Histological Studies of Effect of Dichlorvos on Hypothalamic-Pituitary-Testicular Axis Following the Administration of Mimosa Pudica Extract
Abstract
Plants have played a significant role in maintaining human health and improving the quality of human life. Therefore, this present study seeks to evaluate the role of its alkaloid fraction in the hypothalamic-pituitary-testicular axis after dichlorvos poisoning in mice.
A total of 25 male mice, weighing approximately 20-30 g, were randomly assigned into 5 groups of 5 mice each (Groups A, B, C, D and E). Mice in groups A served as control and received normal saline while groups B, C and E. received 5 mg/kg of dichlorvos daily for 14 days. Mice in group D received 200mg/ kg of Alkaloid extract of Mimosa pudica (AEMM) for 14 days. At the end of the experiment, all animals were sacrificed and Hypothalamus, Pituittary and Testes were excised for histological process. Sperm from cauda epididymidal duct on one side was collected and used for spermatological studies and data were expressed as mean± SEM. Significant difference was set at p<0.05.
Results showed significance difference (p<0.05) in sperm motility, sperm life death ratio groups when compared with their controls and no significance difference in sperm volume when compared with control. Histological studies reveal distortion in hypothalamuss, pituitary and testes in dichlorvos administration group compared with control rats while mimosa showed a ameliorative effect when co-administered with dichlorvos. In conclusion, the alkaloids from Mimosa (AEM) leaves enhanced the released of gonadotropins by the cordial action of the hypothalamus-pituitary axis for the reversed of Dichlorvos effect.
Author Contributions
Academic Editor: Xiangwei Xiao, Children's Hospital of Pittsburgh, University of Pittsburgh, School of Medicine, United States.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Enye Linus Anderson, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Reproduction is the crux for the existence of life. It is a critical biological process and forms a complex and finely tuned system that affects every cell system in the body. It encompasses a wide range of physiological processes, associated behaviours and anatomical structures 1, 2. The reproductive system of the male mammal is composed of the hypothalamic-pituitary-gonadal axis (HPG); the external organs, which include the penis, scrotum, and urethra; and the internal organs, which include the testis, vas deferens, epididymis (caput, corpus, and cauda), seminal vesicles, coagulating glands, prostate, bulbourethral glands, and the ejaculatory ducts 3. The testis contains the seminiferous tubules and interstitial tissue covered by a capsule called tunica. The interstitium contain the Leydig cells, which secrete androgens as well as other steroids including estrogen 4.
The harmonization of male reproduction is controlled by the Hypothalamo-pituitary gonadal axis. The medial basal hypothalamus is responsible for the release of the decapeptide gonadotropin- releasing hormone (GnRH) into the pituitary portal circulation. GnRH stimulates the pituitary synthesis and release of luteinizing hormone (LH) and follicle stimulating hormone (FSH). These pituitary glycoproteins in turn regulate testicular function including spermatogenesis and steroidogenesis 5.
Male reproductive health has been at the center of attention after the research of Carlsen et al., which reported a significant decline in the sperm count from 113×106/mL in 1940 to 66 ×106/mL in 1990 among men without a history of infertility. Several other reports also suggest declining sperm counts, increase of sexual dysfunction, infertility, cryptorchidism, hypospadias and testicular cancer in some areas and consequently an increase in male infertility due to exposure of toxicants 6.
Various environmental contaminants affect human fertility, these include lifestyle, dietary choices, pesticides, substances from various industrial uses and residues such as dioxins, polychlorinated biphenyls (PCBs), ethylenedibromide (EDB), phthalates, polyvinyl chloride (PVC), dioxins (by products of manufacture), organic solvents and heavy metals (lead, mercury, arsenic) 7. Mehta et al., even postulated that the use of pesticides and fluoride in drinking water could be responsible for a high incidence of azoospermia insome Indian towns 8. Dichlorvos, also known as DDVP (2,2-dichlorovinyl dimethyl phosphate) is an organophosphate insecticide and have been applied in northern Nigeria as mosquitoes insecticides over the decades since its commercial manufacture started in 1961 9, 10.
