Abstract
Development of the hand occurs as a complex series of events. In order to understand the mechanisms of developmental hand anomalies, the clinician has to understand the normal embryonic development of the hand. In this review, we present the development of the hand in the light of new literature knowledge.
Author Contributions
Academic Editor: Jianliang jin, Department of Anatomy, School of Basic Medicine, Nanjing Medical University
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Baris Baykal, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
The development of the hand depends on many regulatory molecules secreted by varios regions of the developing upper limb. While some of these molecules have stimulatory effects, others have inhibitory effects. Both proliferation and apoptosis take place in various areas of the developing upper limb in order to form skeletal stuctures and spaces. The development of upper extremity has to be understood in order to understand the development of the hand. Being aware of the development and developmental mechanisms of the hand, may help clinicians in understanding the underlying mechanisms of congenital hand malformations.
Development of the Upper Extremıty
Development of the upper extremity begins with the appearance of the upper extremity bud by means of high levels of proliferation in the lower five cervical and first thoracal myotomes of somatopleural mesenchyme due to the effects of fibroblast growth factor 10 (FGF-10) and T-box transcription factor (Tbx5) on the 27. day of intrauterine life 1, 2, 3, 4. Specialized signaling centers that express and secrete many molecules help in determining and development along three spatial limb axes: proximodistal, anteroposterior, and dorsoventral 5. Three specialized signaling centers occur within the growing limb bud: apical ectodermal ridge, zone of polarizing activity, and nonridge ectoderm 5.
A condensed mesenchyme exists in the center of the extremity bud and a ridge termed as apical ectodermal ridge composed of tall columnar epithelium is present on the apical edge of the extremity bud 1, 2. The location of the apical ectodermal ridge is determined by radical fringe and engrailed-1 molecules secreted from the dorsal ecdoderm and ventral ectoderm of the extremity bud, respectively 3. While apical ectodermal ridge orchestrates the development of the extremity bud 2, the mesenchyme induces and maintains the development of the apical ectodermal ridge 1, 6. Fibroblast growth factors 4, 9 and 17 (FGF-4, FGF-9 and FGF-17) expressed by the apical ectodermal ridge induces outgrowth of the bud 3, 6.
In the guidance of homeobox b8 (Hoxb8), which exists at high concentrations at the posterior edge of the extremity bud 3, mesenchymal cells aggregate forming the zone of polarizing activity 2, 6, 7. The principal zone that controls the development of the extremities along their anteroposterior axes at a normal pattern with sonic hedgehog it secretes 3 is the zone of polarizing activity 2, 7. Gremlin, which is expressed in a domain anterior to the zone of polarizing activity 8 maintains the apical ectodermal ridge 2 and helps determine the anteroposterior axis of the bud 3. Sonic hedgehod also acts in the maintenance of the structure and functions of the apical ectodermal ridge 3, 6.
Development of the Hand
The development of the hand begins with the flattening of the distal ends of the extremity buds on the 34-38th days of development. Thus, paddle-like hand plates occur 1, 2. Development of the digits begin with the fragmentation of apical ectodermal ridge and on the 46th day of development hand plates take a notched shape and digit rays form 4. Apical ectodermal ridges at the tips of each digit, induces the mesenchyme to condense and transform into the primordia of phalanges 7. As a result of this induction cartilaginous primordia is formed. Transforming growth factor beta proteins (TGFB’s) and activins play a specific role in the induction of chondrogenesis in the digit ray 9, 10, 11. Bone morphogenic proteins (BMP’s) have reciprocal effects in this process. They have both growth promoter 8 and apoptotic effects 12 and the effects seem to take place by the changes in receptor expressions 13. The chondrogenic effects of BMP’s are enhancement of cell adhesion between the cell clusters in the regions of chondrogenesis, increasing of expression of Sry-related HMG box (SOX) genes and matrix production 13. SOX9 expression is the first indicator of verging towards chondrogenesis (Figure 1) 13, 14.
Figure 1.Diagram showing the apical ectodermal ridge, areas of cartilage matrix production due to the expression of SOX9, and the areas where apoptosis occurs by the effect of BMP-2, BMP-4, BMP-7, Msx-1 and Msx-2 in a developing digit. (Modified from “Tina V. Hellmann, Joachim Nickel and Thomas D. Mueller (2012). Missense Mutations in GDF-5 Signaling: Molecular Mechanisms Behind Skeletal Malformation, Mutations in Human Genetic Disease, Prof. David Cooper (Ed.), InTech, DOI: 10.5772/35195. OPEN ACCESS”)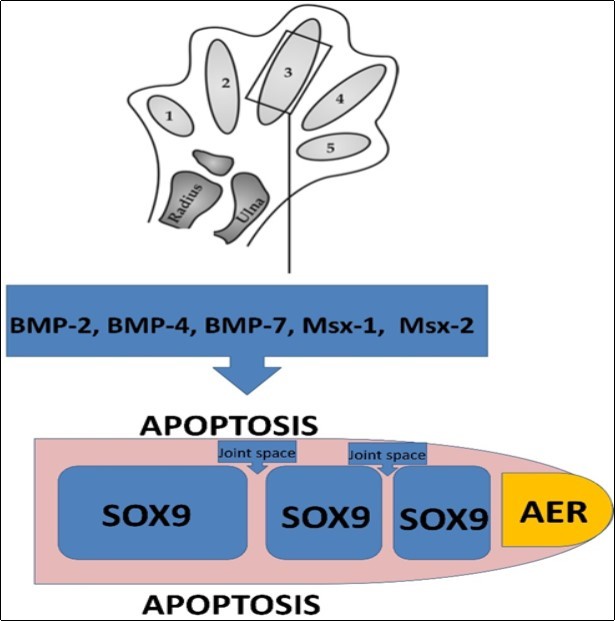
The extracellular matrix plays important roles in chondrogenesis. First of all, it acts as a scaffold for cell aggregation. It possibly effects the local transmission of growth factors both temporally and in concentration gradients. The extracellular matrix components themselves also are present in the developing digital ray at different concentrations. For example, a proteoglycan, syndecan-3, is expressed at high levels restricted to perichondrium in the cell aggregate before chondrogenesis and in the developing joints after differentiation 15, 16.
