi F. (1991) In vivo voltammetry detection of neuropeptides with micro carbon fibre biosensors: possibile selective detection of somatostatin.Analytical. , Biochemistry 194, 1-8.
- 53.Paxinos G, Watson C. (2013) . The Rat Brain in Stereotaxic Coordinates 7th Edition, Academic Press 472pp .
- 54.Crespi F, Romanelli L, Tacconi S, Marien M. (1994) CCK agonist and antagonist effects on dopamine and serotonin metabolism in the rat nucleus accumbens: in vivo studies using voltammetry and microdialysis.Pharmacology. , Communications 4, 153-162.
- 55.J F Flood, J S Garland, J E Morley. (1992) Evidence that CCK-enhanced retention is mediated by changes in opioid activity in the amygdala.Brain Res. 585, 94-104.
- 56.De Wied, D. (1997) Neuropeptides in learning and memory processes. , Behavioural Brain Research 83(12), 83-90.
- 57.R F Lane, C D Blaha, A G Phillips. (1986) In vivo electrochemical analysis of CCK-induced inhibition of dopamine release in the nucleus accumbens.Brain. Res.397 200-204.
- 58.Voigt M, R Y Wang, T C Westfall. (1985) The effects of cholecystokinin on the in vivo release of newly synthesized dopamine from the nucleus accumbens of the rat.J.Neurosci. 5, 2744-2749.
- 59.F H Marshall, Barnes S, Hughes J, G N Woodruff, J C Hunter. (1991) . Cholecystokinin Modulates the Release of Dopamine from the Anterior and Posterior Nucleus Accumbens by Two Different Mechanisms.JNC 56(3), 917-922.
- 60.Rotzinger S, D A Lovejoy, L A Tan. (2010) Behavioral effects of neuropeptides in rodent models of depression and anxiety.Peptides. 31(4), 736-756.
- 61.R Z Liang, Wu M, Yimand C C Y, G J Mogenson. (1991) Effects of dopamine agonists on excitatory inputs to nucleus accumbens neurons from the amygdala: modulatory action of cholecystokinin.Brain Res. 554, 85-94.
- 62.Derrien M, Durieux C, Dauge V, B P Roques. (1993) Involvement of D-2 dopaminergic receptors in the emotional and motivational responses induced by injection of CCK-8 in the posterior partof the rat nucleus accumbens.Brain Res. 617, 181-188.
- 63.Hebb A, J F Poulin, S P Roach, R M Zacharko, Drolet G. (2005) Cholecystokinin and endogenous opioid peptides: Interactive influence on pain, cognition, and emotion.Progress in. , Neuro-Psychopharmacology and Biological Psychiatry 29(8), 1225-1238.
Cited by (29)
This article has been cited by 29 scholarly works according to:
Citing Articles:
Translational Psychiatry (2024) Crossref
Translational Psychiatry (2024) OpenAlex
Muhammad Asim, Huajie Wang, Abdul Waris, Jufang He - Translational Psychiatry (2024) Semantic Scholar
The Medical and Ecological Problems (2023) Crossref
The Medical and Ecological Problems (2023) OpenAlex
Nature Reviews Gastroenterology & Hepatology (2023) Crossref
Nature Reviews Gastroenterology & Hepatology (2022) OpenAlex
A. Gautier-Stein, G. Mithieux - Nature reviews: Gastroenterology & hepatology (2022) Semantic Scholar
Medical Research Archives (2022) OpenAlex