Plants have played a significant role in maintaining human health and improving the quality of human life for thousands of years and have served humans well as valuable components of medicines, seasonings, beverages, cosmetics and dye. One of such plants is Mimosa pudica, which has invited attention of the researchers worldwide for its pharmacological activities such as anti diabetic, antitoxin, antihepatotoxin, antinociceptive, hyperglycemic, antivenom, immunomodulatory, anticonvulsant, antihepatotoxic, antifertility, and diuretic which posses wound healing activity 11. The phytochemical studies of plant have revealed presence of alkaloid, glycoside, flavonoid and tannis and fixed oil 12, 13. Therefore, the present study seeks to evaluate the role of its alkaloid fraction in the hypothalamic-pituitary-testicular axis after the administration of dichlorvos in mice.
Materials and Methods
Animals
A total of 25 male mice, weighing approximately 20-30 g, were used in the study. Mice were procured from ABUAD animal house. The animals were fed with standard rat pellet diet and water ad libitum. They were housed in polypropylene cages (8" x 12" x 8") with metal grill tops, under conventional, controlled standard conditions. All mice were acclimated to laboratory conditions of temperature and photoperiod of 14h light: 10h dark, one week prior to treatment.
Collection of Mimosa Pudica
Leaves of Mimosa pudica were collected from ABUAD farms. They were air-dried, then powdered with a mechanical grinder and kept in a dry, clean container ready for the investigation and extraction of the active constituents.
Extraction of Alkaloid Fraction
The extraction of the alkaloid was done using the continuous extraction method using the Soxhlet apparatus 14. Four hundred grams (400 g) of ground leaves of Mimosa pudica was weighed and packed in a cheesecloth bag which served as an extraction thimble. The thimble was then placed into a suitable jar with cover. The sample was moistened with sufficient amount of 95% ethanol. This was made alkaline with sufficient quantity of ammonia T.S. and mixed thoroughly. The sample in the thimble was macerated overnight, and then placed in the Soxhlet extractor on the next day. Sufficient amount of 95% ethanol was placed in the solvent flask (4.8 liters). The sample was extracted for about 3 – 4 hours. The ethanol extract was filtered and was concentrated in a Soxhlet distilling apparatus at 60oC. The crude alkaloid extract was further treated with 1.0 N hydrochloric acid. This was filtered and the filtrate was collected. The filtrate was alkalinified with ammonia T.S. and placed in a separatory funnel. Measured quantities of chloroform was added into the separatory funnel, mixed and shaken for about five times and allowed to separate into two layers. The lower layer of chloroform contained the alkaloids and the upper layer the aqueous portion. The upper layer was extracted until the last chloroform extract was found negative to Dragendorff’s reagent. The combined chloroform extract was concentrated in Soxhlet distilling apparatus at 60oC and evaporated in water bath maintained at that temperature until semi-dry. The residue was weighed and percentage yield was calculated using the formula:
Alkaloid extract of Mimosa pudica (AEMM) obtained was administered to the animals at a dose of 200mg/kg and concentration of 50mg/ml for 14days through oral gavage (Czapski et al., 2014).
Source of Pesticide
The pesticide, dichlorvos (90% w/w formulation) used in this study were obtained from agrochemical store in Ado-Ekiti
Experimental Design
Pesticide was diluted at 0.05% and then administered at a dose of 5mg/kg to the animals for 14 days orally.
Twenty-five male mice obtained were grouped into five groups of five mice each as expressed below:
Group A- served as normal control group
Group B – received 1ml of dichlorvos daily for 14 days
Group C – received dichlorvos and AEMM daily for 14 days
Group D – received only AEMM daily for 14 days
Group E – received only dichlorvos for 7 days prior to 7days’ administration of only AEMM (Table 1).
Table 1. shows the experimental Design and animal treatment of the effect of mimosa pudica on dichlorvos administration in mice.| S/N | GROUPS | AGENTS | DOSES | ROUTE | DURATION |
| 1 | A | Distilled water | 0.25 ml | Oral | 14 days |
| 2 | B | dichlorvos | 5 mg/kg | Oral | 14 days |
| 3 | C | Dichlorvos + AEMM | 5mg/kg + 200 mg/kg | Oral | 14 days |
| 4 | D | AEMM | 200 mg/kg | Oral | 14 days |
| 5 | E | Dichlorvos + AEMM | 5mg/kg + 200 mg/kg | Oral | 7 days of induction + 7 days of extract administration |
At the end of the experiment, all animals were sacrificed and Hypothalamus, Pituittary and Testes were excised for histological process. Sperm from cauda epididymidal duct on one side was collected and used for spermatological studies.