Spesific phalangeal segments arise as a result of segmentation 3. Activin-Like Kinase 3 (ALK3), a BMP receptor, may play a key regulatory role in joint formation and interdigitation via apoptosis 17. At the 50th day, the digits are webbed 4. The loose mesenchyme between the digit ray undergoes tissue breakdown via apoptosis and at the 52nd day seperate digits form 4, 7. BMP-2, BMP-4, BMP-7, Muscle Segment Homeobox 1 and 2 (Msx-1 and Msx-2) expressed in interdigital areas are the molecules that play a role in interdigital cell death 3. Sonic hedgehog (shh) is the molecule that plays the main role in the determination of digit identity 3 and the formation of digit patterning 2. Ossification of the phalanges occur antenatally. Carpal bones ossify postnatally 18.
The muscles of the hand are thought to develop in situ not by migration 19, 20. Initially, the mesenchyme of the hand divides into superficial and deep layers. Three muscle primordia (radial, middle and ulnar) develop from the superficial layer. Abductor pollicis brevis, flexor digitorum superficialis and abductor digiti minimi muscles develop from these primordia, respectively. The deep layer gives rise to interosseous and contrahentes muscles. The latter develops into the adductor pollicis muscle 20. Tendons, synovial sheets and pulley system of the digits are observed as condensing mesencyme at the 9th week of development. At the 12th week pulleys are identifiable as in the adult 21.
Brachial artery reaches the palm at 16 mm embryo stage. Digital arteries originate from the regressing capillary nets during notching. A digital vessel forms on the two sides of each forming interspace 22. Cells that originate and migrate from the neural crest contribute to the formation of the nerves of the limbs 7. Nerves begin grow into the limb bud at approximately 36th day of development 5.
Conclusıon
Development of the hand occurs in a complex manner by various molecular interactions. The awareness of hand development may help in understanding the formation mechanisms of congenital hand anomalies.
References
- 1.J P Iannotti, R D Parker.Embryology. in Netter Collection of Medical Illustrations: Biology and Systemic Diseases, J.P. Iannotti, Editor. 2013, Saunders: Electronic Version 1-22.
- 2.Standring S. (2008) Development of the limbs. in Gray’s Anatomy,S Standring, Editor,Churchill Livingstone:Spain 899-904.
- 3.Carlson B M. (2014) Limb Development, in Human Embryology and Developmental Biology,BM Carlson,Editor, Saunders:Electronic version. 193-215.
- 4.T N Singh. (2015) Split Forearm and Hand- a case report. , IOSR Journal of Dental and Medical Sciences 14(11), 20-22.
- 6.Baykal B, Korkusuz P. (2016) Development of the Musculoskeletal System. in Musculoskeletal Research and Basic Science,F Korkusuz,Editor.Springer International Publishing:Cham 289-302.
- 7.K L Moore, Persaud T V N, M G Torchia. (2013) Development of Limbs, in The Developing Human,KL Moore,TVN Persaud and MG Torchia,Editors. Saunders: Electronic version. 371-387.
- 8.Ganan Y. (1996) Role of TGF beta s and BMPs as signals controlling the position of the digits and the areas of interdigital cell death in the developing chick limb autopod. Development. 122(8), 2349-57.
- 9.Chimal-Monroy J, Leon L Diaz de. (1999) Expression of N-cadherin, N-CAM, fibronectin and tenascin is stimulated by TGF-beta1, beta2, beta3 and beta5 during the formation of precartilage condensations. , Int.J.Dev.Biol 43(1), 59-67.
- 10.C M Leonard. (1991) Role of transforming growth factor-beta in chondrogenic pattern formation in the embryonic limb: stimulation of mesenchymal condensation and fibronectin gene expression by exogenenous TGF-beta and evidence for endogenous TGF-beta-like activity. Dev Biol. 145(1), 99-109.
- 11.Merino R. (1998) Morphogenesis of digits in the avian limb is controlled by FGFs, TGFbetas, and noggin through BMP signaling. , Dev Biol 200(1), 35-45.
- 12.Merino R. (1999) Bone morphogenetic proteins regulate interdigital cell death in the avian embryo. , Ann N Y Acad Sci 887, 120-32.
- 14.Wagner T. (1994) Autosomal sex reversal and campomelic dysplasia are caused by mutations in and around the SRY-related gene SOX9. Cell. 79(6), 1111-20.
- 15.S E Gould, W B Upholt, R A Kosher. (1995) Characterization of chicken syndecan-3 as a heparan sulfate proteoglycan and its expression during embryogenesis. Dev Biol. 168(2), 438-51.
- 16.Koyama E. (1995) Syndecan-3, tenascin-C, and the development of cartilaginous skeletal elements and joints in chick limbs. , Dev Dyn 203(2), 152-62.
- 18.Jowett C, Barry M. (2016) Paediatric Upper Limb, in Paediatric Orthopaedics in Clinical Practice,NA Aresti,et al.,Editors. Springer:Electronic Version.
- 20.Hita-Contreras F. (2012) Development and morphogenesis of human wrist joint during embryonic and early fetal period. , J Anat 220(6), 580-90.