Spermatological Studies
The Cauda epididymal duct on one side was exposed and incised. The sperm oozing out from the incision was quickly sucked into a capillary tube up to 0.05 µml mark. It was diluted 100 times using physiological saline. After thorough mixing, the dilute semen was used for the spermatological analyses 15.
Assessment of Sperm Motility
A drop of dilute semen was placed on a cover glass. The cover glass was inverted over a cavity slide so that a hanging drop was obtained. The edges of the coverslip were sealed with Vaseline and the hanging drop was observed under a compound microscope at 450 x magnification. The preparation was observed at regular intervals in such a way as to find the duration, in minutes, of the progressive motility of the last motile sperm. For each animal two separate hanging drop preparations were made, and motility was assessed by two independent observers. The data for each animal were used to obtain the average 15.
Sperm Viability (Life/Dead Ratio)
This was carried out by adding two drops of warm Eosin/Nigrosin stain to the semen on a pre-warmed slide, a uniform smear was then made and dried with air, the stained slide was immdediately examined under the microscope using x 400 magnification. The live sperm cells were unstained while the dead sperm cells absorbed the stain. The stained and unstained sperm were counted and the percentage was calculated 16.
Sperm Cell Volume
Sperm cell volume using testis volume. Testis volume was determined volumetrically using the Archimedes principle of water displacement in a measuring cylinder. The testes density was derived thus:
Testis density = Testis Weight (g)/ Testis Volume (cm3) 17.
Photomicrography
Olympus binocular microscope was used. A 5.1 megapixel MV550 research camera for microscopes was mounted in one of the oculars. This was connected to a computer running on image capture and analysis software. The immersion oil was dropped on the slide and gradually adjusted to adapt X100 oil immersion objective. The system was adjusted to obtain clarity and resolution. The image was captured and saved on the computer.
Statistical Analysis
One-way ANOVA was used to analyze data, followed by Student Newman-Keuls test for multiple comparisons. GraphPad Prism 5 (Version 5.03; Graphpad Software Inc., San Diego, CA) was the statistical package used for data analysis. Statistically significant difference was set at p<0.05.
Result
The effect of alkaloid-rich extract of Mimosa enhanced sperm motility in Dichlrovos-treated animals showed significant difference when group B is compared with control group (p < 0.005)(Figure 1). The effect of alkaloid-rich extract of Mimosa enhanced sperm life death ratio in Dichlrovos-treated animals, with significant difference when compared with control group (p < 0.005) (figure 2). Dichlorvos only treated animals showed no effect on the sperm volume in the testes, and also, the treatment with alkaloid-rich extract of Mimosa showed no significant different when compared with the control group(P <0.05)(figure 3).
Figure 1.Effect of Dichlorvos and Mimosa treatments on sperm motility in mice Values are expressed in Standard Error of Mean (SEM). Different alphabets indicate significant difference in each. KEY: DP = Dichlorvos, MP = Mimosa Pudica, DP/MP = Co Administration of DP and MP, DP then MP = Post Treatment of DP Toxicity with MP. # Statistical difference relative to control at p = 0.0036, F = 8.48, n = 5. 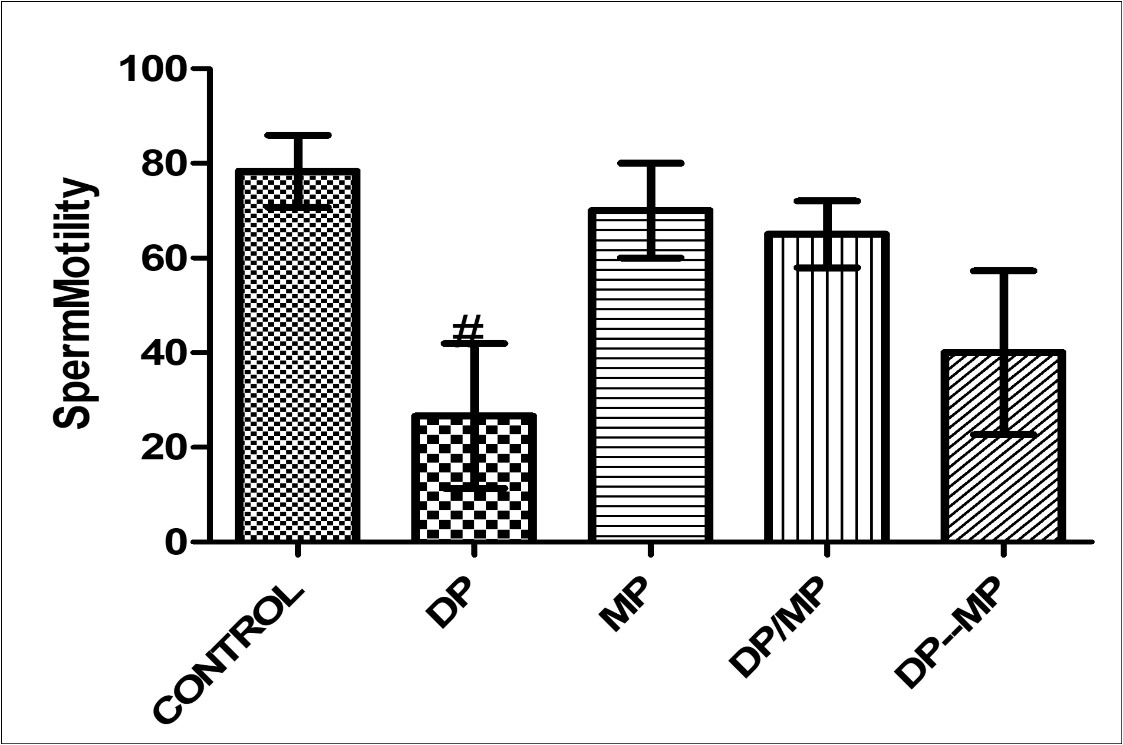
Histological Findings
At the end of the histological study and analysis, toxicity of dichlorvos was established on the Hypothalamic-Pituitary-Testicular axis of mice as remarkable histomorphological alterations were observed in the dichlorvos administration in mice. There was no marked histomorphological alteration in the mimosa treated without dichlorvos whereas, mimosa showed a modulating or ameliorative effect when co-administered with dichlorvos. The post treatment of mimosa after dichlorvos toxicity was not effectively marked, probably due to the dosage and the duration of the administration.
Discussion
The present research and study showed the ameliorative effect of Mimosa pudica on the toxic effect of Dichlorvos on the histoarchitecture of the hypothalamic-pituitary-testicular axis in mice. Histopathology is considerably the best parameter to determine toxic effect on the male reproductive system 18 and use to determine the level of damage, degeneration or disruption of the cellular and vascular response caused by dichlorvos poisoning on the hypothalamic-pituitary-testicular axis of the mice model. Prophylactic and therapeutic treatment of mice with alkaloids-rich extract of mimosa (AEM) in animals in group D and E respectively enhanced the motility of these sperm cells, with significant increase (P <0.05) in sperm motility from animals treated with only Dichlorvos. Administration of animals with only AEM showed no significant difference (P <0.05) from the normal control group. Also, as seen in figure 2, AEM was able to enhance sperm life death ratio on Dichlorvos treated mice, by showing significant difference (P < 0.05) in sperm life death from animals in group B (Dichlorvos treated only). Animals treated with only AEM showed no significant difference (P <0.05) from normal control group, thereby sustaining and maintaining the sperm life death ratio in the testes, hence, enhancing the sperm quality in the male mice. This efficacious effect exerted by AEM could be attributed to its phytochemical constituent, which is basically the alkaloid extracted. Previous study as shown that Aloe vera enhanced sperm quality in mice, and this efficacy was attributed to its alkaloid-rich property, which was suggested that it increased LH and FSH hormones in the HPT, consequentially enhancing testosterone and the sperm quality in male mice 19.
Figure 2.Effect of Dichlorvos and Mimosa treatments on life death ratio in mice Values are expressed in Standard Error of Mean (SEM). Different alphabets indicate significant difference in each. KEY: DP = Dichlorvos, MP = Mimosa Pudica, DP/MP = Co Administration of DP and MP, DP then MP = Post Treatment of DP Toxicity with MP. # Statistical difference relative to control at p = 0.0010, F = 11.34, n = 5.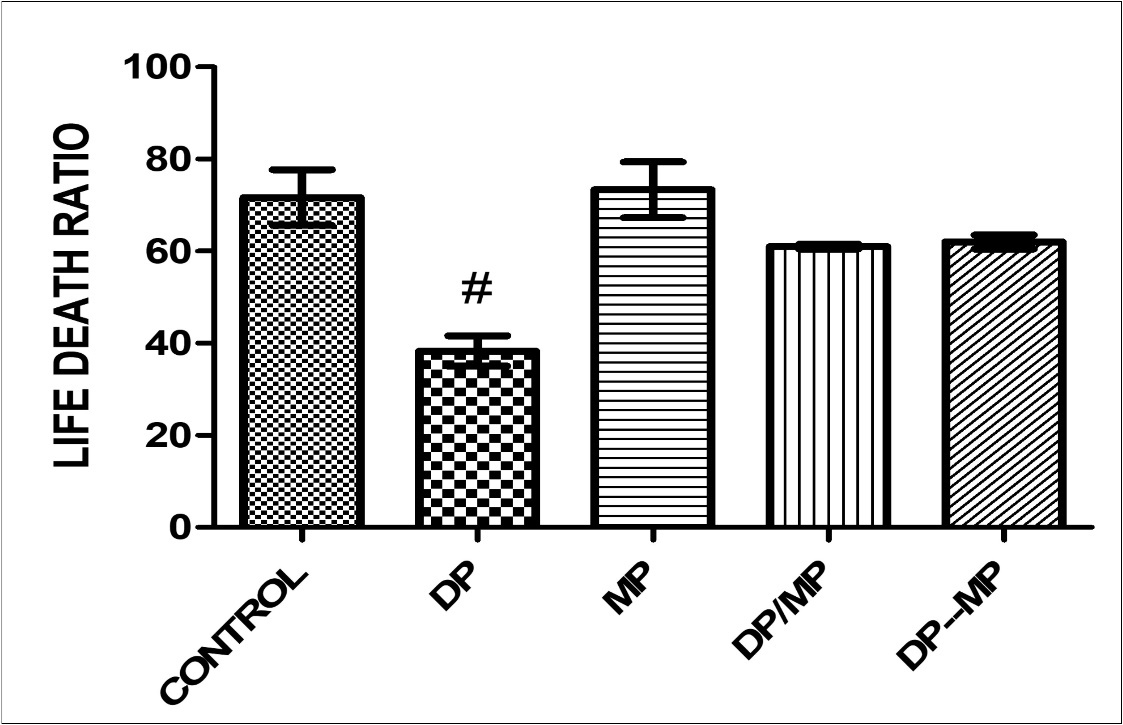
In addition, figure 3, showed the effect of Dichlorvos on the sperm volume in testes. It was observed that there was no significant difference in the volume of sperm present in the testes of Dichlovros only treated groups when compared to normal control group. Also, there was no effect showed by AEM only extracts and Dichlorvos treated with AEM. Although, the sperm volume might not be affected by Divhlorvos, the viability and the functionality of the sperm are weakened, due to the effect on the HPT and hormones levels in group B mice.
Figure 3.Effect of Dichlorvos and Mimosa treatments on sperm volume in mice Values are expressed in Standard Error of Mean (SEM). Different alphabets indicate significant difference in each. KEY: DP = Dichlorvos, MP = Mimosa Pudica, DP/MP = Co Administration of DP and MP, DP then MP = Post Treatment of DP Toxicity with MP. # Statistical difference relative to control at p = 0.0013, F = 10.50, n = 5.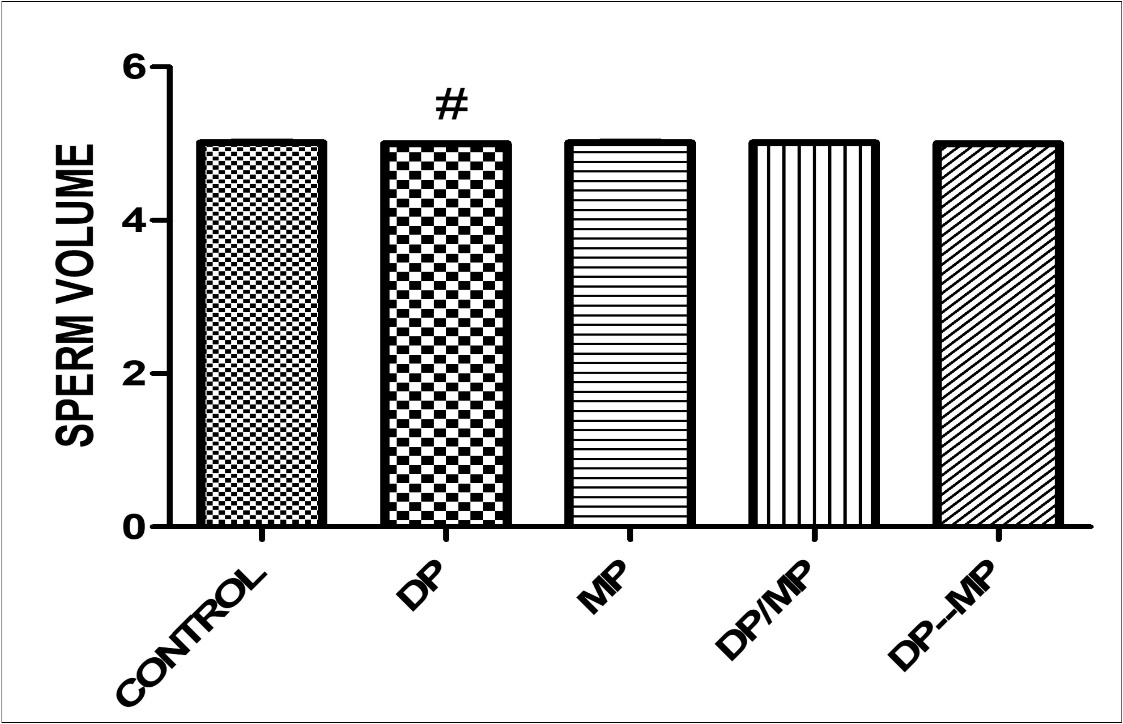
In figure 4, effects of Dichlorvos were studied on the hypothalamus of the experimental mice and the prophylactic and therapeutic effects of AEM when compared to normal control group. Administration of Dichlorvos induced vasculations within the neuroparanchyma and this should be as a result of neurodegenerative property of Dichlorvos on the hypothalamus. The observed reproductive endocrine toxicity on dichlorvos exposure might be due to pesticide interference of the hypothalamus further affecting the pituitary-testicular axis. Weak hypothalamus and reduction of numbers of hypothalamus cells along with decreased sperm qualities indicated interference of hypothalamus gonadotropes. This consequentially would affect the LH and FSH by decreasing their levels, hence affects testicular synthesis and secretion of testosterone whose levels were lowered in the exposure groups 5. Organophosphorus pesticides have been shown to reduce hypothalamus cells in the brain and consequentially testosterone levels 20, 21. Adminstration of animals treated with Dichlorvos with AEM ameliorated the effect of Dichlorvos and prevented the loss of hypothalamus cells. Prophylactic treatment of Dichorvos treated animals with AEM showed capillaries free from cellular and vascular response, the cells are spindle-like showing that the neurodegenerative features still exist but reduced. Therapeutic treated groups (Group E), showed a congested capillary with reduced marked degenerative feature when compared to group B animals (Dichlorvos treated only). Administration of only AEM showed cluster of cells which could be a vascular response to the hypothalamic pituitary portal. Hence, this effect shown by AEM could be attributed to its alkaloid-rich contents 19.
Figure 4.Showing the Hypothalamus of mice. Group A shows clusters of hypothalamic nucleus around the paraventricular region (down arrow); magnocellular nuclei appear normal and unremarkable (4-point star). Group B shows a number of glial cells (up arrow). Group C shows cluster of cells and hypothalamic nuclei (circle). Group D shows the capillary is free from cellular and vascular response (Left Arrow). Group E shows a congested capillary (down arrow) with marked degenerative feature. (x400) H&E
Furthermore, effects of Dichlorvos were also studied on the pituitary cells. As observed in figure 5, group B animals (Dichlorvos treated only-toxicity group) showed capillary is not free collection and congestion which indicates the possibility of cellular response. Organophosphate insecticides have been showed to induce toxicity in the pituitary cells in the brain, consequentially affecting the LH and FSH hormones and the sperm qualities 5. Administration of Dichlorvos treated animals with AEM in group D and Group E (prophylactic and therapeutic respectively) showed enhancement tin the pituitary cells, by suppressing the mechanism of action of Dichlorvos induce toxicity in the HPT axis. Prophylactic section showed the capillary is free from cellular and vascular response and the pituitary cells appear normal and unessentially remarkable. Also, the therapeutic group showed congestion in the capillary showing cellular response, but the pituitary cells are more functional than group B (toxicity) animals.
Figure 5.Showing the pituitary of mice. Group A shows the normal pituitary gland (Pars nervosa) consisting of essentially appearing pituitcytes and the capillaries (up arrow). Group B shows the capillary (up arrow). Group C shows a number of pituitcytes and a remarkably appearing micromophorlogy. Group D shows the capillary (up arrow) free from cellular and vascular response. The pituitcytes appear normal. Group E shows congestion in the capillary (up arrow) showing cellular response. (x400) H&E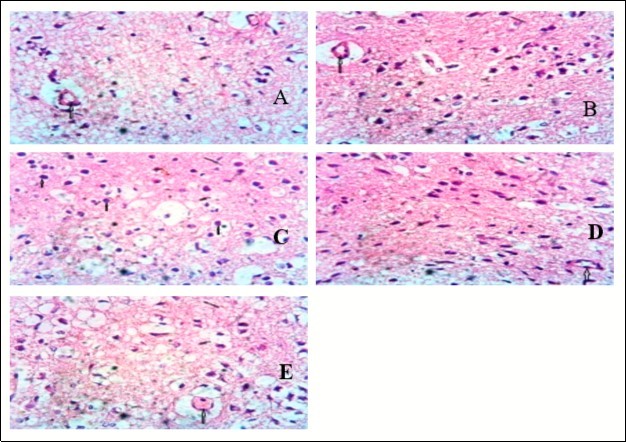
Study on the testes at all treatment levels showed variant influence on the testes by analyzing the testicular tissue morphology as seen in figure 6. In the Dichlorvos treated mice, the cells were irregularly shaped and there was marked intercellular only, space between the spermatogenic cells. Spermatogenesis was still present, but cell disorganization was found. The section showed scanty interstitial cells which could be as a result of testicular atrophy. Seminiferous tubules are separated by wide intersititium showing a possibly degeneration of the spermatids. Dichlorvos might have caused testicular toxicity by interactions between complex networks, involving the inhibition of oxidative stress, which lead to increase in the germ cell apoptosis. Administration of AEM on animals treated with Dichlorvos showed blood vessel is free from congestion and no remarkable inflammatory cell within the interstitial cell as observed in Group D animals (prophylactic group). Also group E animals (therapeutic) shows lesser congestion of interstitial cell (ICG) and proliferation of interstitial cell when compared to the toxicity group. In group C animals (extract only), there was no marked pathology of the spermatids and the seminiferous spermatids are of different maturation.
Figure 6.Showing the testes of mice. Group A shows the normal testes consisting of numerous seminiferous tubules (ST) which compose of spermatogenic cells (germ cells) at varying degree of maturation. Group B shows scanty interstitial cells. Seminiferous tubules are separated by wide interstitium showing a possibility of degeneration of the spermatids. Group C shows a mild proliferation of interstitial cells (up arrow). Also the seminiferous tubules (ST) show spermatids at varying level of maturation Group D shows the blood vessel (up arrow). There is no remarkable inflammatory cell within the interstitial cell. Group E shows congestion of interstitial cell (ICG) and proliferation of interstitial cell. (x400) H&E
Hence, alkaloid-rich extract of Mimosaconsists of properties basically the alkaloids to suppress and inhibit toxicity on the hypothalamus-pituitary axis in the brain and also enhancing their activity, thereby increasing the synthesis of hormones needed for reproduction. Prophylactic treated of AEM on Dichlorvos-treated animals was efficacious than the therapeutic treatment of AEM on Dichlorvos-treated animals as observed from this study 19.
Conclusion
Overall, the alkaloids from Mimosa (AEM) leaves enhanced the released of gonadotropins by the cordial action of the hypothalamus-pituitary axis. Dichlorvos, a common insecticide, suppressed these hormones on the HPT axis. This deprivation effect of Dichlorvos led to a reduction in the secretory and synthetic functions of the testes and seminiferous tubular sperm parameters and an increase in the number of abnormal spermatozoa. The mechanism by which the alkaloids from Mimosa reversed the Dichlorvos effect on the androgenicity may be through enhancement on the hormone levels, androgen elevation to the target organ, increased spermatogenesis, and inhibited malformation of sperm cells.
References
- 1.V C Scanlon. (2006) Essentials of anatomy and physiology, Tina Sanders. FA Davis Company 5th edition .
- 2.T J Evans, V K Gunjam. (2011) Repoductive anatomy and physiology In:. R.C. Gupta, Editor, Reproductive and Developmental toxiciology, Academic press, Sandiego 7-32.
- 3.. Normal development of the male reproductive system.In:R.W.Kapp,R.W.Tyl(eds.),Reproductive toxicology, 3rd Edition,CRC.67-95 (2010). Marty MS, RJ Rasoulpour, Johnson KJ, Carney EW .
- 4.Kerr J B, Loveland K L, O'Bryan M K, DM de Kretse. (2006) Cytology of the testis and intrinsic control mechanisms. In:E.Knobil,J.D.Nil(Eds),Physiology of reproduction,Third edition,Elsevier Publishers. 827-920.
- 5.Gerhard F, Weinbauer C M L, Manuela S, Eberhard N. (2010) Physiology of Testicular Function. In:E.Nieschlag,H.M.Behre,S.Nieschlag(Eds.),Male reproductive health and dysfunction. , Andrology 629.
- 6.Carlsen E, Giwercman A, Keiding S, Skahhehaek N E. (1992) Evidence for decreasing quality of semen during past 50 years.Br Med J1992;305:. 609-613.
- 7.E W Wong, Cheng C Y. (2011) Impacts of environmental toxicants on male reproductive dysfunction.TrendsPharmacol. Sci. 32(5), 290-299.
- 9.Foll C V, Pant C P. (1966) The conditions of malaria transmission in Katsina Province, Northern Nigeria and a discussion of the effects of dichlorvos application. , Bull World Health Organ 34(3), 395-404.
- 10. (1999) BCERF: Breast Cancer and Environmental Risk Factors. Pesticides and Breast Cancer Risk, An Evaluation of Dichlorvos. in New York State Institute for Comparative and Environmental Toxicology Cornell Center for the Environment FACT SHEET #20 1, 4.
- 11.Azmi L, Singh M, Akhtar A. (2011) Pharmacological and biological overview on Mimosa pudica Linn.Int. , J. of Pharm. & Life Sci.2011; 2(11), 1226-1234.
- 12.J F Caius. (1980) Medicinal and poisonous Legumes of India.Pbl.Scientific Publishers,Jodhpur,India. 174-177.
- 14.Czapski G, Szypula W, Kudlik M, Wilenska B, Kania M et al. (2014) Assessment of antioxidative activity of alkaloids from Huperzia selago and Diphasiastrum complanatum using in vitro systems. Folia Neuropathol. 52, 1-13.
- 15.Seyma O K, Emre K, Seyfettin G, Seval Y, M C Ilyas.Effect of Starvation and Refeeding on Spermatological Parameters and Oxidative Stress in Rats. , International Journal of Health Sciences 7(2), 21-28.
- 16.Liang J A. (1979) Fertility and Infertility in Domestic Animas. 3rd Ed. , Baillieere Tindall, London
- 17.Franca L R, Suescun M O, Miranda J R, Giovambattista A, Perello M et al. (2006) Testis Structure and Function in a Nongenetic Hyperadipose Rat Model at Prepubertal and Adult Ages. Endocrinology. 147(3), 1556-1563.
- 18.Creasy D M. (2001) Pathogenesis of Male Reproductive Toxicity. Toxicological Pathology. 29(1), 64-76.
- 19.Zohreh N, Shahla R, Reza M. (2014) The Effect of Aloe Vera Extract on the Sperm Quality. in Male Diabetic Rats. Bulletin of Environment, Pharmacology and Life Sciences 3(3), 223-228.
